Abstract
Malignant ventricular arrhythmias are a major adverse event and worsen the prognosis of patients affected by ischemic and non-ischemic dilated cardiomyopathy. The main parameter currently used to stratify arrhythmic risk and guide decision making towards the implantation of a cardioverter defibrillator is the evaluation of the left ventricular ejection fraction. However, this strategy is characterized by several limitations and consequently additional parameters have been suggested in order to improve arrhythmic risk stratification. The aim of this review is to critically revise the prognostic significance of non-invasive diagnostic tools in order to better stratify the arrhythmic risk prognosis of dilated cardiomyopathy patients.
Keywords: Dilated cardiomyopathy, Major ventricular arrhythmias, Prognosis, Ventricular repolarization, Left ventricular systolic function
Core tip: Arrhythmic risk stratification and decision making towards implantation of a cardioverter defibrillator in dilated cardiomyopathy patients are still open challenges. This review critically revises the possible clinical usefulness of available non-invasive diagnostic tools employed to stratify arrhythmic risk prognosis in dilated cardiomyopathy patients.
INTRODUCTION
The main adverse events affecting the prognosis for both ischemic (IDCM) and non-ischemic (NIDCM) dilated cardiomyopathy patients are the occurrence of malignant ventricular arrhythmias and sudden death and the progression towards heart failure[1]. In order to reduce the incidence of sudden death due to ventricular arrhythmias, the best therapeutic strategy to date is cardioverter defibrillator implantation (ICD)[1-3]. Both for NIDCM and IDCM, the decision to implant an ICD is mainly guided by the evaluation of left ventricular systolic function, i.e., by the calculation of left ventricular ejection fraction (LVEF)[4]. However, its use in defining eligible patients has a number of limitations.
In particular, there are a large number of patients who do not benefit from ICD[5]. In fact, the majority of patients with low LVEF who were enrolled in the main trials evaluating the effect of ICD did not suffer from malignant ventricular arrhythmias. For example, only 26% of the MADIT II patients had malignant ventricular arrhythmias during a 24 mo follow-up[2]. Only 31% of the 829 patients enrolled in the ICD group of the SCD-HeFT trial received shocks from their device for any cause and only 177 (21%) received shocks to arrest rapid ventricular tachycardia or ventricular fibrillation. During a five year follow-up, the annual average rate of ICD shocks was 7.5%; however, the annual average rate for appropriate ICD shocks (i.e., shocks for rapid, sustained ventricular tachycardia or fibrillation) was 5.1%. Moreover, in the SCD-HeFT trial, 32 (4%) patients had their ICD removed during follow-up and ICD complications, defined as clinical events requiring surgical correction, hospitalization, or new and otherwise unanticipated drug therapy, occurred in 5% of patients at the time of implantation and in 9% at a later stage in the trial[3].
It is clear from these data that the need to better assess arrhythmic risk is still a challenge[5]. Better characterization of patients using additional parameters should be able to detect those with a higher or lower risk of arrhythmic events, thus avoiding ICD implantation in patients with low LVEF at low risk and facilitating the implantation of patients with good LVEF at higher risk (Figure 1).
Figure 1.
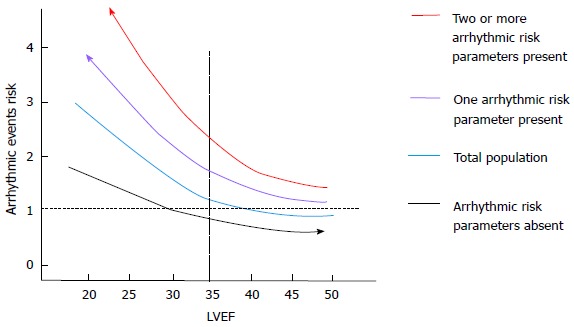
The effect of a better arrhythmic risk stratification are shown. The presence of one or more arrhythmic risk factor allows detection of a population at higher risk of arrhythmic events across all the values of left ventricular ejection fraction. On the other hand, the absence of arrhythmic risk factors is associated with the detection of the group of patients at lower risk of events. LVEF: Left ventricular ejection fraction.
The aim of this review is to critically revise the possible clinical usefulness of the available non-invasive parameters related to the pathophysiology of ventricular arrhythmias (Figure 2) which have been proposed in order to better stratify the arrhythmic risk of dilated cardiomyopathy patients.
Figure 2.
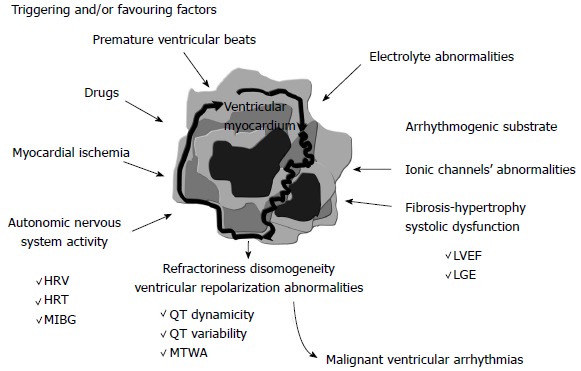
The main parameters proposed in order to better characterize arrhythmic risk are shown. These parameters can reflect arrhythmic substrate by functional (left ventricular systolic function) or anatomical (myocardial fibrosis) information. The parameters assessing sympathetic nervous system activity are also reported, as well as those reflecting the dispersion of ventricular refractoriness, i.e., those based on the analysis of ventricular repolarization. HRV: Heart rate variability; HRT: Heart rate turbulence; LGE: Late gadolinium enhancement; LVEF: Left ventricular ejection fraction; MIBG: Iodine-123 metaiodobenzylguanidine; MTWA: Microvolt T-wave alternans.
THE IMAGING TO DETECT ARRHYTHMIC SUBSTRATES
The assessment of left ventricular systolic function
As previously stated, the use of LVEF to guide decisions on whether to implant ICD leads to only a small percentage that will suffer from ventricular arrhythmias in a selection of a large population. However, the limitation of this approach is also related to several technical and biological aspects.
Firstly, in repeated evaluations, the LVEF calculation is characterized by a wide variability, particularly when an echocardiographic approach is considered. This is even more pronounced when different readers perform the calculation[6]. An improvement in the accuracy of LVEF calculation by echocardiography could be obtained using contrast echocardiography[7] or the 3-dimensional (3D) approach[8], but the gold standard for a more accurate and reproducible 3-D quantification of left ventricular (LV) volumes is cardiac magnetic resonance (CMR)[7,9].
Apart from the technical limitations in LVEF assessment, variability of the measure may also be influenced by biological factors. In particular, LVEF can vary in the different loading conditions due to changes in intravascular volumes and/or adrenergic drive[5,10]. Moreover, LVEF can change over time in response to conventional medical therapy[11].
In this setting, the new echocardiographic measures to evaluate left ventricular systolic function, which are less loading dependent, could be a new, useful tool to improve arrhythmic risk stratification by echocardiography[10]. Among these, two-dimensional (2-D) speckle tracking analysis[12] seems to be a particularly promising technique as it has been validated by sonomicrometry and tagged magnetic resonance imaging[13] and can quantify global and regional cardiac function more accurately and objectively by detecting mild ventricular function abnormalities in both left and right ventricular cardiomyopathies[14-15].
2-D speckle-tracking analysis is based on the detection and the motion tracking of natural acoustic myocardial reflections and interference patterns within an ultrasonic window. The tracking system analyses of echocardiographic grayscale B-mode images permits measurement of the entity of myocardial deformation (strain). Strain parameters can be individualized for each of the myocardial segments or can be expressed as global strain when all the segmental values are averaged. The global longitudinal strain (GLS) is the mean values of myocardial segmental deformation, evaluated using standard apical views. From a technical point of view, the use of 2-D strain measures offers some advantages over routine echocardiographic assessment of LVEF using Simpson’s rule. In particular, strain analysis is not based on any geometrical assumption and should depend less on loading conditions. Moreover, in regional contractility dysfunction, strain measures better correlate with LVEF as assessed by magnetic resonance[16]. Finally, GLS is easy to compute and less dependent on specific training to ensure reproducibility[17].
In order to evaluate the role of this novel technique in stratifying arrhythmic risk prognosis, we recently studied a group of heart failure (HF) outpatients affected by IDCM and NIDCM who had never previously experienced sustained ventricular arrhythmias[18]. During a mean follow-up of 26 ± 13 mo, 31 of 230 patients experienced entricular ventricular tachycardia (VT)/fibrillation (VF) or sudden death. At multivariate analysis, after correction for the univariate predictors, i.e., NYHA class, NT-proBNP and non-sustained ventricular tachycardia (NSVT), GLS remained significantly associated with ventricular arrhythmic events. The best GLS cut-off value detected by ROC curves for the 1 year occurrence of events was -10.0%, with a 73% sensitivity and a 61% specificity in detecting patients prone to experiencing major ventricular arrhythmias. Interestingly, the annual incidence rates of arrhythmic events were significantly greater in the 24 patients with a LVEF > 35% and a GLS above -10% than in the 114 patients with GLS below -10%, whereas no additive value was observed among patients with a LVEF ≤ 35%.
Assessment of myocardial fibrosis
In arrhythmic risk stratification, the usefulness of CMR is related not only to the possibility of more accurately estimating LVEF[19-22], but also to its ability to detect the presence of myocardial replacement fibrosis[23]. CMR assessment of fibrosis is made possible by using late gadolinium enhancement. Gadolinium is a contrast agent that has been shown to be extremely safe. It is an extracellular agent, accumulating in areas of interstitial expansion due to myocardial fibrosis, edema or infiltration. After gadolinium administration, it is possible to assess three phases: the first provides immediate images at rest or during stress, followed by early enhancement after 5 min and late enhancement 5 to 20 min after administration[22]. Late gadolinium enhancement (LGE) imaging allows the detection of contrast accumulation in areas of infarction or fibrosis due to slower contrast kinetics and greater volume or distribution in extracellular matrix. The extent and pattern of LGE enhancement varies according to the underlying pathological process. Fibrosis extent can be quantified as a percentage of total LV mass using dedicated software[22-23]. Moreover, the relative safety of gadolinium agents and tissue characterization sequences allows repeated imaging, follow-up, family screening and serial risk stratification[24].
The presence of fibrosis, as assessed by LGE, is associated with a greater probability of inducible ventricular tachycardia[25]. Moreover, there is considerable evidence that it is also associated with a worse prognosis and an increased arrhythmic risk. Table 1 summarizes the main studies with this evidence[26-33].
Table 1.
The main studies evaluating the association between myocardial fibrosis assessed by cardiac magnetic resonance and the risk of arrhythmic and non-arrhythmic events
| Ref. | Clinical setting | Number of patients | CMR parameters | End-points (mean follow-up) | Results |
| Assomoul et al[26], 2006 | NIDCM | 101 | Midwall fibrosis (LGE) | All-cause death and hospitalization (follow-up 658 ± 355 d) | Independent association with death and hospitalization |
| Wu et al[27], 2008 | NIDCM and LVEF ≤ 35% | 65 | Presence and extent of LGE | Composite end-point (hospitalization for heart failure, appropriate ICD firing, cardiac death) (Follow-up median 24 mo) | Presence of LGE was associated with a greater risk of primary outcome |
| Iles et al[28], 2011 | IDCM/NIDCM before ICD implantation | 103 | Regional fibrosis with LGE | Arrhythmic events and appropriate ICD therapy (follow-up 573 d) | LGE was associated with arrhythmic events and appropriate ICD therapy during follow-up |
| Lehrke et al[29], 2011 | NIDCM | 184 | Presence of LGE | Composite end-point (hospitalization for decompensated heart failure, cardiac death, cardioverter defibrillator discharge) (follow-up 31 mo) | Presence of LGE was associated with composite endpoint |
| Gao et al[30], 2012 | IDCM/NIDCM | 124 | Presence and quantification of LGE | Primary composite outcome: occurrence of appropriate ICD therapy, SCA, SCD (follow-up 632 ± 262 d) | Myocardial scar quantification by LGE-CMR predicts arrhythmic events in patients being evaluated for ICD eligibility |
| Neilan et al[31], 2013 | NIDCM | 162 | Presence and quantification of LGE | Major adverse cardiac events (cardiovascular death and appropriate ICD therapy) (follow-up: 29 ± 18 mo) | Presence of LGE was a strong predictor of major cardiac events |
| Li et al[32], 2013 | NIDCM | 293 | Presence and extent of LGE | All-cause mortality (follow-up: 3.2 yr) | Presence of LGE is an independent predictor of increased all-cause mortality Diffuse LGE is associated with higher mortality |
| Gulati et al[33], 2013 | NIDCM | 472 | Presence and extent of midwall fibrosis | Primary end-point: all cause mortality Secondary end-point: cardiovascular mortality or cardiac transplantation Arrhythmic and HF secondary end-points (follow-up 5.3 yr) | Midwall fibrosis assessed with LGE-CMR provided independent prognostic information and improved risk stratification beyond LVEF for all-cause mortality and SCD |
CMR: Cardiac magnetic resonance; IDCM: Ischemic dilated cardiomyopathy; LGE: Late gadolinium enhancement; NIDCM: Non ischemic dilated cardiomyopathy; SCA Survived cardiac arrest; SCD: Sudden cardiac death; ICD: Implantable cardioverter defibrillator.
Assomull et al[26] first evaluated the prognostic impact of midwall fibrosis in patients diagnosed with NIDCM, prospectively followed up for 658 ± 355 d. Midwall fibrosis was present in 35% of patients and was associated with a higher rate of all-cause death and hospitalization for a cardiovascular event. Multivariate analysis showed that it was the only significant predictor of death or hospitalization. Midwall fibrosis also predicted sudden cardiac death (SCD) or VT and remained predictive of SCD/VT after correction for baseline LVEF.
Iles et al[28] prospectively evaluated 103 patients meeting criteria for ICD implantation for primary prevention of SCD who were affected by both IDCM and NIDCM. Regional fibrosis was identified with LGE in 71% of patients, in all patients with a diagnosis of IDCM and in 51% of those affected by NIDCM. Interestingly, among NIDCM patients, LGE was associated with arrhythmic events during follow-up in 29%, whereas no NIDCM patients without LGE experienced arrhythmic events.
Finally, the relevant role played by LGE in arrhythmic risk stratification has been supported by a study evaluating a large sample of NIDCM patients[33]. In this series, 30% of patients had fibrosis and were characterized by a lower LVEF and a more severe functional limitation. The presence of fibrosis was independently associated with an increased arrhythmic risk as well as an increased probability of death. Moreover, whether fibrosis was present or not, it was possible to detect the group of patients at higher and lower risk across the LVEF spectrum. For example, patients with a LVEF of 35% and fibrosis had a 19.9% estimated risk of death vs 9.4% of patients with the same LVEF but without fibrosis.
Although there is considerable evidence to suggest the relevance of LGE in arrhythmic risk stratification, particularly in NIDCM, this technique has not been recommended yet by current guidelines for the selection of patients who will benefit from ICD implantation.
ELECTROCARDIOGRAPHIC MEASURES OF ARRHYTHMIC RISK
Fragmented QRS
Prolonged QRS duration prevalence in patients with congestive heart failure varies between 20% and 50%[34]. Left bundle branch block and, in general, QRS prolongation (> 120 ms) in heart failure patients independently predict increased overall mortality and SCD[35-36].
However, fragmented QRS complexes (f-QRS) on a routine 12-lead electrocardiogram have also been proposed as a marker of depolarization abnormality[37].
Various studies have suggested that the region of a myocardial scar is associated with alteration in QRS morphology, leading to a terminal conduction delay or a fragmentation of QRS complexes on the 12-lead ECG[38-39].
Fragmented QRS includes various RSR’ patterns with different morphologies of the QRS interval (QRS duration < 120 ms), with or without the Q wave. It is defined by the presence of an additional R wave (R’) or notching in the nadir of the S wave, or the presence of > 1 R’ wave (fragmentation) in 2 contiguous leads, corresponding to a major coronary artery territory[40].
Brenyo et al[41] observed that fragmented QRS (f-QRS), particularly when present in inferior leads, is predictive of SCD, SCD or appropriate ICD shock and all-cause mortality in patients with IDCM.
Sha et al[42] evaluated a population of 128 patients with NIDCM and left ventricular dysfunction (ejection fraction, EF ≤ 40%). They observed that in the group with f-QRS, all-cause mortality and ventricular tachyarrhythmias were significantly more frequent than those observed in the non-fQRS group.
Finally, Das et al[43] tried to assess the prognostic significance of fQRS for an arrhythmic event in 368 patients with IDCM and NIDCM who underwent ICD implantation for primary or secondary prevention of SCD. The authors concluded that fQRS on a 12-lead ECG is a predictor of arrhythmic events but is not associated with a greater probability of death.
Analysis of ventricular repolarization
The analysis of ventricular repolarization is an intriguing way to implement risk stratification of major arrhythmic events. However, in a large study evaluating NIDCM, the electrocardiographic measure of QT intervals and their dispersion at ECG failed to demonstrate any role in predicting arrhythmic events[44].
Compared to the “static” evaluation of QT interval and dispersion at ECG, the possibility of evaluating QT dynamicity and/or variability during a short-term or 24 h period offer a more complete assessment of ventricular depolarization, the expression of the complex interaction between arrhythmic substrate, heart rate and autonomic nervous system activity[45]. Table 2 summarizes the main studies evaluating the prognostic role of QT-dynamicity or variability measures[46-50].
Table 2.
Main studies evaluating the role of dynamic ventricular repolarization measures in predicting arrhythmic and non arrhythmic events
| Ref. | Clinical setting | Number of patients | Parameter evaluated | Cut-off suggested | End-points (mean follow-up) | Results |
| Chevalier et al[46], 2003 | Acute myocardial infarction | 265 | QT dynamicity and HRV (24-h Holter) LVEF Late potential | QTe slope: 0.18 | Sudden death and total mortality (follow-up 81 ± 27 mo) | Increased diurnal QTe dynamicity independently associated with sudden death |
| Haigney et al[47], 2004 | Postinfarction patients (low LVEF) | 871 | QT variability (QTVN) QTVI (QTVN adjusted for heart rate variance) | Arrhythmic events (VT or VF) (follow-up 2 yr) | Increased QT variability associated with an increased risk for VT/VF | |
| Jensen et al[48], 2005 | Postinfarction patients | 481 | QT/RR slope and intercept QT/RR VR LVEF VPB and VT | All-cause mortality (follow-up 3 yr) | VR, LVEF, VPB and age made up the optimal Cox model for risk stratification. VR was a promising risk factor for identifying sudden arrhythmic death | |
| Iacoviello et al[49], 2007 | NIDCM (no history of SVT/VF) | 179 | QTe slope (24 h Holter) LVEF NSVT QRS duration QTc and QTd at ECG | QTe-slope: 0.19 | Major arrhythmic events, (VT or VF or SCD) (follow-up 39 ± 22 mo) | Increased QTe slope is associated with occurrence of major arrhythmic events. The presence of NSVT and/or QTe slope > 0.19 showed 90% sensitivity and 60% specificity in identifying patients with arrhythmic events |
| Cygankiewicz et al[50], 2009 | CHF patients. IDCM/NIDCM LVEF ≥ 35% | 294 | QTe slope SDNN TS LVEF | QTe slope: 0.21 | Primary endpoint: total mortality Secondary endpoint: sudden death (follow-up 44-mo) | Combination of SDNN, TS, and QTe slope is a predictor of increased risk of mortality and sudden death |
BRS: Baroreflex sensitivity; CHF chronic heart failure; EPS Electrophysiological study; ICD Implantable cardioverter defibrillator; IDCM: Ischemic dilated cardiomyopathy; HR Heart rate; HRV: Heart rate variability; LVEF: Left ventricular ejection fraction; MCE: Major cardiac events; NIDCM: Non ischemic dilated cardiomyopathy; NSVT: Non sustained ventricular tachycardia; OHR: Onset heart rate; QTc: QT interval corrected for heart rate; QTe: QT interval calculated at the end of T-wave; SCD: Sudden cardiac death; SDNN: Standard deviation of RR intervals; SR: Sinus rhythm; TS: Turbulence slope; PVB: Premature ventricular beats; VT: Ventricular tachycardia; VR: Variability ratio.
Recently, we studied a series of patients affected by NIDCM to evaluate the role of QT dynamicity in predicting major arrhythmic events as assessed by 24-h ECG recordings[49]. The QT dynamicity index proposed was QT-slope, i.e., the slope of the regression line between QT end and RR during a 24 h period. At univariate analysis, QTe-slope was significantly associated with major arrhythmic events as well as LVEF, NSVT and standard deviation of RR intervals (SDNN). At multivariate analysis, only the QTe-slope, LVEF and NSVT were significant predictors of events, regardless of SDNN, a QRS duration >120 ms or beta-blocker therapy.
The analysis of QT dynamicity has also been found to be associated with an increased arrhythmic risk in patients with IDCM. Chevalier et al[46] demonstrated that QTe slope compared with LVEF, HRV and late potentials was the strongest independent predictor of sudden death in patients with myocardial infarction. In 871 postinfarction patients with severe left ventricular dysfunction enrolled in the MADIT study, Haigney et al[47] demonstrated an increased incidence of malignant ventricular arrhythmias in those with increased QT variability. In this study, QT variability was assessed using a semiautomated algorithm that measured beat-to-beat QT duration. Similarly, in a population of postinfarction patients, Jensen et al[48] demonstrated the prognostic usefulness of a novel QT dynamics parameter: the QT/RR variability ratio (VR), defined as the ratio between the standard deviation of all QT intervals and the standard deviation of all RR intervals. It was evaluated in 481 patients and found to be associated with the occurrence of sudden arrhythmic death.
Finally, the potential usefulness of QT-e slope has also been demonstrated in a large population of 294 patients affected by CHF due to both IDCM and NIDCM and relatively preserved LVEF > 35%[50].
Microvolt T-wave alternans
Microvolt T-wave alternans (MTWA) analysis involves the detection of changes in T-wave morphology occurring on an every-other-beat basis. A wide electrical alternans of T-wave was an ECG abnormality, first described 50 years ago as being associated with cardiac mortality[51-52]. Discordant alternans is responsible for dispersion of repolarization of sufficient magnitude to cause unidirectional block and re-entry. A critical dispersion of repolarization is an important condition for development of re-entrant arrhythmias[53].
Since MTWA is heart rate dependent, it is generally assessed by increasing heart rate with atrial pacing or by exercise stress. The analysis is based on the alignment of ECG cycles to the QRS complex and on the measurement of T-wave amplitude. The beat-to-beat fluctuations of T-wave are then analyzed using fast Fourier transformation and MTWA is represented by the pronounced peak visible in the spectrum at 0.5 cycles/beat. A significant MTWA is present if the alternans voltage is over a threshold (generally 1.9 microV) and if the alternans ratio K is ≥ 3. Generally, an alternans which is longer than 1 min occurring at a heart rate ≤ 110 beats/min is considered positive[54].
In 1994, Rosenbaum et al[55] was the first to demonstrate the efficacy of MTWA in stratifying patients for the risk of ventricular tachyarrhythmic events. However, the studies published to date are not concordant, as summarized in Table 3[56-64].
Table 3.
Main studies evaluating the role of microvolt T-wave alternans in predicting arrhythmic and non arrhythmic events
| Ref. | Clinical setting | Number of patients | Parameter evaluated | End-points (mean follow-up) | Results |
| Adachi et al[56], 1999 | NIDCM | 57 | TWA, LVEF, NYHA, Signal average ECG, QT dispersion | Ventricular tachycardia | MTWA associated with VT |
| Klingenheben et al[57], 2000 | CHF (no history SVT/VF) | 107 | TWA | Arrhythmic events (follow-up 18 mo) | MTWA is an independent predictor of arrhythmic events |
| Kitamura et al[58], 2002 | NIDCM | 146 | Onset heart rate for TWA | SCD, documented sustained ventricular tachycardia/ventricular fibrillation (follow-up 21 ± 14 mo) | TWA and LVEF were independent predictors of arrhythmic events |
| Hohnloser et al[59], 2003 | NIDCM (LVEF 29 ± 11%) | 137 | MTWA, FEVS, mean RR interval, HRV, BRS. | SCD, SCA, SVT or VF (follow-up 14 ± 6 mo) | MTWA is an independent predictor of ventricular tachyarrhythmic events |
| Bloomfield et al[60], 2004 | IDCM (LVEF ≤ 30%) | 177 | MTWA, QRS measurement | All-cause mortality. (follow-up 20 ± 6 mo) | Compared to QRS duration, an abnormal MTWA is a stronger predictor of death |
| Salerno-Uriate et al[61], 2007 | NIDCM (NYHA II-III LVEF ≤ 40%) | 446 | TWA, VO2 peak | Combined primary endpoint of cardiac death and life-threatening ventricular arrhythmias Secondary endpoint: total mortality, combination of arrhythmic death and life-threatening arrhythmias. (follow-up 18 to 24 mo) | Abnormal TWA associated with a 4-fold higher risk of cardiac death and life-threatening arrhythmias |
| Baravelli et al[62], 2007 | NIDCM (NYHA II-III LVEF 29 ± 6.4%) | 70 | MTWA, VO2 peak | Combined primary endpoint of MCE: total cardiac death or VT/VF (including appropriate ICD shock) Secondary endpoint: MAE: SCD or SVT/VF (follow-up 19.2 ± 10.7 mo) | MTWA and peak VO2, but not the two single tests, were significant prognostic markers of both MCE and MA |
| Gold et al[63], 2008 | CHF (IDCM/NIDCM, 71% NYHA II, LVEF 24 ± 7%) | 490 | TWA | Composite primary end point: SCD, SVT / VF, or appropriate ICD discharge (follow-up 30 mo) | MTWA not predictive of MAE or mortality |
| Costantini et al[64], 2009 | IDCM LVEF ≤ 40% | 566 | TWA, EPS | Primary endpoint: appropriate ICD discharge or SCD at 1 yr follow-up (follow-up 1.6 ± 0.6 yr) | Strategies employing MTWA, EPS, or both might identify the subset of patients least likely to benefit from ICD implantation |
BRS: Baroreflex sensitivity; CHF: Chronic heart failure; EPS: Electrophysiological study; HR: Heart rate; HRV: Heart rate variability; ICD: Implantable cardioverter defibrillator; LVEF: Left ventricular ejection fraction; MCE: Major cardiac events; MTWA: Microvolt T-Wave alternans; NYHA: New York Heart Association; NSVT: Non sustained ventricular tachycardia; OHR: Onset heart rate; SR: Sinus rhythm; SCA: Sudden cardiac arrest; SCD: Sudden cardiac death; SDNN: Standard deviation of RR intervals; VF: Ventricular fibrillation; VT: Ventricular tachycardia.
The meta-analysis carried out by Hohnloser et al[65] suggested that MTWA assessed by spectral analysis provides an accurate means of predicting major ventricular arrhythmias. Moreover, the event rate was very low among patients with a negative MTWA test. These results were concordant with the meta-analysis by Calò et al[66] who analyzed fifteen studies involving 5681 patients. A positive MTWA determined an approximately 2.5-fold higher risk of cardiac death and life-threatening arrhythmia and showed a very high NPV in both ischemic and non-ischemic patients. An abnormal MTWA test was associated with a 5-fold increased risk for cardiac mortality in the low-indeterminate group and about a 6-fold increased risk in the beta-blocker group. The potential usefulness of MTWA has also been confirmed by Merchant et al[67] who analyzed the data of five studies with 2883 patients without ICDs. Among patients with an LVEF of ≤ 35%, a negative MTWA test result was associated with a low risk for SCD. Conversely, in patients with a LVEF of > 35%, a positive MTWA test result identified those at a significantly heightened SCD risk. Finally, the Alternans Before Cardioverter Defibrillator (ABCD) trial[64] was the first to use electrophysiological study (EPS) or MTWA to guide prophylactic ICD implantation in patients with a LVEF ≤ 40%, coronary artery disease and NSVT. The authors demonstrated that risk stratification strategies using the non-invasive MTWA are comparable to invasive strategy.
These results seem to encourage the use of MTWA testing in patients who do not have ICDs in order to identify those at higher risk of ventricular arrhythmic events. However, the meta-analysis of Gupta et al[68] concluded that spectrally derived MTWA testing does not sufficiently modify the risk of VTE to change clinical decisions. Moreover, the MTWA technique is characterized by limitation in its feasibility. In an unselected population of 1003 patients with HF, Kraaier et al[69] showed that only half were eligible for MTWA testing and the most common result was an indeterminate test. They concluded that MTWA treadmill testing is not widely applicable in typical HF patients and is unlikely to refine risk stratification for sudden death on a population level.
ASSESSMENT OF AUTONOMIC NERVOUS SYSTEM ACTIVITY
In the genesis of malignant arrhythmias, apart from the presence of a vulnerable substrate, an altered sympathetic nervous activity and the presence of trigger factors, such as ventricular beats, play a fundamental role. The importance of autonomic dysfunction in increasing the risk of death in patients with heart disease may be applicable to all patients with cardiac disease regardless of etiology[70,71]. The pro-arrhythmic effects of the sympathetic nervous system in the normal and ischemic heart are mainly related to the indirect and direct effects of beta-adrenergic receptor activity, but also to the direct effects of alpha-1 adrenergic receptors activity[72].
The direct effects on myocardiocytes are mediated by the activation of cyclic nucleotide and protein kinase regulatory cascade, which can alter spatial heterogeneity of calcium transients and consequently increase the dispersion of repolarization[73]. The major indirect effect of beta-receptors activity is the impairment of oxygen supply caused by increased metabolic activity, coronary vasoconstriction, especially in vessels with damaged endothelium, and changes in preload and afterload. On the other hand, the increase in parasympathetic activity is able to modulate ventricular arrhythmias by means of one of the following three effects: a reduction in sinus heart rate, a direct influence on myocardial electrophysiology and a reduction in myocardial oxygen demand due to the negative inotropic action. However, vagal and sympathetic effects cannot be considered in isolation. Sympathovagal interactions are critical in order to understand the electrophysiological function of the heart. Processes disturbing sympathovagal balance have the potential to facilitate cardiovascular instability, leading to cardiac arrhythmias or even sudden death.
It is clear that every marker of autonomic activity may be used as a clinical prognostic factor. The evaluation of sympathetic nervous system activity can be based on electrocardiographic measures reflecting autonomic control of heart rate, such as the beat-to-beat heart variability (HRV), heart rate turbulence (HRT) and the reflex chronotropic response to a blood pressure change; i.e., baroreflex activity (BRS). Moreover, nuclear imaging techniques can estimate cardiac denervation.
Measures of autonomic control of heart rate
The prognostic role of measures evaluating autonomic control of heart rate has been widely investigated.
HRV is a term which includes a large number of different indices evaluating the beat-to-beat variability by using either time domain or frequency domain analysis[74]. Time domain analysis is based on the detection of each QRS complex and on measurement of all intervals between adjacent QRS complexes, resulting from sinus rhythm, as NN intervals or as instantaneous heart rate. Among the statistical time domain indices, SDNN is the simplest and is the standard deviation of NN intervals generally assessed in 24 h Holter recordings. The prognostic significance of SDNN has been evaluated both in patients with ischemic and non-ischemic diseases, as well as in heart failure patients, but the results are controversial.
Brower et al[75] assessed the prognostic value of HRV measures in patients with mild or moderate chronic heart failure (NYHA class II-III). Ninety-five patients were followed-up for 4 years. None of the conventional time and frequency domains were related to survival. Szabò et al[76] followed-up a group of 159 patients with idiopathic or ischemic dilated cardiomyopathy, selected on the basis of a left ventricular ejection fraction of < 40%. During follow-up, cardiac mortality was subdivided into sudden cardiac death and death due to progressive pump failure. SDNN was found to have an independent predictive value for all cause mortality, while not being related to the type of the death. Fauchier et al[77] designed a study to evaluate HRV in patients with idiopathic dilated cardiomyopathy to determinate its prognostic value. The group of patients with depressed SDNN (< 100 ms.) had an increased risk of cardiac death or heart transplantation during the follow-up (49.5 ± 35.6 mo).
In patients with mild-to-moderate ventricular dysfunction and NIDCM, a low SDNN, combined with an increased QT dynamicity, has been found to be associated with an increased risk of arrhythmic events[50]. However, in other studies, no independent association with arrhythmic events has been found[44].
HRT is another parameter reflecting autonomic control of heart rate. It is the expression of the baroreflex-mediated transient acceleration-deceleration response of the sinus node triggered by a premature ventricular beat (PVB)[78]. HRT is a baroreflex-mediated biphasic reaction of heart rate in response to premature ventricular beats. It is quantified by: turbulence onset (TO) reflecting the initial acceleration of heart rate following premature beat; and turbulence slope (TS) describing subsequent deceleration of heart rate following a premature ventricular beat. TO is the percentage of relative change in the mean of 2 RR intervals after a PVB. TS is the slope of the steepest regression line computed over the sequence of every 5 consecutive RR intervals following a PVB within 15 RR and is expressed in ms/RR. HRT can be calculated only in patients with sinus rhythm presenting with eligible PVBs[79]. Abnormal HRT identifies patients with an autonomic dysfunction or impaired baroreflex sensitivity due to a variety of disorders, but may also reflect changes in the autonomic nervous system induced by different therapeutic modalities such as drugs, revascularization or cardiac resynchronization therapy[80]. HRT has been introduced as an autonomic predictor for cardiac events in heart failure patients and in large cohorts of postinfarction patients[80-91], as summarized in Table 4. The retrospective analysis of the ATRAMI trial[81] showed that HRT identified postinfarction patients at risk of both all-cause death and arrhythmic events. Other large trials confirmed the prognostic role of abnormal HRT for predicting mortality and arrhythmic events in postinfarction patients[85,89] as well as in both NIDCM and IDCM patients[88,90]. However, the results of the studies, particularly in NIDCM, are conflicting. In the Marburg study, Grimm et al[84] observed that in 242 patients with idiopathic cardiomyopathy, HRT onset is a significant predictor of transplant-free survival, but for arrhythmia risk stratification, only LVEF remained a significant risk predictor on multivariate analysis. Moreover, analysis of the Frankfurt DCM database showed that HRT and HRV did not yield predictive power for arrhythmic events[87].
Table 4.
The main studies evaluating the prognostic significance of heart rate turbulence and risk stratification
| Ref. | Clinical setting | Number of patients | Cut-off proposed | End-points (mean follow-up) | Results |
| Schmidt et al[80], 1999 | Postinfarction patients | 577 | TO 0% TS 2.5 ms/RR | All-cause mortality (follow-up 22 mo) | HRT2 predictive for all-cause mortality |
| Ghuran et al[81], 2002 | Postinfarction patients (ATRAMI) | 1212 | TO 0% TS 2.5 ms/RR | Combined end-point of fatal and non fatal cardiac arrhythmias (follow-up 21 mo) | HRT associated with endpoints |
| Barthel et al[83], 2003 | Postinfarction patients (ISAR-HRT) | 1455 | TO 0% TS 2.5 ms/RR | All-cause mortality (follow-up 22 mo) | HRT independent predictor of mortality in patients with LVEF ≥ 30% |
| Grimm et al[84], 2003 | NIDCM, LVEF ≤ 30% | 242 | TO 0% TS 2.5 ms/RR | Transplant-free survival (follow-up: 41 mo) | TO predictor of transplant-free survival. TO and TS only as univariate predictor of MCE |
| Exner et al[85], 2007 | Myocardial infarction (REFINE) | 322 | TO 0% TS 2.5 ms/RR | Cardiac death or resuscitated cardiac arrest (follow-up 47 mo) | HRT (10-14 wk after MI) predictive for cardiac death or resuscitated cardiac arrest |
| Cygankiewicz et al[86], 2008 | CHF (IDCM/and NIDCM) | 607 | TO 0% TS 2.5 ms/RR | All-cause mortality, sudden death and heart failure death (follow-up: 44 mo) | Abnormal TS predictive for all-cause mortality, sudden death and heart failure death |
| Klingenheben et al[87], 2008 | NIDCM (Mean LVEF 28%) | 114 | TO 0% TS 2.5 ms/RR | Arrhythmic events (follow-up 22 mo) | HRT non predictive for arrhythmic events |
| Miwa et al[88], 2009 | IDCM (241) and NIDCM (134) | 375 | TO 0% TS 2.5 ms/RR | Cardiac mortality Combined endpoint of cardiac death and/or stable sustained VT (follow-up 15 mo) | Abnormal HRT predictive for cardiac mortality and combined endpoint Prognostic value observed in both ischemic and non-ischemic cardiomyopathy |
| Huikuri et al[89], 2009 | Postinfarction CARISMA | 312 | TS 2.5 ms/RR | Primary endpoint of documented VT/TV (follow-up 2 yr) | TS evaluated at 6 wk after MI predictive for primary endpoint No prognostic value for HRT evaluated 1 wk after MI |
| Ikeda et al[90], 2011 | NIDC | 134 | TO 0% TS 2.5 ms/RR | Combined endpoint of cardiac mortality and sustained VT (follow-up 15 mo) | Abnormal HRT predictive for combined endpoint |
| Miwa et al[91], 2012 | IDCM / NIDCM (LVEF ≤ 40%) | 299 | TO 0% TS 2.5 ms/RR | Combined endpoint of sudden cardiac death and sustained VT (follow-up 32 mo) | Abnormal HRT predictive for combined endpoint |
HRT: Heart rate turbulence; NIDCM: Non-ischemic dilated cardiomyopathy; TO: Turbulence onset; TS: Turbulence slope; MCE: Major cardiac events; LVEF: Left ventricular ejection fraction; CHF: Chronic heart failure; NYHA ICD: Implantable cardioverter defibrillator.
Cardiac denervation assessed by nuclear imaging
In the pathophysiology of malignant ventricular arrhythmias, a relevant role is played not only by sympathetic autonomic nervous system hyperactivity but also by cardiac sympathetic denervation. The presence of cardiac denervation can cause heterogeneity in a refractory period of the ventricular myocardium, thus favoring the onset and the persistence of ventricular arrhythmias. A scintigraphic approach using 123I-labeled metaiodobenzylguanidine (MIBG) can explore the presence of abnormalities in cardiac sympathetic innervation[92-96].
This radiotracer is administered at rest and planar and single-photon emission computerized tomography images are then acquired after 15 min (early) and 3-5 h (delayed). Generally, the analysis of MIBG distribution is based on the delayed images which reflect overall cardiac sympathetic function, including uptake, re-uptake, storage and release processes of norepinephrine at presynaptic nerve terminals, rather than real time, beat-by-beat sympathetic drive[96]. The quantitative index calculated after MIBG injection is the heart/mediastinal ratio (H/M). This is derived by the mean counts per pixel of the region of interest drawn over the heart and that drawn over the upper mediastinum[97]. The value of H/M range is from 1.9 to 2.8 in a normal subject. A normal H/M ratio reflects the density of receptors and the integrity of presynaptic nerve terminals and uptake function. A low H/M ratio reflects a reduced myocardial uptake and a poor cardiac adrenergic receptor density[95,98].
Besides global myocardial uptake (heart-to-mediastinum ratio), other markers have been used, including washout kinetics and regional uptake heterogeneity. The myocardial washout rate (WR) is expressed as the rate of decrease in myocardial counts over time between early and late imaging, reflecting the neuronal integrity or sympathetic tone[98]. In HF patients, high myocardial WR and low early and delayed H/M are detectable[99-101].
The presence of an altered distribution of MIBG can also be found in NIDCM patients[102] and has been associated with other parameters reflecting arrhythmic risk[103-104].
Over the last three decades a number of studies have reported the relevance of an altered MIBG distribution in predicting increased risk of death and arrhythmic events[105-115]. In a group of patients with heart failure, Nakata et al[101] revealed that impaired cardiac sympathetic innervation assessed by MIBG activity has an incremental and prognostic role for predicting cardiac death and may be useful for identifying a threshold level for selecting patients at risk for death by heart failure, sudden cardiac death and fatal myocardial infarction.
The largest trial evaluating the prognostic role of cardiac denervation assessed by MIBG is the ADMIRE study[108], in which a total of 961 subjects with NYHA functional class II/III HF and LVEF ≤ 35% were evaluated. Time to first occurrence of NYHA functional class progression, a potentially life-threatening arrhythmic event, and cardiac death were the end-points considered. For H/M < 1.60, 2 year probabilities of cardiac death and all-cause mortality were 11.2% and 16.1% vs 1.8% and 3% for the group with H/M ≥ 1.60. Moreover, non-fatal arrhythmic events or sudden cardiac death were observed in patients with H/M < 1.60. ADMIRE-HF provided prospective validation of the independent prognostic value of MIBG in the assessment of patients with HF, in identifying patients at high risk of arrhythmic events, sudden cardiac death and ICD discharge.
Finally, it is worth noting that the prognostic significance of MIBG in predicting sudden death has also been demonstrated in a small population of patients with mild-to-moderate CHF[112].
THE MULTIPARAMETRIC APPROACH TO ARRHYTHMIC RISK STRATIFICATION
Different studies evaluating the role of non-invasive diagnostic tools in predicting arrhythmic events have demonstrated that the combination of the different parameters could be a useful approach in order to better improve arrhythmic risk stratification. Generally, the combination of the different parameters allows the identification of a smaller group of patients at higher risk of arrhythmic events.
In our series of patients[49], by combining LVEF (< 35% vs > 35%), NSVT and QTe-slope (> 0.19 vs < 0.19), arrhythmic events were more frequently observed in patients with NSVT and a low LVEF and in those with a low LVEF and steeper QTe slope. No significantly higher risk was observed in patients with a higher LVEF and NSVT or steeper QTe slope. When all three variables were considered together, the patients with a low LVEF and NSVT or a steeper QTe slope were found to have a higher arrhythmic risk. In the subgroup of patients with LVEF < 35%, the presence of NSVT and QTe slope > 0.19 defined a small population with the highest probability of events.
Also, among HF patients with a LVEF > 35%, the combination of different arrhythmic risk parameters improved prognostic stratification. Cygankiewicz et al[50] demonstrated that in this population of patients, the presence of two or more independent risk parameters (SDNN ≤ 86 ms, HRT < 2.5 ms/RR and QTe slope > 0.21) detected a population at higher risk of death (30% 3 year mortality) and sudden death (12%), with a rate of events similar to that observed among patients with LVEF ≤ 35%.
Merchant et al[114] tried to assess whether a multi-marker strategy would provide more robust SCD risk stratification than LVEF alone. The authors observed that a multivariable model based on the presence of coronary artery disease, LVEF and MTWA status provides a significantly more robust SCD risk prediction than LVEF as a single risk marker. These findings suggest that multi-marker strategies based on different aspects of the electroanatomic substrate may be capable of improving primary prevention implantable cardioverter-defibrillator treatment algorithms.
Finally, Yukinaka et al[115] correlated the incidence of ventricular arrhythmias with mismatches in myocardial 99mTc-methoxyisobutylisonitrile/MIBG accumulation and late ventricular potentials. Patients with late ventricular potentials had greater I-123 MIBG defect scores. The combination of late ventricular potentials and I-123 MIBG uptake could improve the prediction of ventricular arrhythmias after myocardial infarction.
LIMITATIONS OF ALTERNATIVE NON-INVASIVE ARRHYTHMIC RISK PARAMETERS
Although the above mentioned studies have provided evidence about the independent association among a number of parameters and the risk of malignant ventricular arrhythmias, their routine use is still limited for different reasons. In particular, most of the parameters have shown conflicting results, probably related to the methodological differences, such as the studied population (NIDCM or IDCM), the follow-up duration, the end-points considered and the pharmacological treatment at the enrolment. Moreover, all measures are affected by both technical and biological limitations. Finally, almost all these studies were aimed at only evaluating the associations between the studied parameters and the occurrence of ventricular arrhythmias, but not to demonstrate their ability to select patient populations who could benefit from ICD implantation. This ability could be demonstrated only by randomized studies that, to date, are still lacking.
CONCLUSION
Malignant ventricular arrhythmias and sudden death are the main adverse events affecting the prognosis of both NIDCM and IDCM. ICD implantation, i.e., the best therapeutic strategy to reduce the incidence of sudden death, is currently mainly guided by the estimation of LVEF. However, this measure is affected by a number of technical and biological limitations. For these reasons, the best assessment of arrhythmic risk is still a challenge. The use of other non-invasive parameters reflecting functional or anatomical arrhythmic substrate (LGE), sympathetic nervous activity (HRT, SDNN, the presence of sympathetic denervation by MIBG) and the abnormalities in myocardial refractoriness (QT dynamicity/variability, MTWA) could be useful in order to better characterize both patients with reduced and preserved LVEF at higher risk of arrhythmic events.
Although several studies have shown these parameters to be independently associated with events, their routine use is still limited due to the lack of randomized studies demonstrating their ability to select patient populations who could benefit from ICD implantation. Future prospective studies should aim to reduce this gap in the evidence in order to justify the indication of these techniques in daily clinical practice.
Footnotes
P- Reviewer: Al-Biltagi M, Kolettis TM, RamsayM, Sakabe K, Tagarakis G S- Editor: Wen LL L- Editor: Roemmele A E- Editor: Wu HL
References
- 1.Carson P, Anand I, O’Connor C, Jaski B, Steinberg J, Lwin A, Lindenfeld J, Ghali J, Barnet JH, Feldman AM, et al. Mode of death in advanced heart failure: the Comparison of Medical, Pacing, and Defibrillation Therapies in Heart Failure (COMPANION) trial. J Am Coll Cardiol. 2005;46:2329–2334. doi: 10.1016/j.jacc.2005.09.016. [DOI] [PubMed] [Google Scholar]
- 2.Moss AJ, Zareba W, Hall WJ, Klein H, Wilber DJ, Cannom DS, Daubert JP, Higgins SL, Brown MW, Andrews ML. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877–883. doi: 10.1056/NEJMoa013474. [DOI] [PubMed] [Google Scholar]
- 3.Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R, Domanski M, Troutman C, Anderson J, Johnson G, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352:225–237. doi: 10.1056/NEJMoa043399. [DOI] [PubMed] [Google Scholar]
- 4.McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, Falk V, Filippatos G, Fonseca C, Gomez-Sanchez MA, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–869. doi: 10.1093/eurjhf/hfs105. [DOI] [PubMed] [Google Scholar]
- 5.Gehi A, Haas D, Fuster V. Primary prophylaxis with the implantable cardioverter-defibrillator: the need for improved risk stratification. JAMA. 2005;294:958–960. doi: 10.1001/jama.294.8.958. [DOI] [PubMed] [Google Scholar]
- 6.Otterstad JE, Froeland G, St John Sutton M, Holme I. Accuracy and reproducibility of biplane two-dimensional echocardiographic measurements of left ventricular dimensions and function. Eur Heart J. 1997;18:507–513. doi: 10.1093/oxfordjournals.eurheartj.a015273. [DOI] [PubMed] [Google Scholar]
- 7.Malm S, Frigstad S, Sagberg E, Larsson H, Skjaerpe T. Accurate and reproducible measurement of left ventricular volume and ejection fraction by contrast echocardiography: a comparison with magnetic resonance imaging. J Am Coll Cardiol. 2004;44:1030–1035. doi: 10.1016/j.jacc.2004.05.068. [DOI] [PubMed] [Google Scholar]
- 8.Donath L, Roth R, Zahner L, Faude O. Testing single and double limb standing balance performance: comparison of COP path length evaluation between two devices. Gait Posture. 2012;36:439–443. doi: 10.1016/j.gaitpost.2012.04.001. [DOI] [PubMed] [Google Scholar]
- 9.Bellenger NG, Burgess MI, Ray SG, Lahiri A, Coats AJ, Cleland JG, Pennell DJ. Comparison of left ventricular ejection fraction and volumes in heart failure by echocardiography, radionuclide ventriculography and cardiovascular magnetic resonance; are they interchangeable? Eur Heart J. 2000;21:1387–1396. doi: 10.1053/euhj.2000.2011. [DOI] [PubMed] [Google Scholar]
- 10.Thomas JD, Popović ZB. Assessment of left ventricular function by cardiac ultrasound. J Am Coll Cardiol. 2006;48:2012–2025. doi: 10.1016/j.jacc.2006.06.071. [DOI] [PubMed] [Google Scholar]
- 11.Zecchin M, Merlo M, Pivetta A, Barbati G, Lutman C, Gregori D, Serdoz LV, Bardari S, Magnani S, Di Lenarda A, et al. How can optimization of medical treatment avoid unnecessary implantable cardioverter-defibrillator implantations in patients with idiopathic dilated cardiomyopathy presenting with “SCD-HeFT criteria?”. Am J Cardiol. 2012;109:729–735. doi: 10.1016/j.amjcard.2011.10.033. [DOI] [PubMed] [Google Scholar]
- 12.Allgayer H. Translational research on u-PAR. Eur J Cancer. 2010;46:1241–1251. doi: 10.1016/j.ejca.2010.02.029. [DOI] [PubMed] [Google Scholar]
- 13.Amundsen BH, Helle-Valle T, Edvardsen T, Torp H, Crosby J, Lyseggen E, Støylen A, Ihlen H, Lima JA, Smiseth OA, et al. Noninvasive myocardial strain measurement by speckle tracking echocardiography: validation against sonomicrometry and tagged magnetic resonance imaging. J Am Coll Cardiol. 2006;47:789–793. doi: 10.1016/j.jacc.2005.10.040. [DOI] [PubMed] [Google Scholar]
- 14.Saha SK, Kiotsekoglou A, Toole RS, Moggridge JC, Nichols KJ, Govind S, Gopal AS. Value of two-dimensional speckle tracking and real time three-dimensional echocardiography for the identification of subclinical left ventricular dysfunction in patients referred for routine echocardiography. Echocardiography. 2012;29:588–597. doi: 10.1111/j.1540-8175.2011.01631.x. [DOI] [PubMed] [Google Scholar]
- 15.Iacoviello M, Forleo C, Puzzovivo A, Nalin I, Guida P, Anaclerio M, Marangelli V, Sorrentino S, Monitillo F, Ciccone MM, et al. Altered two-dimensional strain measures of the right ventricle in patients with Brugada syndrome and arrhythmogenic right ventricular dysplasia/cardiomyopathy. Eur J Echocardiogr. 2011;12:773–781. doi: 10.1093/ejechocard/jer139. [DOI] [PubMed] [Google Scholar]
- 16.Brown J, Jenkins C, Marwick TH. Use of myocardial strain to assess global left ventricular function: a comparison with cardiac magnetic resonance and 3-dimensional echocardiography. Am Heart J. 2009;157:102.e1–102.e5. doi: 10.1016/j.ahj.2008.08.032. [DOI] [PubMed] [Google Scholar]
- 17.Belghitia H, Brette S, Lafitte S, Reant P, Picard F, Serri K, Lafitte M, Courregelongue M, Dos Santos P, Douard H, et al. Automated function imaging: a new operator-independent strain method for assessing left ventricular function. Arch Cardiovasc Dis. 2008;101:163–169. doi: 10.1016/s1875-2136(08)71798-4. [DOI] [PubMed] [Google Scholar]
- 18.Iacoviello M, Puzzovivo A, Guida P, Forleo C, Monitillo F, Catanzaro R, Lattarulo MS, Antoncecchi V, Favale S. Independent role of left ventricular global longitudinal strain in predicting prognosis of chronic heart failure patients. Echocardiography. 2013;30:803–811. doi: 10.1111/echo.12142. [DOI] [PubMed] [Google Scholar]
- 19.Bloomgarden DC, Fayad ZA, Ferrari VA, Chin B, Sutton MG, Axel L. Global cardiac function using fast breath-hold MRI: validation of new acquisition and analysis techniques. Magn Reson Med. 1997;37:683–692. doi: 10.1002/mrm.1910370510. [DOI] [PubMed] [Google Scholar]
- 20.Semelka RC, Tomei E, Wagner S, Mayo J, Kondo C, Suzuki J, Caputo GR, Higgins CB. Normal left ventricular dimensions and function: interstudy reproducibility of measurements with cine MR imaging. Radiology. 1990;174:763–768. doi: 10.1148/radiology.174.3.2305059. [DOI] [PubMed] [Google Scholar]
- 21.Pennell DJ, Sechtem UP, Higgins CB, Manning WJ, Pohost GM, Rademakers FE, van Rossum AC, Shaw LJ, Yucel EK. Clinical indications for cardiovascular magnetic resonance (CMR): Consensus Panel report. Eur Heart J. 2004;25:1940–1965. doi: 10.1016/j.ehj.2004.06.040. [DOI] [PubMed] [Google Scholar]
- 22.Pennell DJ. Cardiovascular magnetic resonance. Circulation. 2010;121:692–705. doi: 10.1161/CIRCULATIONAHA.108.811547. [DOI] [PubMed] [Google Scholar]
- 23.Spiewak M, Malek LA, Misko J, Chojnowska L, Milosz B, Klopotowski M, Petryka J, Dabrowski M, Kepka C, Ruzyllo W. Comparison of different quantification methods of late gadolinium enhancement in patients with hypertrophic cardiomyopathy. Eur J Radiol. 2010;74:e149–e153. doi: 10.1016/j.ejrad.2009.05.035. [DOI] [PubMed] [Google Scholar]
- 24.Hundley WG, Bluemke D, Bogaert JG, Friedrich MG, Higgins CB, Lawson MA, McConnell MV, Raman SV, van Rossum AC, Flamm S, et al. Society for Cardiovascular Magnetic Resonance guidelines for reporting cardiovascular magnetic resonance examinations. J Cardiovasc Magn Reson. 2009;11:5. doi: 10.1186/1532-429X-11-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nazarian S, Bluemke DA, Lardo AC, Zviman MM, Watkins SP, Dickfeld TL, Meininger GR, Roguin A, Calkins H, Tomaselli GF, et al. Magnetic resonance assessment of the substrate for inducible ventricular tachycardia in nonischemic cardiomyopathy. Circulation. 2005;112:2821–2825. doi: 10.1161/CIRCULATIONAHA.105.549659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Assomull RG, Prasad SK, Lyne J, Smith G, Burman ED, Khan M, Sheppard MN, Poole-Wilson PA, Pennell DJ. Cardiovascular magnetic resonance, fibrosis, and prognosis in dilated cardiomyopathy. J Am Coll Cardiol. 2006;48:1977–1985. doi: 10.1016/j.jacc.2006.07.049. [DOI] [PubMed] [Google Scholar]
- 27.Wu KC, Weiss RG, Thiemann DR, Kitagawa K, Schmidt A, Dalal D, Lai S, Bluemke DA, Gerstenblith G, Marbán E, et al. Late gadolinium enhancement by cardiovascular magnetic resonance heralds an adverse prognosis in nonischemic cardiomyopathy. J Am Coll Cardiol. 2008;51:2414–2421. doi: 10.1016/j.jacc.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Iles L, Pfluger H, Lefkovits L, Butler MJ, Kistler PM, Kaye DM, Taylor AJ. Myocardial fibrosis predicts appropriate device therapy in patients with implantable cardioverter-defibrillators for primary prevention of sudden cardiac death. J Am Coll Cardiol. 2011;57:821–828. doi: 10.1016/j.jacc.2010.06.062. [DOI] [PubMed] [Google Scholar]
- 29.Lehrke S, Lossnitzer D, Schöb M, Steen H, Merten C, Kemmling H, Pribe R, Ehlermann P, Zugck C, Korosoglou G, et al. Use of cardiovascular magnetic resonance for risk stratification in chronic heart failure: prognostic value of late gadolinium enhancement in patients with non-ischaemic dilated cardiomyopathy. Heart. 2011;97:727–732. doi: 10.1136/hrt.2010.205542. [DOI] [PubMed] [Google Scholar]
- 30.Gao P, Yee R, Gula L, Krahn AD, Skanes A, Leong-Sit P, Klein GJ, Stirrat J, Fine N, Pallaveshi L, et al. Prediction of arrhythmic events in ischemic and dilated cardiomyopathy patients referred for implantable cardiac defibrillator: evaluation of multiple scar quantification measures for late gadolinium enhancement magnetic resonance imaging. Circ Cardiovasc Imaging. 2012;5:448–456. doi: 10.1161/CIRCIMAGING.111.971549. [DOI] [PubMed] [Google Scholar]
- 31.Neilan TG, Coelho-Filho OR, Danik SB, Shah RV, Dodson JA, Verdini DJ, Tokuda M, Daly CA, Tedrow UB, Stevenson WG, et al. CMR quantification of myocardial scar provides additive prognostic information in nonischemic cardiomyopathy. JACC Cardiovasc Imaging. 2013;6:944–954. doi: 10.1016/j.jcmg.2013.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li X, Chan CP, Hua W, Ding L, Wang J, Zhang S, Li S, Zhang Y. Prognostic impact of late gadolinium enhancement by cardiac magnetic resonance imaging in patients with non-ischaemic dilated cardiomyopathy. Int J Cardiol. 2013;168:4979–4980. doi: 10.1016/j.ijcard.2013.07.134. [DOI] [PubMed] [Google Scholar]
- 33.Gulati A, Ismail TF, Jabbour A, Alpendurada F, Guha K, Ismail NA, Raza S, Khwaja J, Brown TD, Morarji K, et al. The prevalence and prognostic significance of right ventricular systolic dysfunction in nonischemic dilated cardiomyopathy. Circulation. 2013;128:1623–1633. doi: 10.1161/CIRCULATIONAHA.113.002518. [DOI] [PubMed] [Google Scholar]
- 34.Desai AD, Yaw TS, Yamazaki T, Kaykha A, Chun S, Froelicher VF. Prognostic Significance of Quantitative QRS Duration. Am J Med. 2006;119:600–606. doi: 10.1016/j.amjmed.2005.08.028. [DOI] [PubMed] [Google Scholar]
- 35.Baldasseroni S, Opasich C, Gorini M, Lucci D, Marchionni N, Marini M, Campana C, Perini G, Deorsola A, Masotti G, et al. Left bundle-branch block is associated with increased 1-year sudden and total mortality rate in 5517 outpatients with congestive heart failure: a report from the Italian network on congestive heart failure. Am Heart J. 2002;143:398–405. doi: 10.1067/mhj.2002.121264. [DOI] [PubMed] [Google Scholar]
- 36.Iuliano S, Fisher SG, Karasik PE, Fletcher RD, Singh SN. QRS duration and mortality in patients with congestive heart failure. Am Heart J. 2002;143:1085–1091. doi: 10.1067/mhj.2002.122516. [DOI] [PubMed] [Google Scholar]
- 37.Das MK, El Masry H. Fragmented QRS and other depolarization abnormalities as a predictor of mortality and sudden cardiac death. Curr Opin Cardiol. 2010;25:59–64. doi: 10.1097/HCO.0b013e328333d35d. [DOI] [PubMed] [Google Scholar]
- 38.el-Sherif N. The rsR’ pattern in left surface leads in ventricular aneurysm. Br Heart J. 1970;32:440–448. doi: 10.1136/hrt.32.4.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Peters S, Trümmel M, Koehler B. QRS fragmentation in standard ECG as a diagnostic marker of arrhythmogenic right ventricular dysplasia-cardiomyopathy. Heart Rhythm. 2008;5:1417–1421. doi: 10.1016/j.hrthm.2008.07.012. [DOI] [PubMed] [Google Scholar]
- 40.Das MK, Khan B, Jacob S, Kumar A, Mahenthiran J. Significance of a fragmented QRS complex versus a Q wave in patients with coronary artery disease. Circulation. 2006;113:2495–2501. doi: 10.1161/CIRCULATIONAHA.105.595892. [DOI] [PubMed] [Google Scholar]
- 41.Brenyo A, Pietrasik G, Barsheshet A, Huang DT, Polonsky B, McNitt S, Moss AJ, Zareba W. QRS fragmentation and the risk of sudden cardiac death in MADIT II. J Cardiovasc Electrophysiol. 2012;23:1343–1348. doi: 10.1111/j.1540-8167.2012.02390.x. [DOI] [PubMed] [Google Scholar]
- 42.Sha J, Zhang S, Tang M, Chen K, Zhao X, Wang F. Fragmented QRS is associated with all-cause mortality and ventricular arrhythmias in patient with idiopathic dilated cardiomyopathy. Ann Noninvasive Electrocardiol. 2011;16:270–275. doi: 10.1111/j.1542-474X.2011.00442.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Das MK, Maskoun W, Shen C, Michael MA, Suradi H, Desai M, Subbarao R, Bhakta D. Fragmented QRS on twelve-lead electrocardiogram predicts arrhythmic events in patients with ischemic and nonischemic cardiomyopathy. Heart Rhythm. 2010;7:74–80. doi: 10.1016/j.hrthm.2009.09.065. [DOI] [PubMed] [Google Scholar]
- 44.Grimm W, Christ M, Bach J, Müller HH, Maisch B. Noninvasive arrhythmia risk stratification in idiopathic dilated cardiomyopathy: results of the Marburg Cardiomyopathy Study. Circulation. 2003;108:2883–2891. doi: 10.1161/01.CIR.0000100721.52503.85. [DOI] [PubMed] [Google Scholar]
- 45.Zareba W, Bayes de Luna A. QT dynamics and variability. Ann Noninvasive Electrocardiol. 2005;10:256–262. doi: 10.1111/j.1542-474X.2005.10205.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chevalier P, Burri H, Adeleine P, Kirkorian G, Lopez M, Leizorovicz A, André-Fouët X, Chapon P, Rubel P, Touboul P. QT dynamicity and sudden death after myocardial infarction: results of a long-term follow-up study. J Cardiovasc Electrophysiol. 2003;14:227–233. doi: 10.1046/j.1540-8167.2003.02431.x. [DOI] [PubMed] [Google Scholar]
- 47.Haigney MC, Zareba W, Gentlesk PJ, Goldstein RE, Illovsky M, McNitt S, Andrews ML, Moss AJ. QT interval variability and spontaneous ventricular tachycardia or fibrillation in the Multicenter Automatic Defibrillator Implantation Trial (MADIT) II patients. J Am Coll Cardiol. 2004;44:1481–1487. doi: 10.1016/j.jacc.2004.06.063. [DOI] [PubMed] [Google Scholar]
- 48.Jensen BT, Abildstrom SZ, Larroude CE, Agner E, Torp-Pedersen C, Nyvad O, Ottesen M, Wachtell K, Kanters JK. QT dynamics in risk stratification after myocardial infarction. Heart Rhythm. 2005;2:357–364. doi: 10.1016/j.hrthm.2004.12.028. [DOI] [PubMed] [Google Scholar]
- 49.Iacoviello M, Forleo C, Guida P, Romito R, Sorgente A, Sorrentino S, Catucci S, Mastropasqua F, Pitzalis M. Ventricular repolarization dynamicity provides independent prognostic information toward major arrhythmic events in patients with idiopathic dilated cardiomyopathy. J Am Coll Cardiol. 2007;50:225–231. doi: 10.1016/j.jacc.2007.02.071. [DOI] [PubMed] [Google Scholar]
- 50.Cygankiewicz I, Zareba W, Vazquez R, Bayes-Genis A, Pascual D, Macaya C, Almendral J, Fiol M, Bardaji A, Gonzalez-Juanatey JR, et al. Risk stratification of mortality in patients with heart failure and left ventricular ejection fraction & gt; 35% Am J Cardiol. 2009;103:1003–1010. doi: 10.1016/j.amjcard.2008.11.061. [DOI] [PubMed] [Google Scholar]
- 51.Kalter HH, Schwartz ML. Electrical alternans. N Y State J Med. 1948;48:1164–1166. [PubMed] [Google Scholar]
- 52.Adam DR, Smith JM, Akselrod S, Nyberg S, Powell AO, Cohen RJ. Fluctuations in T-wave morphology and susceptibility to ventricular fibrillation. J Electrocardiol. 1984;17:209–218. doi: 10.1016/s0022-0736(84)80057-6. [DOI] [PubMed] [Google Scholar]
- 53.Pastore JM, Girouard SD, Laurita KR, Akar FG, Rosenbaum DS. Mechanism linking T-wave alternans to the genesis of cardiac fibrillation. Circulation. 1999;99:1385–1394. doi: 10.1161/01.cir.99.10.1385. [DOI] [PubMed] [Google Scholar]
- 54.Klingenheben T, Hohnloser SH. Clinical value of T-wave alternans assessment. Card Electrophysiol Rev. 2002;6:323–328. doi: 10.1023/a:1016305831211. [DOI] [PubMed] [Google Scholar]
- 55.Rosenbaum DS, Jackson LE, Smith JM, Garan H, Ruskin JN, Cohen RJ. Electrical alternans and vulnerability to ventricular arrhythmias. N Engl J Med. 1994;330:235–241. doi: 10.1056/NEJM199401273300402. [DOI] [PubMed] [Google Scholar]
- 56.Adachi K, Ohnishi Y, Shima T, Yamashiro K, Takei A, Tamura N, Yokoyama M. Determinant of microvolt-level T-wave alternans in patients with dilated cardiomyopathy. J Am Coll Cardiol. 1999;34:374–380. doi: 10.1016/s0735-1097(99)00208-9. [DOI] [PubMed] [Google Scholar]
- 57.Klingenheben T, Zabel M, D’Agostino RB, Cohen RJ, Hohnloser SH. Predictive value of T-wave alternans for arrhythmic events in patients with congestive heart failure. Lancet. 2000;356:651–652. doi: 10.1016/s0140-6736(00)02609-x. [DOI] [PubMed] [Google Scholar]
- 58.Kitamura H, Ohnishi Y, Okajima K, Ishida A, Galeano E, Adachi K, Yokoyama M. Onset heart rate of microvolt-level T-wave alternans provides clinical and prognostic value in nonischemic dilated cardiomyopathy. J Am Coll Cardiol. 2002;39:295–300. doi: 10.1016/s0735-1097(01)01718-1. [DOI] [PubMed] [Google Scholar]
- 59.Hohnloser SH, Klingenheben T, Bloomfield D, Dabbous O, Cohen RJ. Usefulness of microvolt T-wave alternans for prediction of ventricular tachyarrhythmic events in patients with dilated cardiomyopathy: results from a prospective observational study. J Am Coll Cardiol. 2003;41:2220–2224. doi: 10.1016/s0735-1097(03)00467-4. [DOI] [PubMed] [Google Scholar]
- 60.Bloomfield DM, Steinman RC, Namerow PB, Parides M, Davidenko J, Kaufman ES, Shinn T, Curtis A, Fontaine J, Holmes D, et al. Microvolt T-wave alternans distinguishes between patients likely and patients not likely to benefit from implanted cardiac defibrillator therapy: a solution to the Multicenter Automatic Defibrillator Implantation Trial (MADIT) II conundrum. Circulation. 2004;110:1885–1889. doi: 10.1161/01.CIR.0000143160.14610.53. [DOI] [PubMed] [Google Scholar]
- 61.Salerno-Uriarte JA, De Ferrari GM, Klersy C, Pedretti RF, Tritto M, Sallusti L, Libero L, Pettinati G, Molon G, Curnis A, et al. Prognostic value of T-wave alternans in patients with heart failure due to nonischemic cardiomyopathy: results of the ALPHA Study. J Am Coll Cardiol. 2007;50:1896–1904. doi: 10.1016/j.jacc.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 62.Baravelli M, Fantoni C, Rogiani S, Farina S, Anzà C, Caltabiano V, Forzani T, Salerno-Uriarte JA. Combined prognostic value of peak O(2) uptake and microvolt level T-wave alternans in patients with idiopathic dilated cardiomyopathy. Int J Cardiol. 2007;121:23–29. doi: 10.1016/j.ijcard.2006.10.026. [DOI] [PubMed] [Google Scholar]
- 63.Gold MR, Ip JH, Costantini O, Poole JE, McNulty S, Mark DB, Lee KL, Bardy GH. Role of microvolt T-wave alternans in assessment of arrhythmia vulnerability among patients with heart failure and systolic dysfunction: primary results from the T-wave alternans sudden cardiac death in heart failure trial substudy. Circulation. 2008;118:2022–2028. doi: 10.1161/CIRCULATIONAHA.107.748962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Costantini O, Hohnloser SH, Kirk MM, Lerman BB, Baker JH, Sethuraman B, Dettmer MM, Rosenbaum DS. The ABCD (Alternans Before Cardioverter Defibrillator) Trial: strategies using T-wave alternans to improve efficiency of sudden cardiac death prevention. J Am Coll Cardiol. 2009;53:471–479. doi: 10.1016/j.jacc.2008.08.077. [DOI] [PubMed] [Google Scholar]
- 65.Hohnloser SH, Ikeda T, Cohen RJ. Evidence regarding clinical use of microvolt T-wave alternans. Heart Rhythm. 2009;6:S36–S44. doi: 10.1016/j.hrthm.2008.10.011. [DOI] [PubMed] [Google Scholar]
- 66.Calò L, De Santo T, Nuccio F, Sciarra L, De Luca L, Stefano LM, Piroli E, Zuccaro L, Rebecchi M, de Ruvo E, et al. Predictive value of microvolt T-wave alternans for cardiac death or ventricular tachyarrhythmic events in ischemic and nonischemic cardiomyopathy patients: a meta-analysis. Ann Noninvasive Electrocardiol. 2011;16:388–402. doi: 10.1111/j.1542-474X.2011.00467.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Merchant FM, Ikeda T, Pedretti RF, Salerno-Uriarte JA, Chow T, Chan PS, Bartone C, Hohnloser SH, Cohen RJ, Armoundas AA. Clinical utility of microvolt T-wave alternans testing in identifying patients at high or low risk of sudden cardiac death. Heart Rhythm. 2012;9:1256–1264.e2. doi: 10.1016/j.hrthm.2012.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gupta A, Hoang DD, Karliner L, Tice JA, Heidenreich P, Wang PJ, Turakhia MP. Ability of microvolt T-wave alternans to modify risk assessment of ventricular tachyarrhythmic events: a meta-analysis. Am Heart J. 2012;163:354–364. doi: 10.1016/j.ahj.2011.11.021. [DOI] [PubMed] [Google Scholar]
- 69.Kraaier K, McCracken T, van der Palen J, Wilde AA, Scholten MF. Is T-wave alternans testing feasible in candidates for prophylactic implantable defibrillators? Neth Heart J. 2011;19:6–9. doi: 10.1007/s12471-010-0053-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zipes DP. Sympathetic stimulation and arrhythmias. N Engl J Med. 1991;325:656–657. doi: 10.1056/NEJM199108293250911. [DOI] [PubMed] [Google Scholar]
- 71.Barron HV, Lesh MD. Autonomic nervous system and sudden cardiac death. J Am Coll Cardiol. 1996;27:1053–1060. doi: 10.1016/0735-1097(95)00615-X. [DOI] [PubMed] [Google Scholar]
- 72.Verrier RL. Autonomic modulation of arrhythmias in animal models. In: Rosen MR, Wit AL, Janse MJ, eds , editors. Cardiac electrophysiology: a textbook in honor of Brian Hoffman. Mount Kiscop, NY: Futura; 1990. pp. 933–949. [Google Scholar]
- 73.Levy MN. Role of calcium in arrhythmogenesis. Circulation. 1989;80:IV23–IV30. [PubMed] [Google Scholar]
- 74.Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation. 1996;93:1043–1065. [PubMed] [Google Scholar]
- 75.Brouwer J, van Veldhuisen DJ, Man in ‘t Veld AJ, Haaksma J, Dijk WA, Visser KR, Boomsma F, Dunselman PH. Prognostic value of heart rate variability during long-term follow-up in patients with mild to moderate heart failure. The Dutch Ibopamine Multicenter Trial Study Group. J Am Coll Cardiol. 1996;28:1183–1189. doi: 10.1016/s0735-1097(96)00279-3. [DOI] [PubMed] [Google Scholar]
- 76.Szabó BM, van Veldhuisen DJ, van der Veer N, Brouwer J, De Graeff PA, Crijns HJ. Prognostic value of heart rate variability in chronic congestive heart failure secondary to idiopathic or ischemic dilated cardiomyopathy. Am J Cardiol. 1997;79:978–980. doi: 10.1016/s0002-9149(97)00026-x. [DOI] [PubMed] [Google Scholar]
- 77.Fauchier L, Babuty D, Cosnay P, Autret ML, Fauchier JP. Heart rate variability in idiopathic dilated cardiomyopathy: characteristics and prognostic value. J Am Coll Cardiol. 1997;30:1009–1014. doi: 10.1016/s0735-1097(97)00265-9. [DOI] [PubMed] [Google Scholar]
- 78.Cygankiewicz I. Heart rate turbulence. Prog Cardiovasc Dis. 2013;56:160–171. doi: 10.1016/j.pcad.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 79.Bauer A, Malik M, Schmidt G, Barthel P, Bonnemeier H, Cygankiewicz I, Guzik P, Lombardi F, Müller A, Oto A, et al. Heart rate turbulence: standards of measurement, physiological interpretation, and clinical use: International Society for Holter and Noninvasive Electrophysiology Consensus. J Am Coll Cardiol. 2008;52:1353–1365. doi: 10.1016/j.jacc.2008.07.041. [DOI] [PubMed] [Google Scholar]
- 80.Schmidt G, Malik M, Barthel P, Schneider R, Ulm K, Rolnitzky L, Camm AJ, Bigger JT Jr, Schömig A. Heart rate turbulence after ventricular premature beats as a predictor of mortality after acute myocardial infarction. Lancet. 1999;335:1390–1396. doi: 10.1016/S0140-6736(98)08428-1. [DOI] [PubMed] [Google Scholar]
- 81.Ghuran A, Reid F, La Rovere MT, Schmidt G, Bigger JT, Camm AJ, Schwartz PJ, Malik M. Heart rate turbulence-based predictors of fatal and nonfatal cardiac arrest (The Autonomic Tone and Reflexes After Myocardial Infarction substudy) Am J Cardiol. 2002;89:184–190. doi: 10.1016/s0002-9149(01)02198-1. [DOI] [PubMed] [Google Scholar]
- 82.Koyama J, Watanabe J, Yamada A, Koseki Y, Konno Y, Toda S, Shinozaki T, Miura M, Fukuchi M, Ninomiya M, et al. Evaluation of heart-rate turbulence as a new prognostic marker in patients with chronic heart failure. Circ J. 2002;66:902–907. doi: 10.1253/circj.66.902. [DOI] [PubMed] [Google Scholar]
- 83.Barthel P, Schneider R, Bauer A, Ulm K, Schmitt C, Schömig A, Schmidt G. Risk stratification after acute myocardial infarction by heart rate turbulence. Circulation. 2003;108:1221–1226. doi: 10.1161/01.CIR.0000088783.34082.89. [DOI] [PubMed] [Google Scholar]
- 84.Grimm W, Schmidt G, Maisch B, Sharkova J, Müller HH, Christ M. Prognostic significance of heart rate turbulence following ventricular premature beats in patients with idiopathic dilated cardiomyopathy. J Cardiovasc Electrophysiol. 2003;14:819–824. doi: 10.1046/j.1540-8167.2003.03085.x. [DOI] [PubMed] [Google Scholar]
- 85.Exner DV, Kavanagh KM, Slawnych MP, Mitchell LB, Ramadan D, Aggarwal SG, Noullett C, Van Schaik A, Mitchell RT, Shibata MA, et al. Noninvasive risk assessment early after a myocardial infarction the REFINE study. J Am Coll Cardiol. 2007;50:2275–2284. doi: 10.1016/j.jacc.2007.08.042. [DOI] [PubMed] [Google Scholar]
- 86.Cygankiewicz I, Zareba W, Vazquez R, Vallverdu M, Gonzalez-Juanatey JR, Valdes M, Almendral J, Cinca J, Caminal P, de Luna AB. Heart rate turbulence predicts all-cause mortality and sudden death in congestive heart failure patients. Heart Rhythm. 2008;5:1095–1102. doi: 10.1016/j.hrthm.2008.04.017. [DOI] [PubMed] [Google Scholar]
- 87.Klingenheben T, Ptaszynski P, Hohnloser SH. Heart rate turbulence and other autonomic risk markers for arrhythmia risk stratification in dilated cardiomyopathy. J Electrocardiol. 2008;41:306–311. doi: 10.1016/j.jelectrocard.2007.10.004. [DOI] [PubMed] [Google Scholar]
- 88.Miwa Y, Ikeda T, Sakaki K, Miyakoshi M, Ishiguro H, Tsukada T, Abe A, Mera H, Yusu S, Yoshino H. Heart rate turbulence as a predictor of cardiac mortality and arrhythmic events in patients with dilated cardiomyopathy: a prospective study. J Cardiovasc Electrophysiol. 2009;20:788–795. doi: 10.1111/j.1540-8167.2009.01438.x. [DOI] [PubMed] [Google Scholar]
- 89.Huikuri HV, Raatikainen MJ, Moerch-Joergensen R, Hartikainen J, Virtanen V, Boland J, Anttonen O, Hoest N, Boersma LV, Platou ES, et al. Prediction of fatal or near-fatal cardiac arrhythmia events in patients with depressed left ventricular function after an acute myocardial infarction. Eur Heart J. 2009;30:689–698. doi: 10.1093/eurheartj/ehn537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ikeda T, Miwa Y, Abe A, Nakazawa K. Usefulness of heart rate turbulence for predicting cardiac events in patients with nonischemic dilated cardiomyopathy. J Electrocardiol. 2011;44:669–672. doi: 10.1016/j.jelectrocard.2011.08.003. [DOI] [PubMed] [Google Scholar]
- 91.Miwa Y, Yoshino H, Hoshida K, Miyakoshi M, Tsukada T, Yusu S, Ikeda T. Risk stratification for serious arrhythmic events using nonsustained ventricular tachycardia and heart rate turbulence detected by 24-hour holter electrocardiograms in patients with left ventricular dysfunction. Ann Noninvasive Electrocardiol. 2012;17:260–267. doi: 10.1111/j.1542-474X.2012.00522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Calkins H, Allman K, Bolling S, Kirsch M, Wieland D, Morady F, Schwaiger M. Correlation between scintigraphic evidence of regional sympathetic neuronal dysfunction and ventricular refractoriness in the human heart. Circulation. 1993;88:172–179. doi: 10.1161/01.cir.88.1.172. [DOI] [PubMed] [Google Scholar]
- 93.Inoue H, Zipes DP. Results of sympathetic denervation in the canine heart: supersensitivity that may be arrhythmogenic. Circulation. 1987;75:877–887. doi: 10.1161/01.cir.75.4.877. [DOI] [PubMed] [Google Scholar]
- 94.Sisson JC, Shapiro B, Meyers L, Mallette S, Mangner TJ, Wieland DM, Glowniak JV, Sherman P, Beierwaltes WH. Metaiodobenzylguanidine to map scintigraphically the adrenergic nervous system in man. J Nucl Med. 1987;28:1625–1636. [PubMed] [Google Scholar]
- 95.Schofer J, Spielmann R, Schuchert A, Weber K, Schlüter M. Iodine-123 meta-iodobenzylguanidine scintigraphy: a noninvasive method to demonstrate myocardial adrenergic nervous system disintegrity in patients with idiopathic dilated cardiomyopathy. J Am Coll Cardiol. 1988;12:1252–1258. doi: 10.1016/0735-1097(88)92608-3. [DOI] [PubMed] [Google Scholar]
- 96.Wieland DM, Wu J, Brown LE, Mangner TJ, Swanson DP, Beierwaltes WH. Radiolabeled adrenergi neuron-blocking agents: adrenomedullary imaging with [131I]iodobenzylguanidine. J Nucl Med. 1980;21:349–353. [PubMed] [Google Scholar]
- 97.Flotats A, Carrió I, Agostini D, Le Guludec D, Marcassa C, Schäfers M, Somsen GA, Unlu M, Verberne HJ. Proposal for standardization of 123I-metaiodobenzylguanidine (MIBG) cardiac sympathetic imaging by the EANM Cardiovascular Committee and the European Council of Nuclear Cardiology. Eur J Nucl Med Mol Imaging. 2010;37:1802–1812. doi: 10.1007/s00259-010-1491-4. [DOI] [PubMed] [Google Scholar]
- 98.Henderson EB, Kahn JK, Corbett JR, Jansen DE, Pippin JJ, Kulkarni P, Ugolini V, Akers MS, Hansen C, Buja LM. Abnormal I-123 metaiodobenzylguanidine myocardial washout and distribution may reflect myocardial adrenergic derangement in patients with congestive cardiomyopathy. Circulation. 1988;78:1192–1199. doi: 10.1161/01.cir.78.5.1192. [DOI] [PubMed] [Google Scholar]
- 99.Merlet P, Valette H, Dubois-Randé JL, Moyse D, Duboc D, Dove P, Bourguignon MH, Benvenuti C, Duval AM, Agostini D. Prognostic value of cardiac metaiodobenzylguanidine imaging in patients with heart failure. J Nucl Med. 1992;33:471–477. [PubMed] [Google Scholar]
- 100.Agostini D, Verberne HJ, Hamon M, Jacobson AF, Manrique A. Cardiac 123I-MIBG scintigraphy in heart failure. Q J Nucl Med Mol Imaging. 2008;52:369–377. [PubMed] [Google Scholar]
- 101.Nakata T, Miyamoto K, Doi A, Sasao H, Wakabayashi T, Kobayashi H, Tsuchihashi K, Shimamoto K. Cardiac death prediction and impaired cardiac sympathetic innervation assessed by MIBG in patients with failing and nonfailing hearts. J Nucl Cardiol. 1998;5:579–590. doi: 10.1016/s1071-3581(98)90112-x. [DOI] [PubMed] [Google Scholar]
- 102.Harada M, Shimizu A, Murata M, Ono K, Kubo M, Mitani R, Dairaku Y, Matsumoto T, Yamagata T, Seki K, et al. Relation between microvolt-level T-wave alternans and cardiac sympathetic nervous system abnormality using iodine-123 metaiodobenzylguanidine imaging in patients with idiopathic dilated cardiomyopathy. Am J Cardiol. 2003;92:998–1001. doi: 10.1016/s0002-9149(03)00988-3. [DOI] [PubMed] [Google Scholar]
- 103.Anastasiou-Nana MI, Terrovitis JV, Athanasoulis T, Karaloizos L, Geramoutsos A, Pappa L, Tsagalou EP, Efentakis S, Nanas JN. Prognostic value of iodine-123-metaiodobenzylguanidine myocardial uptake and heart rate variability in chronic congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol. 2005;96:427–431. doi: 10.1016/j.amjcard.2005.03.093. [DOI] [PubMed] [Google Scholar]
- 104.Bax JJ, Kraft O, Buxton AE, Fjeld JG, Parízek P, Agostini D, Knuuti J, Flotats A, Arrighi J, Muxi A, et al. 123 I-mIBG scintigraphy to predict inducibility of ventricular arrhythmias on cardiac electrophysiology testing: a prospective multicenter pilot study. Circ Cardiovasc Imaging. 2008;1:131–140. doi: 10.1161/CIRCIMAGING.108.782433. [DOI] [PubMed] [Google Scholar]
- 105.Stefanelli A, Treglia G, Giordano A. (123)I-MIBG Scintigraphy as a Powerful Tool to Plan an Implantable Cardioverter Defibrillator and to Assess Cardiac Resynchronization Therapy in Heart Failure Patients. Int J Mol Imaging. 2012;2012:690468. doi: 10.1155/2012/690468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wakabayashi T, Nakata T, Hashimoto A, Yuda S, Tsuchihashi K, Travin MI, Shimamoto K. Assessment of underlying etiology and cardiac sympathetic innervation to identify patients at high risk of cardiac death. J Nucl Med. 2001;42:1757–1767. [PubMed] [Google Scholar]
- 107.Agostini D, Verberne HJ, Burchert W, Knuuti J, Povinec P, Sambuceti G, Unlu M, Estorch M, Banerjee G, Jacobson AF. I-123-mIBG myocardial imaging for assessment of risk for a major cardiac event in heart failure patients: insights from a retrospective European multicenter study. Eur J Nucl Med Mol Imaging. 2008;35:535–546. doi: 10.1007/s00259-007-0639-3. [DOI] [PubMed] [Google Scholar]
- 108.Jacobson AF, Senior R, Cerqueira MD, Wong ND, Thomas GS, Lopez VA, Agostini D, Weiland F, Chandna H, Narula J. Myocardial iodine-123 meta-iodobenzylguanidine imaging and cardiac events in heart failure. Results of the prospective ADMIRE-HF (AdreView Myocardial Imaging for Risk Evaluation in Heart Failure) study. J Am Coll Cardiol. 2010;55:2212–2221. doi: 10.1016/j.jacc.2010.01.014. [DOI] [PubMed] [Google Scholar]
- 109.Boogers MJ, Borleffs CJ, Henneman MM, van Bommel RJ, van Ramshorst J, Boersma E, Dibbets-Schneider P, Stokkel MP, van der Wall EE, Schalij MJ, et al. Cardiac sympathetic denervation assessed with 123-iodine metaiodobenzylguanidine imaging predicts ventricular arrhythmias in implantable cardioverter-defibrillator patients. J Am Coll Cardiol. 2010;55:2769–2777. doi: 10.1016/j.jacc.2009.12.066. [DOI] [PubMed] [Google Scholar]
- 110.Kasama S, Toyama T, Sumino H, Nakazawa M, Matsumoto N, Sato Y, Kumakura H, Takayama Y, Ichikawa S, Suzuki T, et al. Prognostic value of serial cardiac 123I-MIBG imaging in patients with stabilized chronic heart failure and reduced left ventricular ejection fraction. J Nucl Med. 2008;49:907–914. doi: 10.2967/jnumed.107.047548. [DOI] [PubMed] [Google Scholar]
- 111.Tamaki S, Yamada T, Okuyama Y, Morita T, Sanada S, Tsukamoto Y, Masuda M, Okuda K, Iwasaki Y, Yasui T, et al. Cardiac iodine-123 metaiodobenzylguanidine imaging predicts sudden cardiac death independently of left ventricular ejection fraction in patients with chronic heart failure and left ventricular systolic dysfunction: results from a comparative study with signal-averaged electrocardiogram, heart rate variability, and QT dispersion. J Am Coll Cardiol. 2009;53:426–435. doi: 10.1016/j.jacc.2008.10.025. [DOI] [PubMed] [Google Scholar]
- 112.Kioka H, Yamada T, Mine T, Morita T, Tsukamoto Y, Tamaki S, Masuda M, Okuda K, Hori M, Fukunami M. Prediction of sudden death in patients with mild-to-moderate chronic heart failure by using cardiac iodine-123 metaiodobenzylguanidine imaging. Heart. 2007;93:1213–1218. doi: 10.1136/hrt.2006.094524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Nagahara D, Nakata T, Hashimoto A, Wakabayashi T, Kyuma M, Noda R, Shimoshige S, Uno K, Tsuchihashi K, Shimamoto K. Predicting the need for an implantable cardioverter defibrillator using cardiac metaiodobenzylguanidine activity together with plasma natriuretic peptide concentration or left ventricular function. J Nucl Med. 2008;49:225–233. doi: 10.2967/jnumed.107.042564. [DOI] [PubMed] [Google Scholar]
- 114.Merchant FM, Zheng H, Bigger T, Steinman R, Ikeda T, Pedretti RF, Salerno-Uriarte JA, Klersy C, Chan PS, Bartone C, et al. A combined anatomic and electrophysiologic substrate based approach for sudden cardiac death risk stratification. Am Heart J. 2013;166:744–752. doi: 10.1016/j.ahj.2013.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yukinaka M, Nomura M, Ito S, Nakaya Y. Mismatch between myocardial accumulation of 123I-MIBG and 99mTc-MIBI and late ventricular potentials in patients after myocardial infarction: association with the development of ventricular arrhythmias. Am Heart J. 1998;136:859–867. doi: 10.1016/s0002-8703(98)70132-2. [DOI] [PubMed] [Google Scholar]


