Abstract
Irritable bowel syndrome (IBS) is the most frequently diagnosed functional gastrointestinal disorder in primary and secondary care. It is characterised by abdominal discomfort, pain and changes in bowel habits that can have a serious impact on the patient’s quality of life. The pathophysiology of IBS is not yet completely clear. Genetic, immune, environmental, inflammatory, neurological and psychological factors, in addition to visceral hypersensitivity, can all play an important role, one that most likely involves the complex interactions between the gut and the brain (gut-brain axis). The diagnosis of IBS can only be made on the basis of the symptoms of the Rome III criteria. Because the probability of organic disease in patients fulfilling the IBS criteria is very low, a careful medical history is critical and should pay particular attention to the possible comorbidities. Nevertheless, the severity of the patient’s symptoms or concerns sometimes compels the physician to perform useless and/or expensive diagnostic tests, transforming IBS into a diagnosis of exclusion. The presence of alarming symptoms (fever, weight loss, rectal bleeding, significant changes in blood chemistry), the presence of palpable abdominal masses, any recent onset of symptoms in patient aged over 50 years, the presence of symptoms at night, and a familial history of celiac disease, colorectal cancer and/or inflammatory bowel diseases all warrant investigation. Treatment strategies are based on the nature and severity of the symptoms, the degree of functional impairment of the bowel habits, and the presence of psychosocial disorders. This review examines and discusses the pathophysiological aspects and the diagnostic and therapeutic approaches available for patients with symptoms possibly related to IBS, pointing out controversial issues and the strengths and weaknesses of the current knowledge.
Keywords: Irritable bowel syndrome, Pathogenesis, Diagnosis, Therapy
Core tip: The pathophysiology of irritable bowel syndrome (IBS) is not definitely known and many fundamental questions remain unanswered about its pathophysiology, diagnosis and therapy. Conflicting results reflect the largely overlapping data of healthy controls and the wide heterogeneity of the IBS patients. This review summarises the main pathophysiological aspects, practical diagnostic approaches and therapeutic management strategies for patients with symptoms possibly related to IBS, in addition to pointing out some controversial issues and pointing out the strengths and the weaknesses of our current knowledge.
INTRODUCTION
Irritable bowel syndrome (IBS) is quite prevalent in the general population (from 5% to 20%) and represents the functional gastrointestinal (GI) disorder most frequently encountered in primary and secondary care[1,2]. IBS is characterised by abdominal discomfort, pain and changes in bowel habits (constipation and/or diarrhoea)[3] that wax and wane over time. Moreover, it is often associated with other functional digestive and non-digestive disorders[4-8].
The pathophysiology of IBS is not definitely known but most likely involves central and peripheral mechanisms. A disruption of the so called “brain-gut axis” that determines changes in digestive motility and secretion, causes visceral hypersensitivity and leads to cellular and molecular abnormalities in the enteroendocrine and immune systems has been suggested. In addition, genetic factors, infections and alterations of the intestinal microbiota, inflammation and food intolerance and/or hypersensitivity could play a role by altering the integrity of the intestinal barrier and increasing intestinal permeability[9,10]. Up to now, unfortunately, conflicting results have been achieved, most likely reflecting the largely overlapping data of healthy controls and the wide heterogeneity of the IBS population.
The direct and indirect costs of the syndrome are significant, as IBS can have a serious impact on patient quality of life. Because there are not yet any available biological markers or resolving therapies, the patient may undergo expensive tests and treatments[11-13].
The therapeutic approach depends on the intensity of symptoms and the degree of psychosocial comorbidities. Initial treatment is directed towards education, reassurance and lifestyle modification. In a second phase, an appropriate pharmacotherapy can be proposed on the basis of individual or global intestinal symptoms and/or psychological disturbances.
Many different drugs have been suggested for IBS treatment, but their real benefits are very debatable. Based on the multifaceted pathophysiology of the disease, it is unlikely that drugs acting on a single receptor and/or a unique pathophysiologic mechanism would be able to provide any substantial therapeutic gain over a placebo in this disease, for which the placebo response rate is approximately 40%[14].
Essentially, we are still far from having discovered the magic bullet capable of treating all IBS symptoms. Although many papers have been published on this syndrome in recent years, up to now, many fundamental questions remain unanswered about its pathophysiology, diagnosis and therapy.
This review summarises the main pathophysiological aspects, practical diagnostic approaches and therapeutic management strategies for patients with symptoms possibly related to IBS, in addition to pointing out some controversial issues and pointing out the strengths and the weaknesses of our current knowledge.
A search of the literature was carried out using the online databases of PubMed, Medline and Cochrane to identify articles published in English concerning pathophysiology, diagnosis and treatment of IBS.
PATHOPHYSIOLOGICAL ASPECTS
The pathophysiology of IBS, as in all functional digestive disorders, is complicated because there is no clearly identified pathophysiological basis for the disease. In fact, IBS is identified by a combination of chronic or recurrent GI symptoms in the absence of structural abnormalities (radiological/endoscopic) or biomarkers capable of positively identifying this condition. Aside from these drawbacks, the clinical manifestations of IBS are themselves extremely heterogeneous, a sort of “semantic umbrella” under which different clinical situations related to phenotypic aspect (traditionally subtyped as diarrhoea predominant, constipation predominant and mixed type) and the modality of clinical onset (post-infectious, food-related, stress-linked, etc.) fall[15].
The aetiology of IBS is multifactorial. Many pathogenetic factors, in various combinations and not all necessarily present in each patient, can play an important role (Table 1). Genetics, immune factors, environmental influences, inflammatory and infective agents, neurological and psychological factors, hypersensitivity to food and to bile salts and altered intestinal microbiota and permeability can all influence the brain-gut axis, leading to abnormal GI function and motility. It is unclear which among these factors is the trigger or how these conditions converge to initiate the IBS; previous studies aiming to identify a factor as more of a trigger over the others all failed to distinguish any one trigger.
Table 1.
Factors potentially involved in the pathogenesis of irritable bowel syndrome
| Altered intestinal motility |
| Food intolerance/allergy |
| Enteric infection/inflammation |
| Altered intestinal immunity |
| Altered gut microbiota |
| Genetics |
| Psychological distress and disorders; sexual abuse |
The genetic factors have been extensively studied. Up to 33% of IBS patients have a family history of IBS, compared to 2% of controls[16]. There is a higher prevalence of the disease in families of patients with IBS compared to the families of the spouses without IBS[17]. Moreover, some studies have reported a higher prevalence in monozygotic twins compared with heterozygotes, indicating a hypothetical genetic component[18]. However, other studies[19] demonstrated that having a parent with IBS was a better predictive factor than having a twin affected with IBS, suggesting that the environmental factor is more important.
The genetic factors involved in the pathogenesis of IBS has also been evaluated by a number of studies investigating the possible role of gene polymorphisms coding for serotonin (SERT), cholecystokinin (CCK) receptors 1, anti-inflammatory and pro-inflammatory interleukins and alpha 2 adrenergic receptors[20-22]. As serotonin was involved in the regulation of digestive motility, secretion and visceral sensitivity, particular investigative emphasis has been placed on polymorphisms of the gene regulating the reuptake of serotonin (SERT), which can induce a variation of its synaptic concentration[23]. SERT polymorphisms are not related to the development or onset of IBS, but rather to a different clinical expression, a greater perception of abdominal pain and an increased dissatisfaction regarding bowel habits[24].
Recently, a “biopsychosocial” model[25,26] has been introduced, in an attempt to integrate and harmonise the different factors (genetic, environmental and psychological) acting in a synergistic way to produce these symptoms.
These deficiencies in understanding the pathophysiological mechanisms of IBS have a heavy negative effect on clinical practice and may explain the disappointing results of previous therapeutic attempts, as well as the high costs of management. Currently, there is no single drug that is able to treat all of the symptoms related to IBS; rather, a “drug cocktail” is administered, having different effects on different symptoms.
Previous studies[27] have considered this syndrome a result of alterations in the normal digestive motility pattern, the so-called “spastic colon”. Subsequently, much interest was directed toward visceral hypersensitivity, under the hypothesis that IBS patients experienced visceral stimuli more strongly than healthy subjects. Later, IBS came to be considered a two-way interaction between the gut and the brain, with much interest directed not only toward the activation/deactivation of afferent and efferent nervous stimuli but also toward the effects of neuromodulators.
The possibility that IBS could be initiated after an enteric infection and the evidence that, in inflammatory bowel disease limited to the mucosa, patients suffer from enhanced sensory perception and motor dysfunction have driven researchers to study these as further potential causes of IBS.
Some previous studies[28,29] attempted to assess whether an abnormal motility pattern is typical in cases of IBS; however, despite identifying cluster contractions in phase II of the migrating motor complex in the jejunum, propagated ileal contractions related to pain and an increased postprandial motor activity of the colon, up to now, all attempts made have failed to reach a single typical pattern.
An altered colonic transit rate [accelerated in IBS and diarrhoea (IBS-D) and slowed in IBS with constipation (IBS-C)] was described in some studies[30,31] but these results have not been confirmed by more recent studies[32,33]. Salvioli et al[34] reported a decreased capacity of the motor activity in the small intestine to eliminate intestinal gas, resulting in abdominal distension and typical symptoms of IBS. IBS patients likely experience psychological stress, foods, neurotransmitters and/or rectal or bowel distension, which can lead to an altered motor response that leads to the same motor events being perceived more strongly and painfully[35].
Visceral hypersensitivity in IBS patients is supported by several studies[36-38]. Verne et al[39] used functional nuclear magnetic resonance (RMN) to show that a mechanical stimulus (rectal distension) active different regions of the brain in healthy volunteers, compared to patients with IBS. Unfortunately, this technique is expensive and not widely available. Moreover, comorbidities, such as fibromyalgia and psychological disturbances, can significantly affect its outcome.
Psychological disorders, including sexual and physical abuse, result in a high percentage of patients with functional disorders. Even if the disorders are not directly responsible for the onset or progression of the IBS symptoms, they certainly determine a different perception of the symptoms and result in more frequent requests for medical aid. In fact, these disorders are more common in IBS patients who seek medical care than in patients who do not ask for medical help or healthy volunteers[15,40].
Psychological distress and disorders can affect the brain-gut axis, promoting the release of corticotropin-releasing hormone, which is able to influence mood, digestive motility, permeability, visceral sensitivity and inflammatory pathways via neuroendocrine and autonomic outflows[41-44]. Dinan et al[44] showed that physical and mental stress in IBS patients increased the levels of pro-inflammatory interleukins, activating both the hypothalamic-autonomic nervous system and the hypothalamic-pituitary-adrenal (HPA) axes and consequently increasing the serological adreno-cortico-tropic-hormone and cortisol levels. Recent studies[44,45] introduced the hypothesis that IBS could be an inflammatory disorder that is supported by a dysregulation of the HPA.
On the other hand, it has been shown that physical and psychological stress activates different regions of the brain among patients with IBS than among healthy volunteers. In particular, IBS patients have a greater activation of the mid-anterior cingulate cortex, an area linked to anxiety, fear and hypervigilance[46]. This area is the target of many antidepressant drugs and psychotherapy. In healthy controls, stress instead activates the perigenual area, from which originate the descending inhibitory pathways that control visceral afferents to the posterior horn of the spinal cord[47].
A continuous and mutual interaction between the gut and the brain is made possible through the autonomic nervous system and the enteric nervous system via neuroendocrine mediators (VIP, 5HT, Ach, NO, NO, CCK, etc.); this system comprises the so-called “gut - brain axis”. Signals received from the GI tract affect the brain that, in turn, can affect the motility, secretion and immune functions of the digestive tract. Thus, alterations to this system may cause many digestive disorders, and particularly IBS, compared to normal, unaltered subjects[41,48,49].
The neuroendocrine system is potentially involved in the pathogenesis of IBS. This system is very complex and consists essentially of two components.
The endocrine cells (at least 14 endocrine or paracrine cell populations), which are distributed between the epithelial cells of the digestive mucosa and directly in contact with the intestinal lumen and its contents; and the nerve fibres (peptidergic, serotonergic, nitrergic, etc.) of the enteric nervous system[15].
Motility, secretion, absorption and intestinal microcirculation are all influenced by this system by the means of several mediators that have endocrine (released directly into the blood stream), autocrine/paracrine (local effects) or neuroendocrine (released from synapses into the bloodstream) functions[41].
An alteration to this system has been hypothesised, in which a decreased density of cells producing gastric inhibitory polypeptide (GIP) and somatostatin (in D-IBS and C-IBS) and in those producing secretin and CCK (in D-IBS) was reported in the small intestine, whereas a lower expression of cells producing 5-hydroxytryptamine and PYY was detected in the colons of patients with D-IBS and C-IBS[15,41]. An abnormal inflammatory response to different events (stress, infections, food, etc.) could be responsible for the abnormal cellularity in the colonic mucosa and the increased concentration of pro-inflammatory interleukins detected in the colons of some IBS patients[50]. These studies suggest that the activation of mast cells, macrophages or leukocytes producing inflammatory mediators is able to affect the motility, secretion, sensitive nerve endings and ultimate perception of pain.
Biopsies from the colons of IBS patients showed an increased activation of lymphocytes and mast cells in close proximity to the enteric neurons, with increased production of cytokines and other proinflammatory and vasoactive peptides[51,52]. Degranulation of these cells (especially mast cells) has been associated with the onset of the typical abdominal pain endured by IBS patients[53]. Moreover, the density of immunocompetent cells gradually increases on a spectrum from controls to patients with IBS, then to patients with microscopic colitis and, finally, to those with ulcerative colitis[54].
Inflammation can also result from a previous enteric infection. The onset of IBS follows an infection in approximately 10% of patients. In these patients, there are increases in the levels of CD3 serum lymphocytes, CD8 intraepithelial lymphocytes, and macrophage calprotectin-positive cells. Moreover, cells producing serotonin and CCK were found to be increased in the small bowel, while those producing serotonin and PYY were decreased in the colon. These alterations were usually transient but tended to persist in patients who developed IBS[55].
In post-infectious IBS and D-IBS, intestinal permeability has also been studied. The findings included a decreased expression and remodelling of the structural proteins constituting the epithelial “tight junctions” in the cells of the small intestine and colon. These changes increased the intestinal permeability, resulting in an easier passage of antigenic material through the epithelium and a stimulation of the intestinal immune system (especially mast cells) with the production of the proteases, histamine and prostanoids able to maintain the permeability and to produce abnormal neuronal responses, inducing the motor and sensory results typical in IBS[42].
Based on these results, it is evident that preserving, maintaining or restoring the normal composition of the intestinal microbiota is essential for good bowel function[42]. The intestinal microbiota is a major target of many therapeutic options for relieving IBS symptoms. The colon of each individual contains from 300 to 500 different species of bacteria. Thus, each of our microbiota is individual and unique. The microbiota is influenced by the environment, diet, previous infections, genetics, age, and antibiotic therapy. In normal conditions, the lactobacilli and bifidobacteria bind to epithelial cells, inhibiting the binding of pathogens and reinforcing the defences of the mucosal barrier. In addition, lactobacilli and bifidobacteria do not produce gas by fermenting carbohydrates and inhibiting the growth of the Clostridia species, which do produce this effect. Lactobacilli and bifidobacteria were found to be decreased in IBS patients, and their activities were found to be heavily compromised[56]. Moreover, some evidence indicates that probiotics affect intestinal fermentation and stabilise the intestinal microbiota, normalising the relationship between pro-inflammatory and anti-inflammatory cytokines with beneficial effects on intestinal inflammation, permeability and visceral sensitivity[57,58].
Unfortunately, at present, there are intrinsic difficulties in clearly establishing the role of the gut microbiota in the pathophysiology of IBS, both due to the great heterogeneity in the clinical presentation of IBS and to the limitations of the available studies (study design, length of observation, small sample, etc.).
Finally, the role of food in IBS merits specific mention. Patients with IBS tend to declare that their symptoms are often exacerbated by meals or by certain foods (sweeteners, fats, etc.). The classical IgE-mediated food allergy does not seem to play an important role in IBS. In the recent past, high levels of the specific IgG4 for wheat, beef, pork and lamb were found in IBS patients, compared to healthy subjects, and based on this, an exclusion diet was proposed[59]. On the other hand, this subgroup of Ig seems to be only an epiphenomenon of mucosal production, according to recent evidence[60].
In any case, up to 60% of patients with IBS reported a worsening of symptoms after food intake, in particular after specific foods like milk and dairy products, wheat, onions, beans, spices, cabbage, red meat, fried, smoked products, and caffeine. These foods represent the so-called fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs). However, studies supporting this are limited and demonstrate only a partial improvement in patients after the restriction of these foods. More frequently, IBS patients seem to have an exaggerated gastric-colic reflex after eating any item of food.
In recent years, it has been observed that the ingestion of gluten causes abdominal discomfort and IBS-like symptoms in subjects without a diagnosis of celiac disease (the so-called gluten sensitivity).
At the moment, the mechanisms responsible for these symptoms are not clear. Most likely, the gluten, as other well-known factors, alters the intestinal permeability, activating the enteric and autonomous nervous systems and producing the typical symptoms of IBS. Recently, authors have disagreed on the topic of gluten sensitivity, instead attempting to explain the problem with a simpler hypothesis: gluten-rich foods may cause symptoms with the same mechanisms of the FODMAPs[61,62]. The positive effect of the gluten-free diet on abdominal disorders could be due to the drastic reduction of FODMAPs that is inevitable in a diet of this type.
Up to now, the available results in the literature conflict; thus, further studies are needed to clarify this intriguing matter.
DIAGNOSTIC APPROACHES
A careful medical history is critical for the evaluation of a patient with a possible diagnosis of IBS. Particular attention has to be devoted to many different issues, such as dietary habits, therapies (especially the intake of drugs capable of altering the bowel frequency and/or causing abdominal pain), the degree of physical activity, comorbidities, previous surgical interventions, presence of symptoms suggesting anxiety or depression, and recent trips to exotic locations[3,63].
In the absence of accepted and shared biological markers, symptoms remain the cornerstone for the diagnosis of IBS.
Regarding the symptom “pain”, it is useful to assess its type (cramping, tensive, stabbing, burning), localisation, frequency, duration, mode of occurrence and possible changes in relation to defecation, to food intake (or to intake of particular foods), to stressful events and to the menstrual cycle[63,64].
As for abdominal distension or tension, it is mandatory to ask the patient if it is visible from others or if it is otherwise measurable (changes in size, inability to tie the skirt or pants, etc.). Additionally, patients should be asked whether their pain gets worse at certain times or improves with evacuation or emission of the flatus.
It is also necessary to investigate the characteristics of the defecation: difficult or prolonged, painful or simply incomplete, the presence of a sensation of anorectal blocking, the need for manual help, the presence of ineffective attempts or, on the contrary, of an urgency at defecation and real episodes of faecal incontinence[64].
Moreover, it is important to check for the presence of blood, mucus or pus in the faeces and to assess the usual shape of the stool using the Bristol Scale that, by relating the rate of intestinal transit with faecal consistency, provides a visual aid to help the patient better classify a topic otherwise difficult to objectify[65].
Additionally, it is mandatory to look for the possible co-morbidities that can occur in a patient with IBS, because they can increase the perception of the disease severity[8,13,66,67].
In Table 2, the most frequent co-morbidities are represented. These share common characteristics, such as the following: (1) a higher prevalence in females; (2) pathophysiology linked to low-grade inflammation, stress, somatisation, hypersensitivity, changes in the central processing of peripheral afferents and/or alterations of substances acting as neuromodulators; (3) a diagnosis mainly based on symptoms; (4) possible responsiveness to antidepressant medications and cognitive-behavioural therapies; (5) frequent multidisciplinary management; and (6) a considerable reduction of the quality of life and high, direct and indirect, costs.
Table 2.
Most frequently reported comorbidities in irritable bowel syndrome patients
| Functional dyspepsia and functional heartburn |
| Fibromyalgia |
| Chronic fatigue syndrome |
| Back pain |
| Multiple chemical sensitivity syndrome |
| Post-traumatic stress disorder |
| Psychological/psychiatric disorders |
| Sleep disturbances |
| Migraine and tension headaches |
The presence of alarm symptoms, the so-called “red flags” like fever, weight loss, rectal bleeding, and significant changes in blood chemistry, should be investigated, as well as the presence of palpable abdominal masses, any recent onset of symptoms in patients aged over 50 years, the presence of symptoms at night, and a familiar history positive for celiac disease, colorectal cancer and/or inflammatory bowel disease[64,68].
Still, some authors[69] believe that the accuracy of the “alarm symptoms” is disappointing. In particular, rectal bleeding and nocturnal pain would be of little value in discriminating patients with IBS from patients with organic disease, while anaemia and weight loss would have low sensitivity, but high specificity, to identify an organic disease.
A physical examination would not be very rich in information, as it could only detect abdominal tenderness (localised or diffuse) and abdominal hypertympanism or bowel sounds at auscultation, but this practice reassures the patient and can provide a first, coarse exclusion of organic diseases (abdominal masses, etc.). The examination should include the inspection of the anorectal region and a digital rectal examination, preferably in the left-lateral decubitus, which would provide useful information about the dynamics of the pelvic floor, especially if any functional alteration is suspected. Thus, the presence of comorbidities and organic diseases can be detected[63,70-72].
The use of specifically dedicated scores to measure the impairment of the quality of life and symptom severity has been debated in clinical practice, both at the initial stages and later, in order to verify the effectiveness of the therapy administered[73]. Indeed, any such scoring systems are not widely used outside of clinical trials, even if they do not seem time-consuming or difficult to use[74-77].
Can a diagnosis of IBS be made only using only symptom-based criteria? The evidence from the literature seems reassuring in this respect, because the probability of organic disease arising in patients fulfilling the IBS criteria is very low[78]. Nevertheless, the nature and severity of the symptoms themselves, or of the patient’s concerns and fears, sometimes compel the physician to perform unnecessary, useless, and/or expensive diagnostic tests, transforming IBS into a diagnosis of exclusion.
Indeed, in the differential diagnosis, the conditions reported in Table 3 will have to be considered with greater or lesser probability[68].
Table 3.
Diseases and conditions considered in the differential diagnosis
| Celiac disease and malabsorption |
| Lactose intolerance, fructose intolerance |
| Inflammatory bowel disease |
| Lymphocytic and collagenous colitis |
| Whipple's disease |
| Colonic cancer |
| Enteric infections |
| Metabolism disorders (e.g., thyroid, diabetes, etc.) |
| Food allergy and intolerance |
| Endometriosis |
| SIBO |
| Neuroendocrine tumors |
| Drugs |
SIBO: Small intestinal bacterial overgrowth.
Unfortunately, there are no available biological markers that clearly identify IBS patients.
Some recent studies have examined faecal lactoferrin and calprotectin, which seem quite suitable to differentiate between infectious bursal disease and IBS but are not able to provide a certain diagnosis of IBS[79,80].
Recent studies have investigated some biomarkers involved in the pathophysiology of IBS[45,81]. A recent systematic review and meta-analysis examined the placebo response rate in treatment trials for IBS and demonstrated a high placebo response[82].
In the case of a patient with IBS-like chronic recurring abdominal symptoms, the presence of alarming symptoms should first be assessed[68,69,83,84]. In the presence of alarming symptoms, further investigation should be undertaken. On the contrary, in the case of Rome III criteria positivity and in the absence of alarm symptoms, possible comorbidities (which are part of the IBS management) should be considered. Serological screening for celiac disease and a few basal blood tests have to be performed; if a negative result is returned, it is usually sufficient to reassure the patient and to offer advice on drug therapies, lifestyle habits and diet. A check-up after 8-12 wk should be offered, and in cases with sustained improvement, the patient will enter into a follow-up program (Figure 1).
Figure 1.
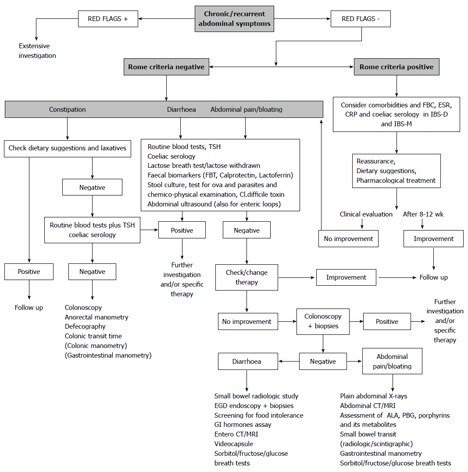
Diagnostic-therapeutic algorithm in a patient with abdominal symptoms possibly related to irritable bowel syndrome. FBT: Faecal blood test; FBC: Full blood count; ESR: Erythrocyte sedimentation rate; CRP: C-reactive protein; PBG: Porphobilinogen; IBS: Irritable bowel syndrome; CT: Computed tomography; MRI: Magnetic resonance imaging.
In the case of a patient with symptoms in any way compatible with irritable bowel syndrome but that did not satisfy the Rome criteria, or in the case of a patient with a poor response to the therapy, depending on the prevailing symptoms (constipation, diarrhoea, abdominal pain/bloating), different options should be considered (Figure 1).
In the case of constipation, dietary habits and behaviours, as well as the use of laxatives, should be checked. In the case of the ineffectiveness of these measures, if not already performed, an assessment of the thyroid function, routine blood tests and screening for celiac disease are recommended. In the case of diarrhoea and abdominal pain/distention, lactose breath test (LBT) (or simply lactose withdrawal), a faecal blood test, faecal Calprotectin or Lactoferrin, stool culture, test for ova and parasites, a chemico-physical examination to test for Clostridium difficile toxins and an abdominal ultrasound aimed at studying the enteric loops should be considered.
If signs of a specific disease emerge from the investigation or from specific treatments, further investigation should be initiated. In the case of a negative outcome, it will become mandatory to proceed to the next steps, as follows (Figure 1): (1) in the case of constipation, the possibility arises of performing a colonoscopy, anorectal manometry, defecography, intestinal transit time and, in carefully selected cases, colonic and gastrojejunal manometry; (2) in the case of diarrhoea and abdominal pain, it will become appropriate to check and eventually change the patients’ drugs; (3) in the case of a failed colonoscopy, biopsies may be useful; and (4) in the case of a negative outcome of a colonoscopy, the further investigations reported in Figure 1 should be considered.
Still, it is mandatory to emphasise that none of these investigations, even those that are costly and unusual, should be performed to achieve the diagnosis of IBS, which is essentially based on the Rome III criteria, as reported above. On the contrary, these tools are to be taken into account only in a patient with abdominal symptoms that are IBS-like but Rome III criteria-negative or -equivocal. They may also be used in IBS patients with very severe symptoms that require a careful reassessment of the clinical situation.
In IBS, the follow up should be tailored to the patient, because the disease is characterised by variable remissions and relapses, with symptoms waxing and waning over time, often oddly and sometimes in coincidence with stressful events, anxiety, the intake of certain foods, etc. IBS patients usually tend to avoid fixed controls, although, at least at the beginning, a clinical visit 2-3 mo after the diagnosis is advised to assess the patient’s adherence to therapy and the dietary and behavioural recommendations.
The aim will be to help IBS patients perceive their symptoms as part of a chronic, intermittent disorder, learning to live with them. Thus, these patients can re-join that “silent majority” of IBS patients who perceive her/his symptoms as no more than a nuisance and do not seek further special care, doctor visits, or additional diagnostic tests.
THERAPEUTIC PERSPECTIVES
Treatment strategies for IBS are based on the nature and severity of the symptoms, the degree of functional impairment of the bowel habits, and the presence of psychosocial comorbidity. In general, milder symptoms relate primarily to visceral hypersensitivity and are commonly treated symptomatically, with pharmacological agents directed at the gut. However, more severe symptoms are associated with greater levels of psychosocial problems and often require psychological and antidepressant medications.
There is limited evidence for the efficacy, safety and tolerability of the therapies currently available for the treatment of IBS. Overall, there is a limited availability of pharmacological agents licensed specifically for the treatment of IBS subtypes, and new agents are eagerly awaited. In any case, it is difficult to achieve a significant therapeutic improvement in global IBS symptoms[64,69,71,85,86].
There is some evidence for improvements in individual IBS symptoms with the use of antidiarrhoeals, antispasmodics, bulking agents, laxatives, tricyclic antidepressants and behavioural therapy. Despite evidence that some pharmaceutical agents benefit the treatment of IBS in the short term, there is no medical intervention that has been proven to alter the long-term natural history of this condition. Further, there is no agreement on a gold-standard for the treatment of IBS. Finally, in functional GI disorders, in which the trial endpoints are likely to be less tangible than organic conditions, the placebo response rate may be very high (over 40%)[82]. Table 4 summarises the various drug categories and their relationships with individual IBS symptoms.
Table 4.
Indication of pharmacological agents in individual irritable bowel syndrome symptoms
| Constipation | Diarrhoea | Pain |
| Soluble fibre | Opioid agents | Antispasmodics |
| Osmotic Laxative | 5-HT3 antagonists | Peppermint oil |
| 5-HT4 agonists | Probiotics | Serotoninergic drugs |
| Secretagogues | Antibiotics | Antidepressants |
| Probiotics | Mesalazine | Herbal therapy |
| SSRI | Colestyramine | Acupuncture |
| Tricyclic antidepressants |
SSRI: Selective serotonin reuptake inhibitors.
Education and reassurance
A strong physician-patient relationship should be the foundation for effective treatment and realistic expectations. Responding to all patient concerns and questions and spending time in the clinical visits validate their condition. A reassurance-based approach permits the patient to understand and accept his or her affliction and to participate in a care strategy. Using this approach, a decrease in the number of health care visits, a reduction in symptoms, and improved patient satisfaction can be easier obtained.
Diet
Patients with IBS commonly believe that specific dietary products contribute to their symptoms of abdominal discomfort, bloating, or alterations of bowel habits. The truth is that no specific food is likely implicated, as true food allergies and intolerances are rare. In many cases, IBS patients have an exaggerated gastric-colic reflex after eating certain foods.
Patients can associate with their complaints the ingestion of certain foods, such as fatty foods, caffeine, alcoholic beverages, carbonated foods, or gas-producing foods. Specifically, symptoms can be related to FODMAPs, such as fructans, galactans, lactose, fructose, sorbitol, xylitol, and mannitol[87]. Studies supporting this are limited and demonstrate a partial improvement in patients after the restriction of these foods. Otherwise, a lactose-restricted diet does not seem to produce a clear clinical benefit in IBS. Beyond this, recent evidence has shown that lactose intolerance was equally prevalent among IBS patients and the general population[64]. Finally, a recent study showed that patients with IBS but without celiac disease may reach satisfactory symptom control with a gluten-free diet but may suffer a symptom relapse after a gluten rechallenge[61]. Only a double-blind gluten challenge can discriminate between IBS and gluten-sensitivity patients. In any case, some care should be taken to avoid an unnecessarily restrictive diet with potentially serious nutritional consequences.
Fibre and bulking agents
Most physicians recommend the use of dietary fibre and bulking agents to regularise bowel function and to reduce meteorism and pain in patients with IBS. The quality of the evidence supporting this recommendation, however, is poor. Some randomised placebo controlled trials have compared the effectiveness of increasing the dietary content of soluble fibre (psyllium and ispaghula) or insoluble fibre (bran) in patients with IBS and constipation. There is some evidence that patients taking psyllium have significant symptom relief, whereas bran shows no clinical benefit and actually may worsen symptoms in many cases[64,69,71,73,85,86,88].
Antispasmodic agents
The rationale for using antispasmodic agents is to attenuate the postprandial abdominal pain seen in patients with IBS. The mechanisms of action of different antispasmodics can be divided broadly into those that directly affect the intestinal smooth muscle and those with anticholinergic/antimuscarinic effects[64,69,71,85,86]. The evidence for the effectiveness of these agents is not compelling.
One meta-analysis demonstrated an advantage of antispasmodics over placebo in terms of abdominal pain and distention[88]. Of all of the drugs studied, the most data were available for otilonium, trimebutine, cimetropium, hyoscine, and pinaverium. Trimebutine seemed to have no benefit over placebo in treating IBS, whereas the other four drugs all significantly reduced the risk of persistent symptoms after treatment. The anticholinergic side effects, including constipation, dry mouth, visual disturbances, and urinary retention, can lead to the discontinuation of these medications. Finally, there is evidence for the efficacy of some peppermint oil preparations (which may also act as antispasmodics) in IBS, but few data are available about the long-term results and adverse effects[88].
Anti-constipation agents
The presence or absence of abdominal pain should be more useful than other associated features for characterising IBS-C in comparison with chronic constipation. However, a clear clinical distinction is not always possible in clinical practice.
Traditional laxatives: Consistent with recent reviews, a therapeutic trial of traditional laxatives (i.e., osmotic laxatives, stimulant laxatives), which are effective, safe, and generally inexpensive, should be considered for managing chronic constipation before newer agents (secretagogues, serotonin 5-HT4 receptor agonists) are used[70,88]. In particular, polyethylene glycol (PEG) is more effective than lactulose in increasing stool frequency and improving stool consistency; thus, it is considered the first choice of treatment for chronic constipation[70].
However, no placebo-controlled, randomised study of laxatives in IBS has been published. Laxatives do not show a significant effect in reducing abdominal pain in IBS. A single small sequential study with PEG in adolescents with IBS-C showed an improvement in stool frequency[89].
Serotonin HT4 agonists: 5-HT4 receptor agonists induce fast excitatory postsynaptic potentials in intrinsic neurons, release acetylcholine, and induce mucosal secretion by activating submucosal neurons.
Tegaserod has been approved by the Food and Drug Administration (FDA) for the treatment of IBS-C in women. Tegaserod is also the only 5-HT4 agonist that has been evaluated in an IBS-mixed population and showed an improvement of global symptoms. However, this drug was removed from the market in 2007 because cardiovascular events were found to be more frequent in tegaserod-treated patients than in placebo-treated patients[89,90].
Among the 5-HT4 agonists for chronic constipation, the most evidence in humans is available for prucalopride[70,90]. The European Agency of Medicinal Products approved this medication for chronic constipation in women for whom laxatives fail to provide an adequate relief of their bowel habits. Prucalopride accelerates GI and colonic transit in constipation, but no placebo-controlled studies have been published, and no conclusive clinical evidence is available for IBS patients[90].
Intestinal secretagogues: By stimulating the efflux of ions and water into the intestinal lumen, secretagogues accelerate transit and facilitate defecation. Both lubiprostone and linaclotide increase intestinal chloride secretion by activating channels on the luminal enterocyte surface[90]. Lubiprostone works by activating apical CIC-2 chloride channels and does not affect colonic motor activity in healthy subjects. It is approved by the FDA for the treatment of women with IBS-C[91,92]. Linaclotide is a guanylyl cyclase C agonist that accelerates colonic transit in patients with IBS-C and chronic constipation[93]. In a recent randomised double-blind trial, linaclotide was shown to improve abdominal pain and discomfort in IBS-C, compared with placebo, over 12 and 26 wk[94]. In the same trial, diarrhoea was the most common adverse effect (19%), although few patients (5.7%) discontinued the drug as a result of this symptom. As of 2012, linaclotide is approved both by the FDA and also by the European Agency for the treatment of IBS-C.
Antidiarrhoeal agents
Opioid analogues: The opioid analogues loperamide and diphenoxylate stimulate inhibitory presynaptic receptors in the enteric nervous system, resulting in the inhibition of peristalsis and secretion. Loperamide has been shown to be effective in decreasing stool frequency and improving stool consistency across all studies[64,69,71,85,95], although it provided no significant improvement in global IBS symptoms (in particular, abdominal pain and distension) compared with placebo.
The simultaneous μ opioid agonist and δ opioid antagonist eluxadoline could reduce abdominal pain and diarrhoea in patients with IBS-D, compared with placebo, in a phase 2 study awaiting publication[96].
Serotonin HT3 antagonists: The 5-HT3 receptor antagonists have been studied in IBS-D because they slow GI transit and decrease discomfort during the distension of the colon[64,69,71,85,86]. Ondansetron is the only 5-HT3 receptor antagonist available in Europe and is licensed as an antiemetic, although it is not approved for use as a treatment for IBS[86]. The selective 5-HT3 receptor antagonist alosetron was currently indicated for the treatment of women with severe IBS-D who had chronic symptoms of IBS[64,69,86,97].
Although it was originally approved by the FDA in 2000, alosetron was withdrawn from the market following reports of serious complications, including constipation, ischemic colitis, and bowel perforation, being associated with its use. Some evidence is available regarding other 5-HT3 antagonists, such as cilansetron and ramosetron. In a recent double-blind randomised trial of 539 IBS-D patients, a positive response to ramosetron treatment was reported compared to patients receiving a placebo[98].
Bile acid binder: Some studies have indicated that a significant number of IBS-D patients can have mild to severe bile acid malabsorption. Several studies have shown a dose-response relationship between the severity of malabsorption and treatment with colestyramine, a bile acid binder[99].
Mesalazine: Mesalazine has intestinal anti-inflammatory properties, including cyclooxygenase and prostaglandin inhibition. A recent study showed that Mesalazine can reduce key symptoms of postinfectious IBS and noninfective IBS-D[100]. The results of an ongoing randomised trial of mesalazine in a group of IBS-D patients will be soon available[101].
Antibiotics and probiotics
Treatments aimed at altering or modifying the gut microbiota, including antibiotics and probiotics, have been the focus of a large number of recent studies on IBS patients[5,97,102,103].
Rifaximin is a semi-synthetic derivative of rifamycin with an additional benzimidazole ring that prevents its systemic absorption. A number of recent clinical trials have evaluated the efficacy and safety of rifaximin in IBS patients (generally IBS-D). A recent systematic review and a meta-analysis[102,103] found rifaximin to be more efficacious than placebo for global IBS symptom improvement. The most common adverse events with rifaximin were headache, upper respiratory infection, diarrhoea, and abdominal pain. Serious side effects, however, were rare, and their prevalences were similar between rifaximin and placebo. Few data are available regarding other antibiotics. A subanalysis of a double-blind, randomised, placebo-controlled trial demonstrated that treatment with neomycin improved global symptoms in individuals with IBS-C compared with placebo[103].
Probiotics have demonstrated benefits for some symptoms, notably bloating and flatulence, and involve a variety of probiotic agents, including lactobacilli, bifidobacteria and streptococcus. Lactobacilli alone had no impact on symptoms, whereas probiotic combinations improved symptoms in IBS patients. Furthermore, there was a positive trend indicating that bifidobacteria improves IBS symptoms[71,85,86,96]. In a recent systematic review[104], probiotics appeared to be efficacious for IBS, but the magnitude of their benefit and the most effective species have not yet been completely established. Finally, probiotics have no serious side effects, and there is no significant difference in the observed adverse events between probiotics and placebo.
Psychological therapies
Among patients with IBS, the majority have anxiety, depression, or features of somatisation. Good patient compliance is necessary to achieve a successful clinical result after a psychotherapeutic approach or after the administration of antidepressants.
Psychotherapy: Among various psychological therapies, there is evidence for a benefit from cognitive behavioural therapy, dynamic psychotherapy, and hypnotherapy, but not from relaxation therapy[105-107]. The abnormal processing and enhanced perception of visceral stimuli in IBS can be normalised by psychological interventions. Psychotherapy is particularly successful in patients who reported a history of sexual abuse. Psychological therapies are not documented to have any serious adverse effects.
Tricyclic antidepressants: Tricyclic antidepressants (TCAs) are drugs with anticholinergic and non-selective serotonin reuptake inhibitor effects. Antidepressants could theoretically provide a benefit in IBS by both central and peripheral mechanisms[64,71,85,86,97]. Five tricyclic agents have been studied formally (amitriptyline, trimipramine, desipramine, clomipramine, and doxepin), and the effects of these agents are primarily related to pain. It has been suggested that patients with IBS-D obtain the greatest benefit from this approach[67]. The side effects of constipation, dry mouth, drowsiness, and fatigue occur in over one-third of IBS patients treated with TCAs, which often precludes good patient compliance.
Selective serotonin reuptake inhibitors, antidepressants: Physicians often prefer selective serotonin reuptake inhibitors (SSRIs) over TCAs because of their lower side-effect profiles. SSRIs, such as paroxetine and fluoxetine, can accelerate whole gut transit and are considered potentially effective in the treatment of IBS-C. A large trial[71] showed that a standard dose of an SSRI antidepressant led to a significant improvement in the health-related quality of life in patients with IBS, but no significant effects were observed in bowel habits or pain. However, in a double-blind randomised trial, fluoxetine was effective in decreasing global symptoms in the short-term therapy of a group of IBS-C patients[104].
Alternative approaches
Chinese herbal preparations have also been the subject of several trials[108]. By combining the effects of Iberis amara on smooth muscle tone with the spasmolytic effects of other plants, Iberogast, a popular combination of nine herbal plants, exerts a dual action on smooth muscle, stimulating or spasmolytic, depending on functional baseline conditions. These plant preparations have been shown to improve overall IBS scores and abdominal pain, but it is unclear which component is the active ingredient. A longer study of 16 wk with Chinese herbal preparations reported significant symptom improvement[109]. No conclusive data are available regarding any toxicity, especially regarding liver failure, of any Chinese herbal mixture.
Another popular alternative treatment concerns the use of acupuncture in IBS. A Cochrane review of six trials with a median sample size of 54 found insufficient evidence to determine whether acupuncture is an effective treatment for IBS[110]. In a recent open randomised trial, acupuncture for IBS provided an additional benefit over the usual care alone in a primary care experience[111].
Further studies are needed before any final recommendations on acupuncture or herbal therapy can be made.
CONCLUSION
Even though there is some evidence that changes in the digestive motility and secretion, visceral hypersensitivity, abnormalities of enteroendocrine and immune systems, genetic factors, infections, alterations of the intestinal microbiota and inflammation could play a role in IBS, its pathogenesis remains only partially understood. Thus, in clinical practice, its management is quite difficult. Because no biological markers are available, diagnoses can be made only on the basis of the symptoms described by the Rome III criteria, for example. Unfortunately, many physicians do not use these criteria in their clinical practice and instead, driven by their own concerns or the concern of their patients, often prescribe many unnecessary diagnostic tests.
Furthermore, IBS therapy is far from satisfactory. The cornerstone for any effective treatment strategy should be a solid patient-physician relationship; indeed, this relationship should be individualised for each patient. To achieve this goal, the use of combination drug therapies may be suggested. The data reviewed here indicate that there is limited evidence to support the individual efficacy of any of the agents currently available.
In conclusion, the pathogenesis, diagnosis and treatment of IBS remain subjects of much ongoing research. Further well-structured studies are needed to improve our knowledge about IBS and its management.
Footnotes
P- Reviewers: Chen JX, Grundmann O, Guglielmetti F, Pasechnikov V, Sharara A, Soares RLS S- Editor: Ma YJ L- Editor: A E- Editor: Ma S
References
- 1.Bellini M, Tosetti C, Costa F, Biagi S, Stasi C, Del Punta A, Monicelli P, Mumolo MG, Ricchiuti A, Bruzzi P, et al. The general practitioner’s approach to irritable bowel syndrome: from intention to practice. Dig Liver Dis. 2005;37:934–939. doi: 10.1016/j.dld.2005.06.011. [DOI] [PubMed] [Google Scholar]
- 2.Videlock EJ, Chang L. Irritable bowel syndrome: current approach to symptoms, evaluation, and treatment. Gastroenterol Clin North Am. 2007;36:665–685, x. doi: 10.1016/j.gtc.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 3.Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders. Gastroenterology. 2006;130:1480–1491. doi: 10.1053/j.gastro.2005.11.061. [DOI] [PubMed] [Google Scholar]
- 4.Burbige EJ. Irritable bowel syndrome: diagnostic approaches in clinical practice. Clin Exp Gastroenterol. 2010;3:127–137. doi: 10.2147/CEG.S12596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frissora CL, Koch KL. Symptom overlap and comorbidity of irritable bowel syndrome with other conditions. Curr Gastroenterol Rep. 2005;7:264–271. doi: 10.1007/s11894-005-0018-9. [DOI] [PubMed] [Google Scholar]
- 6.Whitehead WE, Palsson O, Jones KR. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: what are the causes and implications? Gastroenterology. 2002;122:1140–1156. doi: 10.1053/gast.2002.32392. [DOI] [PubMed] [Google Scholar]
- 7.Aaron LA, Burke MM, Buchwald D. Overlapping conditions among patients with chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221–227. doi: 10.1001/archinte.160.2.221. [DOI] [PubMed] [Google Scholar]
- 8.Nellesen D, Chawla A, Oh DL, Weissman T, Lavins BJ, Murray CW. Comorbidities in patients with irritable bowel syndrome with constipation or chronic idiopathic constipation: a review of the literature from the past decade. Postgrad Med. 2013;125:40–50. doi: 10.3810/pgm.2013.03.2640. [DOI] [PubMed] [Google Scholar]
- 9.Spiller R. Clinical update: irritable bowel syndrome. Lancet. 2007;369:1586–1588. doi: 10.1016/S0140-6736(07)60726-0. [DOI] [PubMed] [Google Scholar]
- 10.Barbara G, Cremon C, Carini G, Bellacosa L, Zecchi L, De Giorgio R, Corinaldesi R, Stanghellini V. The immune system in irritable bowel syndrome. J Neurogastroenterol Motil. 2011;17:349–359. doi: 10.5056/jnm.2011.17.4.349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maxion-Bergemann S, Thielecke F, Abel F, Bergemann R. Costs of irritable bowel syndrome in the UK and US. Pharmacoeconomics. 2006;24:21–37. doi: 10.2165/00019053-200624010-00002. [DOI] [PubMed] [Google Scholar]
- 12.Nyrop KA, Palsson OS, Levy RL, Von Korff M, Feld AD, Turner MJ, Whitehead WE. Costs of health care for irritable bowel syndrome, chronic constipation, functional diarrhoea and functional abdominal pain. Aliment Pharmacol Ther. 2007;26:237–248. doi: 10.1111/j.1365-2036.2007.03370.x. [DOI] [PubMed] [Google Scholar]
- 13.Whitehead WE, Palsson OS, Levy RR, Feld AD, Turner M, Von Korff M. Comorbidity in irritable bowel syndrome. Am J Gastroenterol. 2007;102:2767–2776. doi: 10.1111/j.1572-0241.2007.01540.x. [DOI] [PubMed] [Google Scholar]
- 14.De Ponti F. Drug development for the irritable bowel syndrome: current challenges and future perspectives. Front Pharmacol. 2013;4:7. doi: 10.3389/fphar.2013.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.El-Salhy M. Irritable bowel syndrome: diagnosis and pathogenesis. World J Gastroenterol. 2012;18:5151–5163. doi: 10.3748/wjg.v18.i37.5151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Whorwell PJ, McCallum M, Creed FH, Roberts CT. Non-colonic features of irritable bowel syndrome. Gut. 1986;27:37–40. doi: 10.1136/gut.27.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kalantar JS, Locke GR, Zinsmeister AR, Beighley CM, Talley NJ. Familial aggregation of irritable bowel syndrome: a prospective study. Gut. 2003;52:1703–1707. doi: 10.1136/gut.52.12.1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bengtson MB, Rønning T, Vatn MH, Harris JR. Irritable bowel syndrome in twins: genes and environment. Gut. 2006;55:1754–1759. doi: 10.1136/gut.2006.097287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mohammed I, Cherkas LF, Riley SA, Spector TD, Trudgill NJ. Genetic influences in irritable bowel syndrome: a twin study. Am J Gastroenterol. 2005;100:1340–1344. doi: 10.1111/j.1572-0241.2005.41700.x. [DOI] [PubMed] [Google Scholar]
- 20.Park MI, Camilleri M. Genetics and genotypes in irritable bowel syndrome: implications for diagnosis and treatment. Gastroenterol Clin North Am. 2005;34:305–317. doi: 10.1016/j.gtc.2005.02.009. [DOI] [PubMed] [Google Scholar]
- 21.Barkhordari E, Rezaei N, Ansaripour B, Larki P, Alighardashi M, Ahmadi-Ashtiani HR, Mahmoudi M, Keramati MR, Habibollahi P, Bashashati M, et al. Proinflammatory cytokine gene polymorphisms in irritable bowel syndrome. J Clin Immunol. 2010;30:74–79. doi: 10.1007/s10875-009-9342-4. [DOI] [PubMed] [Google Scholar]
- 22.Gonsalkorale WM, Perrey C, Pravica V, Whorwell PJ, Hutchinson IV. Interleukin 10 genotypes in irritable bowel syndrome: evidence for an inflammatory component? Gut. 2003;52:91–93. doi: 10.1136/gut.52.1.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Van Kerkhoven LA, Laheij RJ, Jansen JB. Meta-analysis: a functional polymorphism in the gene encoding for activity of the serotonin transporter protein is not associated with the irritable bowel syndrome. Aliment Pharmacol Ther. 2007;26:979–986. doi: 10.1111/j.1365-2036.2007.03453.x. [DOI] [PubMed] [Google Scholar]
- 24.Colucci R, Gambaccini D, Ghisu N, Rossi G, Costa F, Tuccori M, De Bortoli N, Fornai M, Antonioli L, Ricchiuti A, et al. Influence of the serotonin transporter 5HTTLPR polymorphism on symptom severity in irritable bowel syndrome. PLoS One. 2013;8:e54831. doi: 10.1371/journal.pone.0054831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Palsson OS, Drossman DA. Psychiatric and psychological dysfunction in irritable bowel syndrome and the role of psychological treatments. Gastroenterol Clin North Am. 2005;34:281–303. doi: 10.1016/j.gtc.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 26.Halpert A, Drossman D. Biopsychosocial issues in irritable bowel syndrome. J Clin Gastroenterol. 2005;39:665–669. doi: 10.1097/01.mcg.0000174024.81096.44. [DOI] [PubMed] [Google Scholar]
- 27.Drossman DA, Lowman BC. Irritable bowel syndrome: epidemiology, diagnosis and treatment. Clin Gastroenterol. 1985;14:559–573. [PubMed] [Google Scholar]
- 28.Evans PR, Bennett EJ, Bak YT, Tennant CC, Kellow JE. Jejunal sensorimotor dysfunction in irritable bowel syndrome: clinical and psychosocial features. Gastroenterology. 1996;110:393–404. doi: 10.1053/gast.1996.v110.pm8566585. [DOI] [PubMed] [Google Scholar]
- 29.Kellow JE. Pathophysiology and management of irritable bowel syndrome. Korean J Intern Med. 2001;16:137–146. doi: 10.3904/kjim.2001.16.3.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vassallo MJ, Camilleri M, Phillips SF, Steadman CJ, Talley NJ, Hanson RB, Haddad AC. Colonic tone and motility in patients with irritable bowel syndrome. Mayo Clin Proc. 1992;67:725–731. doi: 10.1016/s0025-6196(12)60796-4. [DOI] [PubMed] [Google Scholar]
- 31.Stivland T, Camilleri M, Vassallo M, Proano M, Rath D, Brown M, Thomforde G, Pemberton J, Phillips S. Scintigraphic measurement of regional gut transit in idiopathic constipation. Gastroenterology. 1991;101:107–115. doi: 10.1016/0016-5085(91)90466-x. [DOI] [PubMed] [Google Scholar]
- 32.Manabe N, Wong BS, Camilleri M, Burton D, McKinzie S, Zinsmeister AR. Lower functional gastrointestinal disorders: evidence of abnormal colonic transit in a 287 patient cohort. Neurogastroenterol Motil. 2010;22:293–e82. doi: 10.1111/j.1365-2982.2009.01442.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Törnblom H, Van Oudenhove L, Sadik R, Abrahamsson H, Tack J, Simrén M. Colonic transit time and IBS symptoms: what’s the link? Am J Gastroenterol. 2012;107:754–760. doi: 10.1038/ajg.2012.5. [DOI] [PubMed] [Google Scholar]
- 34.Salvioli B, Serra J, Azpiroz F, Lorenzo C, Aguade S, Castell J, Malagelada JR. Origin of gas retention and symptoms in patients with bloating. Gastroenterology. 2005;128:574–579. doi: 10.1053/j.gastro.2004.12.047. [DOI] [PubMed] [Google Scholar]
- 35.Di Stefano M, Miceli E, Missanelli A, Mazzocchi S, Corazza GR. Meal induced rectosigmoid tone modification: a low caloric meal accurately separates functional and organic gastrointestinal disease patients. Gut. 2006;55:1409–1414. doi: 10.1136/gut.2005.076323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ritchie J. Pain from distension of the pelvic colon by inflating a balloon in the irritable colon syndrome. Gut. 1973;14:125–132. doi: 10.1136/gut.14.2.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Camilleri M, Lasch K, Zhou W. Irritable bowel syndrome: methods, mechanisms, and pathophysiology. The confluence of increased permeability, inflammation, and pain in irritable bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2012;303:G775–G785. doi: 10.1152/ajpgi.00155.2012. [DOI] [PubMed] [Google Scholar]
- 38.Mayer EA. The neurobiology of stress and gastrointestinal disease. Gut. 2000;47:861–869. doi: 10.1136/gut.47.6.861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Verne GN, Himes NC, Robinson ME, Gopinath KS, Briggs RW, Crosson B, Price DD. Central representation of visceral and cutaneous hypersensitivity in the irritable bowel syndrome. Pain. 2003;103:99–110. doi: 10.1016/s0304-3959(02)00416-5. [DOI] [PubMed] [Google Scholar]
- 40.Delvaux M, Denis P, Allemand H. Sexual abuse is more frequently reported by IBS patients than by patients with organic digestive diseases or controls. Results of a multicentre inquiry. French Club of Digestive Motility. Eur J Gastroenterol Hepatol. 1997;9:345–352. doi: 10.1097/00042737-199704000-00006. [DOI] [PubMed] [Google Scholar]
- 41.Stasi C, Rosselli M, Bellini M, Laffi G, Milani S. Altered neuro-endocrine-immune pathways in the irritable bowel syndrome: the top-down and the bottom-up model. J Gastroenterol. 2012;47:1177–1185. doi: 10.1007/s00535-012-0627-7. [DOI] [PubMed] [Google Scholar]
- 42.Matricon J, Meleine M, Gelot A, Piche T, Dapoigny M, Muller E, Ardid D. Review article: Associations between immune activation, intestinal permeability and the irritable bowel syndrome. Aliment Pharmacol Ther. 2012;36:1009–1031. doi: 10.1111/apt.12080. [DOI] [PubMed] [Google Scholar]
- 43.Fukudo S, Nomura T, Hongo M. Impact of corticotropin-releasing hormone on gastrointestinal motility and adrenocorticotropic hormone in normal controls and patients with irritable bowel syndrome. Gut. 1998;42:845–849. doi: 10.1136/gut.42.6.845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dinan TG, Quigley EM, Ahmed SM, Scully P, O’Brien S, O’Mahony L, O’Mahony S, Shanahan F, Keeling PW. Hypothalamic-pituitary-gut axis dysregulation in irritable bowel syndrome: plasma cytokines as a potential biomarker? Gastroenterology. 2006;130:304–311. doi: 10.1053/j.gastro.2005.11.033. [DOI] [PubMed] [Google Scholar]
- 45.Stasi C, Bellini M, Costa F, Mumolo MG, Ricchiuti A, Grosso M, Duranti E, Metelli MR, Gambaccini D, Bianchi L, et al. Neuroendocrine markers and psychological features in patients with irritable bowel syndrome. Int J Colorectal Dis. 2013;28:1203–1208. doi: 10.1007/s00384-013-1646-4. [DOI] [PubMed] [Google Scholar]
- 46.Mertz H. Role of the brain and sensory pathways in gastrointestinal sensory disorders in humans. Gut. 2002;51 Suppl 1:i29–i33. doi: 10.1136/gut.51.suppl_1.i29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mertz H. Review article: visceral hypersensitivity. Aliment Pharmacol Ther. 2003;17:623–633. doi: 10.1046/j.1365-2036.2003.01447.x. [DOI] [PubMed] [Google Scholar]
- 48.Naliboff BD, Berman S, Suyenobu B, Labus JS, Chang L, Stains J, Mandelkern MA, Mayer EA. Longitudinal change in perceptual and brain activation response to visceral stimuli in irritable bowel syndrome patients. Gastroenterology. 2006;131:352–365. doi: 10.1053/j.gastro.2006.05.014. [DOI] [PubMed] [Google Scholar]
- 49.Bellini M, Alduini P, Bassotti G, Bove A, Bocchini R, Sormani MP, Bruzzi P, Pucciani F; Italian Constipation Study Group. Self-perceived normality in defecation habits. Dig Liver Dis. 2006;38:103–108. doi: 10.1016/j.dld.2005.09.022. [DOI] [PubMed] [Google Scholar]
- 50.Collins SM. Is the irritable gut an inflamed gut? Scand J Gastroenterol Suppl. 1992;192:102–105. doi: 10.3109/00365529209095988. [DOI] [PubMed] [Google Scholar]
- 51.Barbara G, De Giorgio R, Stanghellini V, Cremon C, Salvioli B, Corinaldesi R. New pathophysiological mechanisms in irritable bowel syndrome. Aliment Pharmacol Ther. 2004;20 Suppl 2:1–9. doi: 10.1111/j.1365-2036.2004.02036.x. [DOI] [PubMed] [Google Scholar]
- 52.Chadwick VS, Chen W, Shu D, Paulus B, Bethwaite P, Tie A, Wilson I. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology. 2002;122:1778–1783. doi: 10.1053/gast.2002.33579. [DOI] [PubMed] [Google Scholar]
- 53.Barbara G, Stanghellini V, De Giorgio R, Cremon C, Cottrell GS, Santini D, Pasquinelli G, Morselli-Labate AM, Grady EF, Bunnett NW, et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004;126:693–702. doi: 10.1053/j.gastro.2003.11.055. [DOI] [PubMed] [Google Scholar]
- 54.Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, Stanghellini V, Corinaldesi R, Barbara G. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392–400. doi: 10.1038/ajg.2008.94. [DOI] [PubMed] [Google Scholar]
- 55.Barbara G, Cremon C, Pallotti F, De Giorgio R, Stanghellini V, Corinaldesi R. Postinfectious irritable bowel syndrome. J Pediatr Gastroenterol Nutr. 2009;48 Suppl 2:S95–S97. doi: 10.1097/MPG.0b013e3181a15e2e. [DOI] [PubMed] [Google Scholar]
- 56.Begtrup LM, de Muckadell OB, Kjeldsen J, Christensen RD, Jarbøl DE. Long-term treatment with probiotics in primary care patients with irritable bowel syndrome--a randomised, double-blind, placebo controlled trial. Scand J Gastroenterol. 2013;48:1127–1135. doi: 10.3109/00365521.2013.825314. [DOI] [PubMed] [Google Scholar]
- 57.Ohman L, Simrén M. Intestinal microbiota and its role in irritable bowel syndrome (IBS) Curr Gastroenterol Rep. 2013;15:323. doi: 10.1007/s11894-013-0323-7. [DOI] [PubMed] [Google Scholar]
- 58.Simrén M, Barbara G, Flint HJ, Spiegel BM, Spiller RC, Vanner S, Verdu EF, Whorwell PJ, Zoetendal EG; Rome Foundation Committee. Intestinal microbiota in functional bowel disorders: a Rome foundation report. Gut. 2013;62:159–176. doi: 10.1136/gutjnl-2012-302167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Atkinson W, Sheldon TA, Shaath N, Whorwell PJ. Food elimination based on IgG antibodies in irritable bowel syndrome: a randomised controlled trial. Gut. 2004;53:1459–1464. doi: 10.1136/gut.2003.037697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zar S, Benson MJ, Kumar D. Food-specific serum IgG4 and IgE titers to common food antigens in irritable bowel syndrome. Am J Gastroenterol. 2005;100:1550–1557. doi: 10.1111/j.1572-0241.2005.41348.x. [DOI] [PubMed] [Google Scholar]
- 61.Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology. 2013;145:320–8.e1-320-8.e3. doi: 10.1053/j.gastro.2013.04.051. [DOI] [PubMed] [Google Scholar]
- 62.Barrett JS, Gibson PR. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs) and nonallergic food intolerance: FODMAPs or food chemicals? Therap Adv Gastroenterol. 2012;5:261–268. doi: 10.1177/1756283X11436241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Quigley EM, Abdel-Hamid H, Barbara G, Bhatia SJ, Boeckxstaens G, De Giorgio R, Delvaux M, Drossman DA, Foxx-Orenstein AE, Guarner F, et al. A global perspective on irritable bowel syndrome: a consensus statement of the World Gastroenterology Organisation Summit Task Force on irritable bowel syndrome. J Clin Gastroenterol. 2012;46:356–366. doi: 10.1097/MCG.0b013e318247157c. [DOI] [PubMed] [Google Scholar]
- 64.Spiller R, Aziz Q, Creed F, Emmanuel A, Houghton L, Hungin P, Jones R, Kumar D, Rubin G, Trudgill N, Whorwell P; Clinical Services Committee of The British Society of Gastroenterology. Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut. 2007;56:1770–1798. doi: 10.1136/gut.2007.119446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32:920–924. doi: 10.3109/00365529709011203. [DOI] [PubMed] [Google Scholar]
- 66.Sperber AD, Drossman DA. Irritable bowel syndrome: a multidimensional disorder cannot be understood or treated from a unidimensional perspective. Therap Adv Gastroenterol. 2012;5:387–393. doi: 10.1177/1756283X12460420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bellini M, Gemignani A, Gambaccini D, Toti S, Menicucci D, Stasi C, Costa F, Mumolo MG, Ricchiuti A, Bedini R, et al. Evaluation of latent links between irritable bowel syndrome and sleep quality. World J Gastroenterol. 2011;17:5089–5096. doi: 10.3748/wjg.v17.i46.5089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.World Gastroenterology Organisation. Global guideline. Irritable bowel syndrome: A global perspective; 2009. Available from: http://www.worldgastroenterology.org/irritable-bowel-syndrome.html. [Google Scholar]
- 69.American College of Gastroenterology Task Force on Irritable Bowel Syndrome; Brandt LJ, Chey WD, Foxx-Orenstein AE, Schiller LR, Schoenfeld PS, Spiegel BM, Talley NJ, Quigley EM. An evidence-based position statement on the management of irritable bowel syndrome. Am J Gastroenterol. 2009;104 Suppl 1:S1–35. doi: 10.1038/ajg.2008.122. [DOI] [PubMed] [Google Scholar]
- 70.Bove A, Pucciani F, Bellini M, Battaglia E, Bocchini R, Altomare DF, Dodi G, Sciaudone G, Falletto E, Piloni V, et al. Consensus statement AIGO/SICCR: diagnosis and treatment of chronic constipation and obstructed defecation (part I: diagnosis) World J Gastroenterol. 2012;18:1555–1564. doi: 10.3748/wjg.v18.i14.1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Drossman DA, Camilleri M, Mayer EA, Whitehead WE. AGA technical review on irritable bowel syndrome. Gastroenterology. 2002;123:2108–2131. doi: 10.1053/gast.2002.37095. [DOI] [PubMed] [Google Scholar]
- 72.Wong RK, Drossman DA, Bharucha AE, Rao SS, Wald A, Morris CB, Oxentenko AS, Ravi K, Van Handel DM, Edwards H, et al. The digital rectal examination: a multicenter survey of physicians’ and students’ perceptions and practice patterns. Am J Gastroenterol. 2012;107:1157–1163. doi: 10.1038/ajg.2012.23. [DOI] [PubMed] [Google Scholar]
- 73.Bijkerk CJ, de Wit NJ, Muris JW, Jones RH, Knottnerus JA, Hoes AW. Outcome measures in irritable bowel syndrome: comparison of psychometric and methodological characteristics. Am J Gastroenterol. 2003;98:122–127. doi: 10.1111/j.1572-0241.2003.07158.x. [DOI] [PubMed] [Google Scholar]
- 74.Mangel AW, Hahn BA, Heath AT, Northcutt AR, Kong S, Dukes GE, McSorley D. Adequate relief as an endpoint in clinical trials in irritable bowel syndrome. J Int Med Res. 1998;26:76–81. doi: 10.1177/030006059802600203. [DOI] [PubMed] [Google Scholar]
- 75.Francis CY, Morris J, Whorwell PJ. The irritable bowel severity scoring system: a simple method of monitoring irritable bowel syndrome and its progress. Aliment Pharmacol Ther. 1997;11:395–402. doi: 10.1046/j.1365-2036.1997.142318000.x. [DOI] [PubMed] [Google Scholar]
- 76.Patrick DL, Drossman DA, Frederick IO, DiCesare J, Puder KL. Quality of life in persons with irritable bowel syndrome: development and validation of a new measure. Dig Dis Sci. 1998;43:400–411. doi: 10.1023/a:1018831127942. [DOI] [PubMed] [Google Scholar]
- 77.Wong E, Guyatt GH, Cook DJ, Griffith LE, Irvine EJ. Development of a questionnaire to measure quality of life in patients with irritable bowel syndrome. Eur J Surg Suppl. 1998;(583):50–56. doi: 10.1080/11024159850191247. [DOI] [PubMed] [Google Scholar]
- 78.Cash BD, Rubenstein JH, Young PE, Gentry A, Nojkov B, Lee D, Andrews AH, Dobhan R, Chey WD. The prevalence of celiac disease among patients with nonconstipated irritable bowel syndrome is similar to controls. Gastroenterology. 2011;141:1187–1193. doi: 10.1053/j.gastro.2011.06.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Pavlidis P, Chedgy FJ, Tibble JA. Diagnostic accuracy and clinical application of faecal calprotectin in adult patients presenting with gastrointestinal symptoms in primary care. Scand J Gastroenterol. 2013;48:1048–1054. doi: 10.3109/00365521.2013.816771. [DOI] [PubMed] [Google Scholar]
- 80.Costa F, Mumolo MG, Bellini M, Romano MR, Ceccarelli L, Arpe P, Sterpi C, Marchi S, Maltinti G. Role of faecal calprotectin as non-invasive marker of intestinal inflammation. Dig Liver Dis. 2003;35:642–647. doi: 10.1016/s1590-8658(03)00381-5. [DOI] [PubMed] [Google Scholar]
- 81.Chang L, Adeyemo M, Karagiannides I, Videlock EJ, Bowe C, Shih W, Presson AP, Yuan PQ, Cortina G, Gong H, et al. Serum and colonic mucosal immune markers in irritable bowel syndrome. Am J Gastroenterol. 2012;107:262–272. doi: 10.1038/ajg.2011.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ford AC, Moayyedi P. Meta-analysis: factors affecting placebo response rate in the irritable bowel syndrome. Aliment Pharmacol Ther. 2010;32:144–158. doi: 10.1111/j.1365-2036.2010.04328.x. [DOI] [PubMed] [Google Scholar]
- 83.Costa F, Mumolo MG, Marchi S, Bellini M. Differential diagnosis between functional and organic intestinal disorders: is there a role for non-invasive tests? World J Gastroenterol. 2007;13:219–223. doi: 10.3748/wjg.v13.i2.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Spiller RC, Thompson WG. Bowel disorders. Am J Gastroenterol. 2010;105:775–785. doi: 10.1038/ajg.2010.69. [DOI] [PubMed] [Google Scholar]
- 85.Lesbros-Pantoflickova D, Michetti P, Fried M, Beglinger C, Blum AL. Meta-analysis: The treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 2004;20:1253–1269. doi: 10.1111/j.1365-2036.2004.02267.x. [DOI] [PubMed] [Google Scholar]
- 86.Tack J, Fried M, Houghton LA, Spicak J, Fisher G. Systematic review: the efficacy of treatments for irritable bowel syndrome--a European perspective. Aliment Pharmacol Ther. 2006;24:183–205. doi: 10.1111/j.1365-2036.2006.02938.x. [DOI] [PubMed] [Google Scholar]
- 87.Staudacher HM, Lomer MC, Anderson JL, Barrett JS, Muir JG, Irving PM, Whelan K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J Nutr. 2012;142:1510–1518. doi: 10.3945/jn.112.159285. [DOI] [PubMed] [Google Scholar]
- 88.Ford AC, Talley NJ, Spiegel BM, Foxx-Orenstein AE, Schiller L, Quigley EM, Moayyedi P. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ. 2008;337:a2313. doi: 10.1136/bmj.a2313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Khoshoo V, Armstead C, Landry L. Effect of a laxative with and without tegaserod in adolescents with constipation predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2006;23:191–196. doi: 10.1111/j.1365-2036.2006.02705.x. [DOI] [PubMed] [Google Scholar]
- 90.Bharucha AE, Pemberton JH, Locke GR. American Gastroenterological Association technical review on constipation. Gastroenterology. 2013;144:218–238. doi: 10.1053/j.gastro.2012.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Drossman DA, Chey WD, Johanson JF, Fass R, Scott C, Panas R, Ueno R. Clinical trial: lubiprostone in patients with constipation-associated irritable bowel syndrome--results of two randomized, placebo-controlled studies. Aliment Pharmacol Ther. 2009;29:329–341. doi: 10.1111/j.1365-2036.2008.03881.x. [DOI] [PubMed] [Google Scholar]
- 92.Chey WD, Drossman DA, Johanson JF, Scott C, Panas RM, Ueno R. Safety and patient outcomes with lubiprostone for up to 52 weeks in patients with irritable bowel syndrome with constipation. Aliment Pharmacol Ther. 2012;35:587–599. doi: 10.1111/j.1365-2036.2011.04983.x. [DOI] [PubMed] [Google Scholar]
- 93.Rao S, Lembo AJ, Shiff SJ, Lavins BJ, Currie MG, Jia XD, Shi K, MacDougall JE, Shao JZ, Eng P, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol. 2012;107:1714–1724; quiz p.1725. doi: 10.1038/ajg.2012.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Quigley EM, Tack J, Chey WD, Rao SS, Fortea J, Falques M, Diaz C, Shiff SJ, Currie MG, Johnston JM. Randomised clinical trials: linaclotide phase 3 studies in IBS-C - a prespecified further analysis based on European Medicines Agency-specified endpoints. Aliment Pharmacol Ther. 2013;37:49–61. doi: 10.1111/apt.12123. [DOI] [PubMed] [Google Scholar]
- 95.Lavö B, Stenstam M, Nielsen AL. Loperamide in treatment of irritable bowel syndrome--a double-blind placebo controlled study. Scand J Gastroenterol Suppl. 1987;130:77–80. doi: 10.3109/00365528709091003. [DOI] [PubMed] [Google Scholar]
- 96.Dove LS, Lembo A, Randall CW, Fogel R, Andrae D, Davenport JM, McIntyre G, Almenoff JS, Covington PS. Eluxadoline benefits patients with irritable bowel syndrome with diarrhea in a phase 2 study. Gastroenterology. 2013;145:329–338.e1. doi: 10.1053/j.gastro.2013.04.006. [DOI] [PubMed] [Google Scholar]
- 97.Chang L, Lacy BE, Spiegel BM. An Evidence-based Approach to Therapy in IBS-D: A Case Study Compendium. Gastroenterol Hepatol (N Y) 2010;6:1–12. [PMC free article] [PubMed] [Google Scholar]
- 98.Chiba T, Yamamoto K, Sato S, Suzuki K. Long-term efficacy and safety of ramosetron in the treatment of diarrhea-predominant irritable bowel syndrome. Clin Exp Gastroenterol. 2013;6:123–128. doi: 10.2147/CEG.S32721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wedlake L, A’Hern R, Russell D, Thomas K, Walters JR, Andreyev HJ. Systematic review: the prevalence of idiopathic bile acid malabsorption as diagnosed by SeHCAT scanning in patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2009;30:707–717. doi: 10.1111/j.1365-2036.2009.04081.x. [DOI] [PubMed] [Google Scholar]
- 100.Bafutto M, Almeida JR, Leite NV, Oliveira EC, Gabriel-Neto S, Rezende-Filho J. Treatment of postinfectious irritable bowel syndrome and noninfective irritable bowel syndrome with mesalazine. Arq Gastroenterol. 2011;48:36–40. doi: 10.1590/s0004-28032011000100008. [DOI] [PubMed] [Google Scholar]
- 101.Leighton MP, Lam C, Mehta S, Spiller RC. Efficacy and mode of action of mesalazine in the treatment of diarrhoea-predominant irritable bowel syndrome (IBS-D): study protocol for a randomised controlled trial. Trials. 2013;14:10. doi: 10.1186/1745-6215-14-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Pimentel M, Lembo A, Chey WD, Zakko S, Ringel Y, Yu J, Mareya SM, Shaw AL, Bortey E, Forbes WP; TARGET Study Group. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364:22–32. doi: 10.1056/NEJMoa1004409. [DOI] [PubMed] [Google Scholar]
- 103.Menees SB, Maneerattannaporn M, Kim HM, Chey WD. The efficacy and safety of rifaximin for the irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol. 2012;107:28–35; quiz 36. doi: 10.1038/ajg.2011.355. [DOI] [PubMed] [Google Scholar]
- 104.Moayyedi P, Ford AC, Talley NJ, Cremonini F, Foxx-Orenstein AE, Brandt LJ, Quigley EM. The efficacy of probiotics in the treatment of irritable bowel syndrome: a systematic review. Gut. 2010;59:325–332. doi: 10.1136/gut.2008.167270. [DOI] [PubMed] [Google Scholar]
- 105.Ford AC, Talley NJ, Schoenfeld PS, Quigley EM, Moayyedi P. Efficacy of antidepressants and psychological therapies in irritable bowel syndrome: systematic review and meta-analysis. Gut. 2009;58:367–378. doi: 10.1136/gut.2008.163162. [DOI] [PubMed] [Google Scholar]
- 106.Lindfors P, Unge P, Arvidsson P, Nyhlin H, Björnsson E, Abrahamsson H, Simrén M. Effects of gut-directed hypnotherapy on IBS in different clinical settings-results from two randomized, controlled trials. Am J Gastroenterol. 2012;107:276–285. doi: 10.1038/ajg.2011.340. [DOI] [PubMed] [Google Scholar]
- 107.Lowén MB, Mayer EA, Sjöberg M, Tillisch K, Naliboff B, Labus J, Lundberg P, Ström M, Engström M, Walter SA. Effect of hypnotherapy and educational intervention on brain response to visceral stimulus in the irritable bowel syndrome. Aliment Pharmacol Ther. 2013;37:1184–1197. doi: 10.1111/apt.12319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Madisch A, Holtmann G, Plein K, Hotz J. Treatment of irritable bowel syndrome with herbal preparations: results of a double-blind, randomized, placebo-controlled, multi-centre trial. Aliment Pharmacol Ther. 2004;19:271–279. doi: 10.1111/j.1365-2036.2004.01859.x. [DOI] [PubMed] [Google Scholar]
- 109.Bensoussan A, Talley NJ, Hing M, Menzies R, Guo A, Ngu M. Treatment of irritable bowel syndrome with Chinese herbal medicine: a randomized controlled trial. JAMA. 1998;280:1585–1589. doi: 10.1001/jama.280.18.1585. [DOI] [PubMed] [Google Scholar]
- 110.Lim B, Manheimer E, Lao L, Ziea E, Wisniewski J, Liu J, Berman B. Acupuncture for treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2006;(4):CD005111. doi: 10.1002/14651858.CD005111.pub2. [DOI] [PubMed] [Google Scholar]
- 111.MacPherson H, Tilbrook H, Bland JM, Bloor K, Brabyn S, Cox H, Kang’ombe AR, Man MS, Stuardi T, Torgerson D, et al. Acupuncture for irritable bowel syndrome: primary care based pragmatic randomised controlled trial. BMC Gastroenterol. 2012;12:150. doi: 10.1186/1471-230X-12-150. [DOI] [PMC free article] [PubMed] [Google Scholar]


