Abstract
The circadian system, driven by the suprachiasmatic nucleus (SCN), regulates properties of cardiovascular function. The dysfunction of this timing system can result in cardiac pathology. The neuropeptide vasoactive intestinal peptide (VIP) is crucial for circadian rhythms in a number of biological processes including SCN electrical activity and wheel running behavior. Anatomic evidence indicates that SCN neurons expressing VIP are well positioned to drive circadian regulation of cardiac function through interactions with the autonomic centers. In this study, we tested the hypothesis that loss of VIP would result in circadian deficits in heart rate (HR) and clock gene expression in cardiac tissue. We implanted radiotelemetry devices into VIP-deficient mice and wild-type (WT) controls and continuously recorded HR, body temperature, and cage activity in freely moving mice. Under light-dark conditions, VIP-deficient mice displayed weak rhythms in HR, body temperature, and cage activity, with onsets that were advanced in phase compared with WT mice. Similarly, clock gene expression in cardiac tissue was rhythmic but phase advanced in mutant mice. In constant darkness, the normal circadian rhythms in HR were lost in VIP-deficient mice; however, most mutant mice continued to exhibit circadian rhythms of body temperature with shortened free-running period. The loss of VIP altered, but did not abolish, autonomic regulation of HR. Analysis of the echocardiograms did not find any evidence for a loss of cardiac function in VIP-deficient mice, and the size of the hearts did not differ between genotypes. These results demonstrate that VIP is an important regulator of physiological circadian rhythmicity in the heart.
Keywords: heart rate, body temperature
circadian rhythms are an endogenous timing mechanism that coordinates behavioral, physiological, and biochemical processes with the 24-h environment of light and dark (55). The site of the core circadian clock lies in a bilaterally paired nucleus located in the hypothalamus called the suprachiasmatic nucleus (SCN). Neurons from this region have the intrinsic ability to generate circadian rhythms in electrical activity, secretion, and metabolism. These intrinsic rhythms are generated by a cell-autonomous molecular feedback loop driving rhythmic transcription and translation of key clock genes such as period (Per1 and Per2), cryptochrome (Cry1 and Cry2), clock (Clk), and Bmal1 (27).
Recently, it has become clear that mammals exhibit robust circadian rhythms in cardiovascular function that are dependent on signals from the SCN. Cardiovascular parameters such as heart rate (HR) and blood pressure are elevated during periods of wake and depressed during periods of sleep in anticipation of the level of physical activity and physiological demands of the organism (16). Cardiac tissue also exhibits rhythmic expression of clock genes (17, 30, 38, 45). The function of such oscillations in the heart is unknown, but a reasonable assumption is that these molecular oscillations serve to gate information from the SCN to heart-specific rhythmic outputs. Circadian rhythms of HR and clock gene expression in the heart disappear when the SCN is lesioned (32, 48, 63).
In humans, the onset of cardiovascular events such as myocardial ischemia, infarctions, and arrhythmias cluster around the morning period, suggesting an influence by the circadian system (22, 29, 36, 37). Furthermore, disruptions in the circadian system caused by shiftwork is associated with an increased incidence of cardiovascular disease and mortality (7, 26, 54, 59). Work using animal models supports the view that circadian rhythmicity may be a critical aspect of cardiovascular health. Circadian perturbations have been shown to exacerbate heart disease and decrease survival in cardiomyopathic hamsters (31, 32, 40). It has also been shown that the deletion of mouse Per2 in all tissues provided a protective measure against myocardial infarction (61). These studies suggest a complex interaction between circadian rhythms and cardiovascular function exists that influences cardiovascular disease progression.
The neuropeptide VIP and its receptor VPAC2R are critical for normal circadian function. Loss of VIP signaling has been shown to abolish electrical rhythmicity in a subgroup of SCN neurons, whereas those that remain rhythmic exhibit a decrease in cellular coupling (2, 6, 34, 62). Many SCN neurons that project to extra-SCN tissues and peripheral outputs are VIPergic, and, therefore, VIP may be required for communication between the SCN and peripheral tissues to drive rhythms in behavior and other biological processes (3, 23, 52). Indeed, studies (4, 11, 28, 39) of circadian output in VIP-deficient mice have demonstrated that VIP is critical for normal circadian rhythms in wheel running behavior, metabolic processes, and plasma corticosterone levels. This study examined the effect of loss of VIP on rhythms and function of the heart. In addition, we concurrently measured rhythms of body temperature and cage activity, which enabled a comparison of the effects of loss of VIP on various physiological measurements.
MATERIALS AND METHODS
Experimental animals.
To study the effects of loss of VIP on circadian rhythms of HR, body temperature, and cage activity, we used adult male C57BL/6J mice lacking the genes encoding for VIP and peptide histidine-isoleucine (VIP−/− mice). As controls, littermate or age-matched mice from other litters were used [wild-type (WT) mice]. All animal experimentations were approved by and conducted in accordance with recommendations and guidelines of the Division of Laboratory Animals of the University of California (Los Angeles, CA) and the National Institutes of Health.
Mice were placed in a cage in the absence of a running wheel. Under light-dark (LD) conditions, Zeitgeber (ZT) 0 was defined as the time of lights on and ZT 12 as the time of lights off. Under constant darkness (DD), circadian time (CT) 12 was defined as the onset of increased activity (point of half-maximal rise) in HR, body temperature, or cage activity. In VIP-deficient mice, we often found circadian arrhythmicity of HR and cage activity but concurrently detected rhythms in body temperature in the same animal. To produce group mean waveforms and analyze subjective day-night differences in these cases, we used the onset of body temperature to define CT 12 for HR and cage activity.
Telemetry measurements.
WT and VIP-deficient mice (3–4 mo) were surgically implanted with a wireless radiofrequency transmitter (ETA-F20, Data Sciences, St. Paul, MN). Mice were housed in individual cages in the absence of a running wheel. Cages were placed atop telemetry receivers (Data Sciences) in a light- and temperature-controlled chamber. Standard rodent chow was provided ad libitum. Data collection began 2 wk postsurgery to allow mice to recover in the 12:12 LD cycle. We recorded 20 s of ECG and measured average body temperature and cage activity every 10 min for ∼15 days. Lights were then turned off, and measurements continued under DD conditions for another 15–20 days. HR was extrapolated from ECG waveforms using the RR interval. Upon completion of LD and DD recordings, mice (5–6 mo old) were used for the light exposure and exercise experiments described below.
ECG and circadian analysis.
A 20-s ECG recording every hour was used to calculate ECG intervals (RR, PR, QRS, QT, and QTc) using Ponemah Analysis Software (Data Sciences) at ZT 2–4 and ZT 14–16 time points (LD conditions). HR variability (HRV) in the time domain (SD of all normal RR intervals) was calculated using the Data Science analysis software. Circadian rhythms of HR, body temperature, and cage activity were analyzed by periodogram analysis combined with a χ2-test (El Temps software, Barcelona, Spain). The strongest amplitude (= power) of periodicities (within 20- and 31-h limits) was compared between WT and VIP-deficient mice. The amplitude of the χ2-test determined the period of a rhythm and its robustness.
Acute effects of light.
To examine the acute, photic regulation of HR, we adapted a protocol previously described in rats (48). Six days of 8-h HR recording from ZT 13 to ZT 20 were averaged and served as controls. On the seventh day, mice were exposed to light starting at ZT 14 and then returned to dark conditions at ZT 15. Recording continued until HR returned to baseline levels. During this experiment, ECG, body temperature, and cage activity data were recorded every minute.
Acute effects of exercise.
Baseline HR was recorded for 20 min before forced exercise. Mice were placed on a motorized running wheel (Rota Rod, Ugo Basile Comerio, Italy) for 20 min at a constant speed of ∼4 m/min. Recording continued for 20 min after exercise. Forced exercise was administered at ZT 2 and ZT 14, and an infrared viewer (FJW Industries) was used to visualize the mice in the dark. Data were recorded continuously.
Echocardiography and heart size.
Middle-aged (12 mo) WT and VIP-deficient mice were subjected to functional analysis using two-dimensional, M-mode echocardiography, and spectral Doppler images were acquired by a Siemens Acuson Sequoia C256 equipped with a 15L8 15-MHz probe (Siemens Medical Solutions, Mountain View, CA). Mice were lightly anesthetized with 1% isoflurane, and HRs were maintained between 450 and 600 beats/min during the procedure. Parameters measured included the following: ventricular septal thickness, end-diastolic diameter, posterior wall thickness, end-systolic diameter, aortic ejection time, left ventricular (LV) fractional shortening (in %), LV ejection fraction, LV mass, and the early diastole-to-atrial contraction ratio. Heart weights and tibia lengths were measured from middle-aged mice (12 mo) perfused with paraformaldehyde.
Real-time PCR of clock genes.
RNA extraction and real-time PCR were carried out as previously described (28). Briefly, RNA was extracted from the heart tissue of young mice (3–6 mo) using the TRIzol (Invitrogen) procedure and treated with DNase (Turbo DNA-free, Ambion, Austin, TX). cDNA was produced from 1 μg of total mRNA (High-Capacity cDNA Reverse Transcription Kit, Applied Biosystems, Foster City, CA), and an equivalent of 50 ng of starting total RNA was used for real-time PCR using SYBR green (SYBR Green PCR Master Mix, Applied Biosystems). The oligonucleotide primer sequences used were as follows: Per2, sense 5′-GGGCATTACCTCCGAGTATA-3′ and antisense 5′-GGCCACTTGGTTAGAGATGTA-3′; Bmal1, sense 5′-CTCAACCATCAGCGACTTCA-3′ and antisense 5′-CTGCCTTTCCTCTTGCGATT-3′; and Gapdh, sense 5′-GGCCTTCCGTGTTCCTAC-3′ and antisense 5′-TGTCATCATACTTGGCAGGTT-3′. Primers for Per2 were designed using Oligo6 (Molecular Biology Insights, Cascade, CO) and Mfold programs (65) to cross intron-exon boundaries and prevent contaminants in the subsequent RT-PCR. Relative levels of Per2 and Bmal1 transcripts were determined using the 2−ΔΔCt method (where Ct is threshold cycle) using Gapdh as the normalizing reference gene. We sampled mRNA at ZT 2, 6, 10, 14, 18, and 22 so that only large phase differences could be detected. The time of peak expression was determined for each gene and compared between WT and VIP-deficient mice.
Statistics.
Genotypic comparisons were analyzed using a Student's t-test, whereas day/night comparisons were analyzed using a paired Student's t-test. We used Student's t-tests to compare the onset of HR, body temperature, and cage activity between WT and VIP-deficient mice. We also used Student's t-tests to compare maximum HR during forced exercise, HR responses to light exposure between WT and VIP-deficient mice, echocardiogram parameters, and heart weight-to-tibia length ratios. To analyze clock gene expression, we used a one-way ANOVA statistical test. Data were tested for normal distribution and variance, but in cases where the data did not pass these tests, we used nonparametric statistical tests (e.g., Wilcoxon signed-rank test or Mann-Whitney rank-sum test) to determine significance. Statistical analyses were performed using Sigma Stat 3.5 software (San Jose, CA). Appropriate t-, Z-, F-, or H-statistics are reported, including the degrees of freedom for each analysis.
RESULTS
Analysis of ECG features.
Surgical implantation of a telemetric device into the abdomen allowed for the continuous recording of ECG waveforms (Fig. 1A), body temperature, and cage activity in freely moving WT and VIP-deficient mice. We compared and analyzed a number of ECG features (RR, PR, QRS, QT, and QTc intervals; Fig. 1B) between WT and VIP-deficient mice under LD conditions to detect any day/night and genotypic differences (Table 1). WT mice displayed significant day/night differences in the RR (Z5 = 2.20, P = 0.031), PR (Z5 = 2.20, P = 0.031), and QRS (t5 = 3.05, P = 0.029) intervals, whereas VIP-deficient mice showed differences only in the PR interval (t4 = 3.67, P = 0.021). A significant elongation of the QTc interval during the day period (t9 = 2.37, P = 0.042) was detected in VIP-deficient mice compared with WT mice. These results indicate a disruption in the diurnal regulation of HR and a number of ECG intervals in VIP-deficient mice. Furthermore, the significant elongation of the QTc interval during the light phase in VIP-deficient mice suggests changes in the regulation of electrical activity, particularly in the repolarization of the heart.
Fig. 1.
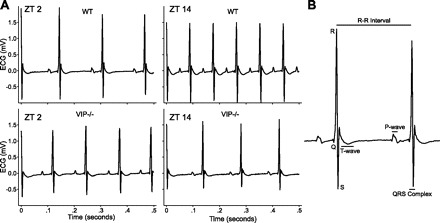
In vivo ECG recordings from freely behaving mice. A: representative examples of ECG tracings from wild-type (WT; top) and VIP-deficient (bottom) mice at Zeitgeber (ZT) 2 and ZT 14. B: magnified example of an ECG tracing labeled with features and intervals of the waveform.
Table 1.
Average duration of ECG features (RR, PR, QRS, QT, and QTc intervals) in WT and VIP-deficient mice under LD conditions
| WT Mice | VIP-Deficient Mice | |
|---|---|---|
| RR, ms | ||
| Day | 129.9 ± 5.5 | 117.5 ± 4.3 |
| Night | 107.6 ± 2.8* | 111.2 ± 6.8 |
| PR, ms | ||
| Day | 39.3 ± 1.1 | 37.4 ± 0.6 |
| Night | 36.0 ± 0.5* | 35.5 ± 0.9* |
| QRS, ms | ||
| Day | 14.1 ± 0.4 | 14.3 ± 0.5 |
| Night | 13.0 ± 0.6* | 13.2 ± 0.8 |
| QT, ms | ||
| Day | 41.9 ± 4.2 | 54.0 ± 3.8 |
| Night | 45.4 ± 3.6 | 53.0 ± 4.9 |
| QTc, ms | ||
| Day | 118.7 ± 13.0 | 159.5 ± 10.5† |
| Night | 139.8 ± 10.5 | 160.6 ± 14.2 |
Values are means ± SE; n = 6 wild-type (WT) mice and 5 VIP-deficient mice. ECG intervals at two time points, day [Zeitgeber (ZT) 2–4] and night (ZT 14–16) were calculated and averaged over 8 days.
Significant difference in intervals between day and night (P < 0.05), as analyzed by a paired Student's t-test;
significant difference in intervals between WT and VIP-deficient mice (P < 0.05), as analyzed by an unpaired Student's t-test.
Circadian analysis of HR, body temperature, and cage activity.
To examine the circadian component of HR, body temperature, and cage activity of individual animals, we plotted the raw data to produce graphs similar to an actogram (Fig. 2, A–C). The raw data were subjected to periodogram analysis (Table 2), and the resulting amplitude was used as an index of rhythm strength. To examine rhythms at a population level, we produced group mean waveforms under LD and DD conditions (Fig. 2, D–F) and compared average values between the day or subjective day (ZT or CT 0–11) and the night or subjective night (ZT or CT 12–23). Under LD conditions, WT mice exhibited robust daily rhythms of HR, body temperature, and cage activity (Table 2) that were synchronized to the LD cycle. The onset of these measurements occurred minutes after lights off (ZT 12): HR onset, 11.5 ± 20 min; body temperature onset, 27 ± 17 min; and cage activity onset, 23.9 ± 12 min (Fig. 2, D–F). Significant day/night differences in HR (t6 = −11.54, P < 0.001), body temperature (t6 = −8.693, P < 0.001), and cage activity (t6 = −5.238, P = 0.002) were detected (Table 3). When WT mice were placed under DD conditions, circadian rhythms of HR, body temperature, and cage activity remained robust with a free-running period of 23.7 ± 0.1 h (Fig. 2 and Table 2). Subjective day/night differences in average HR (t6 = 9.085, P < 0.001), body temperature (t6 = −19.684, P < 0.001), and cage activity (t6 = −5.588, P < 0.001) remained significant (Table 3).
Fig. 2.
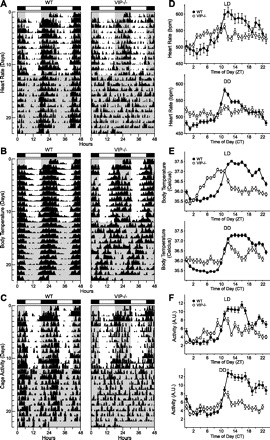
Daily pattern of heart rate [HR; in beats/min (bpm)], body temperature, and cage activity [in arbitrary units (AU)] in WT and VIP-deficient mice. Double-plotted raster plots of HR (A), body temperature (B), and cage activity (C) from WT (left) and VIP-deficient mice (right) were created. Mice were trained in 12:12 light-dark (LD) conditions and then placed in constant darkness (DD). Shaded regions indicated when mice were in the dark. Each horizontal row represents a 24-h period that was plotted twice, and each day/row was plotted in succession. Ten days of HR, body temperature, and cage activity were averaged to produce group mean waveforms of HR (D), body temperature (E), and cage activity (F) for WT (n = 7) and VIP-deficient (n = 8) mice under LD (top) and DD (bottom) conditions. All of the WT mice showed strong diurnal and circadian rhythms of HR, body temperature, and cage activity (7 of 7 mice) under LD and DD conditions. VIP-deficient mice displayed dampened diurnal rhythms in HR (6 of 8 mice), body temperature (8 of 8 mice), and cage activity (7 of 8 mice) under LD conditions. Under DD conditions, VIP-deficient mice displayed a dampened circadian rhythm of body temperature (7 of 8 mice) and cage activity (2 of 8 mice). VIP-deficient mice did not display a circadian rhythm of HR under DD conditions (0 of 8 mice), as assessed by periodogram analysis.
Table 2.
Periodogram analysis using 10 days of HR, body temperature, and cage activity data
| LD Conditions |
DD Conditions |
|||
|---|---|---|---|---|
| WT mice | VIP-deficient mice | WT mice | VIP-deficient mice | |
| HR | 28.6 ± 2.4 | 12.0 ± 0.8* | 24.5 ± 3.0 | 10.0 ± 0.5* |
| Body temperature | 70.5 ± 4.1 | 32.5 ± 3.4* | 75.7 ± 3.3 | 17.7 ± 1.4* |
| Cage activity | 24.5 ± 3.5 | 11.3 ± 0.8* | 23.3 ± 2.3 | 10.9 ± 0.9* |
Values are means ± SE. LD, light-dark; DD, constant darkness; HR, heart rate. Peak amplitudes (= power) were compared between WT (n = 7) and VIP-deficient mice (n = 8) under LD and DD conditions. Genotypic comparisons were analyzed by Student's t-test (
P < 0.05).
Table 3.
Day/night differences in HR, body temperature, and cage activity under LD and DD conditions
| WT Mice |
VIP-Deficient Mice |
|||
|---|---|---|---|---|
| Day | Night | Day | Night | |
| LD conditions | ||||
| HR, beats/min | 508 ± 15 | 580 ± 16† | 517 ± 14 | 532 ± 16 |
| Body temperature, °C | 35.8 ± 0.13 | 37.2 ± 0.09† | 36.2 ± 0.06 | 36.4 ± 0.13 |
| Cage activity | 3.6 ± 0.3 | 8.7 ± 1.1* | 4.2 ± 0.4 | 5.6 ± 0.8* |
| Subjective day | Subjective night | Subjective day | Subjective night | |
|---|---|---|---|---|
| DD conditions | ||||
| HR, beats/min | 486 ± 10 | 549 ± 10† | 504 ± 6 | 510 ± 7 |
| Body temperature, °C | 35.6 ± 0.05 | 37.2 ± 0.09† | 36.1 ± 0.07 | 36.5 ± 0.05† |
| Cage activity | 3.4 ± 0.4 | 9.3 ± 1.3† | 4.1 ± 0.1 | 5.4 ± 0.1* |
Values are means ± SE; n = 7 WT mice and 8 VIP-deficient mice. Day/subjective day (ZT or CT 0–11) and night/subjective night (ZT or CT 12–23) values of HR, body temperature, and cage activity were averaged over 10 days and compared using a paired Student's t-test (* P < 0.05; † P < 0.001).
VIP-deficient mice displayed weak diurnal rhythms in HR, body temperature, and cage activity (Table 2). The onsets of all three physiological measurements were significantly advanced in VIP-deficient mice compared with WT mice (HR: −215 ± 71.8 min, T = 56.6, P = 0.035; body temperature: −244 ± 50.6 min, t12 = 7.165, P < 0.001; and cage activity: −194 ± 51.8 min, T = 69.06, P < 0.001), indicating an advanced phase angle of entrainment (Fig. 2, D–F). Day/night differences in cage activity (Z7 = 2.521, P = 0.008) were significant in VIP-deficient mice, but no differences were detected for HR (t7 = 2.325, P = 0.058) and body temperature (t7 = 1.497, P = 0.18). Under DD conditions, HR was arrhythmic in all VIP-deficient mice tested (8 of 8 mice; Table 2). Rhythms in cage activity were detected in only two of eight mice, and these rhythms were weak with a shortened free-running period (22.3 ± 0.17 h). Most VIP-deficient mice maintained circadian rhythms in body temperature (7 of 8 mice) but with shortened periodicity (22.7 ± 0.25 h). To produce average waveforms of HR and activity despite the absence of a detectable rhythm in individual mice, we used the half-maximal rise of body temperature, which remained rhythmic in VIP-deficient mice, to determine CT 12 for HR and cage activity. We detected significant subjective day/night differences in body temperature (t7 = −6.693, P < 0.001) and cage activity (t7 = −3.636, P = 0.008), but no difference was detected for HR (t7 = −1.511, P = 0.175; Table 3). Under both LD and DD conditions, there were no significant differences in average HR, body temperature, and cage activity between WT and VIP-deficient mice except for a significant decrease of average cage activity in VIP-deficient mice under DD conditions (WT mice: 6.47 ± 0.73 arbitrary units and VIP-deficient mice: 3.75 ± 0.322 arbitrary units, T = 76, P = 0.021). These data indicate that the temporal patterning of HR, body temperature, and cage activity is impacted in VIP-deficient mice.
HR and body temperature rhythms during periods of no/low activity.
While levels of motor activity and HR are closely coupled, previous work has attempted to separate these parameters by measuring HR during times of low activity (48, 49). We undertook a similar analysis and extracted HR and body temperature data during periods of low/no activity (<1% of average activity). In WT mice, there were still clear rhythms in HR and body temperature under both LD and DD conditions during times at which the mice were inactive (Supplemental Fig. 1).1 In contrast, VIP-deficient mice showed no day/night differences in HR under LD or DD conditions in the absence of activity. However, the rhythms in body temperature in VIP-deficient mice persisted under these conditions. These data indicate that, even if we factor out acute activity, we can still measure clear rhythms in body temperature but not HR in VIP-deficient mice.
Rhythms of HRV.
HRV is an index of the balance of sympathovagal signals to the heart and measures the variability of the time between individual heart beats (33). We assessed the temporal patterning of neural signals to the heart by calculating HRV from WT and VIP-deficient mice under LD and DD conditions (Fig. 3). WT mice exhibited significant day/night differences in HRV under LD conditions (t6 = 3.60, P = 0.01) and significant subjective day/night differences under DD conditions (t6 = 4.582, P = 0.005). VIP-deficient mice did not exhibit a day/night difference in HRV under LD conditions (t7 = 1.88, P = 0.10). Under DD conditions, there was no rhythm evident in the average waveform (Fig. 3B); however, if we used body temperature rhythm to define CT, we did find a low amplitude rhythm of HRV in VIP-deficient mice (t7 = 3.96, P = 0.005; Fig. 3D). HRV was higher during the subjective day compared with the subjective night in all VIP-deficient mice (Supplemental Fig. 2), although the difference was small compared with WT mice. Average HRV (measured over 24 h) did not vary between the genotypes, so the autonomic nervous system (ANS) still functioned but the temporal patterning was altered. These results suggest that circadian regulation of sympathovagal signals is dampened but not completely eliminated in VIP-deficient mice.
Fig. 3.
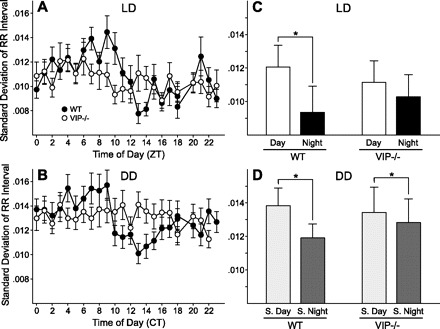
Daily pattern of HR variability (HRV) in WT and VIP-deficient mice. HRV was determined by calculating the variance of the time between individual beats, also known as the interbeat interval (IBI). Group mean waveforms of HRV using 10 days of data were produced for WT (n = 7) and VIP-deficient (n = 8) mice under LD (A) and DD (B) conditions. WT mice displayed significant day/night differences under LD (C) and DD (D) conditions. No significant day/night differences were measured for VIP-deficient mice under LD conditions (C), but significance was detected under DD conditions (D). HRV was consistently decreased during the subjective night (S. night) compared with the subjective day (S. day) in both WT and VIP-deficient mice. These data were analyzed using a paired Student's t-test (*P < 0.05).
Acute regulation of HR.
In addition to a circadian component, HR is acutely regulated via the ANS to allow the organism to respond to various external stimuli. To determine whether the acute regulation of HR is intact in VIP-deficient mice, we exposed mice to light (1 h) at ZT 14. In WT mice, the administration of light resulted in a precipitous drop in HR followed by a rapid recovery to baseline levels (Fig. 4A). In VIP-deficient mice, HR decreased in response to light administration (Fig. 4B), with an amplitude that was not significantly different than WT mice (WT mice: 175 ± 20 beats/min, n = 7; and VIP-deficient mice: 201 ± 27 beats/min, n = 6, t11 = 0.810, P = 0.44) but at a rate that was significantly delayed (rate to maximum response: WT mice, 36 ± 4 min and VIP-deficient mice, 113 ± 10 min; t11 = 8.049, P < 0.001). The recovery rate was determined by measuring the duration from the time of maximum response to the return of HR to baseline levels, which we found was not significantly different between WT (43.6 ± 23 min) and VIP-deficient mice (32.21 ± 21 min). This result suggests that the acute HR response is present in VIP-deficient mice, but the loss of VIP may be delaying the transmission of light information to the heart.
Fig. 4.
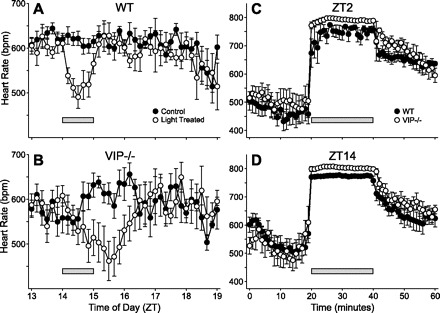
Acute regulation of HR in WT and VIP-deficient mice. A: light exposure (1 h) administered at ZT 14 elicited a precipitous drop of HR in WT mice (n = 7). B: VIP-deficient mice (n = 6) responded to the light treatment, but at a rate that was significantly delayed (P < 0.001). Forced exercise triggered the significant increase of HR in WT (n = 5) and VIP-deficient (n = 5) mice at ZT 2 (C) and ZT 14 (D) time points. VIP-deficient mice exhibited an increase in HR that was larger than WT controls. Bars indicate the times of treatment.
We also challenged the cardiovascular system to respond to an exercise protocol. HRs of WT and VIP-deficient mice increased when mice were forced to run at two time points: ZT 2 and ZT 14 (Fig. 4, C and D). We compared HRs of WT and VIP-deficient mice during exercise (20 min in duration) at both time points (WT mice: ZT 2, 751 ± 6 beats/min and ZT 14, 790 ± 2 beats/min; VIP-deficient mice: ZT2, 773 ± 2 beats/min and ZT 14, 801 ± 2 beats/min). VIP-deficient mice exhibited significantly higher HRs during exercise in both the light (t10 = 2.43, P = 0.036) and dark (t10 = 3.40, P = 0.007) compared with WT mice. These data suggest that acute signaling to the heart is intact in VIP-deficient mice, although their response to exercise is larger than WT mice.
Cardiac function and morphology.
To determine whether loss of VIP may have functional consequences on the heart, we performed echocardiograms on WT and VIP-deficient mice at 12 mo of age. Our results showed no significant differences in any of the measurements by this assay between the various groups (Table 4), indicating that the hearts of VIP-deficient mice are functionally intact. Previous work has suggested that the hearts of these mice exhibit hypertrophy (44), so we calculated the heart weight-to-tibia length ratio to provide an index for hypertrophy (Supplemental Fig. 3, A and B). We did not find significant differences in the heart weight-to-tibia length ratio between WT and VIP-deficient mice at this age.
Table 4.
Echocardiogram analysis of young WT and VIP-deficient mice
| WT Mice | VIP-Deficient Mice | |
|---|---|---|
| HR, beats/min | 529 ± 17 | 527 ± 34 |
| Ventricular septal thickness, mm | 0.60 ± 0.02 | 0.65 ± 0.02 |
| End-diastolic diameter, mm | 4.37 ± 0.05 | 4.43 ± 0.12 |
| Posterior wall thickness, mm | 0.61 ± 0.02 | 0.62 ± 0.03 |
| End-systolic diameter, mm | 2.94 ± 0.09 | 2.88 ± 0.15 |
| Aortic ejection time, ms | 51.4 ± 1.8 | 55.5 ± 1.1 |
| LV fractional shortening, % | 32.7 ± 2.0 | 35.1 ± 2.7 |
| Circumferential fiber shortening velocity | 6.4 ± 0.5 | 6.3 ± 0.5 |
| LV ejection fraction | 67.2 ± 2.6 | 70.6 ± 3.4 |
| LV mass | 96.0 ± 3.1 | 105.5 ± 4.6 |
| E | 0.99 ± 0.18 | 0.71 ± 0.02 |
| A | 0.53 ± 0.10 | 0.41 ± 0.04 |
| E/A | 1.87 ± 0.05 | 1.80 ± 0.14 |
Values are means ± SE; n = 7 WT mice and 4 VIP-deficient mice. LV, left ventricular; E, early diastole contraction; A, atrial contraction. There were no significant differences between the genotypes.
Clock gene expression in the heart.
Most peripheral tissues, including the heart, possess a molecular clock and display circadian rhythms in core clock gene expression. To determine the condition of the molecular clock in the hearts of VIP-deficient mice, we measured mRNA expression levels of the clock genes Per2 and Bmal1 under LD conditions at ZT 2, 6, 10, 14, 18, and 22 (Fig. 5). As measured by one-way ANOVA, Per2 and Bmal1 were rhythmic in both WT mice (Per2: H5 = 21.08, P < 0.001; and Bmal1: H5 = 19.56, P = 0.002) and VIP-deficient mice (Per2: H6 = 21.54, P = 0.001; and Bmal1: H5 = 18.43, P = 0.002). We also found a clear advance in the phase of peak expression of both genes (Per2 peak phase: WT mice, ZT 13 and VIP-deficient mice, ZT 6; t6 = 3.656, P = 0.011; Bmal1 peak phase: WT mice, ZT 1 and VIP-deficient mice, ZT 20; T7 = 10.00, P = 0.029) compared with WT controls. These results indicate that the loss of VIP does not disrupt molecular rhythms in the heart but alters the phase of these rhythms under LD conditions.
Fig. 5.
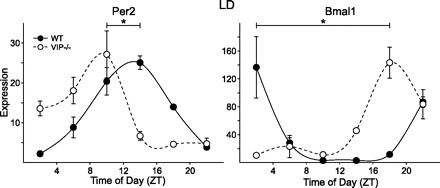
Expression profiles of clock genes period 2 (Per2) and Bmal1 in heart tissues of WT and VIP-deficient mice under LD conditions using RT-PCR techniques. Each time point shows mean results of 3–4 hearts. A one-way ANOVA statistical test was used to determine whether there were significant differences in expression. Bars indicate differences in peak expression between WT and VIP-deficient mice; results were analyzed using Student's t-test (*P < 0.05).
DISCUSSION
In this set of experiments, we used a telemetry system to measure the impact of loss of VIP on daily and circadian rhythms in HR, body temperature, and cage activity in freely moving mice. We found that loss of VIP differentially disrupted these physiological and behavioral rhythms. Most dramatically, VIP-deficient mice exhibited weak rhythms in HR under LD conditions, which were completely lost under DD conditions, confirming a central role for VIP in the circadian regulation of HR. Rhythms of cage activity in VIP-deficient mice were weak under LD conditions, and most mice were arrhythmic under DD conditions. In contrast, VIP-deficient mice exhibited daily rhythms in body temperature, albeit with an altered phase compared with WT controls, and continued to display robust rhythms in body temperature under DD conditions with a shortened free-running period. The changes in profile and the reduction in the amplitude of the average waveform of physiological outputs may also be impacted by the imprecise onset of activity observed in VIP-deficient mice (11). All three peripheral outputs displayed onsets (half-maximal rise) that were phase advanced under an LD cycle, which has also been described for feeding behavior and metabolism in VIP-deficient mice (4). This phase advance in onset under LD conditions was absent when activity was measured using a running wheel (11). Furthermore, the wheel appears to improve rhythms in activity under DD conditions, raising the possibility that running on the wheel may be able to temporally organize activity rhythms in VIP-deficient mice (11, 41). Overall, it appears that the impact of loss of VIP on circadian rhythms varies with the output being measured. Within the SCN, genetic loss of VIP or VPAC2R disrupts circadian rhythms in neural activity in the SCN population (6, 12), where the loss of VIP decreases the number of electrically rhythmic SCN neurons (2) and appears to weaken the coupling among the remaining rhythmic SCN neurons (2, 10). Circadian regulation of peripheral tissues may be specialized at the level of the SCN, an idea that has been suggested by a number of studies (24, 25). In the case of VIP-deficient mice, SCN neurons that regulate body temperature may be among those that do not require VIP to generate daily rhythms. In contrast, arrhythmic physiological processes in VIP-deficient mice, such as the regulation of plasma corticosterone levels (28) and HR, may have circuitry coupled to the arrhythmic SCN neurons. Running on the wheel may provide feedback to the SCN that aids in the coupling of individual neurons. Models considering the role of VIP as a coupling molecule within the SCN (58, 60) will need to take into account the findings that not all outputs are arrhythmic in VIP-deficient mice.
The telemetry system enabled us to record ECG waveforms from WT and VIP-deficient mice under LD conditions. While most of the ECG parameters were not different between WT and VIP-deficient mice, there was a significant increase in the duration of the QTc interval in VIP-deficient mice during the light phase. A long QTc interval indicates a delay in the repolarization of the ventricle after contraction, which is a risk factor for ventricular arrhythmias and sudden death in humans (42, 46). In most cases, long QT syndrome is caused by an ion channelopathy associated with loss-of-function mutations in genes encoding for repolarizing K+ channels or proteins that interact with these channels (21). Expression levels of K+ channels have been shown to fluctuate depending on the time of day (53, 64), and pharmacological administration of nicroandil, a K+ channel opener, can selectively disrupt rhythms in HR but not body temperature or cage activity (15). VIP regulates K+ channels in a variety of tissues, including the colon, coronary arteries, and trachea (47, 51, 57). Within the heart, VIP has effects on the neuroexcitability of intracardiac neurons and can increase the rate and strength of contraction (1, 9, 18). Based on these data, a reasonable hypothesis is that the disruption of circadian rhythms in HR in VIP-deficient mice may be in part due to the misregulation of K+ channel activity within the heart.
Neurons in the SCN containing VIP are well positioned to drive the rhythms in cardiac output through the regulation of the ANS (8, 25). Previous anatomic studies have shown that VIP is expressed in SCN efferents projecting onto preautonomic neurons of the PVN (56) as well as in the parasympathetic postganglion neurons that innervate the heart (18). To assess the ANS and determine the effect of the loss of VIP, we examined HRV in WT and VIP-deficient mice. In WT mice, we found clear evidence for circadian regulation in HRV, whereas this rhythmicity appeared to be lost in the VIP-deficient mice. However, a closer inspection using the animal's own rhythm in body temperature to mark phase indicated that a low amplitude rhythm in HRV persisted even in the absence of VIP. Furthermore, the acute autonomic regulation of HR is still functional in VIP-deficient mice, as determined by their response to light exposure and exercise, although these responses were different compared with WT. In rodents, the light-evoked decrease in HR is largely driven by an increase in parasympathetic tone (48), whereas the exercise-evoked increase in HR is driven by sympathetic activity (35). In the present study, we found that VIP-deficient mice were able to decrease their HR in response to light to the same level as WT mice, albeit at a more sluggish rate. VIP-deficient mice also showed a rapid exercise-induced increase in HR that was even larger than WT controls. VIP's localization in the parasympathetic neurons may result in some reduction in parasympathetic function in mutant mice. Overall, the HRV data as well as the ability of the mice to respond to environmental challenges suggest that the autonomic regulation of HR is altered but still functional in the absence of VIP.
Interacting molecular feedback loops driving rhythmic transcription and translation of key clock genes such as Per and Bmal1 (20) are at the core of the oscillatory mechanism. Several studies (16, 17, 38, 45) have now shown that cardiac tissue exhibits rhythmic expression of clock genes. These molecular rhythms persist in isolated cardiomyocyte cultures, including persistent circadian oscillations in Per1-driven bioluminescence (13, 14). These data are consistent with the conclusion that cardiovascular tissue contains molecular circadian oscillators that are independent from the SCN. The function of such oscillations in the heart is unknown, but a reasonable assumption is that these molecular oscillations serve to gate information from the SCN to heart-specific rhythmic outputs. Microarray studies (30, 43, 53) have found that hundreds of genes (∼10–15% of total genome) oscillate in the heart. These rhythmic genes code for proteins such as ion channels, neurotransmitter receptors, and proteins involved in Ca2+ flux, which are likely to have substantial effects on function. Under LD conditions, we found robust rhythms in the expression of Per2 and Bmal1 genes within the hearts of WT mice. These genes were also rhythmic in VIP-deficient mice but showed an advancement in the phase of peak expression, which nicely paralleled the advanced onset of HR, body temperature, and cage activity under LD conditions. The data demonstrate that loss of VIP can alter, but not eliminate, the daily rhythms in gene expression that underlie circadian oscillations in this peripheral organ. Previous work (19, 49) reported the loss of clock gene expression rhythms in the SCN of VPAC2R knockout mice, yet these mice continued to display gene expression rhythms in peripheral tissues such as the heart and liver. Given the present data demonstrating that clock gene expression is still rhythmic in the hearts of VIP-deficient mice, the impact of loss of VIP on the central clock and other peripheral organs will need to be revisited.
Several recent studies (5, 31, 32, 40) have found evidence that disruption of the circadian system alone is sufficient to lead to the development of cardiovascular disease in circadian compromised animals. Echocardiogram and heart weight-to-tibia length ratio data indicate that the hearts of VIP-deficient mice are normal in size and function in a normal range despite the loss of rhythms in activity, HR, and corticosterone. Perhaps the presence of rhythms in clock gene expression and body temperature in VIP-deficient mice protect the cardiovascular system in some way. More work on the interrelationships between VIP, disruption of circadian function, and cardiac morphology are clearly needed. We propose that the VIP-deficient mice as well as VPAC2R-deficient mice may provide important models to look at these relationships.
GRANTS
This work was supported by the Neuroendocrinology, Sex Differences, and Reproduction Training Grant from the University of California (Los Angeles, CA) as well as the Training Program in Neural Microcircuits Training Grant from UCLA (to A. Schroeder). In addition, this work was supported by CHDI Foundation Grant A-2702 and American Heart Association Grant 10GRNT430078 (to C. S. Colwell).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
Supplementary Material
ACKNOWLEDGMENTS
The authors acknowledge the intellectual support and suggestions of Dr. Block, Dr. Philipson, and Dr. Waschek.
Footnotes
Supplemental Material for this article is available at the American Journal of Physiology-Heart and Circulatory Physiology website.
REFERENCES
- 1. Accili EA, Redaelli G, DiFrancesco D. Activation of the hyperpolarization-activated current (If) in sino-atrial node myocytes of the rabbit by vasoactive intestinal peptide. Pflugers Arch 431: 803–805, 1996. [DOI] [PubMed] [Google Scholar]
- 2. Aton SJ, Colwell CS, Harmar AJ, Waschek J, Herzog ED. Vasoactive intestinal polypeptide mediates circadian rhythmicity and synchrony in distinct subsets of mammalian clock neurons. Nat Neurosci 8: 476–483, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ban Y, Shigeyoshi Y, Okamura H. Development of vasoactive intestinal peptide mRNA rhythm in the rat suprachiasmatic nucleus. J Neurosci 17: 3920–3931, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bechtold DA, Brown TM, Luckman SM, Piggins HD. Metabolic rhythm abnormalities in mice lacking VIP-VPAC2 signaling. Am J Physiol Regul Integr Comp Physiol 294: R344–R351, 2008. [DOI] [PubMed] [Google Scholar]
- 5. Bray MS, Shaw CA, Moore MW, Garcia RA, Zanquetta MM, Durgan DJ, Jeong WJ, Tsai JY, Bugger H, Zhang D, Rohrwasser A, Rennison JH, Dyck JR, Litwin SE, Hardin PE, Chow CW, Chandler MP, Abel ED, Young ME. Disruption of the circadian clock within the cardiomyocyte influences myocardial contractile function, metabolism, and gene expression. Am J Physiol Heart Circ Physiol 294: H1036–H1047, 2008. [DOI] [PubMed] [Google Scholar]
- 6. Brown T, Colwell CS, Waschek J, Piggins H. Disrupted neuronal activity rhythms in the suprachiasmatic nuclei of vasoactive intestinal polypeptide-deficient mice. J Neurophysiol 97: 2553–2558, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Brown DL, Feskanich D, Sánchez BN, Rexrode KM, Schernhammer ES, Lisabeth LD. Rotating night shift work and the risk of ischemic stroke. Am J Epidemiol 169: 1370–1377, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Buijs RM, la Fleur SE, Wortel J, Van Heyningen C, Zuiddam L, Mettenleiter TC, Kalsbeek A, Nagai K, Niijima A. The suprachiasmatic nucleus balances sympathetic and parasympathetic output to peripheral organs through separate preautonomic neurons. J Comp Neurol 464: 36–48, 2003. [DOI] [PubMed] [Google Scholar]
- 9. Chang F, Yu H, Cohen IS. Actions of vasoactive intestinal peptide and neuropeptide Y on the pacemaker current in canine Purkinje fibers. Circ Res 74: 157–162, 1994. [DOI] [PubMed] [Google Scholar]
- 10. Ciarleglio CM, Gamble KL, Axley JC, Strauss BR, Cohen JY, Colwell CS, McMahon DG. Population encoding by circadian clock neurons organizes circadian behavior. J Neurosci 29: 1670–1676, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Colwell CS, Michel S, Itri J, Rodriguez W, Tam J, Lelievre V, Hu Z, Liu X, Waschek JA. Disrupted circadian rhythms in VIP- and PHI-deficient mice. Am J Physiol Regul Integr Comp Physiol 285: R939–R949, 2003. [DOI] [PubMed] [Google Scholar]
- 12. Cutler DJ, Haraura M, Reed HE, Shen S, Sheward WJ, Morrison CF, Marston HM, Harmar AJ, Piggins HD. The mouse VPAC2 receptor confers suprachiasmatic nuclei cellular rhythmicity and responsiveness to vasoactive intestinal polypeptide in vitro. Eur J Neurosci 17: 197–204, 2003. [DOI] [PubMed] [Google Scholar]
- 13. Davidson AJ, London B, Block GD, Menaker M. Cardiovascular tissues contain independent circadian clocks. Clin Exp Hypertens 27: 307–311, 2005. [PubMed] [Google Scholar]
- 14. Durgan DJ, Hotze MA, Tomlin TM, Egbejimi O, Graveleau C, Abel ED, Shaw CA, Bray MS, Hardin PE, Young ME. The intrinsic circadian clock within the cardiomyocyte. Am J Physiol Heart Circ Physiol 289: H1530–H1541, 2005. [DOI] [PubMed] [Google Scholar]
- 15. Gatenbein M, Attolini L, Bruguerolle B. Nicorandil affects diurnal rhythms of body temperature, heart rate and locomotor activity in rats. Eur J Pharmacol 346: 125–130, 1998. [DOI] [PubMed] [Google Scholar]
- 16. Guo YF, Stein PK. Circadian rhythm in the cardiovascular system: chronocardiology. Am Heart J 145: 779–786, 2003. [DOI] [PubMed] [Google Scholar]
- 17. Guo H, Brewer JM, Champhekar A, Harris RB, Bittman EL. Differential control of peripheral circadian rhythms by suprachiasmatic-dependent neural signals. Proc Natl Acad Sci USA 102: 3111–3116, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Halimi F, Piot O, Guize L, Le Heuzey JY. Electrophysiological effects of vasoactive intestinal peptide in rabbit atrium: a modulation of acetylcholine activity. J Mol Cell Cardiol 29: 37–44, 1997. [DOI] [PubMed] [Google Scholar]
- 19. Harmar AJ, Marston HM, Shen S, Spratt C, West KM, Sheward WJ, Morrison CF, Dorin JR, Piggins HD, Reubi JC, Kelly JS, Maywood ES, Hastings MH. The VPAC2 receptor is essential for circadian function in the mouse suprachiasmatic nuclei. Cell 109: 497–508, 2002. [DOI] [PubMed] [Google Scholar]
- 20. Hastings MH, Reddy AB, Maywood ES. A clockwork web: circadian timing in brain and periphery, in health and disease. Nat Rev Neurosci 4: 649–661, 2003. [DOI] [PubMed] [Google Scholar]
- 21. Hedley PL, Jørgensen P, Schlamowitz S, Wangari R, Moolman-Smook J, Brink PA, Kanters JK, Corfield VA, Christiansen M. The genetic basis of long QT and short QT syndromes: a mutation update. Hum Mutat 30: 1486–511, 2009. [DOI] [PubMed] [Google Scholar]
- 22. Hu K, Ivanov PCh, Hilton MF, Chen Z, Ayers RT, Stanley HE, Shea SA. Endogenous circadian rhythm in an index of cardiac vulnerability independent of changes in behavior. Proc Natl Acad Sci USA 101: 18223–18227, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kalsbeek A, Rikkers M, Vivien-Roels B, Pévet P. Vasopressin and vasoactive intestinal peptide infused in the paraventricular nucleus of the hypothalamus elevate plasma melatonin levels. J Pineal Res 15: 46–52, 1993. [DOI] [PubMed] [Google Scholar]
- 24. Kalsbeek A, Palm IF, La Fleur SE, Scheer FA, Perreau-Lenz S, Ruiter M, Kreier F, Cailotto C, Buijs RM. SCN outputs and the hypothalamic balance of life. J Biol Rhythms 21: 458–469, 2006. [DOI] [PubMed] [Google Scholar]
- 25. Kalsbeek A, Perreau-Lenz S, Buijs RM. A network of (autonomic) clock outputs. Chronobiol Int 23: 521–535, 2006. [DOI] [PubMed] [Google Scholar]
- 26. Knutsson A, Akerstedt T, Jonsson BG, Orth-Gomer K. Increased risk of ischaemic heart disease in shift workers. Lancet 2: 89–92, 1986. [DOI] [PubMed] [Google Scholar]
- 27. Ko CH, Takahashi JS. Molecular components of the mammalian circadian clock. Hum Mol Genet 15: R271–R277, 2006. [DOI] [PubMed] [Google Scholar]
- 28. Loh DH, Abad C, Colwell CS, Waschek JA. Vasoactive intestinal peptide is critical for circadian regulation of glucocorticoids. Neuroendocrinology 88: 246–255, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Marler JR, Price TR, Clark GL, Muller JE, Robertson T, Mohr JP, Hier DB, Wolf PA, Caplan LR, Foulkes MA. Morning increase in onset of ischemic stroke. Stroke 20: 473–476, 1989. [DOI] [PubMed] [Google Scholar]
- 30. Martino T, Arab S, Straume M, Belsham DD, Tata N, Cai F, Liu P, Trivieri M, Ralph M, Sole MJ. Day/night rhythms in gene expression of the normal murine heart. J Mol Med 82: 256–264, 2004. [DOI] [PubMed] [Google Scholar]
- 31. Martino TA, Tata N, Belsham DD, Chalmers J, Straume M, Lee P, Pribiag H, Khaper N, Liu PP, Dawood F, Backx PH, Ralph MR, Sole MJ. Disturbed diurnal rhythm alters gene expression and exacerbates cardiovascular disease with rescue by resynchronization. Hypertension 49: 1104–1113, 2007. [DOI] [PubMed] [Google Scholar]
- 32. Martino TA, Oudit GY, Herzenberg AM, Tata N, Koletar MM, Kabir GM, Belsham DD, Backx PH, Ralph MR, Sole MJ. Circadian rhythm disorganization produces profound cardiovascular and renal disease in hamsters. Am J Physiol Regul Integr Comp Physiol 294: R1675–R1683, 2008. [DOI] [PubMed] [Google Scholar]
- 33. Massin MM, Maeyns K, Withofs N, Ravet F, Gérard P. Circadian rhythm of heart rate and heart rate variability. Arch Dis Child 83: 179–182, 2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Maywood ES, Reddy AB, Wong GK, O'Neill JS, O'Brien JA, McMahon DG, Harmar AJ, Okamura H, Hastings MH. Synchronization and maintenance of timekeeping in suprachiasmatic circadian clock cells by neuropeptidergic signaling. Curr Biol 16: 599–605, 2006. [DOI] [PubMed] [Google Scholar]
- 35. Miller TD. Exercise treadmill test: estimating cardiovascular prognosis. Cleve Clin J Med 75: 424–430, 2008. [DOI] [PubMed] [Google Scholar]
- 36. Muller JE, Stone PH, Turi ZG, Rutherford JD, Czeisler CA, Parker C, Poole WK, Passamani E, Roberts R, Robertson T, et al. Circadian variation in the frequency of onset of acute myocardial infarction. N Engl J Med 313: 1315–1322, 1985. [DOI] [PubMed] [Google Scholar]
- 37. Muller JE, Tofler GH, Stone PH. Circadian variation and triggers of onset of acute cardiovascular disease. Circulation 79: 733–743, 1989. [DOI] [PubMed] [Google Scholar]
- 38. Oishi K, Sakamoto K, Okada T, Nagase T, Ishida N. Antiphase circadian expression between BMAL1 and period homologue mRNA in the suprachiasmatic nucleus and peripheral tissues of rats. Biochem Biophys Res Commun 253: 199–203, 1998. [DOI] [PubMed] [Google Scholar]
- 39. Pantazopoulos H, Dolatshad H, David FC. Chronic stimulation of the hypothalamic vasoactive intestinal peptide receptor lengthens circadian period in mice and hamsters. Am J Physiol Regul Integr Comp Physiol 299: R379–R385, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Penev PD, Kolker DE, Zee PC, Turek FW. Chronic circadian desynchronization decreases the survival of animals with cardiomyopathic heart disease. Am J Physiol Heart Circ Physiol 275: H2334–H2337, 1998. [DOI] [PubMed] [Google Scholar]
- 41. Power A, Hughes ATL, Samuels RE, Piggins HD. Rhythm-promoting actions of exercise in mice with deficient neuropeptide signaling. J Biol Rhythms 25: 235–246, 2010. [DOI] [PubMed] [Google Scholar]
- 42. Priori SG, Schwartz PJ, Napolitano C, Bloise R, Ronchetti E, Grillo M, Vicentini A, Spazzolini C, Nastoli J, Bottelli G, Folli R, Cappelletti D. Risk stratification in the long-QT syndrome. N Engl J Med 348: 1866–1874, 2003. [DOI] [PubMed] [Google Scholar]
- 43. Rudic RD, Curtis AM, Cheng Y, FitzGerald G. Peripheral clocks and the regulation of cardiovascular and metabolic function. Methods Enzymol 393: 524–539, 2005. [DOI] [PubMed] [Google Scholar]
- 44. Said SI, Hamidi SA, Dickman KG, Szema AM, Lyubsky S, Lin RZ, Jiang YP, Chen JJ, Waschek JA, Kort S. Moderate pulmonary arterial hypertension in male mice lacking the vasoactive intestinal peptide gene. Circulation 115: 1260–1268, 2007. [DOI] [PubMed] [Google Scholar]
- 45. Sakamoto K, Ishida N. Light-induced phase-shifts in the circadian expression rhythm of mammalian period genes in the mouse heart. Eur J Neurosci 12: 4003–4006, 2000. [DOI] [PubMed] [Google Scholar]
- 46. Sauer AJ, Moss AJ, McNitt S, Peterson DR, Zareba W, Robinson JL, Qi M, Goldenberg I, Hobbs JB, Ackerman MJ, Benhorin J, Hall WJ, Kaufman ES, Locati EH, Napolitano C, Priori SG, Schwartz PJ, Towbin JA, Vincent GM, Zhang L. Long QT syndrome in adults. J Am Coll Cardiol 49: 329, 2007. [DOI] [PubMed] [Google Scholar]
- 47. Sawmiller DR, Ashtari M, Urueta H, Leschinsky M, Henning RJ. Mechanisms of vasoactive intestinal peptide-elicited coronary vasodilation in the isolated perfused rat heart. Neuropeptides 40: 349–355, 2006. [DOI] [PubMed] [Google Scholar]
- 48. Scheer FA, Ter Horst GJ, van Der Vliet J, Buijs RM. Physiological and anatomic evidence for regulation of the heart by suprachiasmatic nucleus in rats. Am J Physiol Heart Circ Physiol 280: H1391–H1399, 2001. [DOI] [PubMed] [Google Scholar]
- 49. Sheward WJ, Maywood ES, French KL, Horn JM, Hastings MH, Seckl JR, Holmes MC, Harmar AJ. Entrainment to feeding but not to light: circadian phenotype of VPAC2 receptor-null mice. J Neurosci 27: 4351–4358, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Sheward WJ, Naylor E, Knowles-Barley S, Armstrong JD, Brooker GA, Seckl JR, Turek FW, Holmes MC, Zee PC, Harmar AJ. Circadian control of mouse heart rate and blood pressure by the suprachiasmatic nuclei: behavioral effects are more significant than direct outputs. PLoS One 5: e9783, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Shuttleworth CW, Koh SD, Bayginov O, Sanders KM. Activation of delayed rectifier potassium channels in canine proximal colon by vasoactive intestinal peptide. J Physiol 493: 651–663, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Sollars PJ, Pickard GE. Restoration of circadian behavior by anterior hypothalamic grafts containing the suprachiasmatic nucleus: graft/host interconnections. Chronobiol Int 15: 513–533, 1998. [DOI] [PubMed] [Google Scholar]
- 53. Storch KF, Lipan O, Leykin I, Viswanathan N, Davis FC, Wong WH, Weitz CJ. Extensive and divergent circadian gene expression in liver and heart. Nature 417: 78–83, 2002. [DOI] [PubMed] [Google Scholar]
- 54. Stoynev AG, Minkova NK. Effect of forward rapidly rotating shift work on circadian rhythms of arterial pressure, heart rate and oral temperature in air traffic controllers. Occup Med Lond 48: 75–79, 1998. [DOI] [PubMed] [Google Scholar]
- 55. Takahashi JS, Hong HK, Ko CH, McDearmon EL. The genetics of mammalian circadian order and disorder: implications for physiology and disease. Nat Rev Genet 9: 764–775, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Teclemariam-Mesbah R, Kalsbeek A, Pevet P, Buijs RM. Direct vasoactive intestinal polypeptide-containing projection from the suprachiasmatic nucleus to spinal projecting hypothalamic paraventricular neurons. Brain Res 748: 71–76, 1997. [DOI] [PubMed] [Google Scholar]
- 57. Thirstrup S, Nielsen-Kudsk JE, Mellemkjaer S. Involvement of K+ channels in the relaxant effect of vasoactive intestinal peptide and atrial natriuretic peptide in isolated guinea-pig trachea. Eur J Pharmacol 319: 253–259, 1997. [DOI] [PubMed] [Google Scholar]
- 58. To TL, Henson MA, Herzog ED, Doyle FJ., 3rd A molecular model for intercellular synchronization in the mammalian circadian clock. Biophys J 92: 3792–3803, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Tüchsen F. Working hours and ischaemic heart disease in Danish men: a 4-year cohort study of hospitalization. Int J Epidemiol 22: 215–221, 1993. [DOI] [PubMed] [Google Scholar]
- 60. Vasalou C, Henson MA. A multiscale model to investigate circadian rhythmicity of pacemaker neurons in the suprachiasmatic nucleus. PLoS Comput Biol 6: e1000706, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Virag JA, Dries JL, Easton PR, Friesland AM, Deantonio JH, Chintalgattu V, Cozzi E, Lehmann BD, Ding JM, Lust RM. Attenuation of myocardial injury in mice with functional deletion of the circadian rhythm gene mPer2. Am J Physiol Heart Circ Physiol 298: H1088–H1095, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Vosko AM, Schroeder A, Loh DH, Colwell CS. Vasoactive intestinal peptide and the mammalian circadian system. Gen Comp Endocrinol 152: 165–175, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Witte K, Schnecko A, Buijs RM, van der Vliet J, Scalbert E, Delagrange P, Guardiola-Lemaître B, Lemmer B. Effects of SCN lesions on circadian blood pressure rhythm in normotensive and transgenic hypertensive rats. Chronobiol Int 15: 135–145, 1998. [DOI] [PubMed] [Google Scholar]
- 64. Yamashita T, Sekiguchi A, Iwasaki YK, Sagara K, Iinuma H, Hatano S, Fu LT, Watanabe H. Circadian variation of cardiac K+ channel gene expression. Circulation 107: 1917–1922, 2003. [DOI] [PubMed] [Google Scholar]
- 65. Zuker M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 31: 3406–3415, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


