Abstract
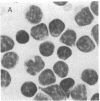
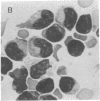
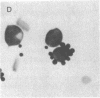
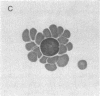
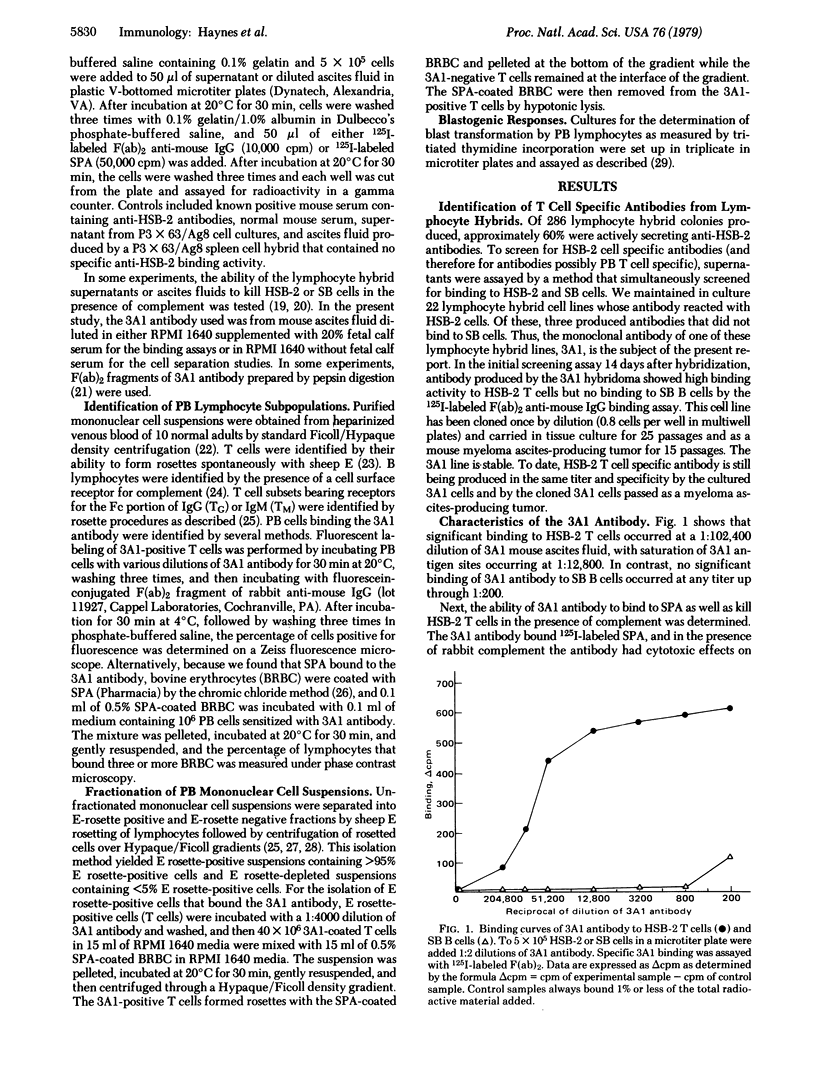
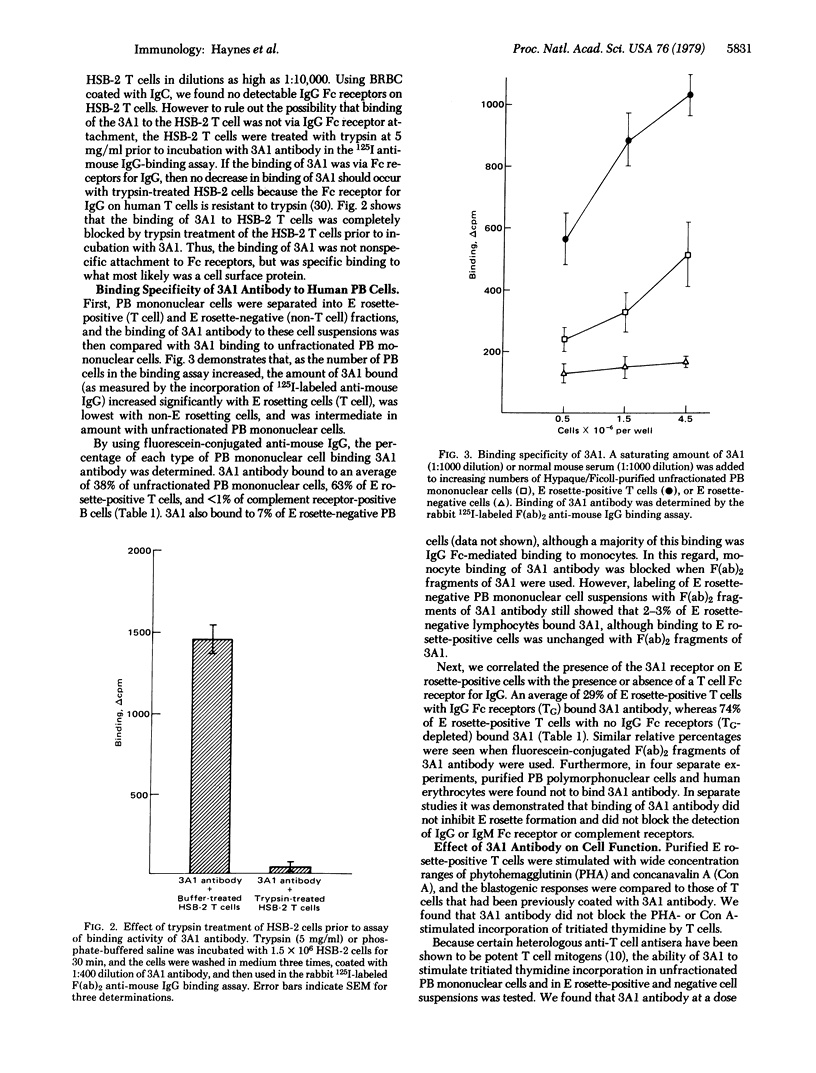
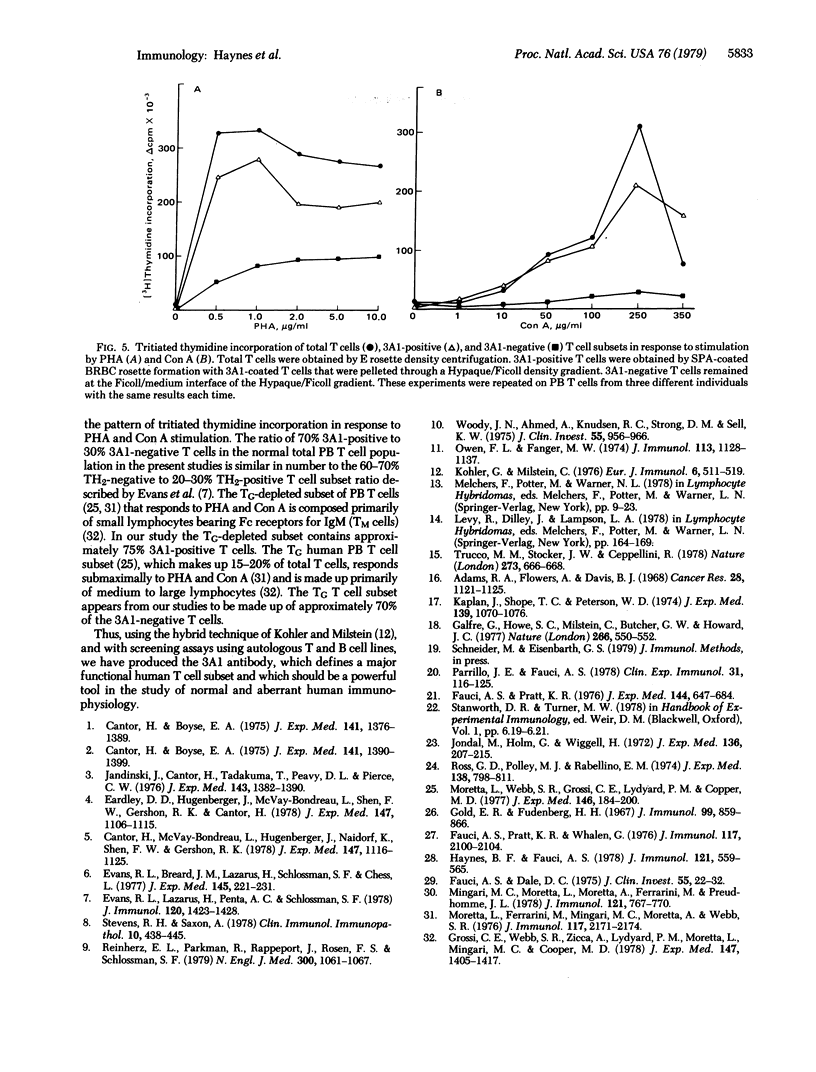
A monoclonal mouse antibody (3A1) that specifically bound to 65% of human peripheral blood (PB) thymus-derived (T) cells but did not bind to complement receptor-positive PB bone marrow-derived (B) cells, polymorphonuclear leukocytes, or human erythrocytes has been produced. The 3AI antibody was synthesized by a stable cloned lymphocyte hybrid cell line. This lymphocyte hybrid line (3AI) was derived from fusion of P3 X 63/Ag8 myeloma cells and spleen cells from BALB/c mice immunized with HSB-2 cells, a human T cell line. The 3A1 lymphocyte hybrid line produced mouse ascites fluid containing 3A1 antibody in saturating titers of up to 1:25,600. Purified PB T cells that carried the 3A1 antigen incorporated tritiated thymidine maximally in response to phytohemagglutinin and concanavalin A stimulation, whereas purified PB T cells that lacked the 3A1 antigen responded suboptimally to phytohemagglutinin and minimally to concanavalin A. Thus, the 3A1 antibody can be easily used to study the role of 3A1-positive and negative T cell subsets in the regulation of normal and abnormal human immune responses.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams R. A., Flowers A., Davis B. J. Direct implantation and serial transplantation of human acute lymphoblastic leukemia in hamsters, SB-2. Cancer Res. 1968 Jun;28(6):1121–1125. [PubMed] [Google Scholar]
- Cantor H., Boyse E. A. Functional subclasses of T lymphocytes bearing different Ly antigens. II. Cooperation between subclasses of Ly+ cells in the generation of killer activity. J Exp Med. 1975 Jun 1;141(6):1390–1399. doi: 10.1084/jem.141.6.1390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantor H., Boyse E. A. Functional subclasses of T-lymphocytes bearing different Ly antigens. I. The generation of functionally distinct T-cell subclasses is a differentiative process independent of antigen. J Exp Med. 1975 Jun 1;141(6):1376–1389. doi: 10.1084/jem.141.6.1376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantor H., McVay-Boudreau L., Hugenberger J., Naidorf K., Shen F. W., Gershon R. K. Immunoregulatory circuits among T-cell sets. II. Physiologic role of feedback inhibition in vivo: absence in NZB mice. J Exp Med. 1978 Apr 1;147(4):1116–1125. doi: 10.1084/jem.147.4.1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eardley D. D., Hugenberger J., McVay-Boudreau L., Shen F. W., Gershon R. K., Cantor H. Immunoregulatory circuits among T-cell sets. I. T-helper cells induce other T-cell sets to exert feedback inhibition. J Exp Med. 1978 Apr 1;147(4):1106–1115. doi: 10.1084/jem.147.4.1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans R. L., Breard J. M., Lazarus H., Schlossman S. F., Chess L. Detection, isolation, and functional characterization of two human T-cell subclasses bearing unique differentiation antigens. J Exp Med. 1977 Jan 1;145(1):221–233. doi: 10.1084/jem.145.1.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans R. L., Lazarus H., Penta A. C., Schlossman S. F. Two functionally distinct subpopulations of human T cells that collaborate in the generation of cytotoxic cells responsible for cell-mediated lympholysis. J Immunol. 1978 Apr;120(4):1423–1428. [PubMed] [Google Scholar]
- Fauci A. S., Dale D. C. Alternate-day prednisone therapy and human lymphocyte subpopulations. J Clin Invest. 1975 Jan;55(1):22–32. doi: 10.1172/JCI107914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fauci A. S., Pratt K. R. Activation of human B lymphocytes. I. Direct plaque-forming cell assay for the measurement of polyclonal activation and antigenic stimulation of human B lymphocytes. J Exp Med. 1976 Sep 1;144(3):674–684. doi: 10.1084/jem.144.3.674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fauci A. S., Pratt K. R., Whalen G. Activation of human B lymphocytes. II. Cellular interactions in the PFC response of human tonsillar and peripheral blood B lymphocytes to polyclonal activation by pokeweed mitogen. J Immunol. 1976 Dec;117(6):2100–2104. [PubMed] [Google Scholar]
- Galfre G., Howe S. C., Milstein C., Butcher G. W., Howard J. C. Antibodies to major histocompatibility antigens produced by hybrid cell lines. Nature. 1977 Apr 7;266(5602):550–552. doi: 10.1038/266550a0. [DOI] [PubMed] [Google Scholar]
- Gold E. R., Fudenberg H. H. Chromic chloride: a coupling reagent for passive hemagglutination reactions. J Immunol. 1967 Nov;99(5):859–866. [PubMed] [Google Scholar]
- Grossi C. E., Webb S. R., Zicca A., Lydyard P. M., Moretta L., Mingari M. C., Cooper M. D. Morphological and histochemical analyses of two human T-cell subpopulations bearing receptors for IgM or IgG. J Exp Med. 1978 May 1;147(5):1405–1417. doi: 10.1084/jem.147.5.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haynes B. F., Fauci A. S. Activation of human B lymphocytes. X. Heterogeneity of concanavalin A-generated suppressor cells of the pokeweed mitogen-induced plaque-forming cell response of human peripheral blood lymphocytes. J Immunol. 1978 Aug;121(2):559–565. [PubMed] [Google Scholar]
- Jandinski J., Cantor H., Tadakuma T., Peavy D. L., Pierce C. W. Separation of helper T cells from suppressor T cells expressing different Ly components. I. Polyclonal activation: suppressor and helper activities are inherent properties of distinct T-cell subclasses. J Exp Med. 1976 Jun 1;143(6):1382–1390. doi: 10.1084/jem.143.6.1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jondal M., Holm G., Wigzell H. Surface markers on human T and B lymphocytes. I. A large population of lymphocytes forming nonimmune rosettes with sheep red blood cells. J Exp Med. 1972 Aug 1;136(2):207–215. doi: 10.1084/jem.136.2.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan J., Shope T. C., Peterson W. D., Jr Epstein-barr virus-negative human malignant T-cell lines. J Exp Med. 1974 May 1;139(5):1070–1076. doi: 10.1084/jem.139.5.1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Derivation of specific antibody-producing tissue culture and tumor lines by cell fusion. Eur J Immunol. 1976 Jul;6(7):511–519. doi: 10.1002/eji.1830060713. [DOI] [PubMed] [Google Scholar]
- Mingari M. C., Moretta L., Moretta A., Ferrarini M., Preud'homme J. L. Fc-receptors for IgG and IgM immunoglobulins on human T lymphocytes: mode of re-expression after proteolysis or interaction with immune complexes. J Immunol. 1978 Aug;121(2):767–770. [PubMed] [Google Scholar]
- Moretta L., Ferrarini M., Mingari M. C., Moretta A., Webb S. R. Subpopulations of human T cells identified by receptors for immunoglobulins and mitogen responsiveness. J Immunol. 1976 Dec;117(6):2171–2174. [PubMed] [Google Scholar]
- Moretta L., Webb S. R., Grossi C. E., Lydyard P. M., Cooper M. D. Functional analysis of two human T-cell subpopulations: help and suppression of B-cell responses by T cells bearing receptors for IgM or IgG. J Exp Med. 1977 Jul 1;146(1):184–200. doi: 10.1084/jem.146.1.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owen F. L., Fanger M. W. Studies on the human T-lymphocyte population. I. The development and characterization of a specific anti-human T-cell antibody. J Immunol. 1974 Oct;113(4):1128–1137. [PubMed] [Google Scholar]
- Parrillo J. E., Fauci A. S. Mechanisms of corticosteroid action on lymphocyte subpopulations. III. Differential effects of dexamethasone administration on subpopulations of effector cells mediating cellular cytotoxicity in man. Clin Exp Immunol. 1978 Jan;31(1):116–125. [PMC free article] [PubMed] [Google Scholar]
- Reinherz E. L., Parkman R., Rappeport J., Rosen F. S., Schlossman S. F. Aberrations of suppressor T cells in human graft-versus-host disease. N Engl J Med. 1979 May 10;300(19):1061–1068. doi: 10.1056/NEJM197905103001901. [DOI] [PubMed] [Google Scholar]
- Ross G. D., Polley M. J., Rabellino E. M., Grey H. M. Two different complement receptors on human lymphocytes. One specific for C3b and one specific for C3b inactivator-cleaved C3b. J Exp Med. 1973 Oct 1;138(4):798–811. doi: 10.1084/jem.138.4.798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens R. H., Saxon A. Anti-human helper T-lymphocyte antiserum: generation and functional characterization. Clin Immunol Immunopathol. 1978 Aug;10(4):438–445. doi: 10.1016/0090-1229(78)90156-3. [DOI] [PubMed] [Google Scholar]
- Trucco M. M., Stocker J. W., Caeppellini R. Monoclonal antibodies against human lymphocyte antigens. Nature. 1978 Jun 22;273(5664):666–668. doi: 10.1038/273666a0. [DOI] [PubMed] [Google Scholar]
- Woody J. N., Ahmed A., Knudsen R. C., Strong D. M., Sell K. W. Human T-cell heterogeneity as delineated with a specific human thymus lymphocyte antiserum. In vitro effects on mitogen response mixed leukocyte culture, cell-mediated lymphocytotoxicity, and lymphokine production. J Clin Invest. 1975 May;55(5):956–966. doi: 10.1172/JCI108025. [DOI] [PMC free article] [PubMed] [Google Scholar]