Abstract
This paper describes a novel method for studying the bio-behavioral basis of addiction to food. This method combines the surgical component of taste reactivity with the behavioral aspects of operant self-administration of drugs. Under very brief general anaesthesia, rats are implanted with an intraoral (IO) cannula that allows delivery of test solutions directly in the oral cavity. Animals are then tested in operant self-administration chambers whereby they can press a lever to receive IO infusions of test solutions. IO self-administration has several advantages over experimental procedures that involve drinking a solution from a spout or operant responding for solid pellets or solutions delivered in a receptacle. Here, we show that IO self-administration can be employed to study self-administration of high fructose corn syrup (HFCS). Rats were first tested for self-administration on a progressive ratio (PR) schedule, which assesses the maximum amount of operant behavior that will be emitted for different concentrations of HFCS (i.e. 8%, 25%, and 50%). Following this test, rats self-administered these concentrations on a continuous schedule of reinforcement (i.e. one infusion for each lever press) for 10 consecutive days (1 session/day; each lasting 3 hr), and then they were retested on the PR schedule. On the continuous reinforcement schedule, rats took fewer infusions of higher concentrations, although the lowest concentration of HFCS (8%) maintained more variable self-administration. Furthermore, the PR tests revealed that 8% had lower reinforcing value than 25% and 50%. These results indicate that IO self-administration can be employed to study acquisition and maintenance of responding for sweet solutions. The sensitivity of the operant response to differences in concentration and schedule of reinforcement makes IO self-administration an ideal procedure to investigate the neurobiology of voluntary intake of sweets.
Keywords: Behavior, Issue 84, Administration, Oral, Conditioning, Operant, Reinforcement (Psychology), Reinforcement Schedule, Taste, Neurosciences, Intraoral infusions, operant chambers, self-administration, high fructose corn syrup, progressive ratio, breakpoint, addiction
Introduction
The study of the neurobiological and behavioral basis of addiction to food relies on the observations that, similarly to drugs of abuse, excessive consumption of palatable foods promotes behavioral dependence1-4 and induces alterations in brain reward circuits in both humans5-6 and laboratory animals7-8. But, while there are several protocols and procedures for studying the addictive properties of drugs of abuse in laboratory rats, adapting these methods to assess "addictive" behaviors induced by food presents unique challenges. The goal of the current study was to apply principles of operant intravenous (IV) drug self-administration to the investigation of operant self-administration of sweet solutions delivered by intraoral (IO) infusion. The sweet employed was high fructose corn syrup (HFCS) because, although controversial, there is evidence that HFCS may be linked to the modern epidemic of obesity9-12.
Traditionally, IO infusions are delivered to study the palatability of tastants in taste reactivity experiments13. Briefly, an IO cannula is surgically implanted into the cheek of rats and IO infusions of various solutions are passively delivered14. The objective is to study orofacial reactions of the animals to the tastants. However, IO cannulas have also been implanted chronically in the palate of rats to determine whether they would learn to press a lever to self-infuse solutions directly into their oral cavity; hence the term IO self-injection15-17. In the current study, we describe a procedure that utilizes minor surgery and that allows long term operant self-administration testing. This procedure has several significant advantages over traditional procedures that involve drinking a solution from a spout (a), or operant responding for solid pellets (b), or operant responding for drops of fluid delivered in a receptacle (c).
In comparison to (a), IO self-administration involves an operant response (i.e. pressing a lever) and therefore it is possible to modify the schedule regulating the relationship between response requirements and delivery of IO infusions. For example, by employing a progressive ratio (PR) schedule, whereby responses required for successive infusions increase exponentially within a session18, it is possible to assess how much an animal "wants" the next infusion19. This important aspect of "seeking" behavior cannot be assessed when animals drink a solution from a spout, unless a special apparatus that controls fluid delivery and monitors responses is employed20. Moreover, IO self-administration provides a mean to compare behaviors motivated by different reinforcing stimuli. That is, it is possible to compare operant responding maintained by sweets and other reinforcers such as drugs of abuse in so called "substitution studies."
In comparison to (b), IO self-administration allows testing of any concentration and any volume of any water-soluble food additive. This is critical to study behavior motivated by sweets such as HFCS because, to the best of our knowledge, there are no commercially available solid pellets of fructose, or fructose-glucose combinations at different ratios, that would be suitable for operant chambers. Also, the importance of controlling and manipulating concentration/volume ratios is mandatory in experiments where intake can be modulated both by the caloric value of a solution (leading to nutrient-specific satiety) and by how much of that solution can be consumed within a given period of time (i.e. fullness)21. IO self-administration also shortens the delay between the operant response and the delivery of the primary reinforcer, a factor that plays an important role in the acquisition and maintenance of operant behavior16,22-23.
Finally, in comparison to (c), IO self-administration allows the delivery of passive IO infusions of controlled quantities of the test solution, and this makes it possible to measure orofacial responses of "liking" (objective hedonic reaction such as tongue protrusions24-25) and whether these responses change during IO self-administration. Furthermore, the ability to administer passive IO infusions has significant applications for the study of relapse to food-seeking. That is, in studies of IV drug self-administration, following periods of extinction, drug primes (i.e. administration of a small dose of the drug26) can "reinstate" responding27-28. Therefore, the ability to deliver IO infusions without any action on the part of the animal can be used to study "food" primed reinstatement, as well as potential cross reinstatement between food and drugs of abuse.
IO self-administration of sweet solutions is also preferable to IV and intra-gastric self-administration. In fact, although IV infusions of fructose in both humans and laboratory animals produce physiological consequences similar to those observed following oral consumption29-31, this is a poor model of how sweets are normally consumed (i.e. orally). Moreover, gustatory signals produced by mastication confer important information about the palatability of food, and when this stage of the digestive process is omitted, the development of maladaptive behaviors such as overeating is reduced32-33.
Protocol
1. Animal Care and Housing
All experiments are approved by the Animal Care Committee of the University of Guelph, and are carried out in accordance with the recommendations of the Canadian Council on Animal Care. The current study includes 22 adult male Sprague-Dawley rats (Charles River, QC) weighing 200-225 g at the beginning of the experiment.
Single house rats and maintain them on a reverse light/dark cycle (7:00 am OFF - 7:00 pm ON) with ad libitum access to food and water except when otherwise indicated, or during behavioral testing which occurs during the active cycle.
Habituate rats to the colony room and human handling by weighing daily for one week prior to surgery.
2. Intraoral Cannulation Surgery: Materials and Procedures
The intraoral (IO) surgery and materials are described in Limebeer et al.14
To construct the IO cannula, cut a piece of polyethylene plastic tubing to approximately 130 mm in length (inner diameter of 0.86 mm and outer diameter of 1.27 mm).
The tube to be secured in the oral cavity requires a flanged end (~5 mm in diameter). Create this flanged end by heating the end of the tube with a soldering iron for 2-5 sec and then promptly flatten the heated edge of the tube against a flat surface. Cut a surgical mesh disc (approximately 6-7 mm diameter to fit around the diameter of the flanged end of the cannula) and thread it onto the length of the tube until the disc rests against the back of the flanged end of the IO cannula.
For each IO cannula, cut one additional surgical mesh disc (8 mm in diameter) and three squares of an elastic band (6 mm x 6 mm; see Figure 1).
Sterilize the IO cannula, surgical mesh discs, and elastic squares in an antibacterial solution for a minimum of 24 hr prior to surgery.
Prior to surgery, autoclave all surgical tools and disinfect the surgical field. Between surgeries, use a glass bead sterilizer to sterilize tools, and wear a hairnet, nitrile gloves, and surgical masks to maintain aseptic conditions.
Analgesia and anaesthesia
Approximately 30 min prior to surgery administer a subcutaneous (SC) injection of the antibiotic Depocillin (0.33 mg/kg) and the analgesic Meloxicam (5 mg/kg SC).
Induce general anaesthesia using Isoflurane (4% induction and 3% maintenance).
Once rats are unresponsive to toe pinch, and prior to making any incisions, use a cotton tipped swab to administer a topical anaesthetic along the inner left cheek of rats where the cannula is to be implanted, and inject a local anaesthetic such as lidocaine HCl at the incision site for the biopsy needle (base of neck) and head cap (scalp).
Apply lubricating eye ointment to both eyes prior to the beginning of surgery.
Surgical procedures
Shave a 10 mm x 10 mm patch of hair at the base of the neck. Use sterile gauzes to swab and cleanse the shaved area in the following order: antibacterial soap, isopropyl 70% alcohol, and Betadine.
Insert a 15 G stainless steel needle at the base of the neck and move it subcutaneously around the ear to the left cheek. Position the needle tip and pass it through the cheek behind the first molar inside of the oral cavity.
Pass the IO cannula through the tip of the needle protruding from the oral cavity and then remove the needle leaving only the IO cannula.
Thread one surgical mesh disc followed by three square elastic discs onto the exterior portion of the IO cannula and draw them to the exposed skin at the back of the neck to stabilize it.
Flush the IO cannula with an antibacterial oral rinse
Prior to the beginning of surgery, modify a Nylon bolt (28 mm length; 4 mm point; 8 mm head) to remove its head and carve a 2 mm groove along the length of the thread (see Figure 2).
Shave a 10 mm x 20 mm strip of hair along the midline of the head. Use sterile gauzes to swab and cleanse the shaved area in the following order: antibacterial soap, isopropyl 70% alcohol, and Betadine.
Use a surgical scalpel to make a 20 mm long incision along the sagittal suture. Use a hand drill to make 4 holes (2 on each side of the suture) and use a hand held screw driver to secure four jewelers screws to the skull.
Place the bolt in the center of the four jewelers screws with the groove positioned towards the posterior of the animal. Mount the bolt to the skull of the rat by building a headcap with dental cement (use sufficient dental cement to cover the surface of the jewelers screws). Allow the dental cement to dry before removing the animal from the surgical field and terminate anaesthesia.
Post-operative monitoring and care
Individually house rats in bedded cages immediately following surgery and closely monitor for 24 hr.
Administer a second injection of the analgesic Meloxicam 24 hr post-surgery and flush the IO cannula with an antibacterial oral rinse once/day for three days.
Give rats 3-5 days to recover from surgery prior to the beginning of the experiment.
3. IO Self-administration Procedures
Apparatus
For self-administration, Plexiglas operant conditioning chambers are used, and each chamber is enclosed in a larger sound-attenuating plywood box. Each operant conditioning chamber has a house light (28 V) and two retractable levers (active and inactive) located on opposite sides of the chamber, 8 cm above the floor. The active lever is connected to an infusion pump positioned outside the sound-insulating chamber and a white light (28 W) is located 3 cm above it to serve as a stimulus light paired with the delivery of IO infusions. The inactive lever serves to control for baseline, nonreinforced operant behavior; pressing this lever has no consequence, but all presses are recorded.
Food
Provide rats with a standard rat chow (3.1 kcal/g) in their home cage.
Dilute high fructose corn syrup (55% formula) to three different concentrations (8% = 0.026 kcal/80 μl infusion; 25% = 0.08 kcal/80 μl infusion; 50% = 0.17 kcal/80 μl infusion) using reverse osmosis water. These concentrations are selected because each produces a viscosity that is appropriate to deliver rapid (2.5 sec) IO infusions given the experimental apparatus (i.e. diameter of syringes and tubing).
Food deprivation
In order to facilitate the acquisition of IO self-administration, but promote normal growth, rats are cycled through periods of food deprivation and ad libitum access to standard rat chow.
Weigh rats daily at 7 pm and remove chow from the feeding trough of the cage.
The following day, weigh rats at 12:00 pm (approximately 18 hr later) and begin IO self-administration by 1:00 pm.
Return rats to their home cages at 4:00 pm and provide ad libitum access to chow for 3 consecutive hr. Then, record the weight of the rats and the total amount of chow consumed over the three hours. Resume food deprivation as described.
General IO self-administration procedure
Operant responding for IO infusions of HFCS is evaluated on PR and continuous schedules of reinforcement. Rats are tested in IO self-administration until intake is stable as defined by: 1) a significant difference in responding between active and inactive levers for at least 2 consecutive sessions; and 2) less than 20% variation in the number of infusions earned per session for 2 consecutive sessions. Each session begins 6 hr into the dark cycle and lasts 3 hr. The acquisition phase consists of 10 consecutive IO self-administration sessions on a continuous schedule of reinforcement, and rats receive a single PR test 24 hr prior to the beginning of acquisition (PR test I) and 24 hr after the final acquisition session (PR Test II). For PR tests, the breakpoint (BP) is defined as the last infusion received prior to cessation of responding on the active lever for at least 1 hr18.
Counterbalance rats on the basis of weights taken for three days prior to the start of the experiment and assign rats to self-administer one of three concentrations of HFCS: 8%, 25%, or 50%.
Weigh rats at 12:00 pm and transfer them from the colony room to the testing room in their home cages. To remove food or bedding that can accumulate in the IO cannula, pump air through the cannula using a 12 ml syringe fixed with a 16 G needle with 50 mm of polyethylene tubing (inner diameter of 1.14 mm and outer diameter of 1.57 mm) attached to the end.
In the operant chamber, connect the IO cannula to Tygon tubing (inner diameter of 0.02 mm and outer diameter of 1.53 mm) by a shaft constructed of a modified 20 G needle (remove the plastic hub and beveled tip). Position the lower portion of the tubing (i.e. closer to the animal) within the grove of the bolt. The higher portion of the tubing is connected to a swivel, and protected by a spring. Connect the base of the spring to the thread of the bolt mounted on the head of the rat.
Give rats five minutes to acclimatize to the operant chamber and then start the IO self-administration session. The IO self-administration session begins with the activation of the house light, entry of the two retractable levers, and illumination of the light stimulus above the active lever for 30 sec.
For PR Test I, use the PR schedule described by Richardson and Roberts18 (response ratio escalation = (5e(injection number x 0.2)) - 5).
For acquisition sessions 1-10, use a continuous schedule of reinforcement whereby each press on the active lever results in the delivery of an 80 μl infusion of the test solution over 2.5 sec.
To allow sufficient time for ingestion, impose a time-out period of 27.5 sec during which the active lever is retracted and the stimulus light is activated (i.e. rats can obtain an IO infusion every 30 sec). Do not impose a limit on the number of infusions obtainable within each IO self-administration session.
If the rat does not make a response on the active lever for 20 consecutive minutes, prime the rat (open the operant chamber and facilitate the rat pressing once on the active lever). Prime the animals every 20 min up to 5x/session during the first three days of acquisition only.
Administer the second PR test using the parameters described in step 3.10.
Following IO self-administration sessions, remove rats from the operant chambers, flush the IO cannula with 2.0 ml water using the same materials described in 3.7, and return rats to their home cage in the colony room.
Following three hours of ad libitum access to rat chow, weight rats at 7:00 pm, remove food from the feeding trough, and resume food deprivation.
Self-administration data are analyzed using two-factor repeated measures Analyses of Variance (ANOVA). Multiple comparisons using the Student-Newman-Keuls method (α=0.05) were employed to explore any significant interactions or main effects.
Representative Results
Acquisition of IO self-administration
Figure 3 represents operant responding on the active and inactive levers during self-administration of 8%, 25%, or 50% HFCS for 10 consecutive sessions. Rats acquire IO self-administration of HFCS within the first three sessions and maintain stable active lever responding (as described above) for the remainder of acquisition. Concentration-dependent differences in active lever responses are also evident: compared to 25%, responding is significantly lower in rats self-administering 50% HFCS. The lowest concentration tested (8%) maintains an intermediate level of operant responding characterized by higher individual variability in intake.
Progressive ratio tests
Active lever responses and BP on the PR tests are represented in Figures 4 and 5, respectively. On Test I, there is modest responding for all concentrations of HFCS. However, group differences emerge on Test II with higher concentrations engendering more responding and higher BPs.
 Figure 1.
Complete IO cannula with flanged end, mesh disc (left) with the additional elastic and mesh discs required to secure the cannula once implanted (right).
Figure 1.
Complete IO cannula with flanged end, mesh disc (left) with the additional elastic and mesh discs required to secure the cannula once implanted (right).
 Figure 2. Nylon bolt prior to modification (left). Nylon bolt (right) following the removal of the head and the addition of a 2 mm groove along the length of the thread.
Figure 2. Nylon bolt prior to modification (left). Nylon bolt (right) following the removal of the head and the addition of a 2 mm groove along the length of the thread.
 Figure 3. Mean (sem) number of active and inactive lever responses made by rats responding on a continuous schedule of reinforcement for 8% (n=6), 25% (n=8) or 50% (n=8) high fructose corn syrup. The single and double asterisk represents significant group differences in active lever responses between rats self-administering 25% and 50%, and 25% and 8%, respectively (p < 0.05).
Figure 3. Mean (sem) number of active and inactive lever responses made by rats responding on a continuous schedule of reinforcement for 8% (n=6), 25% (n=8) or 50% (n=8) high fructose corn syrup. The single and double asterisk represents significant group differences in active lever responses between rats self-administering 25% and 50%, and 25% and 8%, respectively (p < 0.05).
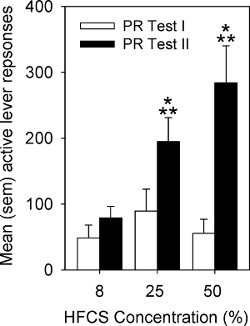 Figure 4. Mean (sem) number of active lever responses by rats responding on a PR schedule for 8%, 25%, or 50% HFCS. Rats were tested twice on the PR schedule; prior to (PR Test I) and following (PR Test II) responding for the same concentration of HFCS for 10 days of IO self-administration on a continuous schedule of reinforcement. The single asterisk indicates a significant difference in active lever responses between PR Test I and PR Test II. The double asterisk indicates significant group differences in active lever responses in comparison to the 8% group (p < 0.05).
Figure 4. Mean (sem) number of active lever responses by rats responding on a PR schedule for 8%, 25%, or 50% HFCS. Rats were tested twice on the PR schedule; prior to (PR Test I) and following (PR Test II) responding for the same concentration of HFCS for 10 days of IO self-administration on a continuous schedule of reinforcement. The single asterisk indicates a significant difference in active lever responses between PR Test I and PR Test II. The double asterisk indicates significant group differences in active lever responses in comparison to the 8% group (p < 0.05).
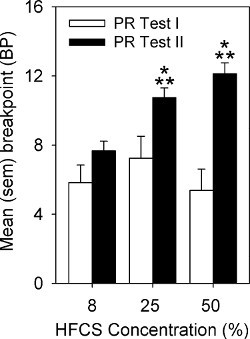 Figure 5. Mean (sem) breakpoint achieved by rats responding on PR for 8%, 25%, or 50% HFCS. The single asterisk indicates a significant difference in active lever responses between PR Test I and PR Test II. The double asterisk indicates significant group differences in active lever responses in comparison to the 8% group (p < 0.05).
Figure 5. Mean (sem) breakpoint achieved by rats responding on PR for 8%, 25%, or 50% HFCS. The single asterisk indicates a significant difference in active lever responses between PR Test I and PR Test II. The double asterisk indicates significant group differences in active lever responses in comparison to the 8% group (p < 0.05).
Discussion
The current study illustrates a novel approach for evaluating the reinforcing properties of sweet solutions by combining methods traditionally employed to study behavior motivated by drugs of abuse (intravenous operant self-administration) and to assess the palatability of tastants (taste reactivity through intraoral infusions). To this end, under very brief isoflurane-induced anesthesia, rats are implanted with an IO cannula that allows for the delivery of test solutions directly into their mouth. Following recovery from surgery, the animals are tested in operant self-administration chambers whereby they can press a lever to receive IO infusions of test solutions.
Using the IO self-administration procedure we established that: 1) rats acquire and maintain IO self-administration of various concentrations of HFCS; 2) rats adjust their self-administration behavior according to different concentrations of HFCS; and 3) higher concentrations of HFCS engender higher responding on a PR schedule of self-administration.
These results clearly indicate that IO self-administration behavior is regulated by the post-ingestional consequences of HFCS. In fact, on a continuous schedule of reinforcement, rats take fewer infusions when the concentration is higher (compare 25% and 50% in Figure 3). Furthermore, a low concentration of HFCS (8%) maintains variable self-administration on a continuous schedule, and the PR test indicates that 8% has lower reinforcing value relative to 25% and 50%. These behavioral patterns closely mirror those that are observed when rats self-administer different doses of drugs of abuse34-36. This is the first study to demonstrate intraoral operant self-administration of high fructose corn syrup in laboratory rats, and to ascertain that self-administration behavior is controlled by the nutritional consequences of this particular sugar.
When employing IO self-administration, it is important to take into consideration a procedural issue. When the animal is placed into the operant chamber and its IO cannula is attached to the Tygon tubing, there is the possibility of disconnection during the session. To prevent and correct this issue, it is essential to frequently observe the animals for leaks (i.e. check for fluid build-up that is apparent on the head cap). In addition, approximately 50-70 mm of the IO cannula exits from the back of the animals, and consequently, the IO cannula can be chewed when the rats are in their home cage. To correct this issue, a connector can be constructed from a 20 G needle in order to replace the missing portion of the IO cannula. In extreme cases, the entire cannula can be replaced by repeating the surgery.
The IO self-administration procedure offers a novel and innovative approach to address important questions regarding the neurobiological and behavioral basis of food addiction. First, it is clear that a contributing factor to overweight and obesity is excessive consumption of foods that are refined with additives such as sweeteners to enhance palatability1-4. There are several sweeteners that are commonly used in foods, differing in structure and hence caloric value and taste37. The role of these differences in promoting excessive consumption can be explored systematically using IO self-administration. Second, availability of addictive foods cannot be the sole determinant of excessive self-administration; as in the case of drugs of abuse, individual differences/vulnerabilities must also play a role38-39. IO self-administration, because of its active and measurable behavioral component, can identify vulnerable phenotypes and hence allow investigation of underlying genetic and epigenetic differences. Third, IO self-administration can be maintained for long periods of time, hence allowing the investigation of neurobiological correlates of voluntary intake of addictive food and diet-induced obesity. Fourth, IV and IO self-administration studies can be performed in parallel to identify shared neurobiological factors that are involved in the regulation of intake and relapse to compulsive reward seeking. Finally, again because of its active and measurable behavioral component, IO self-administration can be employed to test new pharmacological approaches to reduce excessive consumption of highly palatable food.
Disclosures
The authors having nothing to disclose.
Acknowledgments
These studies were supported by grants from the Natural Sciences and Engineering Research Council of Canada (NSERC) to F.L and a Canadian Graduate Scholarship (CGSD) from NSERC to AM.L.
References
- Avena NM, Bocarsly ME, Hoebel BG, Gold MS. Overlaps in the nosology of substance abuse and overeating: the translational implications of "food addiction". Curr. Drug Abuse Rev. 2011;4(3):133–139. doi: 10.2174/1874473711104030133. [DOI] [PubMed] [Google Scholar]
- Ifland JR, Preuss HG, et al. Refined food addiction: a classic substance use disorder. Med. Hyp. 2009;72:518–526. doi: 10.1016/j.mehy.2008.11.035. [DOI] [PubMed] [Google Scholar]
- Volkow N, Wise RA. How can drug addiction help us understand obesity. Nat. Neurosci. 2005;8(5):555–560. doi: 10.1038/nn1452. [DOI] [PubMed] [Google Scholar]
- Gearhardt AN, Davis C, Kuschner R, Brownell KD. The addiction potential of hyperpalatable foods. Curr. Drug Abuse Rev. 2011;4(3):140–145. doi: 10.2174/1874473711104030140. [DOI] [PubMed] [Google Scholar]
- Gearhardt AN, Yokum S, Orr PT, Stice E, Corbin WR, Brownell KD. Neural correlates of food addiction. Arch. Gen. Psychiatry. 2011;68(8):808–816. doi: 10.1001/archgenpsychiatry.2011.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volkow ND, Wang G, Fowler JS, Telang F. Overlapping neuronal circuits in addiction and obesity: evidence of systems pathology. Philos. Trans. Royal Soc. 2008;363:3191–3200. doi: 10.1098/rstb.2008.0107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alsiö J, Olszewski PK, Levine AS, Schiöth HB. Feed-forward mechanisms: addiction-like behavioral and molecular adaptations in overeating. Front. Neuroendocrinol. 2012;33(2):127–139. doi: 10.1016/j.yfrne.2012.01.002. [DOI] [PubMed] [Google Scholar]
- Johnson PM, Kenny PJ. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010;13(5):635–644. doi: 10.1038/nn.2519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bray GA, Nielsen SJ, Popkin BM. Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am. J. Clin. Nutr. 2004;79(4) doi: 10.1093/ajcn/79.4.537. [DOI] [PubMed] [Google Scholar]
- Forshee RA, Storey ML, et al. A critical examination of the evidence relating high fructose corn syrup and weight gain. Crit. Rev. Food Sci. Nutr. 2007;47(6):561–582. doi: 10.1080/10408390600846457. [DOI] [PubMed] [Google Scholar]
- Moeller SM, Fryhofer SA, Osbahr AJ, Robinowitz The effects of high fructose corn syrup. J. Am. Coll. Nutr. 2009;28(6):619–626. doi: 10.1080/07315724.2009.10719794. [DOI] [PubMed] [Google Scholar]
- White JS, Foreyt JP, Melanson KJ, Angelopoulos TJ. High fructose corn syrup: Controversies and common sense. Am. J. Lifestyle Med. 2010;4(6):515–520. [Google Scholar]
- Grill HJ, Norgren R. The taste reactivity test. I. Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res. 1978;143(2):263–279. doi: 10.1016/0006-8993(78)90568-1. [DOI] [PubMed] [Google Scholar]
- Limebeer CL, Vemuri VK, et al. Inverse agonism of cannabinoid CB1 receptors potentiates LiCl-induced nausea in the conditioned gaping model in rats. Br. J. Pharmacol. 2010;161(2):336–349. doi: 10.1111/j.1476-5381.2010.00885.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeBold RC, Miller NE, Jensen DD. Effect of strength of drive determined by a new technique for appetitive classical conditioning of rats. J. Comp. Physiol.Psychol. 1965;59(1):102–108. doi: 10.1037/h0021632. [DOI] [PubMed] [Google Scholar]
- Panksepp J, Trowill JA. Intraoral self injection: I. Effects of delay of reinforcement on resistance to extinction and implications for self-stimulation. Psychonomic Sci. 1967;9(7):405–406. [Google Scholar]
- Panksepp J, Trowill JA. Intraoral self injection: II. The simulation of self-stimulation phenomena with a conventional reward. Psychonomic Sci. 1967;9(7):407–408. [Google Scholar]
- Richardson NR, Roberts DC. Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J. Neurosci. Methods. 1996;66:1–11. doi: 10.1016/0165-0270(95)00153-0. [DOI] [PubMed] [Google Scholar]
- Berridge KC, Robinson TE. Parsing reward. Trends Neurosci. 2003;26(11):507–501. doi: 10.1016/S0166-2236(03)00233-9. [DOI] [PubMed] [Google Scholar]
- Sclafani A, Ackroff K. Reinforcement value of sucrose measured by progressive ratio operant licking in the rat. Physiol. Behav. 2003;79:663–670. doi: 10.1016/s0031-9384(03)00143-4. [DOI] [PubMed] [Google Scholar]
- Houpt KA. Gastrointestinal factors in hunger and satiety. Neurosci. Biobehav. Rev. 1982;6(2):145–164. doi: 10.1016/0149-7634(82)90052-5. [DOI] [PubMed] [Google Scholar]
- Mazur JE. Effects of rate of reinforcement and rate of change on choice behaviour in transition. J. Exp. Psychol. 1997;50(2):111–128. doi: 10.1080/713932646. [DOI] [PubMed] [Google Scholar]
- Samaha AN, Robinson TE. Why does the rapid delivery of drugs to the brain promote addiction. Trends Pharmacol. Sci. 2005;26(2):82–87. doi: 10.1016/j.tips.2004.12.007. [DOI] [PubMed] [Google Scholar]
- Berridge KC, Kringelbach ML. Affective neuroscience of pleasure: reward in humans and animals. Psychopharmacology. 2008;199(3):457–480. doi: 10.1007/s00213-008-1099-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelley AE, Berridge KC. The neuroscience of natural rewards: relevance to addictive drugs. J. Neurosci. 2002;22(9):3306–3311. doi: 10.1523/JNEUROSCI.22-09-03306.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaham Y, Shalev U, Lu L, De Wit H, Stewart J. The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology. 2003;168(1-2):3–20. doi: 10.1007/s00213-002-1224-x. [DOI] [PubMed] [Google Scholar]
- de Wit H, Stewart J. Drug reinstatement of heroin-reinforced responding in the rat. Psychopharmacology. 1983;79(1):29–31. doi: 10.1007/BF00433012. [DOI] [PubMed] [Google Scholar]
- Schmidt HD, Anderson SM, Famous KR, Kumaresan V, Pierce RC. Anatomy and pharmacology of cocaine priming-induced reinstatement of drug seeking. Eur. J. Pharmacol. 2005;526(1-3):65–76. doi: 10.1016/j.ejphar.2005.09.068. [DOI] [PubMed] [Google Scholar]
- Dunnigan MG, Ford JSA. The insulin response to intravenous fructose in relation to blood glucose levels. J. Clin. Endocrinol. Metab. 1975;40(4):629–635. doi: 10.1210/jcem-40-4-629. [DOI] [PubMed] [Google Scholar]
- Crapo PA, Kolterman OG, Olefsky JM. Effects of oral fructose in normal, diabetic, and impaired glucose tolerance subjects. Diabetes Care. 1980;3(5):575–582. doi: 10.2337/diacare.3.5.575. [DOI] [PubMed] [Google Scholar]
- Lê KA, Tappy L. Metabolic effects of fructose. Curr. Opin. Clin. Nutr. Metab. Care. 2006;9(4):469–475. doi: 10.1097/01.mco.0000232910.61612.4d. [DOI] [PubMed] [Google Scholar]
- Scheggi ME, Secci G, Marchese MG, De Montis C, Gambarana Influence of palatability on motivation to operate for caloric and non-caloric food in non food-deprived and food-deprived rats. Neuroscience. 2013;236 doi: 10.1016/j.neuroscience.2013.01.027. [DOI] [PubMed] [Google Scholar]
- Sclafani A, Lucas F, Ackroff K. The importance of taste and palatability in carbohydrate-induced overeating in rats. Am. J. Physiol. 1996;270(6):1197–1202. doi: 10.1152/ajpregu.1996.270.6.R1197. [DOI] [PubMed] [Google Scholar]
- Roberts DCS, Loh EA, Vickers G. Self-administration of cocaine on a progressive ratio schedule in rats: dose-response relationship and effect of haloperidol pretreatment. Psychopharmacology. 1989;97(4):535–538. doi: 10.1007/BF00439560. [DOI] [PubMed] [Google Scholar]
- Reilly S. Reinforcement value of gustatory stimuli determined by progressive ratio Performance. Pharmacol. Biochem. Behav. 1999;63(2):301–311. doi: 10.1016/s0091-3057(99)00009-x. [DOI] [PubMed] [Google Scholar]
- Zittel-Lazarini A, Cador M, Ahmed SH. A critical transition in cocaine self-administration: Behavioral and neurobiological implications. Psychopharmacology. 2007;192(3):337–146. doi: 10.1007/s00213-007-0724-0. [DOI] [PubMed] [Google Scholar]
- Stanhope KL, Havel PJ. Endocrine and metabolic effects of consuming beverages sweetened with fructose, glucose, sucrose, or high fructose corn syrup Am. J. Clin. Nutr. 2008;88(6):17335–17375. doi: 10.3945/ajcn.2008.25825D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deroche-Gamonet V, Belin B, Piazza PV. Evidence for addiction-like behaviour in the rat. Science. 2004;305:1014–1017. doi: 10.1126/science.1099020. [DOI] [PubMed] [Google Scholar]


