Abstract
Purpose
To evaluate the efficacy of anti-vascular endothelial growth factor (VEGF) compared with observation for treating acute central serous chorioretinopathy (CSC).
Methods
A retrospective study of 36 patients with acute CSC, including 21 patients treated with anti-VEGF (anti-VEGF group) and 15 patients with observation (observation group). Patients in the anti-VEGF group received a single dose of bevacizumab or ranibizumab at baseline. Best-corrected visual acuity (BCVA), central foveal thickness (CFT) and resolution of subretinal fluid (SRF) on optical coherence tomography (OCT) were assessed. The integrity of the foveal inner segment/outer segment (IS/OS) line at 12 months was also analyzed.
Results
Resolution of SRF was achieved in 20 of 21 eyes in the anti-VEGF group and in 12 of 15 eyes in the observation group (p = 0.151). Mean BCVA and CFT were not different between the two groups at 12 months (p > 0.05). The amount of change in BCVA, however, differed significantly between the groups (p = 0.044). Final OCT more frequently detected the foveal IS/OS line in the anti-VEGF group than in the observation group (p = 0.012).
Conclusions
In terms of BCVA, anti-VEGF and observation only had similar therapeutic effects in acute CSC patients. In some patients, however, the rapid resolution of SRF by anti-VEGF might reduce the risk of photoreceptor degeneration and improve long-term visual acuity.
Keywords: Anti-vascular endothelial growth factor, Central serous chorioretinopathy, One year
Central serous chorioretinopathy (CSC) is a serous neurosensory detachment that usually involves the macular area. It commonly occurs in patients between the ages of 30 and 50 years and affects men more often than women with a 2 : 1 ratio [1,2]. Risk factors include psychological stress, type A personality, systemic steroid use, hypertension, and pregnancy [3,4,5]. The precise mechanism underlying the development of CSC is not known, but abnormalities in choroidal venous congestion are proposed to play a major role [6,7,8]. CSC may be a response to ischemia and delayed arterial filling, or a consequence of outflow obstruction.
CSC is traditionally managed by observation alone as a spontaneous improvement in symptoms often occurs. Another management option is laser photocoagulation to seal off the leakage site and enhance subretinal fluid (SRF) absorption [9,10]. Although laser treatment may speed up spontaneous recovery, it has no positive impact on the final visual acuity of the patients [4]. More recently, photodynamic therapy (PDT) with verteporfin has been used to treat CSC. Multiple studies have demonstrated beneficial effects of PDT in the majority of subjects [11,12,13], but potential complications, such as retinal pigment epithelial (RPE) atrophy, secondary choroidal neovascularization, and choroidal ischemia, have also been reported [14].
Vascular endothelial growth factor (VEGF) is produced by retinal and choroidal cells damaged by ischemia as a result of abnormal vascular perfusion. VEGF increases vascular permeability and edema [15]. Therefore, anti-VEGF agents (bevacizumab or ranibizumab) are used therapeutically to reduce or reverse the choroidal leakage in acute CSC. Intravitreal injection of bevacizumab (Avastin; Genentech Inc., San Francisco, CA, USA) in patients with CSC was recently reported to be associated with visual improvement and reduced neurosensory detachment without adverse events [16]. Ranibizumab (Lucentis, Genentech Inc.) has potentially better retinal penetration than bevacizumab because of its smaller molecular size and higher binding affinity for VEGF. Reports of the 1-year outcome of intravitreal anti-VEGF injection for acute CSC, however, are scarce [17]. The purpose of the present study is to evaluate the 1-year efficacy of intravitreal anti-VEGF injection as a treatment for patients with acute CSC. Additionally, we investigated the integrity of photoreceptor inner segment/outer segments (IS/OS) in each group.
Materials and Methods
Study design and patient selection
We performed a retrospective study of 50 eyes from 50 patients with acute CSC who were treated at the Kangwon National University Hospital between January 2009 and December 2011. Inclusion criteria included the following: 1) CSC with SRF involving the fovea demonstrated by clinical examination and spectral-domain optical coherence tomography (SD-OCT; Cirrus HD-OCT, Carl Zeiss Meditec, Dublin, CA, USA); 2) active angiographic leakage in fluorescein angiography (FA; HRA+OCT Spectralis, Heidelberg Engineering GmbH, Heidelberg, Germany) caused by CSC but no other retinal diseases, and abnormal dilated choroidal vasculature with hyperpermeability in indocyanine green angiography (ICGA); and 3) the first episode of symptoms and symptom duration of ≤12 weeks. Patients who had evidence of choroidal neovascularization, polypoidal choroidal vasculopathy, or other maculopathies capable of causing macular exudation, such as age-related macular degeneration, pathologic myopia, angioid streaks, and ocular, hereditary or any other disease were excluded. Patients were classified based on the method of treatment as follows: anti-VEGF (n = 21 eyes; 6 eyes bevacizumab and 15 eyes ranibizumab) group and observation only (n = 29 eyes). Informed consent was obtained from all patients. The study was approved by the Kangwon National University Hospital Institutional Research Board.
All patients received a complete ocular examination, including best-corrected visual acuity (BCVA), dilated fundus examination with slit-lamp biomicroscopy, color fundus photography, digital FA, and ICGA at baseline. BCVA was measured with a standard Snellen chart at 6 m and converted to logarithm of the minimum angle of resolution (logMAR) visual acuity for statistical analysis.
Spectral domain-optical coherence tomography
In the present study, a 6 mm × 6 mm (horizontal × vertical) area of the macular region centered on the fovea was examined by an experienced physician using SD-OCT (Cirrus HD-OCT, Carl Zeiss Meditec) with macular cube (512 × 128) scans while monitoring central fixation. Patients were examined 30 min after administration of three drops of intraocular mydriatics (Mydrin-P; Santen, Osaka, Japan) applied with a 5-min interval. The central 1-mm subfield foveal thickness (CFT) was automatically measured using Cirrus analysis software (version 3.0). In addition, to determine the prognostic factors of good visual recovery, we analyzed the integrity of the foveal IS/OS line in both groups. The IS/OS lines beneath the fovea were classified as visible or invisible beneath the fovea. Two retina specialists (MK and SJL) reviewed the OCT images.
Intravitreal anti-vascular endothelial growth factor injection
All patients received a single dose of anti-VEGF (bevacizumab or ranibizumab) at baseline. All intravitreal injections were performed in the operating room under sterile conditions. Topical anesthetic drops were given first, and then a lid speculum was inserted. After cleaning the injection site with 5% povidone iodine, a 30-gauge needle was inserted through the pars plana, and 0.05 mL (0.5 mg) of ranibizumab or 0.05 mL (1.25 mg) of bevacizumab was injected. Indirect ophthalmoscopy and tonometry were performed after the procedure to detect any injection-related complications. Patients were given topical antibiotics 4 times daily for 1 week after each injection.
Follow-up
All patients were followed up at 1, 3, 6, 9, and 12 months with BCVA testing, dilated fundus examination, color fundus photography and SD-OCT. FA and ICGA were performed in all patients at baseline and at 12 months. Additional FA and ICGA were performed in patients with CSC recurrence during the follow-up period. Recurrence was defined as an increase in SRF observed on OCT, leakage noted on FA, and abnormal dilated choroidal vasculature with hyperpermeability in ICGA.
Statistical analysis
All data were collected in a Excel 2007 spreadsheet (Microsoft, Redmond, WA, USA). Statistical analysis was performed using SPSS ver. 16.0 (SPSS Inc., Chicago, IL, USA). Snellen units were converted to logMAR units for statistical analysis. Wilcoxon's rank test was used to compare variables at baseline and after treatment. The Mann-Whitney U-test was used to compare variables between the two groups. Fisher's exact test was used to compare the percentage of a certain feature in each group. A p-value of less than 0.05 was considered statistically significant.
Results
Patient demographics and baseline characteristics
We performed a retrospective study of 50 eyes (21 eyes, anti-VEGF group; 29 eyes, observation group) from 50 patients with acute CSC. Thirty-six eyes of 36 patients with acute CSC underwent treatment and completed at least 12 months of follow-up. The other 14 patients were excluded from the study for the following reasons: 1) lost to follow-up (12 patients), 2) cataract surgery (1 patient), and 3) traumatic hyphema (1 patient). The anti-VEGF group comprised 21 patients (21 eyes; 6 eyes bevacizumab, 15 eyes ranibizumab), and the observation group comprised 15 patients (15 eyes). Analysis of demographic data on sex, age, and symptom duration revealed no significant differences between the two groups (p > 0.05). Baseline visual acuity (logMAR) and baseline CFT were not significantly different between the two groups. The baseline demographic data of both groups are shown in Table 1.
Table 1.
Baseline characteristics of patients with acute central serous chorioretinopathy

Values are presented as mean ± SD or number unless otherwise indicated.
VEGF = vascular endothelial growth factor; BCVA = best-corrected visual acuity; logMAR = logarithm of minimal angle of resolution; CFT = central foveal thickness.
*Mann-Whitney U-test; †Fisher exact test.
Resolution of subretinal fluid
Mean time from baseline to initial complete resolution of SRF was significantly shorter in the anti-VEGF group (1.2 ± 0.6 months) than in the observation group (3.5 ± 2.9 months, p = 0.001). In the anti-VEGF group, 19 of the 21 eyes showed no SRF on OCT at 1 month. Of the remaining 2 eyes, SRF had completely disappeared by 3 months. One eye in the anti-VEGF group in which SRF had completely disappeared at 1 month showed recurrence of the SRF at 9 months. This patient refused any further treatment. In the observation group, complete resolution of SRF on OCT was achieved in 5 of the 15 eyes at 1 month, and SRF resolved completely in the remaining 6 eyes at 3 months and 3 eyes at 6 months. One eye did not show resolution of SRF until the follow-up examination at 12 months. Two eyes in the observation group in which SRF had completely disappeared at 1 month showed recurrence of the SRF at 12 months. The overall success rate without recurrence was similar between groups: 20 eyes (95.2%) in the anti-VEGF group and 12 eyes the observation group (80.0%, p = 0.151).
Visual acuity
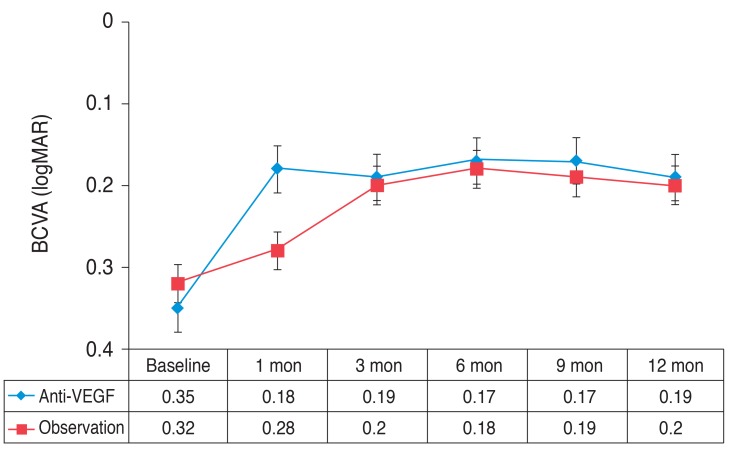
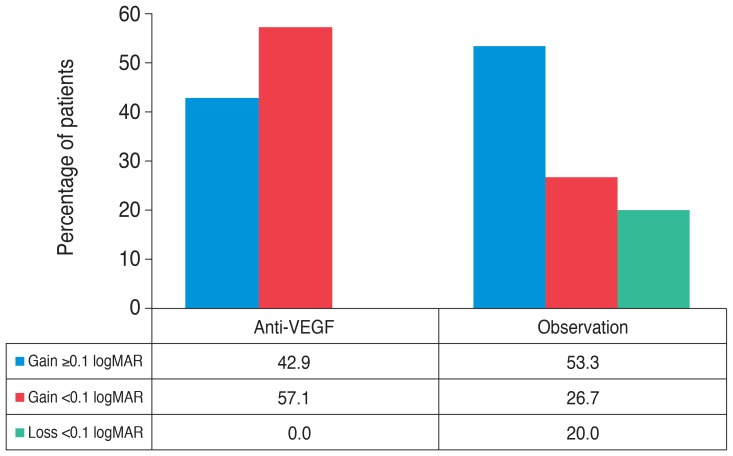
Mean BCVA changes from baseline in both groups are shown in Fig. 1. BCVA significantly improved from baseline over the entire follow-up period in both groups (p < 0.05). Comparison of the two groups revealed no statistically significant difference in BCVA improvement (p > 0.05) during the 12-month period, except at 1 month (p = 0.026). When the groups were stratified by the amount of change in BCVA (improved BCVA ≥0.1 logMAR, improved BCVA <0.1 logMAR, or loss BCVA <0.1 logMAR), however, significantly more patients in the anti-VEGF group (57.1%) had a moderate improvement in BCVA (<0.1 logMAR) than in the comparison group (26.7%), and none of the anti-VEGF group had a loss in BCVA, while 3 (20.0%) of the observation group patients had a loss in BCVA (Fig. 2).
Fig. 1.
Serial changes in the mean logarithm of the minimum angle of resolution (logMAR) for patients in the anti-vascular endothelial growth factor (VEGF) and the observation. Error bars represent the standard error of the mean. BCVA = best-corrected visual acuity.
Fig. 2.
Patients stratified by the amount of best-corrected visual acuity change in the anti-vascular endothelial growth factor (VEGF) and observation groups. logMAR = logarithm of the minimum angle of resolution.
Central foveal thickness
The mean CFT changes from baseline in both groups are summarized in Fig. 3. CFT during the entire follow-up period was significantly smaller than that at baseline in both groups (p < 0.05). Comparison of the two groups revealed that while CFT at baseline, 6 months, and 12 months did not differ significantly between groups, CFT at 1 month and 3 months was significantly smaller in the anti-VEGF group (p = 0.006 and p = 0.045, respectively).
Fig. 3.
Serial changes in mean central foveal thickness (CFT) as measured using optical coherence tomography in the anti-vascular endothelial growth factor (VEGF) and the observation groups. Error bars represent the standard error of the mean.
Integrity of foveal inner segment/outer segment
The photoreceptor IS/OS layer was evaluated, on horizontal and vertical scans, for 500 µm in either direction of the fovea. The IS/OS disruption was graded from 0 to 2: grade 0 meaning intact IS/OS layer, grade 1 focal disruption of the IS/OS junction of ≤200 µm, and grade 2 disruption of the IS/OS junction of >200 µm.
At baseline, all eyes in both groups showed slightly reflective lines in the outer nuclear layer, which was thought to be the external limiting membrane. A highly reflective line that is generally considered to indicate the junction between the photoreceptor IS/OS was not detected. At 12 months, the foveal IS/OS was intact in 20 eyes (grade 1, 95.2%) and focal disruption in 1 eye (grade 2, 4.8%) in the anti-VEGF group. In the observation group, however, the IS/OS line was intact beneath the fovea in 10 eyes (grade 1, 66.7%), 2 eyes had focal disruption (grade 2, 13.37%) and 3 eyes had disruption (grade 2, 20.0%). Statistical analysis revealed that eyes in the anti-VEGF group had higher rates of preservation of the foveal IS/OS line (p = 0.023) than eyes in the observation group.
Adverse effects of anti-vascular endothelial growth factor
None of the patients in the anti-VEGF group developed systemic complications related to anti-VEGF, such as cerebral vascular accidents or thromboembolic events. We did not detect any ocular complications, such as cataracts, increased intraocular pressure, retinal detachment, endophthalmitis, or central retinal artery occlusion. No retinal pigment epithelial tears were detected.
Discussion
CSC has long been considered a condition that will resolve spontaneously and thus the mainstay of management is usually observation alone [18]. There is, however, some evidence to support the benefit of early treatment of acute CSC. A potential benefit of early resolution of SRF might be mediated by a lower rate of RPE degeneration in the treated eye, which might be warranted due to the uncertain relationship between the onset of detachment and the onset of symptoms and special occupational demands for binocular vision function. This has led clinicians to search for a more effective treatment alternative, one less likely than PDT or focal laser photocoagulation to cause side effects. Although focal laser photocoagulation and PDT have proven effective for the treatment of CSC, these treatments can cause permanent damage to the RPE or choriocapillary, retinal thermal injury, and subretinal choroidal neovascularization, often many years following the primary incident [19,20].
VEGF has dramatic effects on vascular permeability. The direct role of VEGF in CSC, however, remains unknown. ICGA in patients with CSC has provided evidence of choroidal lobular ischemia, choroidal venous congestion, and multiple areas of choroidal vascular hyperpermeability [15,20]. Choroidal ischemia in CSC may induce an increase in the concentration of VEGF. Lim et al. [21] demonstrated that aqueous VEGF levels are increased in a portion of patients with chronic CSC, but not in acute CSC. Several reports, however, indicate that intravitreal bevacizumab injection improves vision and reduces the incidence of neurosensory detachment in patients with acute CSC [22,23]. Ranibizumab is a humanized anti-VEGF antibody that inhibits all forms of biologically active VEGF [24]. Treatment with ranibizumab appears to significantly decrease bleeding and exudation in neovascular age-related macular degeneration [25], polypoidal choroidal vasculopathy [26], and retinal vein occlusion [27]. Ranibizumab has several theoretical advantages over bevacizumab. Considering the molecular weight of each medication (ranibizumab is a 48-kDa Fab fragment, whereas bevacizumab is a complete 149-kDa antibody), ranibizumab may be more effective for treating CSC because of its smaller molecular weight and possible deeper penetration into choroidal vascular hyperpermeability lesions in patients with CSC [28].
To compare the efficacy of intravitreal anti-VEGF injections for acute CSC, we investigated the results of other treatment options in several published studies [17,29,30,31] (Table 2). The visual outcomes and resolution of neurosensory retinal detachment in our study were similar to those of previously reported studies. Indeed, the resolution of SRF often leads to improvement in visual acuity. In the clinical setting, however, some eyes with acute CSC have poor visual acuity immediately after resolution of the serous detachment. To study the prognostic factors of good visual recovery, we investigated the integrity of the foveal IS/OS line of both groups at 12 months. OCT reflections from the IS/OS are thought to arise from the abrupt change in the optical index of refraction at the boundary between the inner segments and the highly organized structure of the outer segments [32]. In eyes with active CSC, however, the IS/OS line could not be detected in the detached retina. Ko et al. [33] suggested that the membranous stack of the photoreceptor segment is no longer perpendicular to the incoming OCT beam, and the highly back-reflecting signal at the IS/OS seems to be absent in cases of detached retina. In our study, the foveal IS/OS line was visible in 20 eyes (95.2%) and invisible in 1 eye (4.8%) in the anti-VEGF group at 12 months. In the observation group, however, the IS/OS line was visible beneath the fovea in 10 eyes (66.7%) and invisible in 5 eyes (33.3%). Table 3 summarizes the composition of these 5 eyes. Based on these results, we suggest that decreasing the duration of neurosensory retinal detachment with anti-VEGF could reduce the risk of photoreceptor degeneration. Longstanding detachment of the retina may cause apoptotic cell death or the loss of the outer segments of the foveal photoreceptor cells [34]. These eyes would not be expected to achieve recovery of the IS/OS line or subsequent visual recovery [30].
Table 2.
One-year results of photodynamic therapy, intravitreal anti-VEGF injection, and observation for the treatment of acute central serous chorioretinopathy in published studies

VEGF = vascular endothelial growth factor; BCVA = best-corrected visual acuity; logMAR = logarithm of minimal angle of resolution; PDT = photodynamic therapy.
Table 3.
Analysis of 5 eyes with a disruption of foveal IS/OS line in the observation group

IS/OS = inner segment/outer segment; SRF = subretinal fluid.
The present study has some limitations. First, this is not a prospective, randomized, controlled clinical study, and the follow-up duration was limited to approximately 1 year. Second, our study has a relatively small sample size. Third, we used bevacizumab or ranibizumab as the anti-VEGF agent. Bevacizumab is a larger molecule than ranibizumab, and therefore the penetration of bevacizumab into the choroidal vasculature might be lower than that of ranibizumab.
In conclusion, compared with observation alone, intravitreal anti-VEGF therapy showed no positive effect in terms of visual outcome during the 12-month follow-up period. Intravitreal anti-VEGF may be efficacious, however, in achieving a rapid resolution of neurosensory retinal detachment in acute CSC patients. Decreasing the duration of neurosensory retinal detachment from RPE could reduce the risk of photoreceptor degeneration and ultimately improve long-term visual acuity. Patients with acute CSC in whom SD-OCT images exhibit an intact foveal IS/OS line have better outcomes after the resolution of SRF, suggesting that SD-OCT is an important tool for evaluating and helping predict the visual prognosis in patients with acute CSC based on these specific structural changes. Despite a growing body of evidence for the efficacy of anti-VEGF treatment for acute CSC, the role of anti-VEGF is not yet fully elucidated. Larger, controlled, prospective studies are needed to determine the long-term benefits and risks of intravitreal anti-VEGF for the treatment of acute CSC.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Levine R, Brucker AJ, Robinson F. Long-term follow-up of idiopathic central serous chorioretinopathy by fluorescein angiography. Ophthalmology. 1989;96:854–859. doi: 10.1016/s0161-6420(89)32810-7. [DOI] [PubMed] [Google Scholar]
- 2.Hussain D, Gass JD. Idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 1998;46:131–137. [PubMed] [Google Scholar]
- 3.Loo RH, Scott IU, Flynn HW, Jr, et al. Factors associated with reduced visual acuity during long-term follow-up of patients with idiopathic central serous chorioretinopathy. Retina. 2002;22:19–24. doi: 10.1097/00006982-200202000-00004. [DOI] [PubMed] [Google Scholar]
- 4.Robertson DM, Ilstrup D. Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am J Ophthalmol. 1983;95:457–466. doi: 10.1016/0002-9394(83)90265-9. [DOI] [PubMed] [Google Scholar]
- 5.Taban M, Boyer DS, Thomas EL, Taban M. Chronic central serous chorioretinopathy: photodynamic therapy. Am J Ophthalmol. 2004;137:1073–1080. doi: 10.1016/j.ajo.2004.01.043. [DOI] [PubMed] [Google Scholar]
- 6.Hayashi K, Hasegawa Y, Tokoro T. Indocyanine green angiography of central serous chorioretinopathy. Int Ophthalmol. 1986;9:37–41. doi: 10.1007/BF00225936. [DOI] [PubMed] [Google Scholar]
- 7.Prunte C. Indocyanine green angiographic findings in central serous chorioretinopathy. Int Ophthalmol. 1995;19:77–82. doi: 10.1007/BF00133176. [DOI] [PubMed] [Google Scholar]
- 8.Prunte C, Flammer J. Choroidal capillary and venous congestion in central serous chorioretinopathy. Am J Ophthalmol. 1996;121:26–34. doi: 10.1016/s0002-9394(14)70531-8. [DOI] [PubMed] [Google Scholar]
- 9.Burumcek E, Mudun A, Karacorlu S, Arslan MO. Laser photocoagulation for persistent central serous retinopathy: results of long-term follow-up. Ophthalmology. 1997;104:616–622. doi: 10.1016/s0161-6420(97)30262-0. [DOI] [PubMed] [Google Scholar]
- 10.Robertson DM. Argon laser photocoagulation treatment in central serous chorioretinopathy. Ophthalmology. 1986;93:972–974. doi: 10.1016/s0161-6420(86)33652-2. [DOI] [PubMed] [Google Scholar]
- 11.Cardillo Piccolino F, Eandi CM, Ventre L, et al. Photodynamic therapy for chronic central serous chorioretinopathy. Retina. 2003;23:752–763. doi: 10.1097/00006982-200312000-00002. [DOI] [PubMed] [Google Scholar]
- 12.Yannuzzi LA, Slakter JS, Gross NE, et al. Indocyanine green angiography-guided photodynamic therapy for treatment of chronic central serous chorioretinopathy: a pilot study. Retina. 2003;23:288–298. doi: 10.1097/00006982-200306000-00002. [DOI] [PubMed] [Google Scholar]
- 13.Chan WM, Lam DS, Lai TY, et al. Choroidal vascular remodelling in central serous chorioretinopathy after indocyanine green guided photodynamic therapy with verteporfin: a novel treatment at the primary disease level. Br J Ophthalmol. 2003;87:1453–1458. doi: 10.1136/bjo.87.12.1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Colucciello M. Choroidal neovascularization complicating photodynamic therapy for central serous retinopathy. Retina. 2006;26:239–242. doi: 10.1097/00006982-200602000-00027. [DOI] [PubMed] [Google Scholar]
- 15.Stanga PE, Lim JI, Hamilton P. Indocyanine green angiography in chorioretinal diseases: indications and interpretation: an evidence-based update. Ophthalmology. 2003;110:15–21. doi: 10.1016/s0161-6420(02)01563-4. [DOI] [PubMed] [Google Scholar]
- 16.Torres-Soriano ME, Garcia-Aguirre G, Kon-Jara V, et al. A pilot study of intravitreal bevacizumab for the treatment of central serous chorioretinopathy (case reports) Graefes Arch Clin Exp Ophthalmol. 2008;246:1235–1239. doi: 10.1007/s00417-008-0856-x. [DOI] [PubMed] [Google Scholar]
- 17.Lim JW, Kim MU. The efficacy of intravitreal bevacizumab for idiopathic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2011;249:969–974. doi: 10.1007/s00417-010-1581-9. [DOI] [PubMed] [Google Scholar]
- 18.Yannuzzi LA. Type-A behavior and central serous chorioretinopathy. Retina. 1987;7:111–131. doi: 10.1097/00006982-198700720-00009. [DOI] [PubMed] [Google Scholar]
- 19.Kanyange ML, De Laey JJ. Long-term follow-up of central serous chorioretinopathy (CSCR) Bull Soc Belge Ophtalmol. 2002;(284):39–44. [PubMed] [Google Scholar]
- 20.Piccolino FC, Borgia L. Central serous chorioretinopathy and indocyanine green angiography. Retina. 1994;14:231–242. doi: 10.1097/00006982-199414030-00008. [DOI] [PubMed] [Google Scholar]
- 21.Lim JW, Kim MU, Shin MC. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina. 2010;30:1465–1471. doi: 10.1097/IAE.0b013e3181d8e7fe. [DOI] [PubMed] [Google Scholar]
- 22.Seong HK, Bae JH, Kim ES, et al. Intravitreal bevacizumab to treat acute central serous chorioretinopathy: short-term effect. Ophthalmologica. 2009;223:343–347. doi: 10.1159/000224782. [DOI] [PubMed] [Google Scholar]
- 23.Lim SJ, Roh MI, Kwon OW. Intravitreal bevacizumab injection for central serous chorioretinopathy. Retina. 2010;30:100–106. doi: 10.1097/IAE.0b013e3181bcf0b4. [DOI] [PubMed] [Google Scholar]
- 24.Lantry LE. Ranibizumab, a mAb against VEGF-A for the potential treatment of age-related macular degeneration and other ocular complications. Curr Opin Mol Ther. 2007;9:592–602. [PubMed] [Google Scholar]
- 25.CATT Research Group. Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011;364:1897–1908. doi: 10.1056/NEJMoa1102673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kokame GT, Yeung L, Lai JC. Continuous anti-VEGF treatment with ranibizumab for polypoidal choroidal vasculopathy: 6-month results. Br J Ophthalmol. 2010;94:297–301. doi: 10.1136/bjo.2008.150029. [DOI] [PubMed] [Google Scholar]
- 27.Brown DM, Campochiaro PA, Bhisitkul RB, et al. Sustained benefits from ranibizumab for macular edema following branch retinal vein occlusion: 12-month outcomes of a phase III study. Ophthalmology. 2011;118:1594–1602. doi: 10.1016/j.ophtha.2011.02.022. [DOI] [PubMed] [Google Scholar]
- 28.Gomi F, Tano Y. Polypoidal choroidal vasculopathy and treatments. Curr Opin Ophthalmol. 2008;19:208–212. doi: 10.1097/ICU.0b013e3282fb7c33. [DOI] [PubMed] [Google Scholar]
- 29.Smretschnig E, Ansari-Shahrezaei S, Moussa S, et al. Half-fluence photodynamic therapy in acute central serous chorioretinopathy. Retina. 2012;32:2014–2019. doi: 10.1097/IAE.0b013e318242b9ab. [DOI] [PubMed] [Google Scholar]
- 30.Zhao MW, Zhou P, Xiao HX, et al. Photodynamic therapy for acute central serous chorioretinopathy: the safe effective lowest dose of verteporfin. Retina. 2009;29:1155–1161. doi: 10.1097/IAE.0b013e3181a6c028. [DOI] [PubMed] [Google Scholar]
- 31.Chan WM, Lai TY, Lai RY, et al. Half-dose verteporfin photodynamic therapy for acute central serous chorioretinopathy: one-year results of a randomized controlled trial. Ophthalmology. 2008;115:1756–1765. doi: 10.1016/j.ophtha.2008.04.014. [DOI] [PubMed] [Google Scholar]
- 32.Drexler W, Sattmann H, Hermann B, et al. Enhanced visualization of macular pathology with the use of ultrahigh-resolution optical coherence tomography. Arch Ophthalmol. 2003;121:695–706. doi: 10.1001/archopht.121.5.695. [DOI] [PubMed] [Google Scholar]
- 33.Ko TH, Fujimoto JG, Duker JS, et al. Comparison of ultrahigh-and standard-resolution optical coherence tomography for imaging macular hole pathology and repair. Ophthalmology. 2004;111:2033–2043. doi: 10.1016/j.ophtha.2004.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Matsumoto H, Kishi S, Otani T, Sato T. Elongation of photoreceptor outer segment in central serous chorioretinopathy. Am J Ophthalmol. 2008;145:162–168. doi: 10.1016/j.ajo.2007.08.024. [DOI] [PubMed] [Google Scholar]