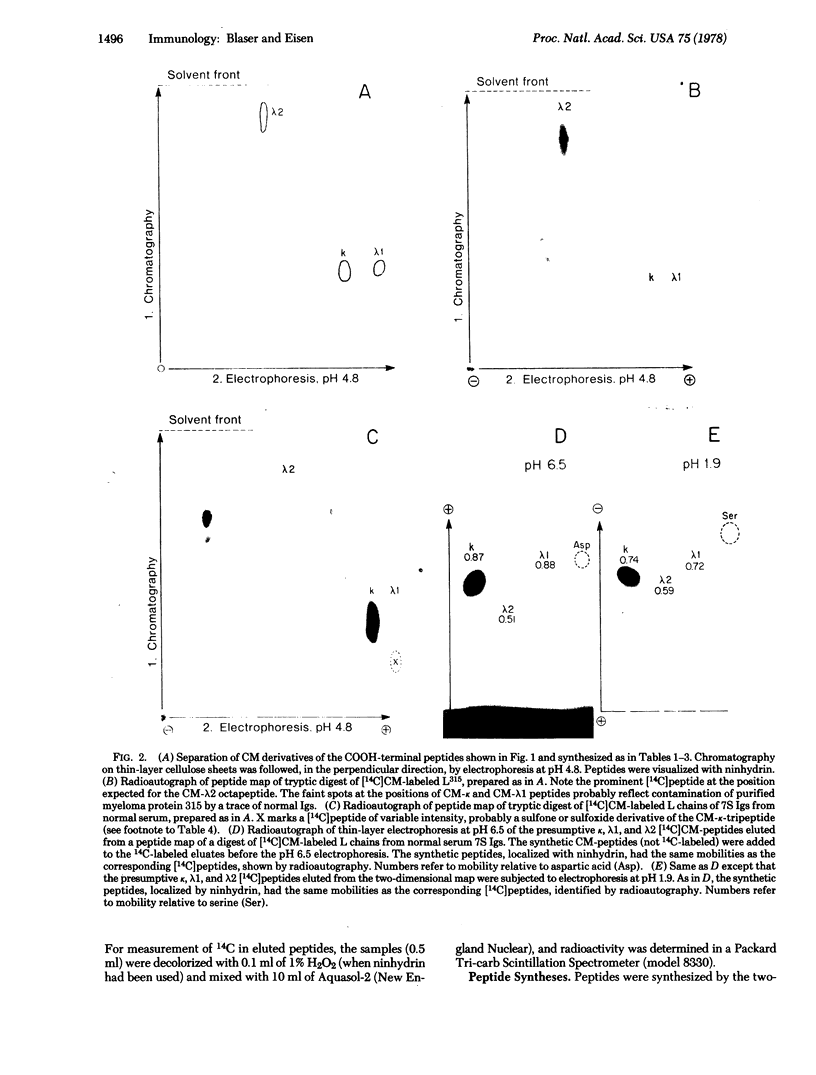
Abstract
Light (L) chains of the lambda type are rare in mouse immunoglobulins. One lambda chain, the L chain of myeloma protein 315 (L315), differs in amino acid sequence at many positions in the COOH-terminal domain from all other lambda chains whose sequences have been determined (called lambda1 chains). To determine whether chains of the L315 type (called lambda2) occur in normal mouse immunoglobulins, we synthesized the COOH-terminal peptides expected in tryptic digests of K, lambda1, and L315 and developed procedures to separate the S-carboxymethyl derivatives of these peptides. Peptide maps of tryptic digests of [14C]carboxymethyl-labeled L chains from normal serum immunoglobulins showed that about 1% of mouse 7S immunoglobulins have L chains of the L315 or lambda2 type.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Appella E. Amino acid sequences of two mouse immunoglobulin lambda chains. Proc Natl Acad Sci U S A. 1971 Mar;68(3):590–594. doi: 10.1073/pnas.68.3.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dugan E. S., Bradshaw R. A., Simms E. S., Eisen H. N. Amino acid sequence of the light chain of a mouse myeloma protein (MOPC-315). Biochemistry. 1973 Dec 18;12(26):5400–5416. [PubMed] [Google Scholar]
- Eisen H. N., Simms E. S., Potter M. Mouse myeloma proteins with antihapten antibody acitivity. The protein produced by plasma cell tumor MOPC-315. Biochemistry. 1968 Nov;7(11):4126–4134. doi: 10.1021/bi00851a048. [DOI] [PubMed] [Google Scholar]
- Frangione B., Milstein C. Variations in the S-S bridges of immunoglobins G: interchain disulfide bridges of gamma G3 myeloma proteins. J Mol Biol. 1968 May 14;33(3):893–906. doi: 10.1016/0022-2836(68)90326-4. [DOI] [PubMed] [Google Scholar]
- Gottlieb P. D. Genetic correlation of a mouse light chain variable region marker with a thymocyte surface antigen. J Exp Med. 1974 Nov 1;140(5):1432–1437. doi: 10.1084/jem.140.5.1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hood L., Talmage D. W. Mechanism of antibody diversity: germ line basis for variability. Science. 1970 Apr 17;168(3929):325–334. doi: 10.1126/science.168.3929.325. [DOI] [PubMed] [Google Scholar]
- Jaffe B. M., Simms E. S., Eisen H. N. Specificity and structure of the myeloma protein produced by mouse plasmacytoma MOPC-460. Biochemistry. 1971 Apr 27;10(9):1693–1699. doi: 10.1021/bi00785a029. [DOI] [PubMed] [Google Scholar]
- Offord R. E. Electrophoretic mobilities of peptides on paper and their use in the determination of amide groups. Nature. 1966 Aug 6;211(5049):591–593. doi: 10.1038/211591a0. [DOI] [PubMed] [Google Scholar]
- Potter M. Immunoglobulin-producing tumors and myeloma proteins of mice. Physiol Rev. 1972 Jul;52(3):631–719. doi: 10.1152/physrev.1972.52.3.631. [DOI] [PubMed] [Google Scholar]
- SELA M., WHITE F. H., Jr, ANFINSEN C. B. The reductive cleavage of disulfide bonds and its application to problems of protein structure. Biochim Biophys Acta. 1959 Feb;31(2):417–426. doi: 10.1016/0006-3002(59)90016-2. [DOI] [PubMed] [Google Scholar]
- Schulenburg E. P., Simms E. S., Lynch R. G., Bradshaw R. A., Eisen H. N. Amino acid sequence of the light chain from a mouse myeloma protein with anti-hapten activity: evidence for a third type of light chain. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2623–2626. doi: 10.1073/pnas.68.11.2623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tonegawa S., Hozumi N., Matthyssens G., Schuller R. Somatic changes in the content and context of immunoglobulin genes. Cold Spring Harb Symp Quant Biol. 1977;41(Pt 2):877–889. doi: 10.1101/sqb.1977.041.01.097. [DOI] [PubMed] [Google Scholar]
- Underdown B. J., Simms E. S., Eisen H. N. Subunit structure and number of combining sites of the immunoglobulin A myeloma protein produced by mouse plasmacytoma MOPC-315. Biochemistry. 1971 Nov 23;10(24):4359–4368. doi: 10.1021/bi00800a002. [DOI] [PubMed] [Google Scholar]
- Weigert M., Riblet R. Genetic control of antibody variable regions. Cold Spring Harb Symp Quant Biol. 1977;41(Pt 2):837–846. doi: 10.1101/sqb.1977.041.01.093. [DOI] [PubMed] [Google Scholar]




