Heparin, a linear highly sulfated polysaccharide, is a member of the glycosaminoglycan (GAG)1 family [1,2]. Since the 1930s, heparin has been widely used as a clinical anticoagulant [3,4]. More recently, low-molecular weight heparin (LMWH) derivatives have come into widespread use as anticoagulant and antithrombotic drugs [5]. Although heparin’s clinical use predated the establishment of the US. Food and Drug Administration (FDA), the approval of LMWH required a number of pharmacological studies on these new drugs. The pharmacologically relevant concentration of heparin and LMWH in plasma is between 0.1 and 1.0 anti-factor XaU/ml, corresponding to approximately 1–10 µg/ml of drug [6]. Plasma is a complex mixture, making the direct analysis of heparin or LMWH at these concentrations exceedingly challenging. Thus, most of the pharmacological studies of heparin and LMWH have relied on bioassays such as anti-factor Xa activity to evaluate these agents, providing only pharmacodynamic data. This inability to obtain pharmacokinetic data on LMWH is a serious concern because the plasma anti-factor Xa activity of LMWH does not correlate well with its therapeutic efficacy [7].
Our laboratory has successfully applied reversed-phase ion-pairing (RPIP) high-performance liquid chromatography (HPLC) in a pharmacokinetic study of heparin in rabbit plasma [8]. Unfortunately, this method required a cumbersome purification procedure that cannot be easily applied to human plasma samples in the high-throughput fashion required to provide pharmacokinetic data for clinical studies. In addition, human plasma samples are much more variable with respect to the presence of interfering substances than rabbit plasma. In particular, human plasma reportedly contains endogenous GAGs [9] that might interfere with heparin analysis. The current study involves the use of a simple recovery and purification scheme for the analysis of heparin from human plasma that relies on proteolysis, an ion-exchange spin column purification, and methanol precipitation.
Pooled, citrated human plasma (Innovative Research, Southfield, MI, USA) was used to assess a new approach for the recovery of exogenously added heparin and for the characterization of endogenous plasma GAGs. Our challenge was to devise a method to recover the anionic heparin polysaccharide from plasma, a complex mixture containing proteins, buffer salts (~150 mM), proteins (~60 g/L), lipids (~1 g/L), and endogenous GAGs [9]. Urea, a nonionic denaturant, is known to solubilize most proteins, and Chaps, a zwitterionic surfactant, is commonly used to solubilize hydrophobic molecules such as triglycerides. Initial studies examined the solubility of lyophilized plasma in 8 M urea containing 2 wt% Chaps, and to our surprise a translucent gel formed. In an alternative approach to prevent gel formation and facilitate heparin recovery, plasma proteins were first degraded with actinase E (Kaken Biochemicals, Tokyo, Japan), a nonspecific protease derived from Streptomyces griseus.
Heparin (porcine intestinal sodium salt, 150 IU/mg, Celsus Laboratories, Cincinnati, OH, USA) was added in amounts of 0, 60, 600, and 3000 µg in 10 ml citrated human plasma. These plasma samples were then individually proteolyzed at 55 °C with 20 mg actinase E for 18 h. After the proteolysis, dry urea (8 g) and dry Chaps (0.36 g) were added to each sample to afford 18 ml solution (2 wt% in Chaps and 8 M in urea). The resulting cloudy solutions were clarified by passing through a 0.2-µm syringe filter (Millipore, Billerica, MA, USA). Vivapure Maxi Q spin columns (20 ml, Viva Science, Edgewood, NJ, USA) were equilibrated with 3 ml of 8 M urea containing 2% Chaps (pH 8.3). The clarified filtered samples were loaded onto and run through the Vivapure Maxi Q spin columns under centrifugal force (500g). The columns were first washed with 3 ml of 8 M urea containing 2% Chaps (pH 8.3) and then washed five times with 5 ml of 500 mM NaCl. Heparin was released from the spin column by washing three times with 1 ml of 2 M NaCl. The 2-M NaCl washes from each sample were combined, and the NaCl concentration was adjusted to 16%. Methanol (12 ml) was added to afford an 80 vol% solution, and the mixture was incubated at 4 °C for 18 h. The resulting precipitate was recovered by centrifugation (2500g) for 15 min, dissolved in 2.5 ml of water, and freeze-dried for further analysis. The same procedure was applied to recover endogenous GAGs from plasma with two exceptions. First, 50 ml of plasma was used. Second, a reduced concentration of NaCl (100 mM) was used to wash the Vivapure Maxi Q column prior to releasing endogenous GAGs with 2 M NaCl. The recovered heparin and endogenous GAGs were subjected to carbazole assay [10], polyacrylamide gel electrophoresis (PAGE) [11], disaccharide analysis [12], and 500 MHz 1H nuclear magnetic resonance (NMR) analysis.
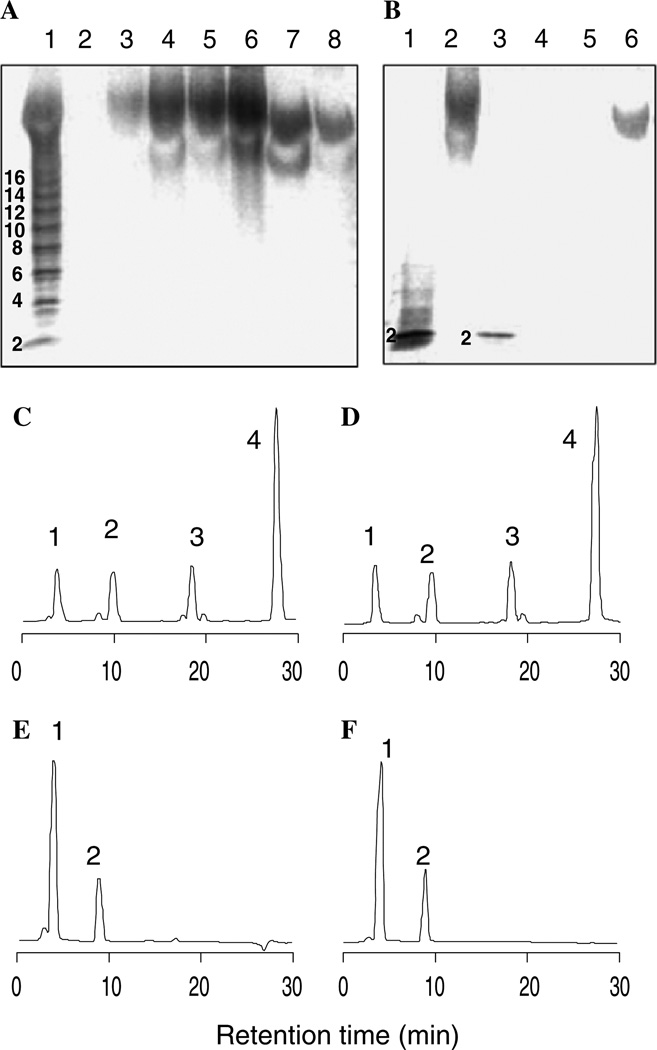
Heparin/Endogenous GAGs recovered from the plasma samples were analyzed by PAGE with Alcian blue staining (Figs. 1A and B). The plasma samples containing added heparin showed a broad band with an average molecular weight (MWavg) of 12,000 [11] and a polydispersity similar to that of the added heparin (Fig. 1A, lanes 3–6). The endogenous GAGs recovered from plasma showed reduced staining intensity, polydispersity, and molecular weight (MWavg ~7500), appearing identical to a peptidoglycan prepared by actinase E treatment of bikunin (Mochida Pharmaceuticals, Tokyo, Japan), a human plasma proteoglycan trypsin inhibitor used in Japan as an anti-tumor drug (Fig. 1A, lanes 7 and 8) [13]. The structure of heparin and endogenous polysaccharide was next examined using specific polysaccharide lyases. PAGE analysis clearly demonstrates that added heparin was sensitive only to heparin lyases (Seikagaku, Tokyo, Japan), affording a prominent trisulfated disaccharide product corresponding to ΔUA2S–GlcNS6S (labeled “2” in Fig. 1B, lanes 2 and 3), whereas endogenous polysaccharide was sensitive only to chondroitin ABC lyase (Seikagaku), affording unsulfated and monosulfated disaccharide products that could not be visualized with Alcian blue staining (Fig. 1B, lanes 4–6) [14].
Fig. 1.
Analysis of GAGs recovered from plasma. (A) Analysis of intact GAGs. Lanes 1: heparin oligosaccharide standards; 2: plasma; 3: plasma + 60 µg heparin; 4: plasma + 600 µg heparin; 5: plasma + 3 mg heparin; 6: heparin standard; 7: peptidoglycan standard; 8: endogenous GAGs. (B) Analysis of lyase treated GAGs. Lanes 1: heparin standard treated with heparin lyases; 2: heparin treated with chondroitin lyase ABC; 3: recovered heparin treated with heparin lyases; 4: peptidoglycan standard treated with chondroitin lyase ABC; 5: endogenous GAGs treated with chondroitin lyase ABC. 6: endogenous GAGs treated with heparin lyases. (C–F) RPIP–HPLC disaccharide analysis [8]. (C) Heparin recovered from plasma. (D) heparin standard. peaks (1–4) and areas (mole %) in panels C and D correspond to ΔUA–GlcNAc (7.2, 6.8), ΔUA–GlcNS (8.4, 8.6), ΔUA–GlcNS6S (12, 11) and ΔUA2S–GlcNS6S (68, 70), respectively. (E) peptidoglycan recovered from plasma. (F) peptidoglycan standard. Peaks (1 and 2) and areas (mole %) in panels E and F correspond to ΔUA–Gal- NAc (62, 68) and ΔUA–GalNAc4S (38, 32), respectively.
The recovered heparin affords a mixture of disaccharides when treated with heparin lyases 1, 2, and 3 (Seikagaku) that was resolved by RPIP–HPLC and detected by postcolumn fluorescence detection [8]. Analysis showed the presence of four major disaccharide products of the structures ΔUA–GlcNAc, ΔUA–GlcNS, ΔUA–GlcNS6S, and ΔUA2S–GlcNS6S (Fig. 1C). The presence of the major disaccharide eluting at 28 min corresponding to ΔUA2S– GlcNS6S is entirely consistent with heparin. Moreover, disaccharide analysis of the pharmaceutical heparin added to the plasma afforded an identical chromatogram (Fig. 1D). The disaccharide analysis of the endogenous polysaccharide recovered from human plasma showed two peaks corresponding to ΔUA–GalNAc and ΔUA–Gal-NAc4S (Fig. 1E). The disaccharide analysis of bikunin was identical (Fig. 1F), further indicating that this represents the major endogenous plasma polysaccharide. Heparin recovered from the sample containing 3 mg/10 ml of plasma was dried, D2O exchanged, and analyzed by 500 MHz 1H NMR. The resulting spectrum (not shown) was identical to that of the pharmaceutical heparin standard.
The recovery of 0, 20, 40, 60, and 100 µg of heparin from 10 ml of citrated human plasma was determined using a carbazole assay for uronic acids based on a heparin standard curve. The amount of recovered GAG was plotted as a function of the amount of heparin added to each sample (y = 0.64x + 8.70, r2 = 0.99) (Fig. 2). The slope of this curve demonstrated a 64% recovery of exogenous heparin across the concentration range tested, and the intercept suggested that human plasma contains 0.87 µg/ml of endogenous bikunin GAG. Smaller Vivapure Mini Q spin columns (0.5 ml) were used to optimize the recovery of heparin (4 µg/ml) and LMWH prepared by oxidative cleavage (MWavg 4800, 74IU/mg, Celsus Laboratories) (4 µg/ml) added to 2 ml of plasma, affording 92.5 and 80.5% recoveries of heparin and LMWH, respectively, with very low assay variability (Fig. 2).
Fig. 2.
Quantification of heparin/LMWH recovered from plasma. The equation of the resulting curve is y = 0.64x + 8.70, r2 = 0.99. ○, Heparin recovery from plasma using Maxi Q column; ■, heparin and ▲, LMWH recovered from plasma using Mini Q column.
In summary, heparin and LMWH can be recovered from human plasma using a simple procedure involving protease digestion and strong anion-exchange chromatography on a spin column, followed by salt release and methanol precipitation. The recovered heparin is free of most contaminants, containing only 0.87 µg/ml of the endogenous chondroitin sulfate plasma peptidoglycan, bikunin. Approximately 90% of heparin, at a concentration of 4 µg/ml, was recovered from 2 ml of plasma and in sufficient purity to be quantified by carbazole assay and to have its molecular weight properties and disaccharide composition determined.
Acknowledgments
This work was supported by the National Institutes of Health (Grant GM 38060 to R. J. Linhardt) and the Ministry of Education, Culture, Sports, Science, and Technology of Japan (Grant 11672136 to T. Toida).
Footnotes
Abbreviations used: LMWH, low-molecular weight heparin; GAG, glycosaminoglycan; CHAPS; 3-[3-cholamidopropyl]-dimethyl ammonio propane sulfonic acid; ΔUA, 4-deoxy-2-l-threo-hex-enepyranosyluronic acid; GlcA, d-glucopyranosyluronic acid; IdoA, l-idopyranosyluronic acid; GlcN, 2-deoxy, 2-amino glucopyranose; GalN, 2-deoxy, 2-amino galactopyranose; S, sulfo; Ac, acetyl; GAG, glycosaminoglycan; MW, molecular weight; MWavg, average molecular weight; RPIP, reversed phase ion pairing.
References
- 1.Casu B, Lindahl U. Structure and biological interactions of heparin and heparan sulfate. Adv. Carbohydr. Chem. Biochem. 2001;57:159–206. doi: 10.1016/s0065-2318(01)57017-1. [DOI] [PubMed] [Google Scholar]
- 2.Linhardt RJ. Heparin: structure and activity. J. Med. Chem. 2003;46:2551–2554. doi: 10.1021/jm030176m. [DOI] [PubMed] [Google Scholar]
- 3.Linhardt RJ. Heparin: an important drug enters its seventh decade. Chem. Ind. 1991;2:45–50. [Google Scholar]
- 4.Linhardt RJ, Toida T. Role of glycosaminoglycans in cellular communication. Acc. Chem. Res. 2004;37:431–438. doi: 10.1021/ar030138x. [DOI] [PubMed] [Google Scholar]
- 5.Munoz EM, Linhardt RJ. Heparin binding domains in vascular biology. Arterioscler. Thromb. Vasc. Biol. 2004;24:1549–1557. doi: 10.1161/01.ATV.0000137189.22999.3f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fareed J, Hoppensteadt D, Schultz C, Ma Q, Kujawski MF, Neville B, Messmore H. Biochemical and pharmacologic heterogeneity in low molecular weight heparins: impact on the therapeutic profile. Curr. Pharm. Des. 2004;10:983–999. doi: 10.2174/1381612043452811. [DOI] [PubMed] [Google Scholar]
- 7.Hoppensteadt DA, Kahn S, Fareed J. Factor X values as a means to assess the extent of oral anticoagulation in patients receiving anti-thrombin drugs. Clin. Chem. 1997;43:1786–1788. [PubMed] [Google Scholar]
- 8.Toyoda H, Kinoshita-Toyoda A, Selleck S. Structural analysis of glycosaminoglycans in Drosophila and Caenorhabditis elegans and demonstration that tout-velu, a Drosophila gene related to EXT tumor suppressors, affects heparan sulfate in vivo. J. Biol Chem. 2000;275:2269–2275. doi: 10.1074/jbc.275.4.2269. [DOI] [PubMed] [Google Scholar]
- 9.Ulpin V, Cusamano M, Venturelli T. Qualitative and quantitative studies of heparin and chondroitin sulfates in normal human plasma. Biochem. Biophys. Acta. 1995;1243:49–58. doi: 10.1016/0304-4165(94)00123-f. [DOI] [PubMed] [Google Scholar]
- 10.Bitter T, Muir HM. A modified uronic acid carbazole reaction. Anal. Biochem. 1962;4:330–334. doi: 10.1016/0003-2697(62)90095-7. [DOI] [PubMed] [Google Scholar]
- 11.Edens RE, Al-Hakim A, Weiler JM, Rethwisch DG, Fareed J, Linhardt RJ. Gradient polyacrylamide gel electrophoresis for determination of the molecular weights of heparin preparations and low-molecular-weight heparin derivatives. J. Pharm. Sci. 1992;81:823–827. doi: 10.1002/jps.2600810821. [DOI] [PubMed] [Google Scholar]
- 12.Ha YW, Jeon BT, Moon SH, Toyoda H, Toida T, Linhardt RJ, Kim YS. Characterization of heparan sulfate from the unossified antler of Cervus elaphus. Carbohydr. Res. 2005;340:411–416. doi: 10.1016/j.carres.2004.11.011. [DOI] [PubMed] [Google Scholar]
- 13.Kobayashi H, Yagyu T, Inagaki K, Kondo T, Suzuki M, Kanayama N, Terao T. Bikunin plus paclitaxel markedly reduces tumor burden and ascites in mouse model of ovarian cancer. Int. J. Cancer. 2004;110:134–139. doi: 10.1002/ijc.20082. [DOI] [PubMed] [Google Scholar]
- 14.Gu K, Liu J, Pervin A, Linhardt RJ. Comparison of the activity of two chondroitin AC lyases on dermatan sulfate. Carbohydr. Res. 1993;244:369–377. doi: 10.1016/0008-6215(83)85014-9. [DOI] [PubMed] [Google Scholar]