Abstract
In a previous study, a synergistic retardation effect of 1,4-cyclohexanediol and 1,2-hexanediol on percutaneous absorption and penetration of metronidazole (MTZ) was discovered. A complex formation between 1,4-cyclohexanediol and 1,2-hexanediol was proposed to be responsible for the observed effect. The objective of this study was to investigate the necessity of hydroxyl group and the ring structure in 1,4-cyclohexanediol on percutaneous absorption and penetration of MTZ. Eleven formulations were studied in an in vitro porcine skin model using glass vertical Frans Diffusion Cell. 1,4-Cyclohexanediol was changed into 1,4-cyclohexanedicarboxylic acid, trans (and cis)-1,2-cyclohexanediol and 1,6-hexanediol, respectively, to study if H-bonding or ring structure would influence the retardation effect. MTZ was applied at infinite dose (100 mg), which corresponded to 750 μg of MTZ. Based on modifier ratios (MR) calculated by the flux values, the retardation effect on percutaneous absorption and penetration of MTZ was found in the formulations containing 1,4-cyclohexanedicarboxylic acid or cis-1,2-cyclohexanediol (MR values were 0.47 for which only contains 1,4-cyclohexanedicarboxylic acid, 0.74 for the formulation containing both 1,4-cyclohexanedicarboxylic acid and 1,2-hexanediol, and 0.90 for the formulation containing cis-1,2-cyclohexanediol and 1,2-hexanediol, respectively). The results showed that the hydroxyl group and structure of 1,4-cyclohexanediol played a significant role in retardation effects and provided valuable insight on the mechanisms of retardation effect through structure–activity relationships.
KEY WORDS: 1,4-cyclohexanediol; hydrogen groups; mechanism; retardation effect; ring structure
INTRODUCTION
Skin was the protective barrier of the body, as the largest organ, it was composed of several layers: the stratum corneum (SC), the epidermis/dermis, and the subcutaneous fat layer. The SC layer was a formidable barrier against the delivery of active molecules and the efficiency of topically applied actives was limited by their penetration through that layer (1).
In recent years, topical and transdermal drug delivery system were designed to cure dermatoses and systemic diseases, respectively. Penetrating into systemic circulation for systematical chemotherapy drug was preferred; however, penetrating across the SC layer and arriving at blood circulation was unwanted for some drugs with specific local-skin targeting as the focal area was the epidermis/dermis layer, therefore, unwanted or toxic side effects might be aroused. In this way, enhancer and retardant were developed. The formulations with enhancer could impart more skin penetration and less skin retention, but retardants militate reversely. Nevertheless, enhancer and retardant could exert an effect by modifying the SC, so they both refered to penetration modifier (2).
The administration of topical delivery remained relatively inefficient, although enhancement strategies had been developed for many years. Many publications on the mechanisms of enhancers suggested that they entered the skin lipids and created disorder (3). However, structure–activity relationships in a series of chemical modifiers were also reported (4–6).
Retardants reported were similar to certain enhancers in structure, and structure–activity relationship of both enhancers and retardants would influence penetration effect (7–10). Most of these retardants reported had the common feature: they formed H-bond crosslinking with ceremides 6, which possess the strongest H-bonding capability in the SC (11). It was found by Roberts et al. that the main determinant of solute diffusion across SC was H-bonding, and the presence of zero to two groups having the most remarkable effect on the diffusion coefficient (12). It was noteworthy that Hadgraft prosposed the alteration of lateral bonding with ceramides 6 in SC lipid was the mechanism of action of enhancers and retardants (13). For example, it was believed that N-0915, which was analog of azone, had a retardation effect by two-sided H-bonding with ceramides and made the SC more impermeable, whereas azone enhanced permeability of metronidazole (MTZ) due to its one-sided H-bonding with ceramides to create disorder in SC. In summary, percutaneous absorption and penetration was sensitive to the interaction between the permeant and the polar head groups of the lipids in the intercellular channels (such as the hydroxyl groups of ceramides), and diffusion parameter is related to effect of H-bonding (14–17).
In the previous time, synergistic effect of 1,4-cyclohexanediol and 1,2-hexanediol on percutaneous absorption and penetration of MTZ had been found (18). The present study was designed to investigate the role of 1,4-cyclohexanediol played in the combined application. Several different structures similar to 1,4-cyclohexanediol, such as 1,4-cyclohexanedicarboxylic acid, 1,2-cyclohexanediol, and 1,6-hexanediol were used in the experiments to change 1,4-cyclohexanediol, respectively. By the above replacement, the necessity and mechanisms of rigid structure and hydroxyls in 1,4-cyclohexanediol which related to the retardation effects would be better understood. MTZ was selected as a model drug, which was an active agent for the treatment of skin disorders, such as rosacea and acne. Percutaneous absorption of MTZ was studied under an in vitro pig skin model, which has similar permeability properties to human membrane.
According to these experiments, mechanisms of transdermal and topical drug delivery could be revealed more thoroughly, and more effective modifiers would be discovered and developed taking consideration of some mechanistic knowledge about permeation process.
Materials
Chemicals
1,4-Cyclohexanedicarboxylic acid, trans-1,2-cyclohexanediol, cis-1,2-cyclohexanediol, and 1,6-hexanediol were obtained from Rui Ding Chemical Co., Ltd. (Tianjin, China). MTZ was kindly provided by ZhongAn Pharmaceutical (Tianjin, China). 1,4-Cyclohexanediol was purchased from Sigma-Aldrich (St. Louis, MO, USA). Klucel® MF was obtained from Hercules, Inc. (Wilmington, DE, USA). All other chemicals used were of analytical grade.
Skin Membranes
Male white pig about 2 to 3 months old and 50 kg was purchased from Hebei Province of China. The back skin was removed from the pig; subcutaneous fat was carefully wiped off and cleaned by normal saline. The skin pieces were stored at −20°C until used but for no longer than 4 months. Prior to each experiment, the skin samples were thawed and hydrated for 1 h in phosphate-buffered saline (PBS), pH 7.4 by mounting on jacketed Franz diffusion cells, which were maintained at 37°C.
Methods
Preparation of Formulations
Eleven formulations shown in Table I were prepared at room temperature. Klucel® MF was used as the gelling agent.
Table I.
Formulations Containing Metronidazole (F1 to F11)
| Formulation | Ingredient (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Metronidazole | Klucel® MF | 1,2-Hexanediol | 1,4-Cyclohexanediol | 1,4-Cyclohexanedicarboxylic acid | Trans-1,2-cyclohexanediol | Cis-1,2-cyclohexanediol | 1,6-Hexanediol | |
| F1 (controlled) | 0.75 | 0.75 | – | – | ||||
| F2 | 0.75 | 0.75 | 4.0 | – | ||||
| F3 | 0.75 | 0.75 | 4.0 | 1.0 | ||||
| F4 | 0.75 | 0.75 | – | 1.0 | ||||
| F5 | 0.75 | 0.75 | 4.0 | 1.0 | ||||
| F6 | 0.75 | 0.75 | – | 1.0 | ||||
| F7 | 0.75 | 0.75 | 4.0 | 1.0 | ||||
| F8 | 0.75 | 0.75 | – | 1.0 | ||||
| F9 | 0.75 | 0.75 | 4.0 | 1.0 | ||||
| F10 | 0.75 | 0.75 | – | 1.0 | ||||
| F11 | 0.75 | 0.75 | 4.0 | 1.0 | ||||
Preparation of F4 and F5
1,4-Cyclohexanedicarboxylic acid was added in water. 1,2-Hexanediol was dissolved in a solution of 1,4-cyclohexanedicarboxylic acid in water. pH was adjusted to 4.9 ± 0.1 with triethanolamine. MTZ was added to the above solution with churning until dissolved. Klucel® MF as the gelling agent was dispersed in the solution while stirring until the solution gelled.
Preparation of Other Formulations
The general procedure was as follows. Taking an example of preparing F6, trans-1,2-cyclohexanediol was dissolved in a solution of 1,2-hexanediol in water, and MTZ was added to the above solution with stirring until dissolved. Then, Klucel® MF was dispersed in the solution while stirring until the solution gelled.
In Vitro Skin Permeation Studies
Porcine skin hydrated with PBS, pH 7.4 was mounted on the glass vertical Franz diffusion cells (Pharmacopoeia Standard Instrument Factory, Tianjin, China). The receptor compartment of 16-ml volume over a surface area of approximately 1.77 cm2 was filled with PBS stirred at 500 rpm and maintained at 37 ± 0.1°C. The donor compartment was covered with parafilm® in order to minimize evaporation of the ingredient in the formulations. The volume of the donor phase was about 2 ml. The model of the vertical Franz diffusion cell is shown in Fig. 1. MTZ (infinite doses approach, 100 mg of the formulations, corresponding to 750 μg of MTZ) was chosen as the model permeant. Six replicates were run for each formulation. MTZ in skin was extracted from four parts: the skin surface, the SC layer, the epidermis and dermis layer, and the receptor medium (RM). The recoveries were higher than 85%, and RSD were less than 10%. The skin surface was wiped with cotton ball soaked with PBS at the end of each time interval (1, 2, 4, 8, 12, 16, 20, and 24 h). The SC layer was removed by an average of ten strips on the bases of tape-stripping method (19). MTZ retained in the epidermis and dermis layer was collected by methanol extraction. The remaining skin was minced after tape stripping, vortexed with 1 ml methanol and centrifuged to collect the supernatant. The supernatants were combined as the extraction step repeated for three times, filtered, and ready for analysis. The RM was withdrawn from the receptor, filtered, and ready for analysis.
Fig. 1.
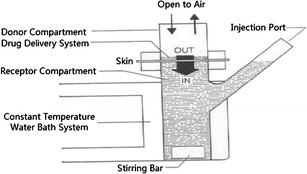
The model of the vertical Franz diffusion cell
HPLC Analytical Method
The analysis of MTZ was performed using HPLC (HP 1100, Agilent Technologies, Inc.) equipped with a stainless steel C18 column having a pore size of 5 μm and dimensions equivalent to 4.6 mm × 250 mm (Thermo, USA). The column was eluted with a mobile phase of double distilled water–methanol (75:25, v/v), which was a filtered (0.45 μm; Millipore) mixture at a flow rate of 1.0 ml/min. Injection volumes of the samples were 20 μl, and the wavelength for detection of MTZ was set at 310 nm on UV detector.
The analytical method was validated for linearity in the range of 0.15 to 75.0 μg/ml, and the correlation coefficient of 0.9997 for linearity of plot was observed in MTZ. LOD was 0.10 μg/ml, and LOQ was 0.15 μg/ml. The method showed good recoveries and the RSD of precision was less 3%. Intraday variability was less than 0.2%, and interday variability was also calculated to be less than 3.0%.
Data and Statistical Analysis
The amount of active present in the samples was determined using validated assay methods. In vitro permeation studies, several transdermal parameters such as mean flux, and cumulative amount after 24 h and lag time were calculated. For determination of the mean flux values of MTZ permeated through the skin membranes into the receptor fluid, slopes of plots of concentration in the receptor phase as a function of time and expressed as micrograms per square centimeter per hour using linear regression (Microsoft Excel) (20). The degree of penetration retardation/enhancement was defined as the modifier ratio (MR, which was calculated from the following equation (21)
 |
The values above unity represented enhancement, and values below 1 represented retardation of the permeant.
The statistical significance was calculated by paired two-tailed Student’s t test (*p < 0.05; **p < 0.01). Values are given as means ± SD.
RESULTS
In Vitro Skin Permeation Study
The composition of F1 to F11 is shown in Table I, and their skin permeation parameters were summarized in Table II.
Table II.
Skin Permeation Parameters of F1 to F11
| Formulation | T lag (h) | Flux (μg cm−2 h−1) | Amount in collection medium at 24 h (μg) | MR |
|---|---|---|---|---|
| F1 (controlled) | 0.72 ± 0.08 | 9.83 ± 0.62 | 394.28 ± 34.85 | – |
| F2 | 0.84 ± 0.16 | 10.52 ± 0.66 | 421.58 ± 35.24 | 1.07 |
| F3 | 1.32 ± 0.17 | 6.68 ± 0.68 | 267.92 ± 36.54 | 0.68 |
| F4 | 0.77 ± 0.11 | 4.58 ± 0.60 | 175.61 ± 41.88** | 0.47 |
| F5 | 1.12 ± 0.16 | 7.23 ± 1.23 | 272.55 ± 77.44* | 0.74 |
| F6 | 1.32 ± 0.06 | 9.60 ± 0.56 | 371.39 ± 96.41 | 0.98 |
| F7 | 0.68 ± 0.06 | 10.96 ± 1.89 | 427.91 ± 98.37 | 1.11 |
| F8 | 1.94 ± 0.18 | 14.01 ± 0.78 | 575.38 ± 59.66** | 1.43 |
| F9 | 1.03 ± 0.25 | 8.81 ± 0.96 | 346.25 ± 67.03 | 0.90 |
| F10 | 0.27 ± 0.10 | 13.90 ± 1.00 | 607.72 ± 66.60** | 1.41 |
| F11 | 1.15 ± 0.29 | 16.89 ± 0.55 | 604.50 ± 39.57** | 1.72 |
Each value represented the mean ± SD (n = 6)
MR modifier ratio (MR = Flux for the formulation containing modifier / Flux for control formulation)
*p < 0.05; **p < 0.01
F1 was conducted as a control formulation without any modifier, whereas F3 was a positive control formulation which showed synergistic retardation effect on a hairless mouse model by Li et al. (18). The MR value of F3 (0.68) indicated that a retardation effect can be observed on pig skin model as well. The presence of 4% 1,2-hexanediol, MTZ, and Klucel® MF at the same dose as the other formulation enhanced diffusion of MTZ mildly on pig skin model on account of the MR value of F2 was 1.07.
MR value of F4 and F5 in comparison with F1 was about 0.47 and 0.74, which means that retardation effect on the penetration of MTZ was quite significant in these two formulations containing 1,4-cyclohexanedicarboxylic acid. Furthermore, the Q24 value of F4 (175.61 ± 41.88 μg) and F5 (272.55 ± 77.44 μg) were prominently lower than F1 (394.28 ± 34.85 μμg), which also demonstrated a retardation effect of the formulation containing 1,4-cyclohexanedicarboxylic acid without and with 1,2-hexanediol. However, it could be observed that the formulation with only 1,4-cyclohexanedicarboxylic acid (F4) retarded penetration of MTZ more significantly than the formulation with both 1,4-cyclohexanedicarboxylic acid and 1,2-hexanediol (F5) did. The amount in collection medium at 24 h of F4 and F5 suggested that F4 retarded more than 50% penetration of MTZ more than half of Q24 of F1 for 24 h; however, F5 retarded about 30% in the same conditions, manifesting a more striking retardation effect of F4 comparing with F5. Interestingly, despite of low flux and Q24, lag time of F4 was not as long as other normal formulations with retardation effect. On the contrary, the lag time of F4 was similar to that of F1 (0.72 ± 0.08 h for F1 and 0.78 ± 0.11 h for F4), indicating of fast onset of action, which was propitious to the treatment of skin disease. In summary, the retardation effect of F4 was more markedly than that of F5, although F4 and F5 both exert a retardation effect.
The MR value of F6 was almost 1, indicating there was nearly no effect on the permeation of MTZ with the only modifier trans-1,2-cyclohexanediol in the formulation. However, the presence of only cis-1,2-cyclohexanediol in F8 made the permeation capability of MTZ markedly increased according to its flux (14.01 ± 0.78 μg cm−2 h−1) and Q24 (575.38 ± 59.66 μg) comparing with flux (9.83 ± 0.62 μg cm−2 h−1) and Q24 (394.28 ± 34.85 μg) of F1. The aforementioned data showed that neither trans-1,2-cyclohexanediol alone nor cis-1,2-cyclohexanediol did not exert a retardation effect significantly, although the effect of the two was not identical. It was interesting to note that the MR value of F7 and F9 were 1.11 and 0.90, respectively, demonstrating trans-1,2-cyclohexanediol and cis-1,2-cyclohexanediol had different effects when combined with 1,2-cyclohexanediol. The lag time of F6 and F8 (0.68 ± 0.06 and 1.03 ± 0.25 h), and Q24 of them (427.91 ± 98.37 and 346.25 ± 67.03 μg) also showed an opposite effect. With the same chemical structure of cis–trans isomer, the physiology activity was different, F7 enhanced absorption and penetration of MTZ, but F9 had a retardation effect.
As was seen in Table I, F10 was the formulation with the only modifier 1,6-hexanediol and F11 was the formulation with both 1,2-hexanediol and 1,6-hexanediol. Both F10 and F11 enhanced the drug delivery significantly according to the MR values (1.41 and 1.72), and Q24 (607.72 ± 66.6 and 604.50 ± 39.57 μg) of them.
In Vitro Epidermis/Dermis Retention Study
The integrity of epidermis was as important as SC, as epidermis also serves as a mean of a barrier to outer substance (22,23). The lesions of most dermatological disorders was the epidermis and dermis, so it was important for active agents in applied formulations to effectively penetrate the SC layer and reach epidermis and dermis layers (24). Conversely, the active ingredient would give rise to an undesired side effect when it reached the blood circulation. In general, taking advantage of more therapeutic action in epidermis and dermis and avoiding toxic and side effect was necessary for a good topical formulation.
In this study, epidermis/dermis retention and the amount of MTZ in the RM of F4–F11 and the control formulation are summarized in Fig. 1. The results of paired two-tailed Student’s t test on epidermis/dermis retention and RM at 8 time points from 1 to 24 h of F4–F11 in comparison with F1 were marked in this picture (**p < 0.01; *p < 0.05).
Although there were no significant statistical difference (p > 0.05) observed between F4–F5 and F1 in Fig. 2a, it could be found that the percent of MTZ in F1 was higher than F4 and F5 at short time points, however, the results were reversed at long time points. Furthermore, retention in epidermis/dermis of both F4 and F5 was more stable than F1 at the time points. In other words, penetration of MTZ would be relatively stable at least within 24 h after administration of F4 and F5, and MTZ could produce a retentive therapeutic effect in epidermis/dermis for a longer time. Moreover, F4 and F5 showed significant statistical difference (p < 0.01) between each other, specifically, MTZ from F4 retained more in epidermis/dermis than F5, which indicated that F4 may be a better topical delivery formulation due to its superior epidermal/dermal retention capability. Less permeation of F4 in the RM than F5 shown in Fig. 2b was also indicative of the above standpoint. In addition, significant statistical difference (p < 0.01) and statistical difference (p < 0.05) calculated by F4 and F5 in comparison with F1 yielded valuable insight in lesser side effect of the formulations containing 1,4-cyclohexanedicarboxylic acid.
Fig. 2.
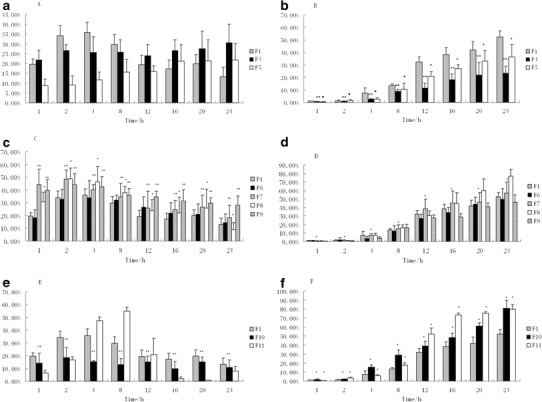
a Percentage of epidermal retention of MTZ: comparison of F1, F4, and F5. Mean ± SD, n = 6. b Percentage of amount of MTZ in receptor medium: comparison of F1, F4, and F5. Mean ± SD, n = 6. c Percentage of epidermal retention of MTZ: comparison of F1, F6, F7, F8, and F9. Mean ± SD, n = 6. d Percentage of amount of MTZ in receptor medium: comparison of F1, F6, F7, F8, and F9. Mean ± SD, n = 6. e Percentage of epidermal retention of MTZ: comparison of F1, F10, and F11. Mean ± SD, n = 6. f Percentage of amount of MTZ in receptor medium: comparison of F1, F10, and F11. Mean ± SD, n = 6. SD standard deviation
The epidermis/dermis retention and RM of formulations containing 1,2-cyclohexanediol was listed in Fig. 2c, d. No statistical difference was investigated of F6 in both epidermis/dermis, and the RM, suggesting neither retardation nor enhancement effect was obvious after administration. Significant statistical difference (p < 0.01) and statistical difference (p < 0.05) were calculated in epidermis/dermis and RM between F7 and F1, respectively, indicating of an enhancement effect. There was a statistical difference (p < 0.05) between F8 and the control formulation in epidermis/dermis retention, and no statistical difference was found in RM. However, on comprehensive consideration of Tlag (1.94 ± 0.18 h) and MR value (1.43), an enhancement effect yielded especially in longer time points by means of administration of F8. MTZ from F9 in epidermis/dermis showed significant statistic difference (p < 0.01) in comparison with F1, it was observed that the epidermis retention was markedly higher than F1 at each time point we studied. Moreover, although there was no statistic difference between F1 and F9 in the RM, less amount of MTZ in F9 was collected at long time point (12, 16, 20, and 24 h) than F1, which investigated in Fig. 1d. Thus, F9 could retard penetration of MTZ on pig skin to a certain extent on consideration of the lower flux (8.81 ± 0.96 μg cm−2 h−1), although the effect was weaker than the formulations containing 1,4-cyclohexanedicarboxylic acid.
It was depicted in Fig. 2e, f that the epidermis/dermis retention of F10 was lower than that of F1, however, the amount of MTZ in the RM was just on the opposite. Thus, an enhancement effect was emerged via administration of F10 which would be a good choice to be an enhancer in transdermal drug delivery. F11 was also a formulation with an enhancement effect despite no statistical difference (p > 0.05) with F1 in epidermis/dermis retention.
DISCUSSION
Porcine skin, which had been used in numerous percutaneous absorption studies, was a good surrogate for human skin. It was reported that porcine skin and human skin were similar histologically and biochemically (25). Furthermore, it had been shown that porcine skin (approximately 600 μm in thickness) had a similar lipid content of SC (17.5 ± 2.4 μm, 4–8% (pig); 18.2 ± 3.3 μm, 2–6.5% (human)) and closer permeability character to human skin (26). Thus, porcine skin was used as skin membrane in this study.
In view of observed synergistic penetration retardation effect of 1,2-hexanediol and 1,4-cyclohexanediol, there were three aspects of the problem to be addressed to understand the role of 1,4-cyclohexanediol played. The first question involved whether the groups that could form H-bonding were necessary for a retardant. The second problem related to the influence of the hydroxyl position of 1,4-cyclohexanediol for the retardation effect. The third aspect dealt with the necessity of ring structure to remain retardation for skin penetration. Thus, 1,4-cyclohexanedicarboxylic acid (change hydroxyls to carboxyls), 1,2-cyclohexanediol (alter the position of hydroxyls), and 1,6-hexanediol (shift the ring structure) were applied in the formulations to realize the key factor in 1,4-cyclohexanediol, and then the mechanism of skin penetration.
The Necessity of Hydrogen Groups in 1,4-Cyclohexanediol
1,4-Cyclohexanedicarboxylic acid was a kind of compound that could form H-bonding with 1,2-hexanediol and ceramides. The proposed retardation mechanism of formulations containing 1,4-cyclohexanedicarboxylic acid could be seen in Fig. 3. The left part of this figure depicted a mechanism that 1,4-cyclohexanedicarboxylic acid exert a retardation effect in terms of forming H-bonding with ceramides by its hydroxyls. In the right part, 1,4-cyclohexanedicarboxylic acid and 1,2-hexanediol formed two-sided H-bonding with ceramides, which prevent the penetration of MTZ to a certain degree. However, F4 acted retardation effects more obviously than F5 suggested that 1,4-cyclohexanedicarboxylic acid without 1,2-hexanediol in the formulation would exert a more intense effect on the penetration of MTZ. Roberts pointed out that passing through skin of a permeant depended on its H-bonding capacity, and the size of the molecule as defined by its molecular weight would also influence the process of diffusion (10). The molecular weight of 1,4-cyclohexanediol,1,2-hexanediol, and 1,4-cyclohexanedicarboxylic acid were estimated to144.21, 118.17, and 168.24, respectively. The MR value of formulation containing 1,2-hexanediol and 1,4-cyclohexanediol (F3) comparing with the control formulation was only 0.68, whereas, the MR value of the combination of 1,2-hexanediol and 1,4-cyclohexanedicarboxylic acid (F4) was 0.74 comparing with the same control, which implied that the presence of 1,4-cyclohexanediol retard skin penetration of MTZ more effective than the presence of 1,4-cyclohexanedicarboxylic acid when maintaining 1,2-hexanediol constant. However, the formulation with only 1,4-cyclohexanedicarboxylic acid (F4) had the most striking retardation effect in accordance with its MR value (0.47). It was speculated that the results may be caused by the follow reasons: First, as the reason of molecular size, the combination of 1,2-hexanediol and 1,4-cyclohexanediol was more suitable for the passageway formed by ceramides than the combination of 1,2-hexanediol and 1,4-cyclohexanedicarboxylic acid and less suitable than 1,4-cyclohexanedicarboxylic acid existed alone in the formulation. Second, the carboxylic acid had stronger capability to form H-bonding than alcohol (12,15). 1,2-Hexanediol may just play a role of solubilizer or enhancer in F5, and 1,4-cyclohexanedicarboxylic acid formed stronger crosslink H-bonding and stayed more stable connection with ceramides by two H bonds than three ones of F3.
Fig. 3.
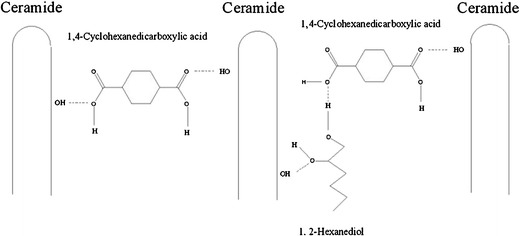
Proposed hydrogen bonds interactions between ceramides and modifier molecules 1,2-hexanediol and 1,4-cyclohexanedicarboxylic acid. Dashed lines, H-bonding
The Influence of Hydrogen Groups Position in 1,4-Cyclohexanediol
Trans-1,2-cyclohexanediol and cis-1,2-cyclohexanediol were isomer. It was interesting to note that trans-1,2-cyclohexanediol and cis-1,2-cyclohexanediol combined with 1,2-hexanediol in F7 and F9 exerted opposite effect on the absorption and penetration of MTZ. It was a common sense that chair-like structure of cyclohexane was more stable than boat-like structure. The position of hydroxyl groups in trans-1,2-cyclehexanediol on e-key was the dominant structure, and the two hydroxyl groups in cis-1,2-cyclohexanediol occupied a-key and e-key, respectively. Figure 4 suggested that forming H-bonding with ceramides was hard for the formulations containing trans-1,2-cyclohexanediol and 1,2-hexanediol (F7), however, it was possible for cis-1,2-cyclohexanediol to form H-bonding with 1,2-hexanediol and ceramides although the chemical bond may be not stable enough. Thus, cis-1,2-cyclohexanediol combined with 1,2-hexanediol retarded penetration of MTZ faintly, whereas trans-1,2-cyclohexanediol and 1,2-hexanediol have the efficiency of enhancement. Conversely, molecular size of 1,2-cyclohexanediol and 1,4-cyclohexanediol were similar, suggesting that the factor of molecular size would not be the reason to explain different effect between formulations containing 1,4-cyclohexanediol or 1,2-cyclohexanediol. In addition, no notable retardation effect was achieved of F6 and F8 may be due to the molecular size of 1,2-cyclohexanediol, which is too small to form two-sided H-bonding with ceramides, and the reason for enhancement effect of the formulation containing only 1% 1,4-cyclohexanediol may be the same (17,27).
Fig. 4.
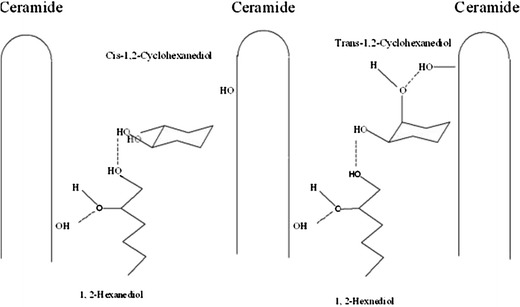
Proposed hydrogen bonds interactions between ceramides and modifier molecules 1,2-hexanediol, trans-1,2-cyclohexanediol, and cis-1,2-cyclohexanediol. Dashed lines, H-bonding
The Influence of Ring Structure in 1,4-Cyclohexanediol
Both of 1,4-cyclohexanediol and 1,6-hexanediol contained six carbons and two hydroxyl groups on both sides of molecular structure, but F11 enhanced absorption and penetration of MTZ compared with retardation effect of F3. The molecular weight of 1,6-hexanediol was just between 1,4-cyclohexanediol and 1,4-cyclohexanedicarboxylic acid. If the mechanism of retardation was considered merely according to molecular weight, F11 would retard the permeation of MTZ because of retardation effect of F3 and F5. On the contrary, F11 enhanced the permeation of MTZ. The reason for that phenomenon maybe, unlike 1,4-cyclohexanediol, 1,6-hexanediol was a kind of compound without a chair-like ring structure. Furthermore, 1,6-hexanediol could form H-bonding with ceramides by stretching or compressing its flexible chain structure in F10 and F11, yet the chemical connection could be destroyed easily because of instability. Thus, it was speculated that the rigid, chair-like structure played a key role in the observed retardation effect.
The in vitro experiments revealed the permeation capability of a formulation and provided invaluable insight into the mechanism of topical absorption and penetration process through structure–activity relationships, resulting in facility of selecting modifier and development of superior topical and transdermal products.
CONCLUSIONS
The retardation effect of percutaneous absorption and penetration of MTZ from formulations was strongly dependent on the groups which could form two-sided H-bonding with ceramides in the modifier. Alternatively, the molecular feature of the modifier, such as the rigid structure and the position of hydroxyls, would also influence the permeation effect. Thus, retardation effect was observed in the formulations containing 1,4-cyclohexanedicarboxylic acid in the absence and presence of 1,2-hexanediol, and the formulation containing 1,2-hexanediol and cis-1,2-cyclohexanediol simultaneously. To sum up, the present study shed light on the mechanism of interactions among molecules from the formulations and ceramides during penetration in SC, which was beneficial to topical or transdermal formulation design.
Contributor Information
Nan Li, Email: linannanli78@163.com.
Jerry Zhang, Email: jerryzhang@zyleris.com.
References
- 1.Melot M, Pudney P, Williamson AM, Caspers PJ, Pol A, Puppels GJ. Study the effectiveness of penetration enhancers to deliver retinal through the stratum cornum by in vivo confocal Raman spectroscopy. J Control Release. 2009;138:32–39. doi: 10.1016/j.jconrel.2009.04.023. [DOI] [PubMed] [Google Scholar]
- 2.Kaushik D, Costache A, Michniak-Kohn B. Percutaneous penetration modifiers and formulation effects. Int J Pharm. 2010;386:42–51. doi: 10.1016/j.ijpharm.2009.10.052. [DOI] [PubMed] [Google Scholar]
- 3.Purdon CH, Azzi CG, Zhang J, Smith EW, Maibach HI. Penetration enhancement of transdermal delivery—current permutations and limitations. Critical review in therapeutic drug carrier systems. Crit Rev Ther Drug Carrier Syst. 2004;21:97–132. doi: 10.1615/CritRevTherDrugCarrierSyst.v21.i2.20. [DOI] [PubMed] [Google Scholar]
- 4.Kanikkannan N, Kandimalla K, Lamba SS, Singh M. Structure-activity relationship of chemical penetration enhancers in transdermal drug delivery. Curr Med Chem. 2000;7(6):593–608. doi: 10.2174/0929867003374840. [DOI] [PubMed] [Google Scholar]
- 5.Patel H, Berge W, Cronin MTD. Quantitative structure–activity relationships (QSARs) for the prediction of skin permeation of exogenous chemicals. Chemosphere. 2002;48(6):603–613. doi: 10.1016/S0045-6535(02)00114-5. [DOI] [PubMed] [Google Scholar]
- 6.Domagala JM. Structure–activity and structure–side-effect relationships for the quinolone antibacterials. J Antimicrob Chemother. 1994;33(4):685–706. doi: 10.1093/jac/33.4.685. [DOI] [PubMed] [Google Scholar]
- 7.Vavrova K, Zbytovska J, Hrabalek A. Amphiphilic transdermal permeation enhancers: structure–activity relationships. Curr Med Chem. 2005;12(19):2273–2291. doi: 10.2174/0929867054864822. [DOI] [PubMed] [Google Scholar]
- 8.Asbill CS, Michniak BB. Percutaneous penetration enhancers: local versus transdermal activity. Pharm Sci Technol Today. 2000;3(1):36–41. doi: 10.1016/S1461-5347(99)00225-4. [DOI] [PubMed] [Google Scholar]
- 9.Cronin MT, Dearden JC, Moss GP, Murray-Dickson G. Investigation of the mechanism of flux across human skin in vitro by quantitative structure–permeability relationships. Eur J Pharm Sci. 1999;7:325–330. doi: 10.1016/S0928-0987(98)00041-4. [DOI] [PubMed] [Google Scholar]
- 10.Chavda S, Bahadur P. Micellization of a cationic Gemini surfactant in aqueous solutions with different alkanols and alkanediols as additives: effect of nonpolar chain and position of hydroxyl groups. J Mol Liq. 2011;161:72–77. doi: 10.1016/j.molliq.2011.04.018. [DOI] [Google Scholar]
- 11.Wertz PW. Epidermal lipids. Semin Dermatol. 1992;11(2):106–113. [PubMed] [Google Scholar]
- 12.Roberts MS, Pugh WJ, Hadgraft J, Watkinson AC. Epidermal permeability–penetrant structure relationships. 1. An analysis of methods of predicting penetration of monofunctional solutes from aqueous solutions. Int J Pharm. 1995;126:219–233. doi: 10.1016/0378-5173(95)04130-3. [DOI] [Google Scholar]
- 13.Hadgraft J, Peck J, Williams DG, Pugh WJ, Allan G. Mechanisms of action of skin penetration enhancers/retarders: azone and analogues. Int J Pharm. 1996;141:17–25. doi: 10.1016/0378-5173(96)04609-1. [DOI] [Google Scholar]
- 14.Roberts MS, Pugh WJ, Hadgraft J. Epidermal permeability: penetrant structure relationships. 2. The effect of H-bonding groups in penetrants on their diffusion through the stratum corneum. Int J Pharm. 1996;132:23–32. doi: 10.1016/0378-5173(95)04278-4. [DOI] [Google Scholar]
- 15.Pugh WJ, Roberts MS, Hadgraft J. Epidermal permeability—penetrant structure relationships: 3. The effect of hydrogen bonding interactions and molecular size on diffusion across the stratum corneum. Int J Pharm. 1996;138:149–165. doi: 10.1016/0378-5173(96)04533-4. [DOI] [Google Scholar]
- 16.Plessis J, Pugh WJ, Judefeind A, Hadgraft J. The effect of the nature of H-bonding groups on diffusion through PDMS membranes saturated with octanol and toluene. Eur J Pharm Sci. 2002;15:63–69. doi: 10.1016/S0928-0987(01)00212-3. [DOI] [PubMed] [Google Scholar]
- 17.Plessis J, Pugh WJ, Judefeind A, Hadgraft J. Physico-chemical determinants of dermal drug delivery: effects of the number and substitution pattern of polar groups. Eur J Pharm Sci. 2002;16:107–112. doi: 10.1016/S0928-0987(02)00085-4. [DOI] [PubMed] [Google Scholar]
- 18.Li N, Jia WB, Zhang Y, Tan FP, Zhang J. Synergistic effect of 1,4-cyclohexanediol and 1,2-hexanediol on percutaneous absorption and penetration of metronidazole. Int J Pharm. 2011;415:169–174. doi: 10.1016/j.ijpharm.2011.05.069. [DOI] [PubMed] [Google Scholar]
- 19.Howes D, Guy R, Hadgraft J. Methods for assessing percutaneous absorption. The report and recommendations of ECVAM Workshop 13. Altern Lab Anim. 1996;24:81–106. [Google Scholar]
- 20.Batheja P, Song Y, Wertz P, Michniak-Kohn B. Effects of growth conditions on the barrier properties of ahumanskin equivalent. Pharm Res. 2009;26:1689–1700. doi: 10.1007/s11095-009-9879-1. [DOI] [PubMed] [Google Scholar]
- 21.Goodman M, Barry BW. Action of penetration enhancers on human skin as assessed by the permeation of model drugs 5-fluorouracil and estradiol. I. Infinite dose technique. J Invest Dermatol. 1988;91:323–327. doi: 10.1111/1523-1747.ep12475655. [DOI] [PubMed] [Google Scholar]
- 22.Elias PM, Goerke J, Friend DS. Mammalian epidermal barrier layer lipids: composition and influence on structure. J Invest Dermatol. 1977;69:535–546. doi: 10.1111/1523-1747.ep12687968. [DOI] [PubMed] [Google Scholar]
- 23.Hughes AL. Evolutionary diversification of the mammalian defensins. Cell Mol Life Sci. 1999;56:94–103. doi: 10.1007/s000180050010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cross SE, Roberts MS. The effect of occlusion on the epidermal penetration of parabens from a commercial test ointment, acetone and ethanol vehicles. J Invest Dermatol. 2000;115:914–918. doi: 10.1046/j.1523-1747.2000.00151.x. [DOI] [PubMed] [Google Scholar]
- 25.Meyer W, Schwarz R, Neurand K. The skin of domestic mammals as a model for the human skin, with reference to the domestic pig. Curr Prob Dermatol. 1978;7:39–52. doi: 10.1159/000401274. [DOI] [PubMed] [Google Scholar]
- 26.Sato K, Sugibayashi K, Morimoto Y. Species differences in percutaneous absorption of nicorandil. J Pharm Sci. 1991;80:104–107. doi: 10.1002/jps.2600800203. [DOI] [PubMed] [Google Scholar]
- 27.Li N, Su Q, Tan FP, Zhang J. Effect of 1,4-cyclohexanediol on percutaneous absorption and penetration of azelaic acid. Int J Pharm. 2010;387:167–171. doi: 10.1016/j.ijpharm.2009.12.025. [DOI] [PubMed] [Google Scholar]


