Abstract
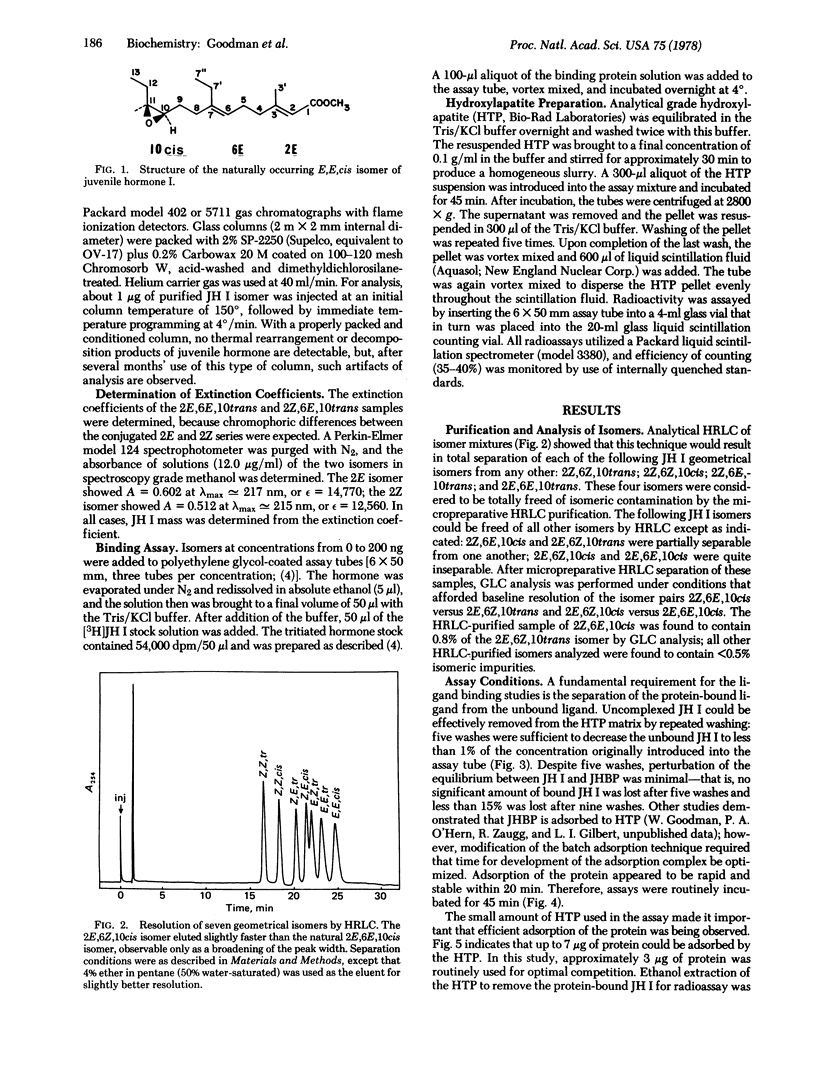
The binding of the geometrical isomers (≥99% pure) of juvenile hormone I to the hemolymph juvenile hormone binding protein of Manduca sexta (Lepidoptera, Sphingidae) was analyzed. A technique is described for isomer separation by micropreparative high-resolution liquid chromatography. Analysis of competition was performed by using a “batch adsorption” hydroxylapatite binding assay. Competition studies indicate that the naturally occurring isomer, 2E,6E,10cis, is bound with the highest affinity. Optimal binding appears to depend most heavily upon the configuration of the 2,3 double bond. Juvenile hormone binding protein shows a higher affinity for the 2E than for the 2Z configuration. The 6,7 double bond is of less importance in determining binding activity, and isomerism about the epoxide appears least important in conferring binding activity. The binding site may be a groove along the surface of the binding protein interacting with the side chains of juvenile hormone, including the ester methyl group. The grouping of the side chains and the ester methyl group thus constitutes a distinct hydrophobic face, and the hydrophobic interactions are essential in maintenance of the bound ligand.
Keywords: racemates, insect hormone, high-resolution liquid chromatography, binding site
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Kramer K. J., Sanburg L. L., Kézdy F. J., Law J. H. The Juvenile Hormone Binding Protein in the Hemolymph of Manduca sexta Johannson (Lepidoptera: Sphingidae). Proc Natl Acad Sci U S A. 1974 Feb;71(2):493–497. doi: 10.1073/pnas.71.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low R. B., Wool I. G., Martin T. E. Skeletal muscle ribosomal proteins: general characteristics and effect of diabetes. Biochim Biophys Acta. 1969 Nov 11;194(1):190–202. doi: 10.1016/0005-2795(69)90194-9. [DOI] [PubMed] [Google Scholar]
- Pavlik E. J., Coulson P. B. Hydroxylapatite "batch" assay for estrogen receptors: increased sensitivity over present receptor assays. J Steroid Biochem. 1976 May;7(5):357–368. doi: 10.1016/0022-4731(76)90095-9. [DOI] [PubMed] [Google Scholar]
- Peterson R. C., Reich M. F., Dunn P. E., Law J. H., Katzenellnbogen J. A. Binding specificity of the juvenile hormone carrier protein from the hemolymph of the tobacco hornworm Manduca sexta Johannson (Lepidoptera: Sphingidae). Biochemistry. 1977 May 17;16(10):2305–2311. doi: 10.1021/bi00629a040. [DOI] [PubMed] [Google Scholar]
- Röller H., Dahm K. H. The chemistry and biology of juvenile hormone. Recent Prog Horm Res. 1968;24:651–680. doi: 10.1016/b978-1-4831-9827-9.50018-6. [DOI] [PubMed] [Google Scholar]
- Trautmann K. H., Schuler A., Suchý M., Wipf H. K. Eine Methode zur qualitativen und quantitativen Bestimmung von drei Juvenilhormonen von Insekten. Nachweis von 10.11-Epoxy-3.7.11-trimethyl-2-trans-6-trans-dodecadiensäurementylester in Mellontha melolontha. Z Naturforsch C. 1974 Mar-Apr;29(3):161–168. [PubMed] [Google Scholar]


