Abstract
The light-triggered release of deoxyribonucleic acid (DNA) from gold nanoparticle-based, plasmon resonant vectors, such as nanoshells, shows great promise for gene delivery in living cells. Here we show that intracellular light-triggered release can be performed on molecules that associate with the DNA in a DNA host-guest complex bound to nanoshells. DAPI (4′,6-diamidino-2-phenylindole), a bright blue fluorescent molecule that binds reversibly to double-stranded DNA, was chosen to visualize this intracellular light-induced release process. Illumination of nanoshell-dsDNA-DAPI complexes at their plasmon resonance wavelength dehybridizes the DNA, releasing the DAPI molecules within living cells, where they diffuse to the nucleus and associate with the cell's endogenous DNA. The low laser power and irradiation times required for molecular release do not compromise cell viability. This highly controlled co-release of nonbiological molecules accompanying the oligonucleotides could have broad applications in the study of cellular processes and in the development of intracellular targeted therapies.
Keywords: nanoshell, DAPI, controlled release, plasmon, DNA
Strategies for the directed release of controlled quantities of molecules inside living cells are in high demand for drug delivery,1 gene therapy,2, 3 and tissue engineering.4, 5 The release mechanisms of most delivery vectors depend on processes such as diffusion, dissolution, chemical and enzymatic reactions, or changes in various environmental factors such as temperature, pH, solvent, and ionic concentrations.2, 6-9 For example, transfection reagents such as polyethylenimine act as a proton sponges following endocytosis, absorbing protons in the low-pH environment of the endosome, causing it to swell and eventually rupture, facilitating gene delivery.10 This type of environmental control of molecular release varies with cellular location and cell type, and can result in unpredictable release. A physical release mechanism that does not rely on the specific chemical properties of the cellular environment would be highly useful and more easily generalizable to various cell types.11 Light-induced release is a particularly attractive option: the high spatial and temporal control that lasers provide would be highly useful for initiating and following intracellular processes dynamically, at the single cell level.12-18
Plasmonic nanoparticles, metal-based nanostructures supporting collective electronic oscillations, are highly promising potential candidates for facilitating controlled light-triggered release, due to their large optical cross sections, their geometrically-tunable optical resonances19-21 and their strong photothermal response.22, 23 Because of their large cross sections and extremely low quantum yield, metallic nanoparticles convert optical energy to thermal energy with high efficiency upon resonant optical illumination.24, 25 Resonant optical illumination of the nanoparticle triggers the controlled dehybridization and release of DNA molecules adsorbed onto the nanoparticle surface.16, 18 Gold nanoparticles are also biocompatible, easy to fabricate, and can be functionalized with a wide variety of host-carrier molecules capable of noncovalent accommodation of guest molecules.6, 18
Au nanoshells, metallodielectric nanoparticles comprised of a spherical dielectric core covered by a thin Au shell, have been successfully employed in a range of biomedical applications26-28 including photothermal cancer therapy.23, 29-32 The plasmon resonance wavelength of a nanoshell can be varied by changing the core size and shell thickness.20, 21, 33 This tunability is particularly important for biomedical applications because the nanoshell can easily be designed to have a large absorption efficiency in the near infrared (NIR) water window (690-900 nm), where blood and tissue are maximally transparent and light can penetrate tissue to depths of several inches.34
Recently we demonstrated light-induced dehybridization of double-stranded DNA (dsDNA) attached to Au nanoshells.18 Nanoshells of dimensions [r1, r2] = [63, 78] nm with the plasmon resonance wavelength at 800 nm were coated with dsDNA, where one strand of the DNA had a thiol moiety on its 5’ end, facilitating covalent attachment to the nanoshell surface by a Au-thiol bond. The complement DNA sequence was nonthiolated, and therefore bound only to its complementary DNA sequence and not to the nanoparticle surface. Upon illumination with NIR light at the nanoshell plasmon resonance wavelength, the dsDNA was dehybridized, releasing the antisense sequence. This process is highly efficient, resulting in the dehybridization and release of nominally 50% of the DNA from the complexes upon illumination, with no apparent temperature increase in the solution ambient. DNA antisense therapy has been explored extensively as a class of gene therapy and has highly promising potential to provide safe and effective treatments for a multitude of diseases and genetic disorders.35
Here we show that, in addition to light-controlled release of DNA, the nanoshell-dsDNA complex serves as an effective host and light-triggered release vector for other types of molecules. Many types of guest molecules can associate with dsDNA, either by intercalating between adjacent base pairs or by binding in either the major or minor groove of the DNA double helix.36 The driving forces for association can include π-stacking, hydrogen bonding, van der Waals forces, hydrophobic and polar interactions, and electrostatic attractions; therefore, dsDNA can host a large variety of guest molecules via noncovalent bonds.36, 37 DAPI (4′,6-diamidino-2-phenylindole), a water soluble blue fluorescent dye that binds reversibly with dsDNA (Figure S4) is the molecule we chose to deliver, to demonstrate and clearly visualize the light-induced intracellular release. DAPI was chosen because of its bright fluorescent properties, stability and negligible toxicity.38 DAPI binds preferentially to the minor grooves of dsDNA: its association with DNA causes a large increase in its quantum yield.38-42 The selectivity of DAPI to dsDNA makes it a frequently used, standard stain for cell nuclei in fluorescence microscopy.41, 42
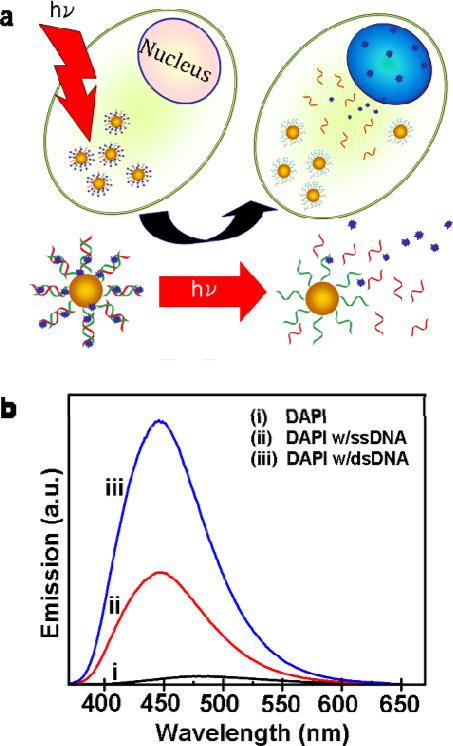
A schematic of the light-triggered molecular release is shown in Figure 1. Initially, nanoshell-dsDNA complexes were loaded with DAPI by incubation of DAPI with the nanoshell-dsDNA complexes (see supplemental information). Next, the nanoshell-dsDNA-DAPI complexes were incubated with H1299 lung cancer cells (see supplemental information), where intracellular uptake was verified using both dark-field and bright-field microscopy. Upon illumination with an 800 nm CW laser, corresponding to the peak resonant wavelength of the nanoshell complexes (Figure S3), the DAPI molecules were released from the nanoshell complexes. Subsequent to release, the DAPI diffused through the cytoplasm and into the cell nucleus, where it preferentially bound and stained the nuclear DNA. To the best of our knowledge, this is the first light-controlled delivery system that can be tailored to release quantifiable amounts of nonbiological molecules, within living cells by remote means, on demand.
Figure 1.
Light-induced DAPI release. (a) Schematic diagram of the light-induced DAPI release and diffusion inside the cell. (b) Fluorescence emission of (i) DAPI only, (ii) DAPI with ssDNA, and (iii) DAPI with dsDNA.
The DAPI fluorescence emission intensity drastically increases as a result of DAPI molecules binding to DNA (Figure 1b). As an isolated molecule, DAPI has a low quantum yield (Fig. 1b, i), 39 however, when DAPI is attached to single stranded DNA (ssDNA) (Fig. 1b, ii), a weak electrostatic attraction binds the cationic DAPI molecules to the negatively charged phosphate backbone of the DNA, resulting in a slight increase in its fluorescence intensity. 39 When the DAPI molecules bind to the minor grooves of the dsDNA (Fig. 1b, iii),39, 42 the increased rigidity and stabilization significantly increases its quantum yield.38 DAPI binding to the dsDNA also displaces H2O molecules initially solvating the DNA oligomers, significantly reducing intermolecular proton transfer between H2O and DAPI, resulting in an additional increase of DAPI fluorescence intensity.39, 40, 43 (Additional characterization of DAPIDNA binding is provided in the supporting information, Fig. S1).
The specific base-pair composition of the dsDNA plays an important role in determining the number of DAPI molecules that will bind to the dsDNA.44 Previous studies have shown that DAPI preferentially binds to regions rich with adenine (A) and thymine (T) nucleotide bases because DAPI forms hydrogen bonds with A-T bases pairs.42 The DAPI molecule is 14-15 Å long, corresponding to an overlap of three base pairs.39-41, 44 In our experiments, since it is desirable to bind as many DAPI molecules as possible to improve the staining of the nucleus after light-induced release, we designed a 26-base pair sequence with multiple A-T-rich regions with segments of three or more consecutive A-T base pairs to specifically enhance DAPI loading (Figure S2, supporting information).
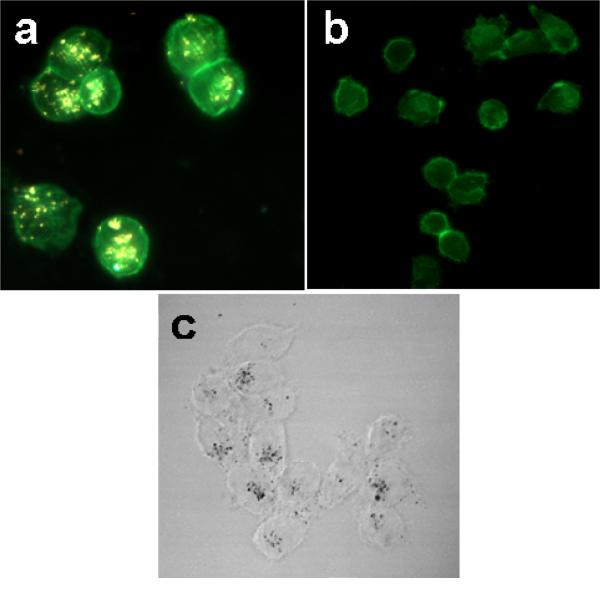
To use this light-triggerable complex for molecular release in live cells, the complex must first be effectively taken up by the cells of interest. To facilitate cell uptake, the nanoshell-dsDNA-DAPI complexes were incubated with H1299 lung cancer cells in serum containing cell culture medium for 1 hour. After incubation, the cells were fixed and internalization of the nanoshell-dsDNA-DAPI complexes was imaged using both dark field (Fig. 2a-b) and bright field (Fig. 2c) microscopy. Nanoshells in this size range both absorb and scatter light: their strong scattering cross section enables them to be easily visualized by optical microscopy. In Figure 2a, a H1299 cell with its cell membrane marked by the green fluorescence dye Alexa Fluor 488 WGA (wheat germ agglutinin) is shown. Internalized nanoshells are easily seen as diffraction-limited bright spots in this image. As a control, cells not incubated with the nanoshell-dsDNA-DAPI complexes showed no observable bright spots when imaged in the same manner (Fig. 2B).
Figure 2.
Nanoshell-dsDNA-DAPI Cell uptake. Dark field/epifluorescence images of (a) H1299 lung cancer cells incubated with nanoshell-dsDNA-DAPI complexes, (b) nonincubated cells (control). (c) Bright field image of middle slice of H1299 lung cancer cells incubated with nanoshell-dsDNA-DAPI complex.
Because the dark-field images are two-dimensional, these images alone do not give clear evidence whether the nanoshell complexes have been endocytosed, or are merely adsorbed onto the outer membrane of the cell. Bright-field imaging was used to further investigate cellular uptake. Obtaining images at varying depths of field within an individual cell allows us to clearly visualize in three dimensions the nanoshell distribution within the cell. Figure 2c is a slice from the middle of the cell showing clear diffraction-limited dark spots corresponding to nanoshell complexes, verifying that the nanoparticles are internalized within the cell. A video sequence of two-dimensional projections obtained as the depth of field is scanned through the cell resolves the 3D distribution of nanoshell complexes (provided in the supporting information). Internalization of nanoshells is in agreement with observations by Ochsenkühn et al, who used TEM sections of NIH-3T3 fibroblast cells to confirm nanoshell uptake.45
At first thought, it is surprising that the nanoshell-dsDNA-DAPI complex is internalized into cells because the negatively-charged phosphate backbone on the DNA should experience electrostatic repulsions with the negatively-charged cell membrane.46 However, previous studies by Chithrani et al and Giljohann et al suggest that Au nanoparticles functionalized both with and without DNA adsorb extracellular serum proteins from the cell culture media.46, 47 The adsorbed extracellular proteins then interact with the cell membrane and facilitate cellular uptake in an adsorptive endocytosis pathway. Conversely, recent studies by Ochsenkühn et al show that nanoshell uptake increases in the absence of extracellular proteins, suggesting the possibility of a passive, nonendocytotic uptake mechanism.45 While in our studies nanoshell complex uptake is clearly visualizable in H1299 cells, the precise uptake mechanism is not clearly identifiable, and is likely to depend on a variety of factors including cell type, functionalization of the nanoparticle, and incubation conditions.
To investigate intracellular light-induced molecular release, the H1299 cells incubated with nanoshell-dsDNA-DAPI complexes were illuminated with a NIR CW laser (1 W/cm2, 800 nm) for 5 minutes. This irradiation time and laser power level were determined from previous experiments,18 which demonstrated after 5 minutes of laser irradiation no additional dehybridization of the DNA occurred. This laser power and time allow the DAPI to be released while minimizing laser exposure to the H1299 cells. After laser irradiation, samples were placed in an incubator for one hour to allow time for released DAPI molecules to diffuse to the nucleus. Next, the nuclei of the cells were isolated by lysing the cell membrane (see supplemental information) and the DAPI fluorescence intensity was quantified by flow cytometry. Nuclei isolation is necessary to ensure that flow cytometry only measures fluorescence from DAPI molecules bound to genomic DNA in the nucleus and does not measure fluorescence from DAPI molecules in the cytoplasm.
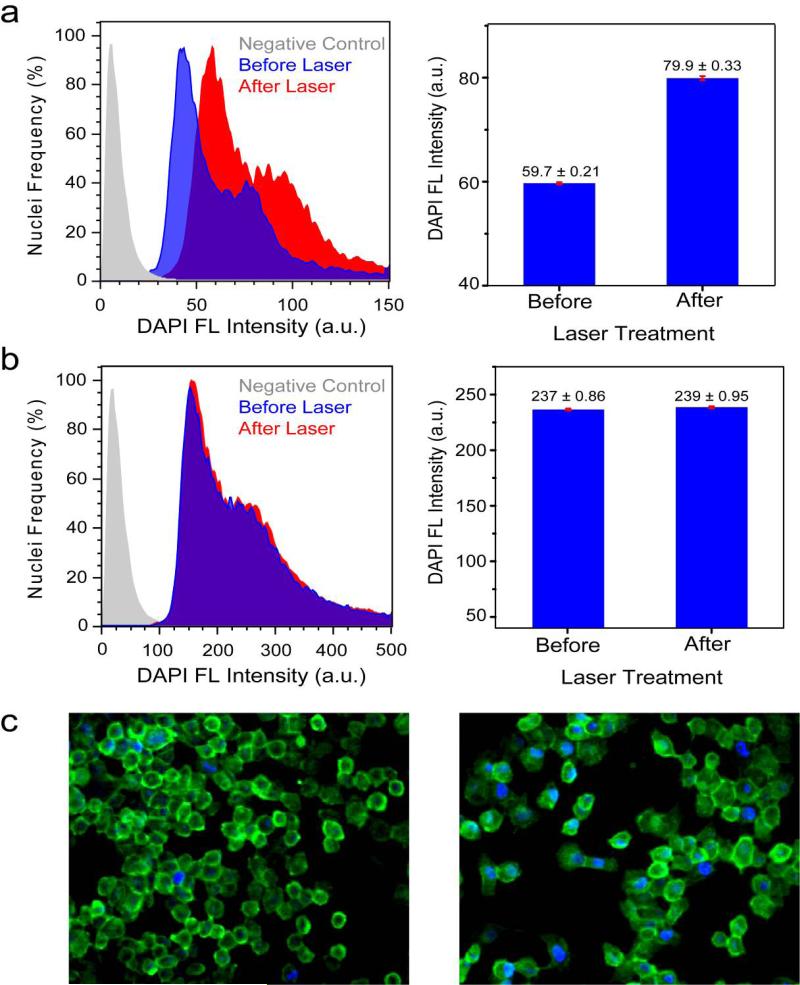
Evidence of DAPI release is shown by the normalized flow cytometry histograms of DAPI fluorescence intensity versus number of nuclei from H1299 cells incubated with nanoshell-dsDNADAPI before and after laser treatment (Figure 3). After laser treatment, the fluorescence intensity of the nuclei increased, demonstrating that DAPI molecules were released from the nanoshells, diffused through the cytoplasm and into the cell nuclei, binding with the genomic DNA. Prior to laser irradiation, some DAPI fluorescence is observed within the cells (Figure 3c, left) and measured by flow cytometry (Fig. 3a-b, before laser). This DAPI fluorescence signal originates from both excess DAPI molecules present in the sample and DAPI molecules which were non-controllably released from the complexes during the incubation and prior to laser irradiation.
Figure 3.
Light-induced DAPI release. (a-b) Flow cytometry histograms of DAPI Fluorescence (Ex: 355nm/Em: 460 nm) versus number of isolated nuclei from H1299 cells incubated with a) nanoshelldsDNA-DAPI and b) DAPI (control). Negative control (gray), treated cells without laser irradiation (blue) and treated cells with laser irradiation (red). Bar graphs display the mean DAPI fluorescence intensity ± SEM before and after laser irradiation. (c) Epifluorescence images of H1299 cells incubated with nanoshell-dsDNA-DAPI (left) before and (right) after laser treatment. The cell membrane is marked by the green dye, Alexa-Fluor 488.
The bar graph depicts the mean DAPI fluorescence intensity ± SEM (standard error of the mean) increase from before laser (59.7±0.21) to after laser (79.8±0.33) (Fig. 3a). A ~33% increase in fluorescence intensity. An unpaired t-test of the two means was performed at a 95% confidence level, which resulted in a two-tailed p value of p < 0.0001, which is statistically significant. This observed increase in DAPI fluorescence after laser treatment (~33%) demonstrates that the nanoshell-dsDNA complex effectively released its guest molecules from the dsDNA host carriers inside the cells. Epifluorescence images of H1299 cells incubated with nanoshell-dsDNA-DAPI before (Figure 3c, left) and after (Figure 3c, right) laser treatment visually show the increase in DAPI fluorescence intensity. The cell membrane is marked by the green dye, Alexa-Fluor 488 wheat germ agglutinin.
The plasmon resonant illumination of the nanoshells is crucial for DAPI release into the cells. To test this hypothesis, a control experiment consisting of H1299 cells incubated with DAPI only (no nanoshells) was conducted (Figure 3b). The cells were irradiated with the NIR laser under conditions identical to the previous experiment. The mean DAPI fluorescence intensity ± SEM did not significantly increase after laser irradiation (237±0.86 to 239 ± 0.95, p = 0.1188), indicating that DAPI release does not occur without the presence of the nanoshell-dsDNA complex. It is important to note that the mean fluorescence intensity is higher for the control (Fig. 3b) compared to the nanoshell-dsDNA-DAPI sample (Fig. 3a) due to multiple washings of the nanoshell-dsDNA-DAPI sample.
The shape of the flow cytometry histograms for both before laser and after laser are consistent with nuclei stained with DAPI.48, 49 DAPI is routinely used to study the cell cycle because it binds to DNA stoichiometrically. Looking at the before laser histogram in figure 3a as an example, the tallest peak (~40) originates from nuclei with two sets of chromosomes. This peak is the tallest because in a typical cell cycle, a cell spends the longest portion of time with two sets of chromosomes; therefore, the probability of a cell having two sets of chromosomes is the highest. The second, smaller peak (~80), double the fluorescence intensity of the tallest peak, indicates nuclei which have exactly double the amount of DNA, four sets of chromosomes, and are ready to enter mitosis and divide. The nuclei with fluorescence intensities in between these two peaks indicate cells which are currently synthesizing DNA prior to mitosis. The negative control histogram (gray) has a single peak because in the absence of DAPI every nuclei essentially fluoresces identically resulting in a signal which is attributed to autofluorescence (background).
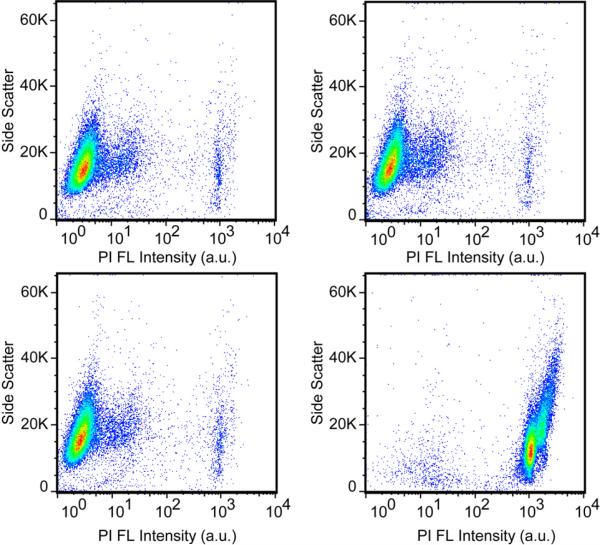
To ensure that this method for light-triggered intracellular molecular release would be useful for biomedical applications, such as drug delivery, a cytotoxicity assay was performed to investigate both the effects of nanoshells and laser irradiation on cell viability. Propidium iodide (PI) was chosen as a marker to distinguish viable from non-viable cells, because it is a membrane-impermeable dye which is excluded from viable healthy cells.50 When a cell membrane is damaged, PI enters the cell, stains the dsDNA in the nuclei and emits red fluorescence; however, undamaged cells will not fluoresce. Flow cytometry was used to observe changes in PI fluorescence intensity for a large sample size of 30,000 cells. The negative control (Figure 4a) consisted of cells which were not incubated with nanoshell-dsDNA-DAPI and did not undergo laser treatment. The fluorescence observed in Figure 4a is attributed to autofluorescence and PI staining caused by apoptotic and necrotic cells already present in the experiment, with damaged membranes. The nanoshell-dsDNA-DAPI complexes were then incubated with H1299 cells for 12 hours. Following incubation, the cells were divided into two samples: cells not treated with the laser (Figure 4b) and cells treated with the laser for 10 minutes (Figure 4c). Figure 4b shows no significant increase in PI fluorescence intensity, demonstrating that nanoshell-dsDNA-DAPI complexes are not cytotoxic under the experimental conditions of the study.
Figure 4.
Flow cytometry cytotoxicity assay. All plots are side-scattered light (SS) versus Propidium Iodide (PI) intensity. (a) Negative control: H1299 cells not incubated with nanoshell-dsDNA-DAPI and no laser treatment. Cells incubated with nanoshell-dsDNA-DAPI for 12 hours: (b) without laser treatment and (c) with laser treatment. (d) Positive Control: Cells were treated with 0.1%Citrate/0.1%Triton, which permeates the cell membrane, allowing PI to stain the dsDNA in the nucleus.
More interestingly, cells incubated with nanoshell-dsDNA-DAPI complexes and irradiated with the laser for 10 minutes also show no significant increase in PI fluorescence intensity. This demonstrates that the light-triggered release procedure did not adversely affect the cells. Considering nanoshells are well-known for their use in photothermal therapy, this result may be surprising; however, the illumination conditions for this experiment (1W/cm2, 5 minutes) were significantly below those used for photothermal induction of cell death in cell culture (4W/cm2, 4-6 minutes).23 Figure 4d represents a positive control sample of cells treated with 0.1% Citrate/0.1% Triton solution, which permeates the cell membrane, allowing PI to enter the cell and stain the nucleus, resulting in a large increase in PI fluorescence intensity.
In conclusion, nanoshells functionalized with dsDNA were successfully used to transport DAPI molecules into living cells. Successful uptake of nanoshells into H1299 cells was achieved. DAPI molecules, initially bound to the dsDNA on the NS surface, are released due to the illumination of the nanoshell-dsDNA-DAPI complex with the appropriate NIR light. DAPI molecules initially released in the cell cytoplasm diffuse into the cell nucleus and bind to the genomic DNA of the cell. The staining of the cell nucleus with the released DAPI was quantified using flow cytometry. A cytotoxicity assay demonstrated that nanoshell uptake is nontoxic and that laser irradiation of nanoshell-laden cells under the conditions where DAPI release occurs does not induce cell death.
This nanoshell-dsDNA system could be extended to a multitude of other guest molecules that associate with the host dsDNA carrier including small organic fluorophores,37 steroid hormones,51 and therapeutic molecules.36, 37, 51 For example, the quest to find dsDNA intercalators that inhibit the uncontrollable replication of tumor cells comprises an entire field of cancer research. Currently, there are more than 130 FDA approved anti-cancer drugs that specifically target DNA.52 For in vivo clinical applications, however, before the DNA intercalator can reach the genomic DNA, it must overcome several hurdles, such as metabolic pathways and cytoplasmic and nuclear membranes. As a result, the failure of DNA therapies to offer successful clinical treatments is primarily due a lack of viable delivery methods rather than effectiveness of the DNA intercalator to treat cancer.36 This nanoshell-dsDNA delivery vector preserves the guest molecule by minimizing non-desired interactions with other molecules and it provides light-triggered release with controllable delivery.
Supplementary Material
Acknowledgments
The work was supported by the Robert A. Welch Foundation (C-1220).
We thank Dr. Lin Ji from University of Texas MD Anderson Cancer Center for help in providing and growing H1299 lung cancer cells, Karen Ramirez of the University of Texas MD Anderson Cancer, Center Flow Cytometry Facility NCI #: P30CA16672, Walter Hittelman of the Department of Experimental Therapeutics M. D. Anderson Cancer Center Epifluorescence Microscope Facility, Katherine Regan for help with bright-field images, Pernilla Wittung-Stafshede, Benjamin G. Janesko, and Surbhi Lal for helpful discussions.
Footnotes
Supporting Information Available
Experimental details, sample preparation, instrumentation used, and experimental results: CD, UV-Vis absorbance spectra, fluorescence of DAPI binding to DNA, base specificity of DAPI binding, and the effect of buffers on DAPI fluorescence intensity are included. This material is available free of charge via the Internet at: http://pubs.acs.org.
References
- 1.Langer R. Nature. 1998;392(6679):5–10. [PubMed] [Google Scholar]
- 2.Panyam J, Labhasetwar V. Adv. Drug Delivery Rev. 2003;55(3):329–347. doi: 10.1016/s0169-409x(02)00228-4. [DOI] [PubMed] [Google Scholar]
- 3.Niidome T, Huang L. Gene Ther. 2002;9(24):1647–1652. doi: 10.1038/sj.gt.3301923. [DOI] [PubMed] [Google Scholar]
- 4.Shea LD, Smiley E, Bonadio J, Mooney DJ. Nat. Biotechnol. 1999;17(6):551–554. doi: 10.1038/9853. [DOI] [PubMed] [Google Scholar]
- 5.Richardson TP, Peters MC, Ennett AB, Mooney DJ. Nature Biotechnology. 2001;19(11):1029–1034. doi: 10.1038/nbt1101-1029. [DOI] [PubMed] [Google Scholar]
- 6.Kim CK, Ghosh P, Pagliuca C, Zhu ZJ, Menichetti S, Rotello VM. J. Am. Chem. Soc. 2009;131(4):1360–1361. doi: 10.1021/ja808137c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.LaVan DA, McGuire T, Langer R. Nat. Biotechnol. 2003;21(10):1184–1191. doi: 10.1038/nbt876. [DOI] [PubMed] [Google Scholar]
- 8.Kim SY, Shin ILG, Lee YM, Cho CS, Sung YK. J. Controlled Release. 1998;51(1):13–22. doi: 10.1016/s0168-3659(97)00124-7. [DOI] [PubMed] [Google Scholar]
- 9.Chilkoti A, Dreher MR, Meyer DE, Raucher D. Adv. Drug Delivery Rev. 2002;54(5):613–630. doi: 10.1016/s0169-409x(02)00041-8. [DOI] [PubMed] [Google Scholar]
- 10.Boussif O, Lezoualch F, Zanta MA, Mergny MD, Scherman D, Demeneix B, Behr JP. Proc. Natl. Acad. Sci. U S A. 1995;92(16):7297–7301. doi: 10.1073/pnas.92.16.7297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shalek AK, Robinson JT, Karp ES, Lee JS, Ahn DR, Yoon MH, Sutton A, Jorgolli M, Gertner RS, Gujral TS, MacBeath G, Yang EG, Park H. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(5):1870–1875. doi: 10.1073/pnas.0909350107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen CC, Lin YP, Wang CW, Tzeng HC, Wu CH, Chen YC, Chen CP, Chen LC, Wu YC. J. Am. Chem. Soc. 2006;128(11):3709–3715. doi: 10.1021/ja0570180. [DOI] [PubMed] [Google Scholar]
- 13.Takahashi H, Niidome Y, Yamada S. Chem. Commun. 2005;(17):2247–2249. doi: 10.1039/b500337g. [DOI] [PubMed] [Google Scholar]
- 14.Wijaya A, Schaffer SB, Pallares IG, Hamad-Schifferli K. Acs Nano. 2009;3(1):80–86. doi: 10.1021/nn800702n. [DOI] [PubMed] [Google Scholar]
- 15.Braun GB, Pallaoro A, Wu GH, Missirlis D, Zasadzinski JA, Tirrell M, Reich NO. Acs Nano. 2009;3(7):2007–2015. doi: 10.1021/nn900469q. [DOI] [PubMed] [Google Scholar]
- 16.Lee SE, Liu GL, Kim F, Lee LP. Nano Lett. 2009;9(2):562–570. doi: 10.1021/nl802689k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Han G, You CC, Kim BJ, Turingan RS, Forbes NS, Martin CT, Rotello VM. Angew. Chem. Int. Ed. 2006;45(19):3165–3169. doi: 10.1002/anie.200600214. [DOI] [PubMed] [Google Scholar]
- 18.Barhoumi A, Huschka R, Bardhan R, Knight MW, Halas NJ. Chem. Phys. Lett. 2009;482(4-6):171–179. [Google Scholar]
- 19.Prodan E, Nordlander P, Halas NJ. Nano Lett. 2003;3(10):1411–1415. [Google Scholar]
- 20.Wang H, Brandl DW, Nordlander P, Halas NJ. Acc. Chem. Res. 2007;40(1):53–62. doi: 10.1021/ar0401045. [DOI] [PubMed] [Google Scholar]
- 21.Prodan E, Radloff C, Halas NJ, Nordlander P. Science. 2003;302:419–422. doi: 10.1126/science.1089171. [DOI] [PubMed] [Google Scholar]
- 22.El-Sayed IH, Huang X, El-Sayed MA. Cancer Lett. (Amsterdam, Netherlands) 2006;239:129–135. doi: 10.1016/j.canlet.2005.07.035. [DOI] [PubMed] [Google Scholar]
- 23.Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, Price RE, Hazle JD, Halas NJ, West JL. Proc. Natl. Acad. Sci. USA. 2003;100:13549–13554. doi: 10.1073/pnas.2232479100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Richardson HH, Carlson MT, Tandler PJ, Hernandez P, Govorov AO. Nano Lett. 2009;9(3):1139–1146. doi: 10.1021/nl8036905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Govorov AO, Richardson HH. Nano Today. 2007;2(1):30–38. [Google Scholar]
- 26.Hirsch LR, Jackson JB, Lee A, Halas NJ, West JL. Anal.Chem. 2003;75:2377–2381. doi: 10.1021/ac0262210. [DOI] [PubMed] [Google Scholar]
- 27.Sershen SR, Westcott SL, Halas NJ, West JL. J. Biomed. Mater. Res. 2000;51:293–298. doi: 10.1002/1097-4636(20000905)51:3<293::aid-jbm1>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 28.Hirsch LR, Gobin AM, Lowery AR, Tam F, Drezek RA, Halas NJ, West JL. Ann. Biomed. Eng. 2006;34(1):15–22. doi: 10.1007/s10439-005-9001-8. [DOI] [PubMed] [Google Scholar]
- 29.Loo C, Lowery A, Halas NJ, J W, Drezek R. Nano Lett. 2005;5(4):709–711. doi: 10.1021/nl050127s. [DOI] [PubMed] [Google Scholar]
- 30.O'Neal DP, Hirsch LR, Halas NJ, Payne JD, West JL. Cancer Lett. (Amsterdam, Netherlands) 2004;209:171–176. doi: 10.1016/j.canlet.2004.02.004. [DOI] [PubMed] [Google Scholar]
- 31.Choi MR, Stanton-Maxey KJ, Stanley JK, Levin CS, Bardhan R, Akin D, Badve S, Sturgis J, Robinson JP, Bashir R, Halas NJ, Clare SE. Nano Letters. 2007;7:3759–3765. doi: 10.1021/nl072209h. [DOI] [PubMed] [Google Scholar]
- 32.Lal S, Clare SE, Halas NJ. Accounts of Chemical Research. 2008;41:1842–1851. doi: 10.1021/ar800150g. [DOI] [PubMed] [Google Scholar]
- 33.Oldenburg SJ, Jackson JB, Westcott SL, Halas NJ. Appl. Phys. Lett. 1999;75(19):2897–2899. [Google Scholar]
- 34.Weissleder R. Nature Biotechnology. 2001;19:316–317. doi: 10.1038/86684. [DOI] [PubMed] [Google Scholar]
- 35.Stephens AC, Rivers RP. A. Curr. Opin. Mol. Ther. 2003;5(2):118–122. [PubMed] [Google Scholar]
- 36.Neto BAD, Lapis AA. M. Molecules. 2009;14(5):1725–1746. doi: 10.3390/molecules14051725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ihmels H, Otto D. Top. Curr. Chem. 2005;258:161–204. [Google Scholar]
- 38.Barcellona ML, Gratton E. Eur Biophys J. 1990;17(6):315–323. doi: 10.1007/BF00258380. [DOI] [PubMed] [Google Scholar]
- 39.Manzini G, Xodo L, Barcellona ML, Quadrifoglio F. J. Biosci. 1985;8(3-4):699–711. [Google Scholar]
- 40.Kapuściński J, Szer W. Nucl. Acids Res. 1979;6(11):3519–3534. doi: 10.1093/nar/6.11.3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kubista M, Aakerman B, Norden B. Biochemistry. 1986;26(14):4545–4553. doi: 10.1021/bi00388a057. [DOI] [PubMed] [Google Scholar]
- 42.Lin MS, Comings DE, Alfi OS. Chromosoma. 1977;60(1):15–25. doi: 10.1007/BF00330407. [DOI] [PubMed] [Google Scholar]
- 43.Jung KS, Kim MS, Lee GJ, Cho TS, Kim EK, Yi SY. Bull. Korean Chem. Soc. 1997;18(5):510–514. [Google Scholar]
- 44.Breusegem SY, Clegg RM, Loontiens FG. J. Mol. Biol. 2002;315:1049–1061. doi: 10.1006/jmbi.2001.5301. [DOI] [PubMed] [Google Scholar]
- 45.Ochsenkuhn MA, Jess PRT, Stoquert H, Dholakia K, Campbell CJ. Acs Nano. 2009;3(11):3613–3621. doi: 10.1021/nn900681c. [DOI] [PubMed] [Google Scholar]
- 46.Chithrani BD, Ghazani AA, Chan WCW. Nano Lett. 2006;6(4):662–668. doi: 10.1021/nl052396o. [DOI] [PubMed] [Google Scholar]
- 47.Giljohann DA, Seferos DS, Patel PC, Millstone JE, Rosi NL, Mirkin CA. Nano Lett. 2007;7(12):3818–3821. doi: 10.1021/nl072471q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lydon MJ, Keeler KD, Thomas DB. J. Cell. Physiol. 1980;102:175–181. doi: 10.1002/jcp.1041020208. [DOI] [PubMed] [Google Scholar]
- 49.Taylor IW. J. Histochem. Cytochem. 1980;28:1021–1024. doi: 10.1177/28.9.6157714. [DOI] [PubMed] [Google Scholar]
- 50.Dengler WA, Schulte J, Berger DP, Mertelsmann R, Fiebig HH. Anti-Cancer Drugs. 1995;6(4):522–532. doi: 10.1097/00001813-199508000-00005. [DOI] [PubMed] [Google Scholar]
- 51.Hendry LB, Mahesh VB, Bransome ED, Ewing DE. Mutat. Res. 2007;623:53–71. doi: 10.1016/j.mrfmmm.2007.03.009. [DOI] [PubMed] [Google Scholar]
- 52.Wheate NJ, Brodie CR, Collins JG, Kemp S, Aldrich-Wright JR. Mini-Rev. Med. Chem. 2007;7(6):627–648. doi: 10.2174/138955707780859413. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.