Abstract
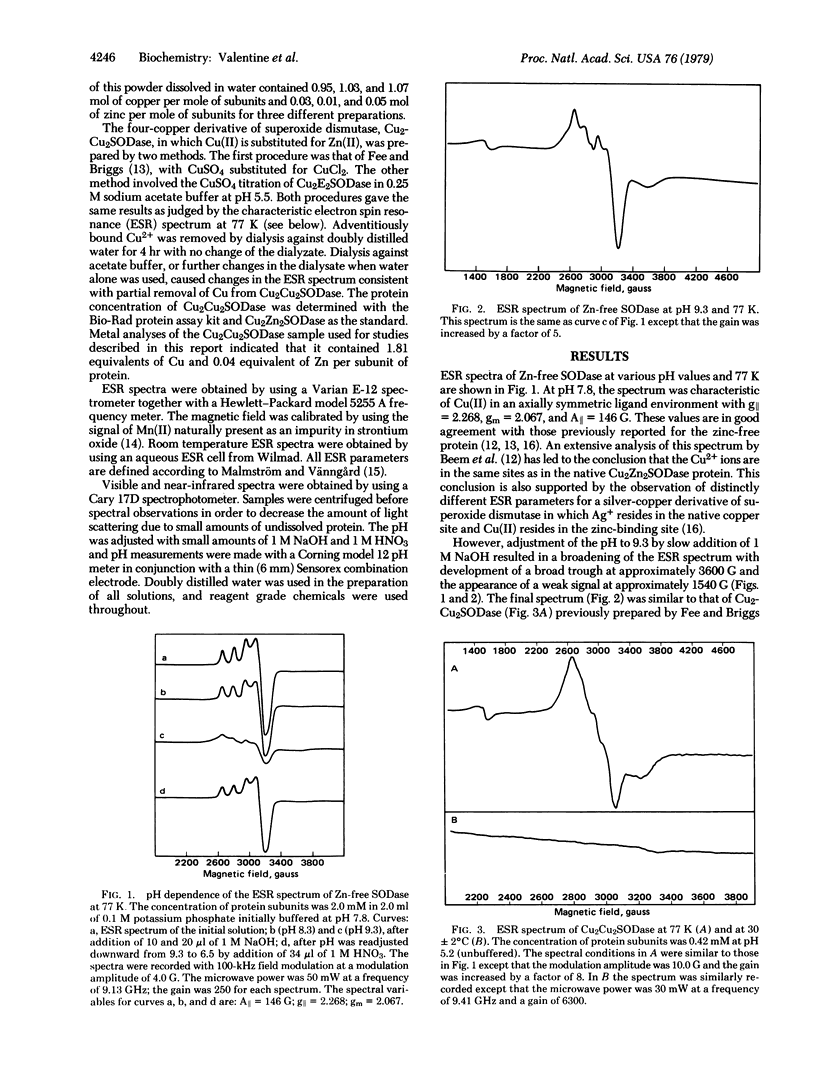
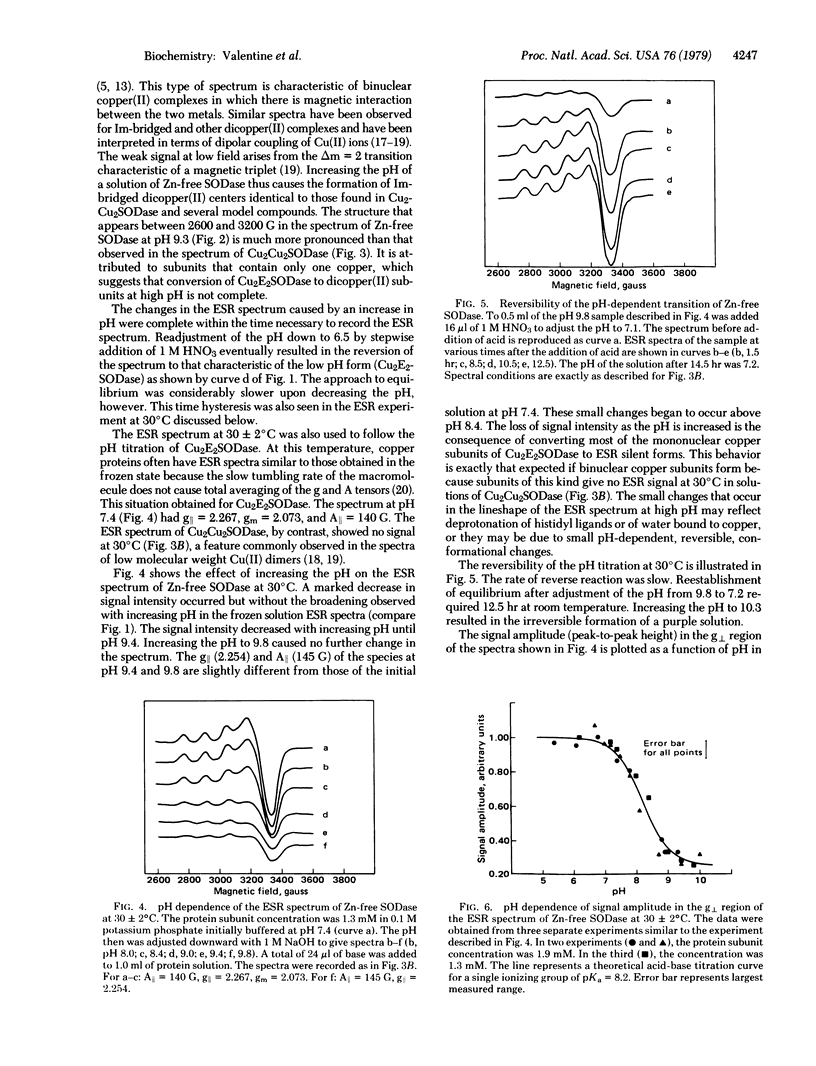
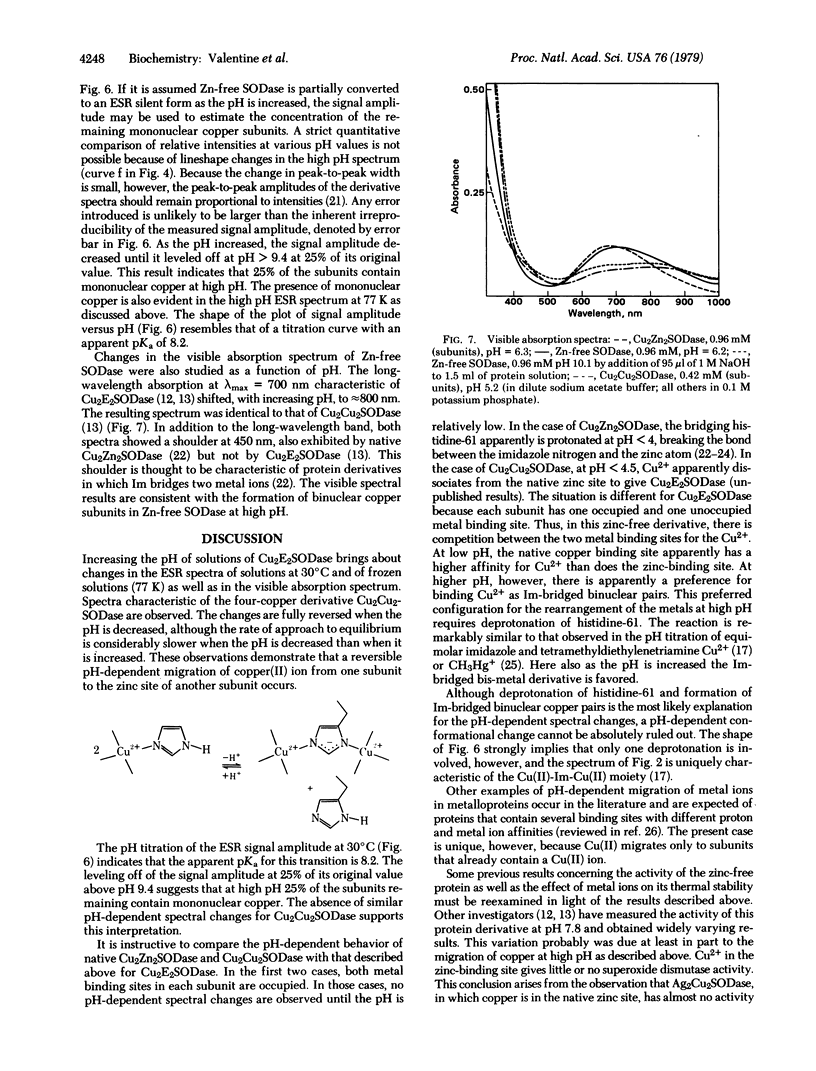
Bovine erythrocyte superoxide dismutase (Cu2Zn2SODase; superoxide:superoxide oxidoreductase, EC 1.15.1.1) consists of two identical subunits each containing Cu2+ and Zn2+ in close proximity. We describe here electron spin resonance (ESR) and visible absorption spectroscopic studies of the zinc-free derivative of this protein, Cu2E2SODase (E = empty) over the pH range 6-10. The ESR spectrum of the zinc-free protein at 77 K is markedly pH dependent. At pH < 8.0 the ESR spectrum is axial in appearance. At pH > 8.0, the lineshape becomes increasingly distorted with increasing pH until, at pH = 9.5, the spectrum is very broad and resembles that of the four-copper derivative Cu2Cu2SODase and of model imidazolate-bridged binuclear Cu(II) complexes. ESR spectra at 30°C are also consistent with formation of Cu(II)-Im-Cu(II). A plot of changes in the signal amplitude of g⊥ for Cu2E2SODase as a function of pH gives an apparent pKa of 8.2 for the transition. The long-wavelength absorption with λmax = 700 nm characteristic of Cu2E2SODase shifts with increasing pH to 800 nm and the resulting visible spectrum is identical to that of Cu2Cu2SODase. All of the above-mentioned spectroscopic changes induced by additions of NaOH are reversed when the pH is decreased with HNO3, although the approach to equilibrium is slow in the latter case. The results of these experiments are consistent with a reversible, pH-dependent migration of Cu2+ from the native copper site of one subunit of the zinc-free protein to the empty zinc site of another subunit. By contrast, native protein, Cu2Zn2SODase, and the four-copper protein, Cu2Cu2SODase, show no variation in visible or ESR spectral properties in this pH range. Some previous results concerning the activity of Cu2E2SODase and its thermal stability are reexamined in light of these new findings.
Keywords: metalloprotein, electron spin resonance, histidyl imidazolate
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beem K. M., Rich W. E., Rajagopalan K. V. Total reconstitution of copper-zinc superoxide dismutase. J Biol Chem. 1974 Nov 25;249(22):7298–7305. [PubMed] [Google Scholar]
- Beem K. M., Richardson D. C., Rajagopalan K. V. Metal sites of copper-zinc superoxide dismutase. Biochemistry. 1977 May 3;16(9):1930–1936. doi: 10.1021/bi00628a027. [DOI] [PubMed] [Google Scholar]
- Calabrese L., Cocco D., Morpurgo L., Mondovì B., Rotilio G. Reversible uncoupling of the copper and cobalt spin systems in cobalt bovine superoxide dismutase at low pH. FEBS Lett. 1975 Nov 1;59(1):29–31. doi: 10.1016/0014-5793(75)80333-4. [DOI] [PubMed] [Google Scholar]
- Carrico R. J., Deutsch H. F. The presence of zinc in human cytocuprein and some properties of the apoprotein. J Biol Chem. 1970 Feb 25;245(4):723–727. [PubMed] [Google Scholar]
- Fee J. A., Briggs R. G. Studies on the reconstitution of bovine erythrocyte superoxide dismutase. V. Preparation and properties of derivatives in which both zinc and copper sites contain copper. Biochim Biophys Acta. 1975 Aug 19;400(2):439–450. doi: 10.1016/0005-2795(75)90200-7. [DOI] [PubMed] [Google Scholar]
- Fee J. A., Phillips W. D. The behavior of holo- and apo-forms of bovine superoxide dismutase at low pH. Biochim Biophys Acta. 1975 Nov 18;412(1):26–38. doi: 10.1016/0005-2795(75)90336-0. [DOI] [PubMed] [Google Scholar]
- Fee J. A. Studies on the reconstituion of bovine erythrocyte superoxide dismutase. 3. Evidence for a strong interdependence between Cu 2+ and Zn 2+ binding in the expression of the spectroscopic properties of the native protein and for a close proximity of the Zn 2+ and Cu 2+ sites. Biochim Biophys Acta. 1973 Jan 25;295(1):107–116. [PubMed] [Google Scholar]
- Forman H. J., Evans H. J., Hill R. L., Fridovich I. Histidine at the active site of superoxide dismutase. Biochemistry. 1973 Feb 27;12(5):823–827. doi: 10.1021/bi00729a006. [DOI] [PubMed] [Google Scholar]
- Forman H. J., Fridovich I. On the stability of bovine superoxide dismutase. The effects of metals. J Biol Chem. 1973 Apr 25;248(8):2645–2649. [PubMed] [Google Scholar]
- Fridovich I. Superoxide dismutases. Adv Enzymol Relat Areas Mol Biol. 1974;41(0):35–97. doi: 10.1002/9780470122860.ch2. [DOI] [PubMed] [Google Scholar]
- Heikkila R. E., Cabbat F. A sensitive assay for superoxide dismutase based on the autoxidation of 6-hydroxydopamine. Anal Biochem. 1976 Oct;75(2):356–362. doi: 10.1016/0003-2697(76)90089-0. [DOI] [PubMed] [Google Scholar]
- Lippard S. J., Burger A. R., Ugurbil K., Pantoliano M. W., Valentine J. S. Nuclear magnetic resonance and chemical modification studies of bovine erythrocyte superoxide dismutase: evidence for zinc-promoted organization of the active site structure. Biochemistry. 1977 Mar 22;16(6):1136–1141. doi: 10.1021/bi00625a017. [DOI] [PubMed] [Google Scholar]
- McCord J. M., Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem. 1969 Nov 25;244(22):6049–6055. [PubMed] [Google Scholar]
- Misra H. P., Fridovich I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem. 1972 May 25;247(10):3170–3175. [PubMed] [Google Scholar]
- Richardson J. S., Thomas K. A., Richardson D. C. Alpha-carbon coordinates for bovine Cu,Zn superoxide dismutase. Biochem Biophys Res Commun. 1975 Apr 21;63(4):986–992. doi: 10.1016/0006-291x(75)90666-x. [DOI] [PubMed] [Google Scholar]
- Richardson J., Thomas K. A., Rubin B. H., Richardson D. C. Crystal structure of bovine Cu,Zn superoxide dismutase at 3 A resolution: chain tracing and metal ligands. Proc Natl Acad Sci U S A. 1975 Apr;72(4):1349–1353. doi: 10.1073/pnas.72.4.1349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigo A., Viglino P., Calabrese L., Cocco D., Rotilio G. The binding of copper ions to copper-free bovine superoxide dismutase. Copper distribution in protein samples recombined with less than stoicheiometric copper ion/protein ratios. Biochem J. 1977 Jan 1;161(1):27–30. doi: 10.1042/bj1610027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rotilio G., Calabrese L., Bossa F., Barra D., Agrò A. F., Mondovì B. Properties of the apoprotein and role of copper and zinc in protein conformation and enzyme activity of bovine superoxide dismutase. Biochemistry. 1972 May 23;11(11):2182–2187. doi: 10.1021/bi00761a027. [DOI] [PubMed] [Google Scholar]



