Abstract
Non-alcoholic fatty liver disease (NAFLD) is a chronic liver disease that might affect up to one-third of the adult population in industrialised countries. NAFLD incorporates histologically and clinically different non-alcoholic entities; fatty liver (NAFL, steatosis hepatis) and steatohepatitis (NASH-characterised by hepatocyte ballooning and lobular inflammation ± fibrosis) might progress to cirrhosis and rarely to hepatocellular cancer. NAFL increasingly affects children (paediatric prevalence is 4.2%-9.6%). Type 2 diabetes mellitus (T2DM), insulin resistance (IR), obesity, metabolic syndrome and NAFLD are particularly closely related. Increased hepatic lipid storage is an early abnormality in insulin resistant women with a history of gestational diabetes mellitus. The accumulation of triacylglycerols in hepatocytes is predominantly derived from the plasma nonesterified fatty acid pool supplied largely by the adipose tissue. A few NAFLD susceptibility gene variants are associated with progressive liver disease, IR, T2DM and a higher risk for hepatocellular carcinoma. Although not approved, pharmacological approaches might be considered in NASH patients.
Keywords: Non-alcoholic fatty liver disease, Non-alcoholic steatohepatitis, Liver cirrhosis, Hepatocellular cancer, Dysfunctional adipose tissue, Type 2 diabetes mellitus, Insulin resistance, Obesity, Genetics, Therapy
Core tip: In this review article, non-alcoholic fatty liver disease (NAFLD) spectrum disease is discussed in detail. The epidemiology of NAFLD/nonalcoholic steatohepatitis and the relationship of NAFLD to different forms of diabetes mellitus including type 2 diabetes mellitus and gestational diabetes mellitus are reviewed. Attention is paid to the main biochemical events, to dysfunctional adipose tissue and visceral adiposity associated with NAFLD and insulin resistance, to mitochondrial dysfunction and to the role of the entero-insular axis in NAFLD. Genetics and potential pharmacological approaches are discussed.
HISTORY
The work regarding non-alcoholic fatty liver disease (NAFLD) was launched only three decades ago, when Ludwig et al[1] described an “unnamed” and “poorly understood” liver disease they named non-alcoholic steatohepatitis (NASH) in 20 patients that histologically reminded authors of alcoholic hepatitis with a potential of progression to cirrhosis. Although 20 patients with NAFLD today could likely be recruited within one day in a lobby of a hotel, their observation that NASH is an obesity-associated disease largely accompanied by diabetes mellitus presenting with hepatomegaly and mild abnormalities of liver tests still accurately describes the most common clinical findings. There are a number of confounding factors in determining the true prevalence and incidence of NAFLD.
EPIDEMIOLOGY OF NAFLD/NASH BASED ON HISTOLOGY
NAFLD incorporates histologically and clinically different non-alcoholic entities as fatty liver (NAFL, steatosis hepatis) and steatohepatitis (NASH) with or without fibrosis that might progress to liver cirrhosis and in a few cases to hepatocellular cancer[2,3].
The gold standard methodologies are difficult to apply in a general population based study. Histological assessment had an exclusive role for decades as the only method for grading hepatic steatosis; however this method has been recently challenged and not only due to the difficulties in everyday use for diagnostic purposes in this spectrum of diseases. Hepatic steatosis is defined as intrahepatic fat content above 5.5%[4,5] or when more than 5% of the hepatocytes contain typically macrovesicular fat on the histology. Steatosis might be graded as-S1, mild (up to 10% of hepatocytes); S2, moderate (10% to 30% of hepatocytes); S3, severe (more than 30% of hepatocytes)-according to the proportion of the cells with macrovesicular changes in the liver cells containing fat.
Under routine clinical circumstances, liver biopsy is typically indicated when liver tests (LTs) are repeatedly and chronically elevated (e.g., for six months) and are of unexplained origin. A few biopsy-based studies report the NASH prevalence in the general population. Williams et al[6] recruited 400 outpatients aged 18 to 70 years and performed 134 liver biopsies when the screening abdominal ultrasound suggested hepatic steatosis. Although ultrasound pre-screening theoretically might implicate a selection bias, the reported prevalence of NAFLD was 46% in this United States cohort. The recalculated NAFLD prevalence-taking into account those who refused the biopsy and those with normal liver histology-is 40% based on the biopsy findings. NASH was diagnosed in 12.2% of the entire United States cohort and 29.9% of the NAFLD patients (Table 1). Differences were found according to ethnic origin, with the highest risk for NAFLD and NASH presented in patients of Hispanic origin (> Caucasian > African-American) and according to the presence of diabetes mellitus (NAFLD prevalence, 74%, NASH prevalence, 22.2%); the NAFLD patients were more likely to be male (58.9%) and of older age, to have a higher BMI and to present with hypertension.
Table 1.
Prevalence of non-alcoholic fatty liver disease, non-alcoholic fatty liver and non-alcoholic steatohepatitis
| NAFLD/NAFL prevalence | NASH prevalence | Population studied | Population size | Method of diagnosis | Remark | Ref. |
| 46 (40)% NAFLD in the entire US cohort | 12.2% in the US entire cohort | 18-70 yr aged US cohort | 328 | Liver biopsy (in 134 ultrasound pre-screend patients) | [6] | |
| 29.9% in patients with NAFLD | Greatest risk for both NAFLD and NASH in Hispanics and with diabetes | |||||
| NAFL: 49.3%-mild, moderate, and severe NAFL in 38.9%, 9.0% and 1.4% donor candidates respectively (mild steatosis was defined as fatty changes in 5%-30% of hepatocytes, moderate steatosis in 30% to 60% of hepatocytes, and severe steatosis in > 60% of hepatocytes without significant inflammation on liver histology) | 2.2% (Asian population) | Korean living liver donor candidates | 589 | 589 US guided liver biopsy | [126] | |
| NAFL: 4.2% in pediatric European population | 1% (pediatric population) | European children (6 mo-18 yr old) | 342 medicolegal autopsy reports/265 children died from trauma | Histopathology at autopsy (and typical macroscopic imaging) | Excess body weight was observed in 55.6% of children with NAFL | [11] |
| NAFLD 9.6% in pediatric US population fatty liver was defined as > or = 5% of hepatocytes containing macrovesicular fat | NR | United States children (2-19 yr old) | 742 children (2-19 yr old) who had autopsy (form 1993 to 2003) | Histopathology at autopsy | Different prevalence according to subpopulation (Asians: 10.2%; Black: 1.5%; Hispanic: 11.8%; White: 8.6%). The highest rate of NAFLD was seen in obese children (38%) | [12] |
| NAFLD 31% in adult Urban US population | NR | Large, ethnically diverse, probability-based adult population sample from Dallas, Texas, United States -participants in the Dallas Heart Study | 2349 | 1H-MRS of the liver to quantify HTGC | 79% of patients with hepatic steatosis had normal levels of serum alanine aminotransferase. Different prevalence of hepatic steatosis in different sub-populations: 45% in Hispanics, 33% in Whites and 24% in Blacks | [4] |
Based on histological examination or proton magnetic resonance spectroscopy (1H-MRS) measurement in Hallmark Studies. HTGC: Hepatic triglyceride content; NAFLD: Non-alcoholic fatty liver disease; NAFL: Non-alcoholic fatty liver; NASH: Non-alcoholic steatohepatitis; NR: Not reported.
We might conclude that the prevalence of NAFLD and NASH is highly dependent on the structure of the study population due to the significant differences in prevalence among different sub-populations. The differences in the prevalence and clinical-pathological presentation are influenced by gender as follows: the males among NAFLD patients are more prone to present with elevated LTs, histologically determined NASH, hepatic fibrosis and higher overall mortality according to the majority of studies[7-9].
The NAFLD spectrum diseases do not differ from the demographic trends observed in metabolic diseases including type 2 diabetes mellitus that affect increasingly younger generations decade by decade[10]. The prevalence of NAFL in Poland and NAFLD in the United States was 4.2% and 9.6% based on histopathology at autopsy in pediatric populations, respectively[11,12]. Excess bodyweight was found in 55.6% of the children with NAFL in the European study, and the highest rate of NAFLD was observed in obese United States children (38%); the latter study found a difference in the NAFLD prevalence among different sub-populations (ethnic origin) in children[11,12].
EPIDEMIOLOGY OF NAFLD BASED ON PROTON MAGNETIC RESONANCE SPECTROSCOPY
Grading hepatic steatosis by histological examination potentially holds other biases, including sampling and observation biases. The amount of triglyceride accumulation in the liver could be assumed to be too low to allow the formation of macrovesicles and might not be assessed at histology. Proton magnetic resonance spectroscopy (1HMRS) has appropriate sensitivity for the quantification of the intrahepatic lipid content and correlates better with the biochemical analysis results of liver specimens even if such a small lipid accumulation is in question[13,14]. Compared to traditional histology, 1HMRS based steatosis (intrahepatocellular lipid content, IHCL) grading is based on a much larger mass of hepatic tissue that is investigated without the risks of liver biopsy (27 g vs 100 mg), providing additional advantages over the invasive method[13,14].
The NAFLD prevalence from the studies using 1HMRS based IHCL measurements are indicated in Table 1. The prevalence of fatty liver disease in a population-based study in the United States using a 1HMRS-based measurement for the determination of the intrahepatic triglyceride content (HTGC) was 34%, and in over 90% of the 2287 enrolled individuals, it was because of non-alcoholic causes[4]. The results of this multi-ethnic study demonstrated differences in the prevalence of hepatic steatosis among the different ethnic groups studied, with a higher prevalence in Hispanic patients, explained by the higher prevalence of obesity and insulin resistance. The authors concluded that ethnic differences in the prevalence of hepatic steatosis in the study reflected those observed previously for NAFLD-related cirrhosis (Hispanics > whites > blacks). The finding that the majority of subjects (79%) with NAFLD had normal levels of serum alanine aminotransferase should be taken into account when evaluating population-based studies using score systems that incorporate LT in the diagnosis of NAFLD[4].
In a recent study, biochemically measured hepatic triglyceride levels correlated significantly with the hepatic lipid level (IHCL) measured with 1HMRS. The histologic and 1HMRS grading of fatty liver was in agreement in the majority (65%) of these C-virus infected patients. In contrast, no linear correlation between the biochemically determined liver triglyceride content and histological examination was found[15].
ULTRASOUND BASED NAFLD PREVALENCE
The diagnostic plethora of NAFLD is further complicated by many studies using ultrasonography data to identify patients with NAFLD, although ultrasonography might not be regarded as accurate radiologic modality as the 1HMRS in measuring the intrahepatic lipid level. The third National Health and Nutrition Examination Survey (NAHNES) assessed the prevalence of NAFLD from 1988 to 1994 in the United States based on the ultrasonography data of 12454 adults. They estimated that 28.8 million adults might be diagnosed with NAFLD in the United States; the corresponding prevalence is 19%[16]. The data obtained from the NAHNES study confirmed that NAFLD occurs with a higher prevalence in Mexican-Americans compared to non-Hispanic whites and non-Hispanic blacks. NAFLD was independently associated with insulin resistance and diabetes; and, among people without diabetes, with dyslipidaemia and obesity. The study confirmed that NAFLD is more common in males[16]. The NAFLD prevalence in the Italian Dionysos project in adults with and without suspected liver disease was 25% and 20%, respectively using the US based method for identification[17]. The prevalence of NAFLD in Japan increased to 2.4-fold from the 12.6% prevalence found in 1989 to the 30.3% prevalence observed in 1998[18]. A lower prevalence was reported in India, using ultrasonography for the identification of NAFLD: the prevalence of NAFLD was 18.9% in adults above 20 years of age, with a higher prevalence of NAFLD in males than females (24.6% vs 13.6%)[19].
A number of studies report the aminotransferase-based approach to diagnose; due to the observation that more than 75% of the patients with steatosis might have normal LT values, these studies are not discussed in detail here[4].
ASSESSMENT OF FIBROSIS AND STEATOHEPATITIS
In most patients with NAFLD, a non-invasive score proposed by Angulo et al[20] could be applied to assess liver fibrosis because advanced fibrosis could be diagnosed with accuracy in 90% of the cases. The authors analysed the clinical and liver biopsy data of more than 700 patients to construct this simple scoring system. Transient elastography (Fibroscan), which is a radiological modality used with high accuracy to non-invasively assess the degree of fibrosis based on the measurement of liver stiffness[21,22], might detect even low-grade steatosis because of a recently described novel ultrasonic controlled attenuation parameter of the machine[23].
Although there are promising biomarkers such as cytokeratin-18 fragments for NASH to be potentially applied as diagnostic tools in the future[24], the accurate diagnosis of NASH in some cases might require a liver biopsy. A biopsy is typically indicated in the cases in which the liver tests (LTs) are repeatedly elevated in a chronic manner (e.g., for six months) and are of unexplained origin and when the patient must be carefully investigated for alternative diagnoses (e.g., to measure assess hepatic iron concentration in a C282Y HFE heterozygote individual; when the case is suggestive of autoimmune liver disease; rarely, when the diagnosis of Wilson disease may not be established without quantitation of the liver copper content; in other storage diseases; or in rare distinct forms of liver disease)[25-29].
WHY IS NAFLD WORRISOME?
Type 2 diabetes mellitus
Type 2 diabetes mellitus (T2DM) and NAFLD are particularly closely related. This relationship among T2DM, insulin resistance (IR) and NAFLD is expected because insulin is subsequently delivered directly to the portal vein after secretion, taking the same route as the absorbed glucose, and the liver eliminates a large portion of portal insulin at the first pass.
Obesity in NAFLD is associated with dysfunctional adipose tissue, and lipotoxicity promotes insulin resistance and pancreatic β-cell dysfunction.
The prevalence of ultrasonographic NAFLD was 69.4% in 180 patients with T2DM[30]. NAFLD was associated with obesity (abdominal), hypertriglyceridemia and high-normal ALT levels. The authors concluded that the progression of NAFLD is independent of the diabetes progression[30].
The ultrasonography results of 204 patients with T2DM showed fatty infiltration in 62.2% of the patients; NAFLD was confirmed by liver biopsy with subsequent histology in 87% of the patients, indicating a 54.11% histologically confirmed prevalence in T2DM. Steatohepatitis and fibrosis were found in 38.9% and 23.2%, respectively, of Indian patients with T2DM[31].
Leite et al[32] found a 78% NASH prevalence at the histological examination in nearly 100 patients with T2DM and US evidence of NAFLD. The presence of high triglyceride, low HDL-cholesterol and increased ALT levels were independently associated with a higher risk of histologically confirmed NASH. The prevalence of advanced fibrosis (≥ stage 2) was found in 38% and 55% of the patients depending on the pathologist who conducted the histological examination. The presence of NASH was independently correlated with high serum γGT levels, older age and male gender[32].
Gestational diabetes mellitus
Recent novel findings have emerged to confirm the relationship between diabetes mellitus and NAFLD: Women with a history of gestational diabetes mellitus (GDM) have an increased risk of developing T2DM decades later. Prikoszovich et al[33] recruited women with a history of GDM (pGDM) four to five years after delivery and assessed the glucose tolerance and oral glucose insulin sensitivity to measure the whole-body insulin sensitivity during a 75 g CH OGTT. The lipid storage in the muscle, liver and flux through the ATP synthase were measured using 1H/31P magnetic resonance spectroscopy. In a comparison with women without any risk factor for T2DM, the hepatic content of lipids (HCL) was doubled in the insulin resistant pGDM women, despite they had normal glucose tolerance. HCL correlated positively with the body fat mass and inversely with insulin sensitivity. The authors concluded that increased hepatic lipid storage is an early and predominant abnormality in insulin resistant women with a history of GDM.
The fatty liver index (FLI) -measured using 1HMRS- in women with previous GDM predicted further metabolic deterioration and subjects with the highest FLI values showed significant alterations in FFA kinetics with a higher risk to develop T2DM in the future[34]. The results of these studies should be taken into consideration when the role of NAFLD in determining the hepatic and whole body insulin sensitivity is under scrutiny in glucose tolerant individuals with insulin resistance.
In 2013, Brumbaugh et al[35] assessed the intrahepatic lipids in the neonatal offspring of obese women with gestational diabetes. The neonates born to obese women with GDM underwent MRS for intrahepatic lipid content determination at 1-3 wk of age and demonstrated a mean 68% increase in the IHCL compared with infants born to normal-weight mothers. The intrahepatic fat deposition in the neonates positively correlated with the maternal pre-pregnancy BMI and not with subcutaneous adiposity.
"Metabolic syndrome"
Although many authors agree that the “metabolic syndrome” is a cluster of risk factors, but whether it may correctly be considered a syndrome is strongly questioned[36]. Provided the term “metabolic syndrome” is accepted, insulin resistance should be at the core of the “syndrome”, and many authors agree that NAFLD that is generally asymptomatic is frequently associated with obesity, type 2 diabetes and the “metabolic syndrome”[17]. Although a statement was published for synchronising the various definitions for metabolic syndrome[37], our experience confirmed that a large proportion of patients with NAFLD did not fulfill the “metabolic syndrome” criteria (49%-46% depending on the NCEP in ATP-III[38] or the AHA and IDF joint criteria[37] were applied). The high proportion of NAFLD patients without “metabolic syndrome” was observed despite that the HOMA2-IR values of the NAFLD patients were even higher in this study population than those with T2DM, provided that the NAFLD patients were excluded from the latter study group. None of the definitions of “metabolic syndrome” could be appraised as a consensus due to that the significant proportion of insulin-resistant NAFLD patients are excluded by these criteria[39].
BIOCHEMISTRY
The major biochemical event in NAFLD is the accumulation of triacylglycerols (TAG) in the hepatocytes. Because of the strong associations described above and to understand the relation of NAFLD to whole-body metabolic status, Donnelly et al[40] conducted a study using gas chromatography/mass spectrometry. Hepatic TAG might accumulate from different sources in the hepatocytes as follows: TAG as a nutrient after absorption from the intestine are delivered via chylomicrons to the liver, where they might subsequently be secreted as lipoproteins. Hepatic TAG synthesis is possible, and this process requires fatty acids and glycerol in the liver The required fatty acids might be from the plasma non-esterified fatty acid pool (NEFA), and they might be produced in the liver as de novo hepatic lipogenesis. It has been reported that approximately 60% of the TAG accumulated in the liver is derived from the plasma NEFA pool, even in the fed state, and that adipose tissue is the largest contributor to the fatty acid content of the plasma NEFA pool (80% in the fasted state). One-quarter of the TAG accumulation is derived from hepatic de novo lipogenesis that is elevated in the fasting state and demonstrates no diurnal variation, whereas approximately 15% is derived from the dietary intake. The situation is even worse when insulin resistance - a hallmark of metabolic syndrome - is also present because of the lack of (insulin induced) down-regulation of the hormone sensitive lipase that eventually results in enhanced lipolysis and an increased efflux of free fatty acids to the plasma NEFA pool from the adipocytes. Hyperglycaemia (and hyperinsulinaemia) induces SREBP-1c and ChREBP in the liver, and these transcription factors subsequently activate genes that are required for lipogenesis, eventually resulting in increased hepatic de novo lipogenesis. Hepatic de novo lipogenesis is increased in insulin-resistant states and in NAFLD[41]. Beta-oxidation of fatty acids is increased in patients with NASH; the oxidation might not overcome the increased hepatic TAG production, and the increased NEFA oxidation might result in increased oxidative stress, enhancing the transition of NAFL to NASH[42,43]. This (patho)biochemical path provides the reason that the association is remarkably strong between fatty liver and obesity-related insulin resistance[40,44].
NAFLD CONCURRENTLY WITH VISCERAL AND SUBCUTANEOUS ADIPOSITY AND INSULIN RESISTANCE
Is there accurate morphological evidence to support this biochemistry-driven hypothesis that the intrahepatic lipid content is hand-in-hand with different adipose tissue deposits, particularly the visceral adipose tissue accumulation that is strongly associated in impaired glucose metabolism?
Bosy-Westphal et al[45] assessed the fat volume including the visceral fat volumes (VAT), the pericardial adipose tissue (PAT, a well-known marker of visceral adiposity) and the abdominal subcutaneous adipose tissue using MRI and compared the results to the IHCL quantified by the highly sensitive 1H-MRS method in thirty overweight, not yet diabetic women. The participating individuals were restricted to a low calorie diet for three months; at baseline, the visceral adipose tissue volume and PAT correlated with the IHCL as well as with the insulin resistance measured with the euglycaemic hyperinsulinaemic clamp and the homeostatic model assessment (HOMA)-IR. The strength of the relationship between the visceral fat volume and IHCL is shown by the finding that the reductions in IHCL induced by the dietary intervention and loss of body weight were only correlated with the decrease in VAT. The exceptional role of NAFLD in determining insulin resistance is confirmed by the improvements in HOMA-IR and HOMA2-%B after three months of diet and weight loss that were only related to the decrease in IHCL[45].
The interpretation of HOMA-IR has been recently challenged. Traditionally, HOMA-IR reflects the degree of insulin resistance; however, these estimates based on the fasting plasma insulin and glucose concentrations[46] do not take into account whether the secretion of insulin by the pancreatic β cells is altered or if an alteration is in the insulin removal (clearance). The differentiation of total body insulin resistance, peripheral insulin resistance and hepatic insulin resistance merits research attention. A recent novel interpretation of HOMA-IR suggests that it is not a precise estimate of peripheral insulin action rather it might reflect the ability of insulin to suppress hepatic glucose production in the fasting state[47]. The clamp technique used in the previously mentioned study measures the peripheral insulin action in non-diabetic individuals resulting from hyperinsulinaemia during the measurement that is high enough to inhibit the hepatic glucose production completely[48]. This concept should be validated in studies using an insulin concentration that is lower than that regularly used during an euglycaemic-hyperinsulinaemic clamp measurement to avoid the absolute inhibition of hepatic glucose production[47]. Provided that this hypothesis regarding HOMA-IR is accurate, the 1H-MRS based follow-up observation that improvements in HOMA-IR and HOMA2-%B after dietary intervention were only related to the decrease in the IHCL indicates that a decrease in the intrahepatocellular lipid content increases the ability of insulin to suppress the hepatic glucose production in the fasting state, which is a major determinant of fasting plasma glucose levels[45].
In addition the contribution of the subcutaneous adipose tissue (SAT) to the whole-body adipose tissue dysfunction has been also recently confronted against the broadly accepted “innocent bystander to VAT” concept. The results of an in vivo human study that functionally assessed SAT in patients with histology-confirmed NASH provided evidence that the abdominal SAT in NASH patients is highly insulin resistant and required > 6 × more insulin to the gain similar degree of glycerol release suppression (referring to impaired suppression of lipolysis in SAT by insulin) than in healthy subjects. Authors therefore suggested that abdominal SAT is dysfunctional in NASH and plays a profound role in NASH development and lipotoxicity[49].
LIPOTOXICITY
Free fatty acids (FFA) are directly hepatotoxic, and FFA levels are elevated in patients with NASH and correlate with disease severity[50]. Patients with severe fibrosis shown by liver biopsy had significantly greater serum concentration of free fatty acids than did the patients without severe fibrosis[50]. Saturated FFAs (e.g., palmitate) are apparently more hepatotoxic than unsaturated (mono-unsaturated) FFAs (e.g., palmitoleate); palmitoleate (known as a lipokine) was demonstrated to suppress hepatic steatosis[51,52]. Unsaturated fatty acids do not induce endoplasmic reticulum (ER) stress or apoptosis and are able to rescue the palmitate-induced ER stress and apoptosis in liver cells. It has been proposed that the difference in toxicity between saturated and unsaturated fatty acids is that unsaturated FFAs are more easily esterified into neutral triglycerides[53,54].
The impairment of liver cellular capacity in FFA utilisation, incorporation to TAGs and export contributes to the development of NASH, and hepatic injury is further accentuated by pathological FA oxidation and altered cell membrane composition[55]. Lipotoxicity induces hepatocellular apoptosis; Kupffer cell activation; impaired insulin signaling and hepatic insulin resistance; and hepatic stellate (Ito) cell activation with subsequent fibrosis. These pathological processes might eventually lead to cirrhosis[55].
DIACYLGLYCEROL ACYLTRANSFERASE 2: DISSOCIATION OF STEATOSIS AND INFLAMMATION-FIBROSIS
The hepatotoxicity of saturated FFAs is further supported by the genetic deletion of diacylglycerol acyltransferase 2 (DGAT2) in mice models as follows: in parallel with the decreased TAG synthesis due to the the increased oxidative stress from the lack of intracellular FFA esterification, hepatocellular apoptosis with subsequent fibrosis occurs, resulting in the dissociation of hepatic steatosis and hepatic fibrosis in the NASH model[56,57]. Overexpression of DGAT2 in the experimental model causes hepatic steatosis without concomitant liver fibrosis or insulin resistance, providing evidence for the different roles that toxic saturated FFAs and TAGs have in the development of NAFLD[57,58].
The pathological effect of lipotoxicity is not limited to the liver cells, and it might affect the pancreatic β-cells, contributing to the β-cell dysfunction that is frequently observed in T2DM[59]. This pathology that affects both the liver and the pancreatic β-cells is highly important in determining the plasma glucose levels.
Lipotoxicity in the pancreatic β-cells
The intracellular signaling pathways altered because of lipotoxicity should partially overlap in the liver cells and in the pancreatic β-cells (Figure 1).
Figure 1.
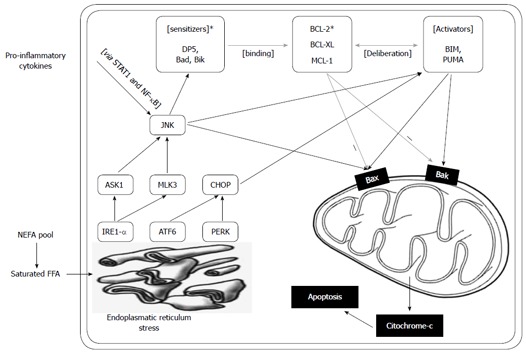
Endoplasmatic reticulum stress caused by saturated free fatty acids. Endoplasmatic reticulum stress caused by saturated free fatty acids, via three main mediators [inositol-requiring endoplasmic reticulum-to-nucleus signal kinase 1α (IRE1α), activating transcription factor 6 (ATF6) and RNA-dependent protein kinase (PRK)-like endoplasmic roticulum (ER) kinase (PERK)], results in the activation of c-Jun N-terminal kinase (JNK) and C/ CCAAT/enhancer binding protein (EBP) homologous protein. The sequence of BH3 protein activation based on a sensitizer and an activator group was described by Gurzov and Eizirik in β-cells[62,63]. The sensitizers bind the anti-apoptotic proteins Bcl-2, Bcl-XL and Mcl-1 and release the activators from this bond. JNK both mediates the induction of the sensitizer and the activator BH3 proteins and also activates Bax. Upregulation of Bcl-2 interacting mediator of cell death (BIM) and p53 upregulated modulator of apoptosis (PUMA) was also demonstrated in liver cells as a ruslt of free fatty acid (FFA) induction. Proinflammatory cytokines also activate JNK. ER sterss also results in CHOP actvation and subsequently the activation of the activator BH3 proteins. This complex signaling pathway might link the metabolic (saturated FFAs) stress and the effect of pro-inflammatory cytokines both in the pancreatic β-cell as well as in liver cells.
Saturated fatty acids, among other factors, induce endoplasmic reticulum stress that induce JNK activation via the IRE1α/ASK1, a signal that has been described in pancreatic β-cells as well as in liver cells[60,61]. Subsequently, the activation of the JNK pathway induces the “sensitiser” BH3 proteins (DP5, Bad, Bik), which bind to the anti-apoptotic Bcl-2 proteins such as Bcl-2 and Bcl-XL; these proteins designate the pro-apoptotic BH3-only proteins, BIM and PUMA (“activators”), eventually leading to their activation, which results in the apoptotic death of the pancreatic β and liver cells via Bak and Bax[61-63]. There is a substantial homology of this unsaturated FFA induced path and the path of cytokine induced β cell apoptosis. Palmitoleate (a mono-unsaturated fatty acid) could inhibit lipoapoptosis by blocking the endoplasmic reticulum stress-associated increases of the BH3-only proteins, Bim and PUMA, in hepatocytes[64]. The deteriorating β cell function, in combination with the increasing hepatic IR and the decreasing suppression of hepatic glucose output, leads to hyperglycaemia that eventually might directly (glucotoxicity) and by biochemical and metabolic consequences further promote this pathologic process. This cross-talk between the metabolic and cytokine induced pathways might facilitate the identification of novel drug targets (e.g., DP5, Bim) that would inhibit the unsaturated FFA induced ER stress mediated apoptotic liver cell death and possess protective properties against cytokine induced pancreatic β cell death[60-63]. The cytokines, growth factors and inflammatory mediators that are important in NAFLD are summarised in Table 2.
Table 2.
Adipokine hormones, cytokines, growth factors and other mediators that play important role in non-alcoholic fatty liver disease pathology
| Mediator/pathway | Observation /proposed mechanism | Ref. |
| Adiponectin | Shown to have anti-inflammatory and antifibrotic activity, serum levels were found to be decreased in NAFLD and NASH patients. Plasma adiponectin in NAFLD is related to hepatic insulin resistance and hepatic lipid content - not to liver disease severity | [127] |
| Ghrelin | Serum levels were found to be diminished in NAFLD and NASH patients - no correlation with histological grade. Ghrelin administration attenuated oxidative stress, inflammation and apoptosis in high fat diet induced NAFLD animal model | [128,129] |
| Leptin | Leptin levels are generally known to be higher in the sera of NASH patients, except for a subgroup; serum levels were shown to negatively correlate with AST/ALT levels. The livers of leptin-deficient mice were found to be unusually sensitive to LPS-induced injury. Recombinant leptin therapy was in clinical trial in patients with NASH and low leptin levels - no results were posted | [128] and ClinicalTrials.gov Identifier: NCT00596934 |
| Resistin | Serum levels were shown to be significantly higher in patients with NAFLD and NASH | [130] |
| Small bowel bacterial overgrowth (SIBO) | Increased gut permeability and tight junction alterations in NAFLD. Higher prevalence of small intestinal bacterial overgrowth in NAFLD patients - correlated with the severity of liver steatosis | [131] |
| Toll-like receptor-4 (TLR4) | Both the TLR4 (endotoxin-receptor) protein and RNA levels were found to be elevated in liver in NASH | [132] |
| Nuclear factor-κB (NF-κB) | Increased activation of NF-κB was found in NASH | [132] |
| Tumor necrosis factor-α (TNF-α) | Key mediator of inflammation, serum levels are elevated in NASH. TNF-α expression in adipose tissue is upregulated in several models of obesity. In patients, TNF-α levels were shown to be higher in obese than in lean individuals, and were correlated with insulin resistance | [133] |
| Interleukin-6 (IL-6) | Increased plasma levels and hepatic expression was described in NASH patients. Increased hepatic IL-6 production may play an important role in NASH, insulin resistance and diabetes development | [134] |
| Transforming growth factor-β (TGF-β) | A key growth factor and a major inductor of hepatic stellate cell activation and therefore hepatic fibrosis, TGF-β signaling in hepatocytes may contribute to hepatocyte death and lipid accumulation via Smad signaling and ROS production | [135] |
| Th17 cells and IL-17 | In the livers of mice on a high fat diet and also NASH patients an increased number of hepatic Th17 cells could be detected. In mice Il-17 neutralization ameliorated LPS induced liver injury | [136] |
| Interleukin-17 exacerbates hepatic steatosis and inflammation in non-alcoholic fatty liver disease | ||
| Notch-mTOR pathway | Liver-specific ablation of Notch signaling, or its acute inhibition with a decoy Notch1 receptor, prevents hepatosteatosis by blocking mTor complex 1 (mTorc1) activity. Notch gain of function induces NAFL through constitutive activation of mTorc1 | [137] |
| Mastocyte chymase | This enzyme is important in the convertion of angiotenzin-I to angiotenzin-II and the activation of matrix metalloproteinase-9, which both are involved in the development of liver fibrosis. Chymase inhibitor prevents the nonalcoholic steatohepatitis in a hamster model | [138] |
| Galectin 3 | Galectin 3 is a β-galactoside-binding lectin with a multiple functions. It is also a receptor of advanced lipoxidation endproducts and plays important role in inflammation, fibrosis and carcinogenesis. Its role is suspected in NASH. Regression of fibrosis by galectin inhibitors in thioacetamide-induced liver disease animal model. Phase 1 Study with a Galectin inhibitor GR-MD-02 in patients with NASH and advanced fibrosis | [139,140], ClinicalTrials.gov Identifier: NCT01899859 |
| Fibroblast geowth factor -19 (FGF19) | Both the intestinal FGF19 production and the hepatic response is impaired in NAFLD patients. A decrease in fasting FGF19 levels is associated with the development of non-alcoholic fatty liver disease in obese adolescents | [141] |
A few of these molecules are therapeutic targets (e.g., galectin 3) in early phase clinical trials. NAFLD: Non-alcoholic fatty liver disease; NASH: Non-alcoholic steatohepatitis; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase.
MITOCHONDRIAL DYSFUNCTION
A defective hepatic mitochondrial respiratory chain (MRC) was described in NASH[65]. The mitochondrial dysfunction, as measured by the activity of the MRC complexes in liver tissue, was correlated with the serum TNF-alpha levels and with the degree of insulin resistance that were higher in NASH and with the BMI[65]. In addition to the mitochondrial dysfunction, Sanyal et al[66] described structural mitochondrial defects including the loss of the mitochondrial cristae and paracrystalline inclusions, the presence of linear crystalline inclusions and mitochondrial swelling in patients with NASH. Patients with T2DM of long duration might have decreased ATP production after fasting and after fructose administration. The mitochondrial dysfunction in NAFLD, from lipotoxicity, oxidative stress and as a result of inflammatory mediator effect, alters the hepatic energy metabolism, as recently reported by Koliaki and Roden[67].
ENTERO-INSULAR AXIS (DPP-4 AND INCRETINS) IN NAFLD
A number of studies have assessed both the glucagon-like peptide-1 (GLP-1) and glucose dependent insulinotropic peptide induced insulin secretion in T2DM patients and concluded that the response to the incretin hormone stimuli might be compromised as an early phenomenon in T2DM[68-71]. In the carriers of the transcription factor 7-like 2 gene polymorphism (TCF7L2-rs7903146, a widely known risk polymorphism in T2DM development) the GLP-1 induced insulin secretion, and not the GLP-1 secretion, is impaired[72]. Similar findings were presented for a common genetic variant in the WFS1 gene, and carriers of the polymorphism had impaired GLP-1-induced insulin secretion independently of insulin sensitivity[73]. Variants of the KCNQ1 (rs151290, rs2237892 and rs2237895) gene altered the endogenous GLP-1 secretion[74,75].
Despite these well-designed, elegant studies that assessed the role of the entero-insular axis in T2DM and its association with the risk gene polymorphisms in T2DM, there is limited data regarding the role of the entero-insular axis in NAFLD. Recently, β cell connectedness has been reported to influence the incretin-induced insulin secretion in human islets, and lipotoxicity was demonstrated to be able to disrupt this incretin-regulated human β cell connectivity that might result in the loss of the coordinated islet response to metabolic stimuli[76].
Increased serum DPP-4 activity (the soluble form of the enzyme in human sera) was described in patients with NAFLD in comparison to healthy controls and T2DM patients, provided that T2DM patients who presented with clinically obvious (with an US and biochemical based diagnosis) NAFLD were excluded from the study group[39]. We found a positive correlation among the γGT, ALAT and serum DPP-4 activities in NAFLD that supports the finding that the excess DPP-4 found in the serum of NAFLD patients is of hepatic origin. We concluded that it is the presence of (fatty) liver disease that primarily influenced the serum DPP-4 enzymatic activity and not hyperglycaemia alone[39]. Subsequently, higher hepatic expression of DPP-4 at the mRNA level was described in NAFLD patients, and high glucose concentrations increased the DPP-4 expression in the HepG2 cells, in contrast to the insulin and FFAs that did not alter the DPP-4 expression[77]. The correlation among the serum levels of soluble CD26/DPP-4 activity and other liver disease biomarkers was also confirmed in Asian patients[78-80]. A positive correlation between the serum DPP-4 activity and insulin resistance (HOMA2-IR) in NAFLD was also found that is not surprising provided that the serum DPP-4 activity is considered as a novel liver disease biomarker. The existence of such a correlation further supported the recent suggestion that the soluble form of DPP-4 is a novel adipokine hormone that could induce insulin resistance without the presence of incretin hormones in an experimental system[81]. In a meta-analysis, Fadini et al[82] found that the DPP-4 activity was increased in T2D and was not lowered by glycemic control; the study confirmed that hyperglycaemia was not a direct determinant of DPP-4 activity and was lower in metformin-treated patients. A significant role of DPP-4 in hepatic glucose metabolism is supported by the study of Edgerton et al[83], who demonstrated that during DPP-4 inhibitor therapy (vildagliptin) and GLP-1 co-treatment, the net hepatic glucose uptake was three-fold greater in the DPP-4 inhibitor treated group than in the control group that was treated with portal vein GLP-1 infusion and not with the DPP-4 inhibitor; this effect was greater than that predicted by the change in insulin. This finding indicates that the DPP-4 inhibitor was able to increase the hepatic glucose disposal beyond the effects of GLP-1 on insulin and glucagon secretion.
The effect of DPP-4 inhibitor therapy on liver fibrosis should be assessed, particularly because the fibroblast activation protein (FAP), which is a duplicate molecule of DPP-4 (FAP-DPP-4 shows 88% homology at the cDNA level), is present at the tissue remodelling interface on hepatic stellate cells (HSCs, ITO cells) that primarily produce the accumulating extracellular matrix proteins (including collagens) in chronic liver diseases, eventually leading to fibrosis and cirrhosis of the liver[84].
DPP-4 INHIBITORS AND GLP-1 MIMETICS IN NAFLD TREATMENT
DPP-4 inhibitors were reported to improve hepatic steatosis and adipose tissue inflammation in mice[85,86]. Human treatments are also documented, in which the DPP-4 inhibitor, sitagliptin, was able to provide benefit for a refractory case of NAFLD[87]. Sitagliptin improved hepatocyte ballooning in a diabetic patient with NASH[88]. The available data on the use of GLP-1 mimetics is limited; the following effects were reported in animal models: improvement of the FA beta-oxidation; a decrease in the liver disease biomarker ALAT and hepatic TAGs; reduced ER-stress related hepatocyte cell death; and enhanced beneficial macroautophagy[89-92]. The GLP-1 derived nonpeptide (by the cleavage of the neutral endopeptidase), GLP-1 (28-36) amide, was shown to improve glucose disposal and decrease hepatic steatosis in high fat diet mice. The GLP-1 (28-36) amide suppressed the hepatic gluconeogenesis and improved the pyruvate tolerance in this model[93]. The large randomised controlled trials (RCTs) that would also assess safety issues of the DPP-4 inhibitors and the GLP-1 mimetics are missing in NAFL patients, as well as in NASH patients, in whom the indication for (an auxiliary) drug treatment (in addition to diet, weight loss and exercise) might be more obvious.
OTHER THERAPIES
There is consensus that the most effective treatment of NAFL in the overwhelming majority of cases is lifestyle change, including a supervised diet, exercise and weight loss. However doctors are not always successful in having patients reach this goal, and the potential pharmacological approaches that should be considered, predominantly in NASH patients, are briefly reviewed.
Metformin
Metformin is experiencing a booming renaissance in the treatment of T2DM; however, this drug has no significant effect on liver histology, probably in part because of the limited anti-steatogenic effect and failure to increase the adiponectin levels[94]. The guidelines do not recommend this biguanide specifically for the treatment of NASH (AASLD: Strength 1/Evidence: A)[26,95].
Thiazolidinediones
In contrast to metformin, thiazolidinediones (TZDs) are experiencing difficulty as pharmacological agents in human medicine. In certain countries there are no remaining agents from this drug class (e.g., the French Agency for the Safety of Health Products requested a pharmaceutical company to suspend the use of pioglitazone containing products for the treatment of type 2 diabetes in France in 2011 because of an increased risk of urinary bladder cancer). EASL and AASLD discussed pioglitazone, outlining that, from the hepatologist point of view, glitazones consistently provided benefit for patients with NASH and could be used to treat biopsy proven steatohepatitis (AASLD: Strength: 1, Evidence: B)[26,95]. The associations emphasise that the long-term safety of pioglitazone in NASH is not established, and it is likely that no doctor is treating NASH isolated from other safety issues[26].
Vitamin E
The nature of the dilemma with vitamin E is somewhat similar to that with the TZDs. A meta-analyses (of over 100000 participants) reported increased all-cause mortality with a dose of vitamin E ≥ 400 IU/d, and one meta-analysis found a statistically significant relationship between vitamin E dosage and all-cause mortality in a dose-response analysis[96,97]. Although others have questioned the results obtained from these meta-analyses[98,99], recently vitamin E (400 IU/d) was shown to significantly increase the risk of prostate cancer among healthy men[100]. According to the AASLD recommendation, this vitamin E dose should be doubled (800 IU/d) to improve liver histology in non-diabetic adults with biopsy proven NASH (Strength: 1, Quality: B), and it is not recommended in NASH patients with T2DM (Strength: 1, Quality: C)[26]. Histological improvement in NASH in this context should be interpreted for all lesions, except for improvement in fibrosis, and vitamin E is not recommended for the treatment of NAFLD-cirrhosis or cryptogenic cirrhosis[26,95].
Ursodeoxycholic acid-nor-ursodeoxycholic acid
The AASLD does not recommend ursodeoxycholic acid (UDCA) A for the treatment of NAFLD and NASH (Strength 1, Quality: B), and long term, high dose UDCA increased the rate of serious adverse events (the development of cirrhosis, varices, cholangiocarcinoma, liver transplantation or death) in patients with primary sclerosing cholangitis[26,101]. The pharmacological properties of norUrsodeoxycholic acid (nor-UDCA) might be more attractive and might later be studied in patients with NASH[102].
Farnesoid X receptor agonists
The bile acids secreted upon feeding undergo enterohepatic circulation and serve as endogenous ligands to a class of nuclear hormone receptors that function as ligand-activated transcription factors. Farnesoid X receptor (FXR) belongs to this class and serves as a receptor for hydrophobic bile acids. In T2DM, the bile acid composition is altered. The bile acid taurochenodeoxycholic acid increases insulin release via the FXR dependent inhibition of the KATP channels, and FXR has been described to improve insulin sensitivity and glucose uptake in adipose tissue, liver and skeletal muscle by regulating the genes that are important in metabolic control[103,104]. The FXR agonist WAY-362450 was able to decrease inflammation and fibrosis in a murine model of NASH[105]. The following results of a 6-wk double-blind, randomised, placebo-controlled clinical phase II trial of a semi-synthetic bile acid derivative, obeticholic acid (OCA), in patients with T2DM and presumed NAFLD has recently been published: administration of 25 or 50 mg of OCA for six weeks increased insulin sensitivity and reduced the markers of liver inflammation and fibrosis. A longer trial is ongoing with OCA in biopsy-proven NASH patients (Clinical trials identifier: NCT01265498)[106].
Despite the primary enthusiasm regarding the FXR agonists, the controversy might not be ignored due to the recent finding that under high fat diet conditions, FXR knockout (FXR-KO) mice benefited from the receptor deficiency, and FXR-KO protected against the HFD-induced impairment of fasting plasma glucose levels and glucose tolerance[107]. In parallel with the clinical trials, a better understanding of bile acid enterohepatic circulation and more research on the FXR-dependent and independent signaling pathways are warranted.
Pre and probiotics
The intestinal microbiome has an increasing role in the understanding of T2DM pathology. In a recent study, 20 patients with histology-proven NASH were randomised to receive probiotics or usual care for six months. The probiotic treatment decreased the ASAT, and the decrease in IHCL was confirmed using 1H-MRS[108]. Other studies have suggested a potential role for prebiotic fibres (non-digestible carbohydrates modulating the human microbiome) in NAFLD treatment, and larger RCTs might provide conclusive evidence[109].
Omega 3 polyunsaturated FA
Di Minno et al[110] recently summarised the potential of omega-3 fatty acids for the treatment of NAFLD reported in seven human trials, the largest of which was a 6-mo follow-up RCT of 144 patients with NAFLD; however, the authors concluded that well-designed RCTs of adequate size and duration, with histological endpoints, are needed to assess the long-term safety and efficacy of such treatment.
Others
The phase 1 study to evaluate GR-MD-02, a Galectin-3 inhibitor in patients with NASH and advanced fibrosis (NCT01899859) could on one day be an excellent example of how a long term molecular research[139,140], may potentially pay out in benefiting certain patients. Galectin-3 protein (binding to terminal galactose residues in glycoproteins) is implicated in the pathogenesis of liver fibrosis and the results with compounds inhibiting the protein suggest a potential role of these drugs in human liver fibrosis and even in cirrhosis in the NAFLD spectrum as well (Table 2). Hypothetical approaches, including the development of a peripheral cannabinoid 1 receptor agonist without psychiatric side effects or the supplementation of vitamin D3 to overcome vitamin D3 deficiency that is highly prevalent in NAFLD[92,111,112], might have an effect on future therapies; however, they have yet to be tested and even limited evidence from concept studies is missing. There are ongoing randomised, controlled studies with potentially promising compounds such as resveratrol (500 mg three times daily for six months vs placebo-NCT01464801) in obese patients with NAFLD/NASH; however, conclusions from these studies would be premature.
Surgical interventions
Surgical interventions including Roux-en-Y gastric bypass might have beneficial effects on NAFLD from calorie intake reduction that could increase hepatic insulin sensitivity and augment postprandial GLP-1 secretion with a subsequently improved β cell function[26,113].
GENETICS
Familial clustering and prevalence differences according to ethnic origin
Although screening of family members is not recommended in NAFLD[26] based on a retrospective review of 90 cases, the authors concluded that familial clustering is common, and 18% of NASH patients had a first degree relative with a similar phenotype[114]. Schwimmer et al[115] found that NAFLD was more common in siblings (59%) and parents (78%), using 1H-MRS based diagnostics of children with biopsy proven NAFLD in a familial aggregation study. After adjustment for age, sex, race and BMI, the study concluded that familial factors are a major determinant in NAFLD. Gene-environment interactions might have a role in the data e.g. the condition that family members are living in a common household might indicate common environmental risk factors (type of oil used in the diet etc.). The role of genetic risk factors is supported by the differences based on ethnic origin that were observed in the NAFLD prevalence in multi-ethnic cohorts (see the Prevalence chapter and Table 1), with a higher prevalence in Hispanics and a lower prevalence in African Americans compared to non-Hispanic whites[4,16].
Genome wide association studies
Five genome wide association scans (GWAS) are reported for NAFLD in the GWAS catalogue. Although these studies did not include case numbers that are typically employed in other GWA studies including T2DM, they provide remarkable evidence for genetic factors predisposing to or protecting from NAFLD. The risk polymorphisms with the highest ORs are summarised in Table 3, from the aspect of the NAFLD binary outcomes. Discussion of all the candidate genes is beyond the scope of this review.
Table 3.
Single nucleotide polymorphisms associated with non-alcoholic fatty liver disease in genome wide association studies
| Region | SNPs | Reported gene(s) | Mapped gene | OR | Risk allele frequency in controls | P value | Gene product function | Context | Initial sample size | Replication sample size | Platform (SNPs passing QC) | Ref. |
| 22q13.3 | rs738409 (I148M), rs2896019-G | PNPLA3 | PNPLA3 | 2.02 | 0.450 | 2 × 10-20 | Adiponutrin (PNPLA3)–nutritionally regulated lysophosphatidic acyltransferase: expressed in liver and adipose tissue. High CH diet increases expression. Has TAG hydrolase and DG transacylase activity. Strong predictor of steatosis, inflammation and fibrosis. Dysfunctional PNPLA3 promotes accumulation of lipotoxic substrates. I148M–association with HCC in severely obese individuals | Intron | 392 Japanese cases, 934 Japanese controls | 172 Japanese cases, 1012 Japanese control | Illumina [261540] | [119,120] |
| rs738491, rs3761472, rs2143571 | SAMM50 | SAMM50 | ||||||||||
| rs6006473, rs5764455, rs6006611 | PARVB | PARVB | ||||||||||
| 1p35 | rs6691847-C | PTPRU | PTPRU - MATN1 | 1.32 | 0.770 | 7 × 10-6 | Member of the protein tyrosine phosphatase (PTP) family. PTPs are signaling molecules that regulate cell growth, differentiation, mitotic cycle, and oncogenic transformation | |||||
| 4q13.3 | rs222054-C | GC | LDHAL6EP - GC | 2.54 | 0.301 | 1 × 10-6 | Albumin gene family. Multifunctional protein found in plasma, ascitic fluid, cerebrospinal fluid and on the surface of many cell types. It binds to vitamin D and its plasma metabolites and transports them to target tissues | 126 European adolescent cases - 802 European adolescent controls | NR | Illumina [2078805] (imputed) | [142] | |
| 16q23 | rs11864146-A | SLC38, A8 | SLC38A8 | 3.14 | 0.100 | 2 × 10-6 | Amino acid transport: Putative sodium-coupled neutral amino acid transporter 8 | Intron | ||||
| 13q14.1 | rs7324845-A | LCP1 | LCP1 | 3.29 | 0.096 | 3 × 10-6 | L-plastin, an actin binding protein expressed in hemopoetic cell lineages. It is expressed by many solid tumor types of non-hemopoetic origin, suggesting its role in tumorigenesis | Intron | ||||
| 1p21 | rs12743824-C | LPPR4 | LPPR4 - PALMD | 2.30 | 0.441 | 5 × 10-6 | LPPR4: a lipid phosphate phosphatase, catalyzes the dephosphorylation of a number of bioactive lipid mediators, found to be important for axonal outgrowth; PALMD: cytosolic isoform of paralemmin-1, a lipid raft-associated protein implicated in cell shape control | NR | PALMD gene product - [143] | |||
| 7p14 | rs343064-A | Intergenic | TBX20-HERPUD2 | 1.31 | 0.400 | 3 × 10-8 | TBX20: transcription factor; HERPUD2: not reported | 236 non-Hispanic Caucasian women | NR | Illumina [324623] | SNPs of other genes were also associated with qulatitative traits (e.g. NAFLD activity score and FDFT1 - rs2645424) as discussed in the text [123] | |
| 2q31 | rs1529093-A | Intergenic | RPL29P8 - KRT8P40 | 4.13 | 0.410 | 2 × 10-6 | RPL29P8, KRT8P40: pseudogenes | NR | Illumina [324623] | |||
| 4p15.2 | rs959903-A | 23231 | SEL1L3 | 3.81 | 0.290 | 7 × 10-6 | Intron | NR | Illumina [324623] |
Not all GWAS studies were replicated. Risk gene variants with OR higher than 1.31 are indicated in the table. Based on the National Human Genome Research. SNPs: Single nucleotide polymorphisms; NR: Not reported.
PATATIN-LIKE PHOSPHOLIPASE DOMAIN CONTAINING 3 GENE (PNPLA3-ADIPONUTRIN) IN NAFLD SPECTRUM DISEASES -BINARY TRAITS
We should outline the single nucleotide polymorphism in the patatin-like phospholipase domain containing a 3-gene (PNPLA3) that is the most studied genetic risk variant in NAFLD. Adiponutrin is a nutritionally regulated lysophosphatidic-acyltransferase possessing the TAG hydrolase and DG transacylase activity. This gene is expressed in liver and adipose tissues, and a high carbohydrate diet increases the gene expression at the mRNA level in the liver. Dysfunctional PNPLA3 promotes the accumulation of lipotoxic substrates[116,117]. After the description that in carriers of the rs738409 C/G polymorphism of the PNPLA3 the hepatic lipid content was more than two-fold higher, PNPLA3 was subsequently confirmed in a Japanese GWAS as an NAFLD binary trait candidate gene[118,119] (Table 3). Polymorphisms in the SAMM50 and PARVB genes were associated with the development and progression of NAFLD in this Japanese GWA study[119].
The quality trait data demonstrate the following findings: PNPLA3 I148M (rs738409) is a genetic marker of progressive liver disease that is characterised with steatosis, inflammation and fibrosis; carriers are more insulin resistant and more susceptible to T2DM[120,121]; and, in a 15-year-follow-up, there is an association of this genetic variant and hepatocellular carcinoma incidence in severely obese individuals, with a hazard ratio of HCC of 5.9x for each PNPLA3 148M allele carried (reaching the HR- 16x in the PNPLA3 148M homozygotes even after adjustment for age, gender, BMI, type 2 diabetes status and ALAT)[121,122].
OTHER CANDIDATE GENES (SQUALENE SYNTHASE AND COLLAGEN XIII A1)-QUALITATIVE TRAITS
In the cases in which additional candidate genes were identified, qualitative traits were also assessed in GWAS. The NAFLD activity score was associated with the rs2645424 polymorphism of farnesyl diphosphate farnesyl transferase 1 (FDFT1), the degree of fibrosis was associated with the rs343062 SNP, and the lobular inflammation was associated with the rs1227756 polymorphism of the Collagen 13 A1 gene[123].
FDFT1 is a membrane-associated enzyme located at a branch point in the mevalonate pathway. The encoded protein is the first specific enzyme in cholesterol biosynthesis, catalysing the dimerisation of two molecules of farnesyl diphosphate in a two-step reaction to form squalene. In addition to the linkage of FDFT1 to the NAFLD activity score, a coding variant in the FDFT1 gene influences the plasma cholesterol levels, likely via alteration of the intracellular production of cholesterol[123,124].
Collagen XIII is one of the nonfibrillar collagens and belongs to the transmembrane collagens, and a number of alternatively spliced transcript variants have been described; integrins mediate the cell adhesion to the type XIII collagen[125]. There is no validation of these associations in larger studies.
In summary, we may conclude that genetic and molecular research might lead to the identification of additional risk and protective gene variants and this together with deeper understanding of gene-environment interactions might provide better insight into the molecular pathology and identification of molecular targets in NAFLD, which is the most common chronic liver disease affecting up to one-third of the adult population in industrialised countries. In addition, little is known regarding the long-term effect of the increasing NAFLD prevalence in paediatric populations.
ACKNOWLEDGEMENTS
EFSD New Horizons grant facilitated the construction of this manuscript. I am grateful to my colleagues, especially Prof. A. Somogyi, Dr. G. Lengyel, Prof. K. Rácz and Prof. Zs Tulassay, who made it possible to conduct research on this topic in the our institution. I am also grateful to my PhD-fellow, Dr. A. Zóka and my co-worker Dr. Z. Kukor for their comments.
Footnotes
P- Reviewers: Fontana L, Rocha R S- Editor: Gou SX L- Editor: A E- Editor: Ma S
References
- 1.Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434–438. [PubMed] [Google Scholar]
- 2.Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221–1231. doi: 10.1056/NEJMra011775. [DOI] [PubMed] [Google Scholar]
- 3.Smith BW, Adams LA. Non-alcoholic fatty liver disease. Crit Rev Clin Lab Sci. 2011;48:97–113. doi: 10.3109/10408363.2011.596521. [DOI] [PubMed] [Google Scholar]
- 4.Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, Grundy SM, Hobbs HH. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387–1395. doi: 10.1002/hep.20466. [DOI] [PubMed] [Google Scholar]
- 5.Roden M. Mechanisms of Disease: hepatic steatosis in type 2 diabetes--pathogenesis and clinical relevance. Nat Clin Pract Endocrinol Metab. 2006;2:335–348. doi: 10.1038/ncpendmet0190. [DOI] [PubMed] [Google Scholar]
- 6.Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, Landt CL, Harrison SA. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology. 2011;140:124–131. doi: 10.1053/j.gastro.2010.09.038. [DOI] [PubMed] [Google Scholar]
- 7.Ong JP, Pitts A, Younossi ZM. Increased overall mortality and liver-related mortality in non-alcoholic fatty liver disease. J Hepatol. 2008;49:608–612. doi: 10.1016/j.jhep.2008.06.018. [DOI] [PubMed] [Google Scholar]
- 8.Kallwitz ER, Kumar M, Aggarwal R, Berger R, Layden-Almer J, Gupta N, Cotler SJ. Ethnicity and nonalcoholic fatty liver disease in an obesity clinic: the impact of triglycerides. Dig Dis Sci. 2008;53:1358–1363. doi: 10.1007/s10620-008-0234-x. [DOI] [PubMed] [Google Scholar]
- 9.Papatheodoridis GV, Goulis J, Christodoulou D, Manolakopoulos S, Raptopoulou M, Andrioti E, Alexandropoulos N, Savvidou S, Papachristou A, Zervou E, et al. High prevalence of elevated liver enzymes in blood donors: associations with male gender and central adiposity. Eur J Gastroenterol Hepatol. 2007;19:281–287. doi: 10.1097/MEG.0b013e328011438b. [DOI] [PubMed] [Google Scholar]
- 10.Dabelea D, Pihoker C, Talton JW, D’Agostino RB, Fujimoto W, Klingensmith GJ, Lawrence JM, Linder B, Marcovina SM, Mayer-Davis EJ, et al. Etiological approach to characterization of diabetes type: the SEARCH for Diabetes in Youth Study. Diabetes Care. 2011;34:1628–1633. doi: 10.2337/dc10-2324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rorat M, Jurek T, Kuchar E, Szenborn L, Golema W, Halon A. Liver steatosis in Polish children assessed by medicolegal autopsies. World J Pediatr. 2013;9:68–72. doi: 10.1007/s12519-012-0387-8. [DOI] [PubMed] [Google Scholar]
- 12.Schwimmer JB, Deutsch R, Kahen T, Lavine JE, Stanley C, Behling C. Prevalence of fatty liver in children and adolescents. Pediatrics. 2006;118:1388–1393. doi: 10.1542/peds.2006-1212. [DOI] [PubMed] [Google Scholar]
- 13.Schwenzer NF, Machann J, Martirosian P, Stefan N, Schraml C, Fritsche A, Claussen CD, Schick F. Quantification of pancreatic lipomatosis and liver steatosis by MRI: comparison of in/opposed-phase and spectral-spatial excitation techniques. Invest Radiol. 2008;43:330–337. doi: 10.1097/RLI.0b013e31816a88c6. [DOI] [PubMed] [Google Scholar]
- 14.Szczepaniak LS, Babcock EE, Schick F, Dobbins RL, Garg A, Burns DK, McGarry JD, Stein DT. Measurement of intracellular triglyceride stores by H spectroscopy: validation in vivo. Am J Physiol. 1999;276:E977–E989. doi: 10.1152/ajpendo.1999.276.5.E977. [DOI] [PubMed] [Google Scholar]
- 15.Krssák M, Hofer H, Wrba F, Meyerspeer M, Brehm A, Lohninger A, Steindl-Munda P, Moser E, Ferenci P, Roden M. Non-invasive assessment of hepatic fat accumulation in chronic hepatitis C by 1H magnetic resonance spectroscopy. Eur J Radiol. 2010;74:e60–e66. doi: 10.1016/j.ejrad.2009.03.062. [DOI] [PubMed] [Google Scholar]
- 16.Lazo M, Hernaez R, Eberhardt MS, Bonekamp S, Kamel I, Guallar E, Koteish A, Brancati FL, Clark JM. Prevalence of nonalcoholic fatty liver disease in the United States: the Third National Health and Nutrition Examination Survey, 1988-1994. Am J Epidemiol. 2013;178:38–45. doi: 10.1093/aje/kws448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bedogni G, Miglioli L, Masutti F, Tiribelli C, Marchesini G, Bellentani S. Prevalence of and risk factors for nonalcoholic fatty liver disease: the Dionysos nutrition and liver study. Hepatology. 2005;42:44–52. doi: 10.1002/hep.20734. [DOI] [PubMed] [Google Scholar]
- 18.Kojima S, Watanabe N, Numata M, Ogawa T, Matsuzaki S. Increase in the prevalence of fatty liver in Japan over the past 12 years: analysis of clinical background. J Gastroenterol. 2003;38:954–961. doi: 10.1007/s00535-003-1178-8. [DOI] [PubMed] [Google Scholar]
- 19.Amarapurkar D, Kamani P, Patel N, Gupte P, Kumar P, Agal S, Baijal R, Lala S, Chaudhary D, Deshpande A. Prevalence of non-alcoholic fatty liver disease: population based study. Ann Hepatol. 2007;6:161–163. [PubMed] [Google Scholar]
- 20.Angulo P, Hui JM, Marchesini G, Bugianesi E, George J, Farrell GC, Enders F, Saksena S, Burt AD, Bida JP, et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology. 2007;45:846–854. doi: 10.1002/hep.21496. [DOI] [PubMed] [Google Scholar]
- 21.Musso G, Gambino R, Cassader M, Pagano G. Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med. 2011;43:617–649. doi: 10.3109/07853890.2010.518623. [DOI] [PubMed] [Google Scholar]
- 22.Myers RP, Pomier-Layrargues G, Kirsch R, Pollett A, Duarte-Rojo A, Wong D, Beaton M, Levstik M, Crotty P, Elkashab M. Feasibility and diagnostic performance of the FibroScan XL probe for liver stiffness measurement in overweight and obese patients. Hepatology. 2012;55:199–208. doi: 10.1002/hep.24624. [DOI] [PubMed] [Google Scholar]
- 23.Wong GL. Transient elastography: Kill two birds with one stone? World J Hepatol. 2013;5:264–274. doi: 10.4254/wjh.v5.i5.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Feldstein AE, Wieckowska A, Lopez AR, Liu YC, Zein NN, McCullough AJ. Cytokeratin-18 fragment levels as noninvasive biomarkers for nonalcoholic steatohepatitis: a multicenter validation study. Hepatology. 2009;50:1072–1078. doi: 10.1002/hep.23050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Musso G, Gambino R, Bo S, Uberti B, Biroli G, Pagano G, Cassader M. Should nonalcoholic fatty liver disease be included in the definition of metabolic syndrome? A cross-sectional comparison with Adult Treatment Panel III criteria in nonobese nondiabetic subjects. Diabetes Care. 2008;31:562–568. doi: 10.2337/dc07-1526. [DOI] [PubMed] [Google Scholar]
- 26.Chalasani N, Younossi Z, Lavine JE, Diehl AM, Brunt EM, Cusi K, Charlton M, Sanyal AJ. The diagnosis and management of non-alcoholic fatty liver disease: practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012;55:2005–2023. doi: 10.1002/hep.25762. [DOI] [PubMed] [Google Scholar]
- 27.Ferenci P, Steindl-Munda P, Vogel W, Jessner W, Gschwantler M, Stauber R, Datz C, Hackl F, Wrba F, Bauer P, et al. Diagnostic value of quantitative hepatic copper determination in patients with Wilson’s Disease. Clin Gastroenterol Hepatol. 2005;3:811–818. doi: 10.1016/s1542-3565(05)00181-3. [DOI] [PubMed] [Google Scholar]
- 28.Pietrangelo A, Schilsky M. Metal storage disorders. Forward. Semin Liver Dis. 2011;31:231–232. doi: 10.1055/s-0031-1286053. [DOI] [PubMed] [Google Scholar]
- 29.Fink S, Schilsky ML. Inherited metabolic disease of the liver. Curr Opin Gastroenterol. 2007;23:237–243. doi: 10.1097/MOG.0b013e3280ef68e4. [DOI] [PubMed] [Google Scholar]
- 30.Leite NC, Salles GF, Araujo AL, Villela-Nogueira CA, Cardoso CR. Prevalence and associated factors of non-alcoholic fatty liver disease in patients with type-2 diabetes mellitus. Liver Int. 2009;29:113–119. doi: 10.1111/j.1478-3231.2008.01718.x. [DOI] [PubMed] [Google Scholar]
- 31.Prashanth M, Ganesh HK, Vima MV, John M, Bandgar T, Joshi SR, Shah SR, Rathi PM, Joshi AS, Thakkar H, et al. Prevalence of nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus. J Assoc Physicians India. 2009;57:205–210. [PubMed] [Google Scholar]
- 32.Leite NC, Villela-Nogueira CA, Pannain VL, Bottino AC, Rezende GF, Cardoso CR, Salles GF. Histopathological stages of nonalcoholic fatty liver disease in type 2 diabetes: prevalences and correlated factors. Liver Int. 2011;31:700–706. doi: 10.1111/j.1478-3231.2011.02482.x. [DOI] [PubMed] [Google Scholar]
- 33.Prikoszovich T, Winzer C, Schmid AI, Szendroedi J, Chmelik M, Pacini G, Krssák M, Moser E, Funahashi T, Waldhäusl W, et al. Body and liver fat mass rather than muscle mitochondrial function determine glucose metabolism in women with a history of gestational diabetes mellitus. Diabetes Care. 2011;34:430–436. doi: 10.2337/dc10-1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bozkurt L, Göbl CS, Tura A, Chmelik M, Prikoszovich T, Kosi L, Wagner O, Roden M, Pacini G, Gastaldelli A, et al. Fatty liver index predicts further metabolic deteriorations in women with previous gestational diabetes. PLoS One. 2012;7:e32710. doi: 10.1371/journal.pone.0032710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brumbaugh DE, Tearse P, Cree-Green M, Fenton LZ, Brown M, Scherzinger A, Reynolds R, Alston M, Hoffman C, Pan Z, et al. Intrahepatic fat is increased in the neonatal offspring of obese women with gestational diabetes. J Pediatr. 2013;162:930–936.e1. doi: 10.1016/j.jpeds.2012.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Borch-Johnsen K, Wareham N. The rise and fall of the metabolic syndrome. Diabetologia. 2010;53:597–599. doi: 10.1007/s00125-010-1659-2. [DOI] [PubMed] [Google Scholar]
- 37.Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–1645. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 38.Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III) JAMA. 2001;285:2486–2497. doi: 10.1001/jama.285.19.2486. [DOI] [PubMed] [Google Scholar]
- 39.Firneisz G, Varga T, Lengyel G, Fehér J, Ghyczy D, Wichmann B, Selmeci L, Tulassay Z, Rácz K, Somogyi A. Serum dipeptidyl peptidase-4 activity in insulin resistant patients with non-alcoholic fatty liver disease: a novel liver disease biomarker. PLoS One. 2010;5:e12226. doi: 10.1371/journal.pone.0012226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1343–1351. doi: 10.1172/JCI23621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Schwarz JM, Linfoot P, Dare D, Aghajanian K. Hepatic de novo lipogenesis in normoinsulinemic and hyperinsulinemic subjects consuming high-fat, low-carbohydrate and low-fat, high-carbohydrate isoenergetic diets. Am J Clin Nutr. 2003;77:43–50. doi: 10.1093/ajcn/77.1.43. [DOI] [PubMed] [Google Scholar]
- 42.Bugianesi E, Leone N, Vanni E, Marchesini G, Brunello F, Carucci P, Musso A, De Paolis P, Capussotti L, Salizzoni M, et al. Expanding the natural history of nonalcoholic steatohepatitis: from cryptogenic cirrhosis to hepatocellular carcinoma. Gastroenterology. 2002;123:134–140. doi: 10.1053/gast.2002.34168. [DOI] [PubMed] [Google Scholar]
- 43.Bugianesi E, Moscatiello S, Ciaravella MF, Marchesini G. Insulin resistance in nonalcoholic fatty liver disease. Curr Pharm Des. 2010;16:1941–1951. doi: 10.2174/138161210791208875. [DOI] [PubMed] [Google Scholar]
- 44.Tamura S, Shimomura I. Contribution of adipose tissue and de novo lipogenesis to nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1139–1142. doi: 10.1172/JCI24930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bosy-Westphal A, Kossel E, Goele K, Blöcker T, Lagerpusch M, Later W, Heller M, Glüer CC, Müller MJ. Association of pericardial fat with liver fat and insulin sensitivity after diet-induced weight loss in overweight women. Obesity (Silver Spring) 2010;18:2111–2117. doi: 10.1038/oby.2010.49. [DOI] [PubMed] [Google Scholar]
- 46.Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004;27:1487–1495. doi: 10.2337/diacare.27.6.1487. [DOI] [PubMed] [Google Scholar]
- 47.Reaven GM. What do we learn from measurements of HOMA-IR? Diabetologia. 2013;56:1867–1868. doi: 10.1007/s00125-013-2948-3. [DOI] [PubMed] [Google Scholar]
- 48.Pisprasert V, Ingram KH, Lopez-Davila MF, Munoz AJ, Garvey WT. Limitations in the use of indices using glucose and insulin levels to predict insulin sensitivity: impact of race and gender and superiority of the indices derived from oral glucose tolerance test in African Americans. Diabetes Care. 2013;36:845–853. doi: 10.2337/dc12-0840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Armstrong MJ, Hazlehurst JM, Hull D, Guo K, Borrows S, Yu J, Gough SC, Newsome PN, Tomlinson JW. Abdominal subcutaneous adipose tissue insulin resistance and lipolysis in patients with non-alcoholic steatohepatitis. Diabetes Obes Metab. 2014;16:651–660. doi: 10.1111/dom.12272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nehra V, Angulo P, Buchman AL, Lindor KD. Nutritional and metabolic considerations in the etiology of nonalcoholic steatohepatitis. Dig Dis Sci. 2001;46:2347–2352. doi: 10.1023/a:1012338828418. [DOI] [PubMed] [Google Scholar]
- 51.Wei Y, Wang D, Topczewski F, Pagliassotti MJ. Saturated fatty acids induce endoplasmic reticulum stress and apoptosis independently of ceramide in liver cells. Am J Physiol Endocrinol Metab. 2006;291:E275–E281. doi: 10.1152/ajpendo.00644.2005. [DOI] [PubMed] [Google Scholar]
- 52.Cao H, Gerhold K, Mayers JR, Wiest MM, Watkins SM, Hotamisligil GS. Identification of a lipokine, a lipid hormone linking adipose tissue to systemic metabolism. Cell. 2008;134:933–944. doi: 10.1016/j.cell.2008.07.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li ZZ, Berk M, McIntyre TM, Feldstein AE. Hepatic lipid partitioning and liver damage in nonalcoholic fatty liver disease: role of stearoyl-CoA desaturase. J Biol Chem. 2009;284:5637–5644. doi: 10.1074/jbc.M807616200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Listenberger LL, Han X, Lewis SE, Cases S, Farese RV, Ory DS, Schaffer JE. Triglyceride accumulation protects against fatty acid-induced lipotoxicity. Proc Natl Acad Sci USA. 2003;100:3077–3082. doi: 10.1073/pnas.0630588100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Trauner M, Arrese M, Wagner M. Fatty liver and lipotoxicity. Biochim Biophys Acta. 2010;1801:299–310. doi: 10.1016/j.bbalip.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 56.Yamaguchi K, Yang L, McCall S, Huang J, Yu XX, Pandey SK, Bhanot S, Monia BP, Li YX, Diehl AM. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology. 2007;45:1366–1374. doi: 10.1002/hep.21655. [DOI] [PubMed] [Google Scholar]
- 57.Monetti M, Levin MC, Watt MJ, Sajan MP, Marmor S, Hubbard BK, Stevens RD, Bain JR, Newgard CB, Farese RV, et al. Dissociation of hepatic steatosis and insulin resistance in mice overexpressing DGAT in the liver. Cell Metab. 2007;6:69–78. doi: 10.1016/j.cmet.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 58.Ibrahim SH, Kohli R, Gores GJ. Mechanisms of lipotoxicity in NAFLD and clinical implications. J Pediatr Gastroenterol Nutr. 2011;53:131–140. doi: 10.1097/MPG.0b013e31822578db. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Marchetti P, Bugliani M, Boggi U, Masini M, Marselli L. The pancreatic beta cells in human type 2 diabetes. Adv Exp Med Biol. 2012;771:288–309. doi: 10.1007/978-1-4614-5441-0_22. [DOI] [PubMed] [Google Scholar]
- 60.Miani M, Barthson J, Colli ML, Brozzi F, Cnop M, Eizirik DL. Endoplasmic reticulum stress sensitizes pancreatic beta cells to interleukin-1β-induced apoptosis via Bim/A1 imbalance. Cell Death Dis. 2013;4:e701. doi: 10.1038/cddis.2013.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ibrahim SH, Gores GJ. Who pulls the trigger: JNK activation in liver lipotoxicity? J Hepatol. 2012;56:17–19. doi: 10.1016/j.jhep.2011.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Eizirik DL, Colli ML, Ortis F. The role of inflammation in insulitis and beta-cell loss in type 1 diabetes. Nat Rev Endocrinol. 2009;5:219–226. doi: 10.1038/nrendo.2009.21. [DOI] [PubMed] [Google Scholar]
- 63.Gurzov EN, Eizirik DL. Bcl-2 proteins in diabetes: mitochondrial pathways of β-cell death and dysfunction. Trends Cell Biol. 2011;21:424–431. doi: 10.1016/j.tcb.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 64.Akazawa Y, Cazanave S, Mott JL, Elmi N, Bronk SF, Kohno S, Charlton MR, Gores GJ. Palmitoleate attenuates palmitate-induced Bim and PUMA up-regulation and hepatocyte lipoapoptosis. J Hepatol. 2010;52:586–593. doi: 10.1016/j.jhep.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pérez-Carreras M, Del Hoyo P, Martín MA, Rubio JC, Martín A, Castellano G, Colina F, Arenas J, Solis-Herruzo JA. Defective hepatic mitochondrial respiratory chain in patients with nonalcoholic steatohepatitis. Hepatology. 2003;38:999–1007. doi: 10.1053/jhep.2003.50398. [DOI] [PubMed] [Google Scholar]
- 66.Sanyal AJ, Campbell-Sargent C, Mirshahi F, Rizzo WB, Contos MJ, Sterling RK, Luketic VA, Shiffman ML, Clore JN. Nonalcoholic steatohepatitis: association of insulin resistance and mitochondrial abnormalities. Gastroenterology. 2001;120:1183–1192. doi: 10.1053/gast.2001.23256. [DOI] [PubMed] [Google Scholar]
- 67.Koliaki C, Roden M. Hepatic energy metabolism in human diabetes mellitus, obesity and non-alcoholic fatty liver disease. Mol Cell Endocrinol. 2013;379:35–42. doi: 10.1016/j.mce.2013.06.002. [DOI] [PubMed] [Google Scholar]
- 68.Kjems LL, Holst JJ, Vølund A, Madsbad S. The influence of GLP-1 on glucose-stimulated insulin secretion: effects on beta-cell sensitivity in type 2 and nondiabetic subjects. Diabetes. 2003;52:380–386. doi: 10.2337/diabetes.52.2.380. [DOI] [PubMed] [Google Scholar]
- 69.Højberg PV, Vilsbøll T, Rabøl R, Knop FK, Bache M, Krarup T, Holst JJ, Madsbad S. Four weeks of near-normalisation of blood glucose improves the insulin response to glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide in patients with type 2 diabetes. Diabetologia. 2009;52:199–207. doi: 10.1007/s00125-008-1195-5. [DOI] [PubMed] [Google Scholar]
- 70.Holst JJ, Knop FK, Vilsbøll T, Krarup T, Madsbad S. Loss of incretin effect is a specific, important, and early characteristic of type 2 diabetes. Diabetes Care. 2011;34 Suppl 2:S251–S257. doi: 10.2337/dc11-s227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Knop FK, Aaboe K, Vilsbøll T, Vølund A, Holst JJ, Krarup T, Madsbad S. Impaired incretin effect and fasting hyperglucagonaemia characterizing type 2 diabetic subjects are early signs of dysmetabolism in obesity. Diabetes Obes Metab. 2012;14:500–510. doi: 10.1111/j.1463-1326.2011.01549.x. [DOI] [PubMed] [Google Scholar]
- 72.Schäfer SA, Tschritter O, Machicao F, Thamer C, Stefan N, Gallwitz B, Holst JJ, Dekker JM, ‘t Hart LM, Nijpels G, et al. Impaired glucagon-like peptide-1-induced insulin secretion in carriers of transcription factor 7-like 2 (TCF7L2) gene polymorphisms. Diabetologia. 2007;50:2443–2450. doi: 10.1007/s00125-007-0753-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Schäfer SA, Müssig K, Staiger H, Machicao F, Stefan N, Gallwitz B, Häring HU, Fritsche A. A common genetic variant in WFS1 determines impaired glucagon-like peptide-1-induced insulin secretion. Diabetologia. 2009;52:1075–1082. doi: 10.1007/s00125-009-1344-5. [DOI] [PubMed] [Google Scholar]
- 74.Smushkin G, Sathananthan M, Sathananthan A, Dalla Man C, Micheletto F, Zinsmeister AR, Cobelli C, Vella A. Diabetes-associated common genetic variation and its association with GLP-1 concentrations and response to exogenous GLP-1. Diabetes. 2012;61:1082–1089. doi: 10.2337/db11-1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Müssig K, Staiger H, Machicao F, Kirchhoff K, Guthoff M, Schäfer SA, Kantartzis K, Silbernagel G, Stefan N, Holst JJ, et al. Association of type 2 diabetes candidate polymorphisms in KCNQ1 with incretin and insulin secretion. Diabetes. 2009;58:1715–1720. doi: 10.2337/db08-1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hodson DJ, Mitchell RK, Bellomo EA, Sun G, Vinet L, Meda P, Li D, Li WH, Bugliani M, Marchetti P, et al. Lipotoxicity disrupts incretin-regulated human β cell connectivity. J Clin Invest. 2013;123:4182–4194. doi: 10.1172/JCI68459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Miyazaki M, Kato M, Tanaka K, Tanaka M, Kohjima M, Nakamura K, Enjoji M, Nakamuta M, Kotoh K, Takayanagi R. Increased hepatic expression of dipeptidyl peptidase-4 in non-alcoholic fatty liver disease and its association with insulin resistance and glucose metabolism. Mol Med Rep. 2012;5:729–733. doi: 10.3892/mmr.2011.707. [DOI] [PubMed] [Google Scholar]
- 78.Aso Y, Ozeki N, Terasawa T, Naruse R, Hara K, Suetsugu M, Takebayashi K, Shibazaki M, Haruki K, Morita K, et al. Serum level of soluble CD26/dipeptidyl peptidase-4 (DPP-4) predicts the response to sitagliptin, a DPP-4 inhibitor, in patients with type 2 diabetes controlled inadequately by metformin and/or sulfonylurea. Transl Res. 2012;159:25–31. doi: 10.1016/j.trsl.2011.09.005. [DOI] [PubMed] [Google Scholar]
- 79.Firneisz G, Somogyi A. Serum level of soluble CD26/dieptidyl peptidase-4 (DPP-4) activity correlates with other liver disease biomarkers both in Asian and European patients. Transl Res. 2012;160:95–96. doi: 10.1016/j.trsl.2012.01.016. [DOI] [PubMed] [Google Scholar]
- 80.Aso Y. Re: Serum level of soluble CD26/dipeptidyl peptidase-4 (DPP-4) Transl Res. 2012;160:164. doi: 10.1016/j.trsl.2012.01.017. [DOI] [PubMed] [Google Scholar]
- 81.Lamers D, Famulla S, Wronkowitz N, Hartwig S, Lehr S, Ouwens DM, Eckardt K, Kaufman JM, Ryden M, Müller S, et al. Dipeptidyl peptidase 4 is a novel adipokine potentially linking obesity to the metabolic syndrome. Diabetes. 2011;60:1917–1925. doi: 10.2337/db10-1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fadini GP, Albiero M, Menegazzo L, de Kreutzenberg SV, Avogaro A. The increased dipeptidyl peptidase-4 activity is not counteracted by optimized glucose control in type 2 diabetes, but is lower in metformin-treated patients. Diabetes Obes Metab. 2012;14:518–522. doi: 10.1111/j.1463-1326.2011.01550.x. [DOI] [PubMed] [Google Scholar]
- 83.Edgerton DS, Johnson KM, Neal DW, Scott M, Hobbs CH, Zhang X, Duttaroy A, Cherrington AD. Inhibition of dipeptidyl peptidase-4 by vildagliptin during glucagon-like Peptide 1 infusion increases liver glucose uptake in the conscious dog. Diabetes. 2009;58:243–249. doi: 10.2337/db08-0515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Levy MT, McCaughan GW, Abbott CA, Park JE, Cunningham AM, Müller E, Rettig WJ, Gorrell MD. Fibroblast activation protein: a cell surface dipeptidyl peptidase and gelatinase expressed by stellate cells at the tissue remodelling interface in human cirrhosis. Hepatology. 1999;29:1768–1778. doi: 10.1002/hep.510290631. [DOI] [PubMed] [Google Scholar]
- 85.Shirakawa J, Fujii H, Ohnuma K, Sato K, Ito Y, Kaji M, Sakamoto E, Koganei M, Sasaki H, Nagashima Y, et al. Diet-induced adipose tissue inflammation and liver steatosis are prevented by DPP-4 inhibition in diabetic mice. Diabetes. 2011;60:1246–1257. doi: 10.2337/db10-1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kern M, Klöting N, Niessen HG, Thomas L, Stiller D, Mark M, Klein T, Blüher M. Linagliptin improves insulin sensitivity and hepatic steatosis in diet-induced obesity. PLoS One. 2012;7:e38744. doi: 10.1371/journal.pone.0038744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Itou M, Kawaguchi T, Taniguchi E, Oriishi T, Sata M. Dipeptidyl Peptidase IV Inhibitor Improves Insulin Resistance and Steatosis in a Refractory Nonalcoholic Fatty Liver Disease Patient: A Case Report. Case Rep Gastroenterol. 2012;6:538–544. doi: 10.1159/000341510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Yilmaz Y, Yonal O, Deyneli O, Celikel CA, Kalayci C, Duman DG. Effects of sitagliptin in diabetic patients with nonalcoholic steatohepatitis. Acta Gastroenterol Belg. 2012;75:240–244. [PubMed] [Google Scholar]
- 89.Mells JE, Fu PP, Sharma S, Olson D, Cheng L, Handy JA, Saxena NK, Sorescu D, Anania FA. Glp-1 analog, liraglutide, ameliorates hepatic steatosis and cardiac hypertrophy in C57BL/6J mice fed a Western diet. Am J Physiol Gastrointest Liver Physiol. 2012;302:G225–G235. doi: 10.1152/ajpgi.00274.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Trevaskis JL, Griffin PS, Wittmer C, Neuschwander-Tetri BA, Brunt EM, Dolman CS, Erickson MR, Napora J, Parkes DG, Roth JD. Glucagon-like peptide-1 receptor agonism improves metabolic, biochemical, and histopathological indices of nonalcoholic steatohepatitis in mice. Am J Physiol Gastrointest Liver Physiol. 2012;302:G762–G772. doi: 10.1152/ajpgi.00476.2011. [DOI] [PubMed] [Google Scholar]
- 91.Sharma S, Mells JE, Fu PP, Saxena NK, Anania FA. GLP-1 analogs reduce hepatocyte steatosis and improve survival by enhancing the unfolded protein response and promoting macroautophagy. PLoS One. 2011;6:e25269. doi: 10.1371/journal.pone.0025269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Schuppan D, Schattenberg JM. Non-alcoholic steatohepatitis: pathogenesis and novel therapeutic approaches. J Gastroenterol Hepatol. 2013;28 Suppl 1:68–76. doi: 10.1111/jgh.12212. [DOI] [PubMed] [Google Scholar]
- 93.Ip W, Shao W, Chiang YT, Jin T. GLP-1-derived nonapeptide GLP-1(28-36)amide represses hepatic gluconeogenic gene expression and improves pyruvate tolerance in high-fat diet-fed mice. Am J Physiol Endocrinol Metab. 2013;305:E1348–E1358. doi: 10.1152/ajpendo.00376.2013. [DOI] [PubMed] [Google Scholar]
- 94.Tiikkainen M, Häkkinen AM, Korsheninnikova E, Nyman T, Mäkimattila S, Yki-Järvinen H. Effects of rosiglitazone and metformin on liver fat content, hepatic insulin resistance, insulin clearance, and gene expression in adipose tissue in patients with type 2 diabetes. Diabetes. 2004;53:2169–2176. doi: 10.2337/diabetes.53.8.2169. [DOI] [PubMed] [Google Scholar]
- 95.Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol. 2010;53:372–384. doi: 10.1016/j.jhep.2010.04.008. [DOI] [PubMed] [Google Scholar]
- 96.Miller ER, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142:37–46. doi: 10.7326/0003-4819-142-1-200501040-00110. [DOI] [PubMed] [Google Scholar]
- 97.Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis. JAMA. 2007;297:842–857. doi: 10.1001/jama.297.8.842. [DOI] [PubMed] [Google Scholar]
- 98.Berry D, Wathen JK, Newell M. Bayesian model averaging in meta-analysis: vitamin E supplementation and mortality. Clin Trials. 2009;6:28–41. doi: 10.1177/1740774508101279. [DOI] [PubMed] [Google Scholar]
- 99.Gerss J, Köpcke W. The questionable association of vitamin E supplementation and mortality--inconsistent results of different meta-analytic approaches. Cell Mol Biol (Noisy-le-grand) 2009;55 Suppl:OL1111–OL1120. [PubMed] [Google Scholar]
- 100.Klein EA, Thompson IM, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT) JAMA. 2011;306:1549–1556. doi: 10.1001/jama.2011.1437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lindor KD, Kowdley KV, Luketic VA, Harrison ME, McCashland T, Befeler AS, Harnois D, Jorgensen R, Petz J, Keach J, et al. High-dose ursodeoxycholic acid for the treatment of primary sclerosing cholangitis. Hepatology. 2009;50:808–814. doi: 10.1002/hep.23082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Beraza N, Ofner-Ziegenfuss L, Ehedego H, Boekschoten M, Bischoff SC, Mueller M, Trauner M, Trautwein C. Nor-ursodeoxycholic acid reverses hepatocyte-specific nemo-dependent steatohepatitis. Gut. 2011;60:387–396. doi: 10.1136/gut.2010.223834. [DOI] [PubMed] [Google Scholar]
- 103.Düfer M, Hörth K, Wagner R, Schittenhelm B, Prowald S, Wagner TF, Oberwinkler J, Lukowski R, Gonzalez FJ, Krippeit-Drews P, et al. Bile acids acutely stimulate insulin secretion of mouse β-cells via farnesoid X receptor activation and K(ATP) channel inhibition. Diabetes. 2012;61:1479–1489. doi: 10.2337/db11-0815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Adorini L, Pruzanski M, Shapiro D. Farnesoid X receptor targeting to treat nonalcoholic steatohepatitis. Drug Discov Today. 2012;17:988–997. doi: 10.1016/j.drudis.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 105.Zhang S, Wang J, Liu Q, Harnish DC. Farnesoid X receptor agonist WAY-362450 attenuates liver inflammation and fibrosis in murine model of non-alcoholic steatohepatitis. J Hepatol. 2009;51:380–388. doi: 10.1016/j.jhep.2009.03.025. [DOI] [PubMed] [Google Scholar]
- 106.Mudaliar S, Henry RR, Sanyal AJ, Morrow L, Marschall HU, Kipnes M, Adorini L, Sciacca CI, Clopton P, Castelloe E, et al. Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and nonalcoholic fatty liver disease. Gastroenterology. 2013;145:574–582.e1. doi: 10.1053/j.gastro.2013.05.042. [DOI] [PubMed] [Google Scholar]
- 107.Abstracts of the 49th EASD (European Association for the Study of Diabetes) Annual Meeting. September 23-27, 2013. Barcelona, Spain. Diabetologia. 2013;56 Suppl 1:1–566. doi: 10.1007/s00125-013-3012-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wong VW, Won GL, Chim AM, Chu WC, Yeung DK, Li KC, Chan HL. Treatment of nonalcoholic steatohepatitis with probiotics. A proof-of-concept study. Ann Hepatol. 2013;12:256–262. [PubMed] [Google Scholar]
- 109.Parnell JA, Raman M, Rioux KP, Reimer RA. The potential role of prebiotic fibre for treatment and management of non-alcoholic fatty liver disease and associated obesity and insulin resistance. Liver Int. 2012;32:701–711. doi: 10.1111/j.1478-3231.2011.02730.x. [DOI] [PubMed] [Google Scholar]
- 110.Di Minno MN, Russolillo A, Lupoli R, Ambrosino P, Di Minno A, Tarantino G. Omega-3 fatty acids for the treatment of non-alcoholic fatty liver disease. World J Gastroenterol. 2012;18:5839–5847. doi: 10.3748/wjg.v18.i41.5839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Targher G, Scorletti E, Mantovani A, Byrne CD. Nonalcoholic fatty liver disease and reduced serum vitamin D(3) levels. Metab Syndr Relat Disord. 2013;11:217–228. doi: 10.1089/met.2013.0044. [DOI] [PubMed] [Google Scholar]
- 112.Eliades M, Spyrou E, Agrawal N, Lazo M, Brancati FL, Potter JJ, Koteish AA, Clark JM, Guallar E, Hernaez R. Meta-analysis: vitamin D and non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2013;38:246–254. doi: 10.1111/apt.12377. [DOI] [PubMed] [Google Scholar]
- 113.Dirksen C, Jørgensen NB, Bojsen-Møller KN, Jacobsen SH, Hansen DL, Worm D, Holst JJ, Madsbad S. Mechanisms of improved glycaemic control after Roux-en-Y gastric bypass. Diabetologia. 2012;55:1890–1901. doi: 10.1007/s00125-012-2556-7. [DOI] [PubMed] [Google Scholar]
- 114.Willner IR, Waters B, Patil SR, Reuben A, Morelli J, Riely CA. Ninety patients with nonalcoholic steatohepatitis: insulin resistance, familial tendency, and severity of disease. Am J Gastroenterol. 2001;96:2957–2961. doi: 10.1111/j.1572-0241.2001.04667.x. [DOI] [PubMed] [Google Scholar]
- 115.Schwimmer JB, Celedon MA, Lavine JE, Salem R, Campbell N, Schork NJ, Shiehmorteza M, Yokoo T, Chavez A, Middleton MS, et al. Heritability of nonalcoholic fatty liver disease. Gastroenterology. 2009;136:1585–1592. doi: 10.1053/j.gastro.2009.01.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Dubuquoy C, Robichon C, Lasnier F, Langlois C, Dugail I, Foufelle F, Girard J, Burnol AF, Postic C, Moldes M. Distinct regulation of adiponutrin/PNPLA3 gene expression by the transcription factors ChREBP and SREBP1c in mouse and human hepatocytes. J Hepatol. 2011;55:145–153. doi: 10.1016/j.jhep.2010.10.024. [DOI] [PubMed] [Google Scholar]
- 117.Li JZ, Huang Y, Karaman R, Ivanova PT, Brown HA, Roddy T, Castro-Perez J, Cohen JC, Hobbs HH. Chronic overexpression of PNPLA3I148M in mouse liver causes hepatic steatosis. J Clin Invest. 2012;122:4130–4144. doi: 10.1172/JCI65179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Romeo S, Kozlitina J, Xing C, Pertsemlidis A, Cox D, Pennacchio LA, Boerwinkle E, Cohen JC, Hobbs HH. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat Genet. 2008;40:1461–1465. doi: 10.1038/ng.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kitamoto T, Kitamoto A, Yoneda M, Hyogo H, Ochi H, Nakamura T, Teranishi H, Mizusawa S, Ueno T, Chayama K, et al. Genome-wide scan revealed that polymorphisms in the PNPLA3, SAMM50, and PARVB genes are associated with development and progression of nonalcoholic fatty liver disease in Japan. Hum Genet. 2013;132:783–792. doi: 10.1007/s00439-013-1294-3. [DOI] [PubMed] [Google Scholar]
- 120.Dubuquoy C, Burnol AF, Moldes M. PNPLA3, a genetic marker of progressive liver disease, still hiding its metabolic function? Clin Res Hepatol Gastroenterol. 2013;37:30–35. doi: 10.1016/j.clinre.2012.06.014. [DOI] [PubMed] [Google Scholar]
- 121.Palmer CN, Maglio C, Pirazzi C, Burza MA, Adiels M, Burch L, Donnelly LA, Colhoun H, Doney AS, Dillon JF, et al. Paradoxical lower serum triglyceride levels and higher type 2 diabetes mellitus susceptibility in obese individuals with the PNPLA3 148M variant. PLoS One. 2012;7:e39362. doi: 10.1371/journal.pone.0039362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Burza MA, Pirazzi C, Maglio C, Sjöholm K, Mancina RM, Svensson PA, Jacobson P, Adiels M, Baroni MG, Borén J, et al. PNPLA3 I148M (rs738409) genetic variant is associated with hepatocellular carcinoma in obese individuals. Dig Liver Dis. 2012;44:1037–1041. doi: 10.1016/j.dld.2012.05.006. [DOI] [PubMed] [Google Scholar]
- 123.Chalasani N, Guo X, Loomba R, Goodarzi MO, Haritunians T, Kwon S, Cui J, Taylor KD, Wilson L, Cummings OW, Chen YD, Rotter JI, Nonalcoholic Steatohepatitis Clinical Research Network. Genome-wide association study identifies variants associated with histologic features of nonalcoholic Fatty liver disease. Gastroenterology. 2010;139:1567–1576, 1576.e1-6. doi: 10.1053/j.gastro.2010.07.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Do R, Paré G, Montpetit A, Hudson TJ, Gaudet D, Engert JC. K45R variant of squalene synthase increases total cholesterol levels in two study samples from a French Canadian population. Hum Mutat. 2008;29:689–694. doi: 10.1002/humu.20702. [DOI] [PubMed] [Google Scholar]
- 125.Nykvist P, Tu H, Ivaska J, Käpylä J, Pihlajaniemi T, Heino J. Distinct recognition of collagen subtypes by alpha(1)beta(1) and alpha(2)beta(1) integrins. Alpha(1)beta(1) mediates cell adhesion to type XIII collagen. J Biol Chem. 2000;275:8255–8261. doi: 10.1074/jbc.275.11.8255. [DOI] [PubMed] [Google Scholar]
- 126.Lee JY, Kim KM, Lee SG, Yu E, Lim YS, Lee HC, Chung YH, Lee YS, Suh DJ. Prevalence and risk factors of non-alcoholic fatty liver disease in potential living liver donors in Korea: a review of 589 consecutive liver biopsies in a single center. J Hepatol. 2007;47:239–244. doi: 10.1016/j.jhep.2007.02.007. [DOI] [PubMed] [Google Scholar]
- 127.Bugianesi E, Pagotto U, Manini R, Vanni E, Gastaldelli A, de Iasio R, Gentilcore E, Natale S, Cassader M, Rizzetto M, et al. Plasma adiponectin in nonalcoholic fatty liver is related to hepatic insulin resistance and hepatic fat content, not to liver disease severity. J Clin Endocrinol Metab. 2005;90:3498–3504. doi: 10.1210/jc.2004-2240. [DOI] [PubMed] [Google Scholar]
- 128.Yalniz M, Bahcecioglu IH, Ataseven H, Ustundag B, Ilhan F, Poyrazoglu OK, Erensoy A. Serum adipokine and ghrelin levels in nonalcoholic steatohepatitis. Mediators Inflamm. 2006;2006:34295. doi: 10.1155/MI/2006/34295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Li Y, Hai J, Li L, Chen X, Peng H, Cao M, Zhang Q. Administration of ghrelin improves inflammation, oxidative stress, and apoptosis during and after non-alcoholic fatty liver disease development. Endocrine. 2013;43:376–386. doi: 10.1007/s12020-012-9761-5. [DOI] [PubMed] [Google Scholar]
- 130.Senateş E, Colak Y, Yeşil A, Coşkunpinar E, Sahin O, Kahraman OT, Erkalma Şenateş B, Tuncer I. Circulating resistin is elevated in patients with non-alcoholic fatty liver disease and is associated with steatosis, portal inflammation, insulin resistance and nonalcoholic steatohepatitis scores. Minerva Med. 2012;103:369–376. [PubMed] [Google Scholar]
- 131.Miele L, Valenza V, La Torre G, Montalto M, Cammarota G, Ricci R, Mascianà R, Forgione A, Gabrieli ML, Perotti G, et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009;49:1877–1887. doi: 10.1002/hep.22848. [DOI] [PubMed] [Google Scholar]
- 132.Singh R, Bullard J, Kalra M, Assefa S, Kaul AK, Vonfeldt K, Strom SC, Conrad RS, Sharp HL, Kaul R. Status of bacterial colonization, Toll-like receptor expression and nuclear factor-kappa B activation in normal and diseased human livers. Clin Immunol. 2011;138:41–49. doi: 10.1016/j.clim.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 133.Braunersreuther V, Viviani GL, Mach F, Montecucco F. Role of cytokines and chemokines in non-alcoholic fatty liver disease. World J Gastroenterol. 2012;18:727–735. doi: 10.3748/wjg.v18.i8.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Wieckowska A, Papouchado BG, Li Z, Lopez R, Zein NN, Feldstein AE. Increased hepatic and circulating interleukin-6 levels in human nonalcoholic steatohepatitis. Am J Gastroenterol. 2008;103:1372–1379. doi: 10.1111/j.1572-0241.2007.01774.x. [DOI] [PubMed] [Google Scholar]
- 135.Yang L, Roh YS, Song J, Zhang B, Liu C, Loomba R, Seki E. Transforming growth factor beta signaling in hepatocytes participates in steatohepatitis through regulation of cell death and lipid metabolism in mice. Hepatology. 2014;59:483–495. doi: 10.1002/hep.26698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tang Y, Bian Z, Zhao L, Liu Y, Liang S, Wang Q, Han X, Peng Y, Chen X, Shen L, et al. Interleukin-17 exacerbates hepatic steatosis and inflammation in non-alcoholic fatty liver disease. Clin Exp Immunol. 2011;166:281–290. doi: 10.1111/j.1365-2249.2011.04471.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Pajvani UB, Qiang L, Kangsamaksin T, Kitajewski J, Ginsberg HN, Accili D. Inhibition of Notch uncouples Akt activation from hepatic lipid accumulation by decreasing mTorc1 stability. Nat Med. 2013;19:1054–1060. doi: 10.1038/nm.3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Tashiro K, Takai S, Jin D, Yamamoto H, Komeda K, Hayashi M, Tanaka K, Tanigawa N, Miyazaki M. Chymase inhibitor prevents the nonalcoholic steatohepatitis in hamsters fed a methionine- and choline-deficient diet. Hepatol Res. 2010;40:514–523. doi: 10.1111/j.1872-034X.2010.00627.x. [DOI] [PubMed] [Google Scholar]
- 139.Iacobini C, Menini S, Ricci C, Blasetti Fantauzzi C, Scipioni A, Salvi L, Cordone S, Delucchi F, Serino M, Federici M, et al. Galectin-3 ablation protects mice from diet-induced NASH: a major scavenging role for galectin-3 in liver. J Hepatol. 2011;54:975–983. doi: 10.1016/j.jhep.2010.09.020. [DOI] [PubMed] [Google Scholar]
- 140.Traber PG, Chou H, Zomer E, Hong F, Klyosov A, Fiel MI, Friedman SL. Regression of fibrosis and reversal of cirrhosis in rats by galectin inhibitors in thioacetamide-induced liver disease. PLoS One. 2013;8:e75361. doi: 10.1371/journal.pone.0075361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wojcik M, Janus D, Dolezal-Oltarzewska K, Kalicka-Kasperczyk A, Poplawska K, Drozdz D, Sztefko K, Starzyk JB. A decrease in fasting FGF19 levels is associated with the development of non-alcoholic fatty liver disease in obese adolescents. J Pediatr Endocrinol Metab. 2012;25:1089–1093. doi: 10.1515/jpem-2012-0253. [DOI] [PubMed] [Google Scholar]
- 142.Adams LA, White SW, Marsh JA, Lye SJ, Connor KL, Maganga R, Ayonrinde OT, Olynyk JK, Mori TA, Beilin LJ, et al. Association between liver-specific gene polymorphisms and their expression levels with nonalcoholic fatty liver disease. Hepatology. 2013;57:590–600. doi: 10.1002/hep.26184. [DOI] [PubMed] [Google Scholar]
- 143.Hu B, Petrasch-Parwez E, Laue MM, Kilimann MW. Molecular characterization and immunohistochemical localization of palmdelphin, a cytosolic isoform of the paralemmin protein family implicated in membrane dynamics. Eur J Cell Biol. 2005;84:853–866. doi: 10.1016/j.ejcb.2005.07.002. [DOI] [PubMed] [Google Scholar]


