Abstract
Objective: To investigate the potential harmful effects of potassium dichromate and magnesium sulphate causing oxidative stress and reproductive toxicity in adult male mice model.
Methods: The experimental work was conducted on sixty male mice (Mus musculus) divided into three groups. Mice in group B and C received potassium dichromate and magnesium sulphate of 5.0 and 500 mg/Kg body weight/ml respectively, for sixty days. The blood sample was analyzed to assess oxidative stress and cellular damage.
Results: Results showed high malondialdehyde (MDA) and low levels of antioxidant enzymes [catalase (CAT), superoxide dismutase (SOD) and glutathione peroxidase (GPx)] in both potassium dichromate and magnesium sulphate administrated groups as compared to control group. Reduced number of sperm count and excessive destruction of testicular follicles, including destruction of spermatids, leydig cells and sertoli cells, were also seen in both groups.
Conclusion: We concluded from present study that potassium dichromate and magnesium sulphate causes oxidative stress by generation of reactive oxygen species (ROS) and causing DNA damage in testicular cells leading to adverse reproductive abnormalities.
Key Words: Magnesium sulphate (MgSO4), Malondialdehyde (MDA), Oxidative stress, Potassium dichromate (K2Cr2O7), Superoxide dismutase (SOD)
INTRODUCTION
Worldwide distribution and extensive use of chemical agents, is associated with concern of the highest priority for environmental and industrial exposure which may have imposing effects on male reproductive function. Chromium has been identified to be one of these toxic metals. Its common salt, potassium dichromate (K2Cr2O7) is most commonly used as an oxidizing agent in various laboratory and industrial applications.1,2 Chromium VI is more toxic than in trivalent form because it readily enters the cells producing various pathological conditions, including reproductive dysfunction, induce toxicity provoke lipid peroxidation, DNA damage, cytotoxicity, mutagenesis and carcinogenesis.3 Magnesium sulfate (MgSO4) is a chemical compound often encountered as the heptahydrate epsomite (MgSO4·7H2O) commonly called Epsom salt, is used as a first line of treatment in the majority of cases. It is used in replacement therapy for hypomagnesaemia and as a bronchodilator in severe exacerbations of asthma. It is commonly administered via the intravenous route for the management of severe asthma attacks.4,5
Lipid peroxidation is one of the major outcomes of free radical-mediated to cellular injury or beneficial biological effects. K2Cr2O7 and MgSO4 both induce oxidative stress due to which high lipid peroxidation occurs and in result MDA level is increased and antioxidant enzymes SOD and CAT activity is decreased.6,7 The evidence indicates that the careless use of toxic heavy metals in the past twenty years have shown very alarming trend in male reproductive health.8 Testicular tissues are major target organ for metals that induce oxidative damage because of its high contents of polyunsaturated membrane lipids. Ingestion of hexavalent chromium compounds produces uncertain levels of degeneration in the outer most cellular layers in several somniferous tubules, reducing the number of sperm count and spermatogonia per tubule, leading to considerable increases in the morphologically abnormal sperms percentage.9,10 Oral administration of vanadyl sulphate for 60 days caused a decrease in the weights of testes, accessory reproductive organs and the diameter of seminiferous tubules and leydig cells nuclei are reduced.11 The aim of this research work was to check the adverse effects of K2Cr2O7 and MgSO4 on the testes of adult male mice.
METHODS
Experimental design: Sixty adult male mice were included in the study, divided into three groups, comprising twenty mice in each group with average weight ranging from 25-40 gm. Group A served as control, while in group B mice were treated with K2Cr2O7 and in group C mice were administered with MgSO4. All the laboratory work was performed at the Institute of Molecular Biology & Biotechnology, the University of Lahore, during March 2013 to September 2013.
Administration of K 2 Cr 2 O 7 and MgSO 4 : K2Cr2O7 was administrated at the dose of 5mg/Kg body weight while MgSO4 was administrated at the dose of 500mg/Kg body weight.12 Both the salts were administrated orally for the period of 60 days.
Samples collection and sample analysis: 2 ml blood was taken from each mouse at 1st, 30th and 60th day of the experiment and serum of the sample was separated by centrifugation at 3000 rpm. Then sample were processed and analyzed for the estimation of MDA, SOD, CAT and GPx activity.
Estimation of MDA : Thiobarbituric acid reactive substance test was used for the estimation of MDA level in serum.13 Total 1ml of serum was taken and a 10% (w/v) homogenate was prepared in 10 mM buffer (pH 7.4). The supernatant was used for immediate thiobarbituric acid reactive substances test. In this test 200µl of serum sample, 200µl of 8.1% sodium dodecyl sulfate (SDS), 1.5ml of 0.8% TBA, 1.5ml of 20% acetic acid solution (pH 3.5) and 4.0ml distilled water and 5.0 ml of n-butanol were used and absorbance at 532nm was recorded.
Estimation of SOD: All procedure for estimation of SOD was performed in ice bath.14 Blood serum was taken and homogenate was prepared in 50% TCA and centrifuged at 13000 rpm. The supernatant was used for immediate SOD and other enzyme activity evaluation. 100µl of serum sample, 1.2ml of sodium phosphate buffer (pH 8.3, 0.052M), 100µl of phenazine methosulphate (186µm), 300 µl of nitro blue tetrazolium (300µm), 200 µl of NADH (750µm) and 4.0 ml of n- butanol were used and absorbance was recorded at 560nm.
Estimation of CAT activity: Catalase activity was measured by the method of Aebi.15 The supernatant was used for the estimation of catalase. The rate of decomposition of H2O2 was measured at 240nm.
Estimation of GPx activity: Glutathione peroxidase was determined by homogenizing 0.1 ml of serum with 2.4ml of 0.02 M EDTA first and then the test tubes were kept in an ice bath for 10 minutes. 2ml of distilled water and 0.5 ml of TCA (50%) was added and was kept in an ice bath for 10-15 minutes. The mixture was centrifuged at 3000-3500rpm for 10 minutes. 1ml of the supernatant was taken in test tube and 2ml of 0.15M Tris HCL plus 0.05ml DTNB were added in it and absorbance was taken at 412nm.
Histopathology of testicles: Two mice from each group were randomly selected for histopathology of testis at 1st, 30th and 60th day of the experiment. These mice were sacrificed and the specimens from testicular tissues were fixed in 10% neutral buffer formalin, dehydrated in ascending grades of ethanol alcohols, cleared in xylol, casted, blocked, cut at 2-5 μm thickness and stained with hematoxylin-eosin for microscopic examination.16
Statistical analysis: All the data thus obtained was statistically analyzed by applying one way analysis of variance (ANOVA). The differences of the means were considered significant at p < 0.05.
RESULTS
MDA levels in male mice receiving K 2 Cr 2 O 7 and MgSO 4 : MDA levels in different groups at different days were studied. It was concluded from the descriptive statistics that at first day insignificant differences in MDA levels between all the groups were observed as compared to control group (Table-Ia). While significant differences in MDA levels were observed in all the groups at 30th and 60th day as compared to control group. The results showed that both of these salts cause significant elevation in MDA levels, resulting in high lipid peroxidation.
Table-Ia.
MDA levels in different groups at different days
| Days | Groups | Means ± SD ( μmol/ml ) | (P-Value) |
|---|---|---|---|
| 1st day | A B C |
24.64+.34 24.16+.30 24.48+0.56 |
0.426 |
| 30th day | A B C |
24.49+0.31 26.80+0.76 26.57+0.20 |
0.004* |
| 60th day | A B C |
24.50+0.62 27.69+0.25 26.78+0.21 |
0.001* |
Significant (p < 0.05)
SOD levels in male mice receiving K 2 Cr 2 O 7 and MgSO 4 : Further, SOD levels in different groups at different days were studied. The SOD level decreased significantly in the groups B and C at 30th and 60th day as compared to control groups (Table-Ib). It is concluded that both of these salts cause significant depletion of SOD levels, resulting in high lipid peroxidation. SOD level in MgSO4 treated mice is even lower than with K2Cr2O7 treated mice.
Table-Ib.
SOD levels in different groups at different days
| Days | Groups | Means ± SD ( μg/ml ) | (P-Value) |
|---|---|---|---|
| 1st day | A B C |
28.77+1.18 28.70+0.65 28.27+0.64 |
0.329 |
| 30th day | A B C |
28.35+1.13 26.16+0.43 19.43+2.04 |
0.004* |
| 60th day | A B C |
28.51+0.48 22.82+0.91 22.44+5.08 |
0.001* |
Significant (p < 0.05)
Levels of CAT in male mice receiving K 2 Cr 2 O 7 and MgSO 4 : Table-IIa shows the CAT levels in different groups at different days. Significant differences in CAT levels were observed in all the groups at 60th day as compared to control group. CAT level was found to be more reduced with MgSO4 as compared to K2Cr2O7.
Table-IIa.
Catalase levels in different groups at different days
| Days | Groups | Means ± SD ( μmol/mol of protein ) | (P-Value) |
|---|---|---|---|
| 1st day | A B C |
191.47+0.62 190.41+0.69 191.29+0.44 |
0.147 |
| 30th day | A B C |
190.78+0.98 183.63+6.70 173.30+2.79 |
0.357 |
| 60th day | A B C |
190.52+0.94 186.52+1.32 179.53+4.08 |
0.005* |
Significant (p < 0.05)
GP x level in male mice receiving K 2 Cr 2 O 7 and MgSO 4 : Table-IIb shows the GPx levels in different groups at different days. Significant differences in GPx levels were observed in all the groups at 30th and 60th day as compared to control group. So it was concluded that both of these salts cause significant decrease of GPx levels, resulting in high oxidative stress. In MgSO4 treated mice the GPx level reduced substantially, making it more lethal than K2Cr2O7.
Table-IIb.
Glutathione peroxidase levels in different groups at different days
| Days | Groups |
Means ± SD
(µmol/g protein) |
(P-Value) |
|---|---|---|---|
| 1st day | A B C |
33.5+0.62 32.7+0.69 34.9+0.44 |
0.321 |
| 30th day | A B C |
31+0.98 19.9+6.70 09.7+2.79 |
0.005* |
| 60th day | A B C |
31.4+0.94 15.3+1.32 07.7+4.08 |
0.001* |
Significant (p < 0.05)
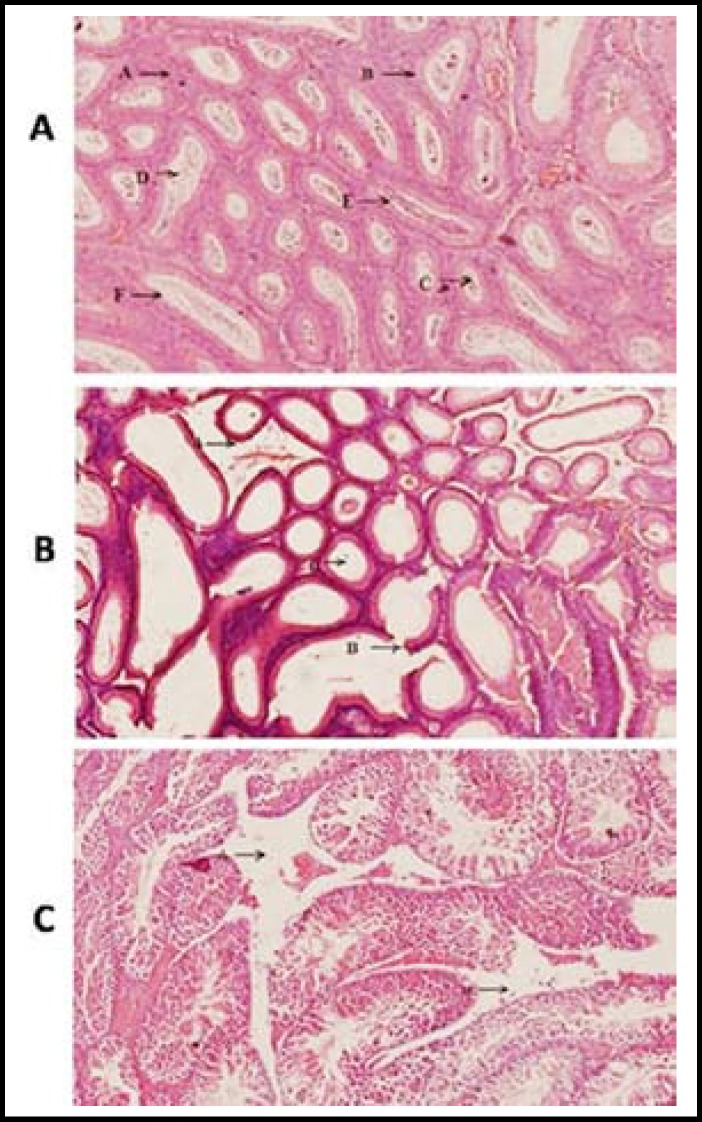
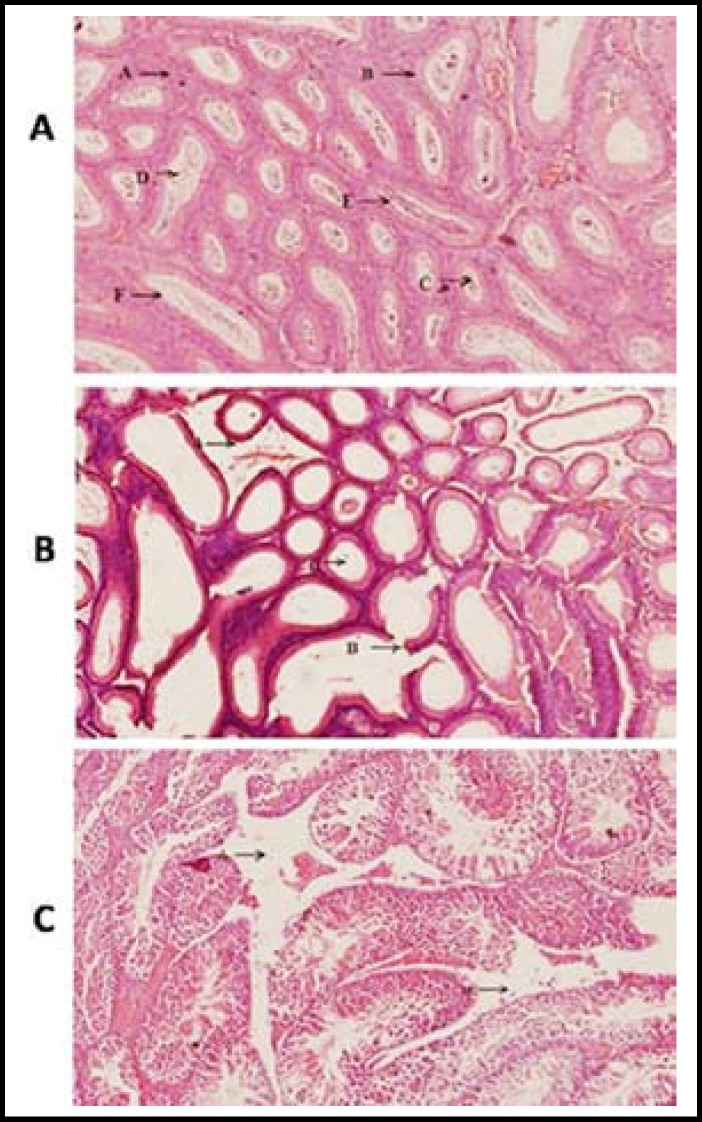
Morphological analysis of Testicular tissue: We also studied the morphology of the cross sectional structure of testicles of normal follicles with intact germinal layer and leydig cell and the results showed complete destruction of leydig cells, partially damaged germinal layer and destruction of sperms leading to empty spaces in the lumen of follicle of testicles in which 30% destruction of follicle was observed in the group at 30th day after the administration of K2Cr2O7 and 35% destruction of follicle was observed in the group at 30th day after administration the MgSO4 (Fig. 1a, b, c). Furthermore, 55% destruction of follicle was observed in this cross section of group B at 60th day after administration the K2Cr2O and 64% destruction of follicle was observed in this group at 60th day after administration the MgSO4 (Fig. 2a, b, c).
Fig.1.
a) Cross section of testicle of mice of control group at 30th day of experiment. A. Leydig Cells, B. Germinal Layer, C. Spermatids, D. Spermatocytes, E. Spermatogonia, F. Sertoli cell.
b) Cross section of testicle of mice of group B at 30th day. A. Destruction of Leydig cells, B. Partially damaged germinal layer, C. Empty central lumen of follicle of testis without sperms.
c) Cross section of testicle of mice of group C at 30th day. A. Destruction of Leydig cells B. partially damaged germinal layer.
Fig.2.
a) Cross section of testicle of mice of control group at 60th day of experiment. A. Leydig Cells, B. Germinal Layer, C. Spermatids, D. Spermatocytes, E. Spermatogonia, F. Sertoli cell.
b) Cross section of testicle of mice of group B at 60th day. A. Destruction of Leydig cells and D. destruction of complete follicles.
c) Cross section of testicle of mice of group C at 60th day. A. Destruction of Leydig cells, B. Empty central lumen of follicle of testis without sperms, C. partially damaged germinal layer D. destruction of complete follicle.
DISCUSSION
Hexavalent chromium is very toxic and readily enters the cells and induces toxicity provoke lipid peroxidation, DNA damage, cytotoxicity, mutagenesis and carcinogenesis. MgSO4 effects on the lipid peroxidation and significantly increases MDA.3,6 The results of lipid peroxidation, MDA, SOD, CAT and GPx showed significant difference in MDA, SOD, CAT and GPx levels in K2Cr2O7 and MgSO4 administrated group after 30th and 60th days treatment. The outcomes were in line with the Goulart et al.17. However, further research is needed to estimate the effect of MgSO4 on lipid peroxidation. In K2Cr2O7 and MgSO4 administrated group ROS were produced and these reactive molecules are able to remove hydrogen from the lipid membrane and imitating a series of reaction leading to the membrane destruction.
Salts of heavy metals when ingested in high dose produced oxygen reactive species which may lead to adverse clinical outcomes such as DNA damage, destruction of testicular cells and germ line of testis at initial levels. Findings of the present study represent similar results by investigating the destruction of germ cells, spermatocytes, spermatogonia, sertoli cells, sperms and ultimate’s whole follicles after administration of the both the salts for two months. However, heavy destruction was observed in the MgSO4 administrated group (being 35% and 66% at 30th and 60th day respectively) as compared to K2Cr2O7 administrated group where 30% destruction of follicles were observed at 30th day and 56% destruction of follicle at 60th day of experiment as previously explained by Acharya et al.9
The destructive effects of both the salts on testicles of male mice could be due to generation of ROS in exposure to the K2Cr2O7 and MgSO4. Oxygen reactive species generated are active in causing DNA damage. The DNA damage in soft tissue of follicles leads to excessive destruction of follicles, germ line leading to severe reproductive abnormality resulting in production of sterile individuals.18 The significant changes in lipid peroxidation and testicular histology suggested that K2Cr2O7 and MgSO4 exposure increase the level of lipid peroxidation leading towards generation of ROS and severe destructive consequences of testicular cells in male mice.
CONCLUSION
We conclude that K2Cr2O7 and MgSO4 have a deleterious effect on the histology of the testis of male mice. The effects of MgSO4 found to be more lethal and destructive as compared to K2Cr2O7. Thus, we suggest further detailed studies in human to corroborate these findings and assuming that K2Cr2O7 and MgSO4 at normal dose could be a potential male anti fertility agent. The self medication involving K2Cr2O7 and MgSO4 should also be discouraged.
Authors Contributions:
AM, KZ and MR: Designed the study. UH, AM, and MA: Collected the data and performed the experiments. MHQ, MIN, MR and AM: Analyzed the data critically, performed statistical analysis and wrote the manuscript.
ACKNOWLEDGEMENT
This study was funded by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah. The authors, acknowledge with thanks DSR technical and financial support. We are also thankful to the Institute of Molecular Biology & Biotechnology (IMBB), the University of Lahore, for providing research facilities. We are grateful to Rabail Alam, Mahwish Arooj and Amir Saeed for help in data collection and statistical analysis.
Conflict of interest: The authors have no competing financial interests.
References
- 1.Celis DR, Pedron N, Feria VA. Toxicology of male reproduction in animals and Humans. Arch Androl. 1996;37:201–218. doi: 10.3109/01485019608988523. DOI:10.3109/01485019608988523. [DOI] [PubMed] [Google Scholar]
- 2.Barceloux DG, Krenzelok EP, Olson K, Watson W. American Academy of Clinical Toxicology Practice Guidelines on the Treatment of Ethylene Glycol Poisoning Ad Hoc Committee. J Toxicol Clin Toxicol. 1999;37(5):537–560. doi: 10.1081/clt-100102445. DOI:10.1081/CLT-100102445. [DOI] [PubMed] [Google Scholar]
- 3.Bagchi D, Stohs SJ, Downs BW, Bagchi M, Preuss HG. Cytotoxicity and oxidative mechanisms of different forms of chromium. Toxicology. 2002;180:5–22. doi: 10.1016/s0300-483x(02)00378-5. DOI: 10.1016/S0300-483X(02)00378-5. [DOI] [PubMed] [Google Scholar]
- 4.Mittendorf R, Pryde P, Khoshnood B, Lee KS. If tocolytic magnesium sulfate is associated with excess total pediatric mortality, what is its impact? Obstet Gynecol. 1998;92:308–311. doi: 10.1016/s0029-7844(98)00163-x. DOI: 10.1016/S0029-7844(98)00163-X. [DOI] [PubMed] [Google Scholar]
- 5.Blitz M, Blitz S, Hughes R, Beasley R, Knopp J, Rowe BH. Aerosolized magnesium sulfate for acute asthma A systematic review. Chest. 2005;128:37–44. doi: 10.1378/chest.128.1.337. DOI:10.1378/chest.128.1.337. [DOI] [PubMed] [Google Scholar]
- 6.Agnieszka S, Halina Z, Irmina N. Lipid peroxidation in the kidney of rats treated with V and/or Mg in drinking water. Toxicology. 2010;30:487–496. doi: 10.1002/jat.1520. DOI: 10.1002/jat.1520. [DOI] [PubMed] [Google Scholar]
- 7.Chelikani P, Ramana T, Radhakrishnan TM. Catalase: a repertoire of unusual features. Indian J Clin Biochem. 2005;20:131–135. doi: 10.1007/BF02867412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chowdhury RA. Recent advanced in heavy metals induced effect on male reproductive Function. J Med Sci. 2009;2:37–42. [Google Scholar]
- 9.Acharya UR, Mishra M, Tripathy R, Mishra I. Testicular dysfunction and anti oxidative defense system of Swiss mice after chromic acid exposure. Reproductive Toxicolog. 2006;22:87–91. doi: 10.1016/j.reprotox.2005.11.004. DOI: 10.1016/j.reprotox.2005.11.004. [DOI] [PubMed] [Google Scholar]
- 10.Zuhair RZ, Zuhair SA, Abdul Hussain V. Comparative effects of trivalent and hexavalent chromium on spermatogenesis of the mouse. Toxicological & Environmental Chemistry. 1990;25:131–136. [Google Scholar]
- 11.Gyan CJ, Hemant P, Sameer S, Mamta B, Balvant SK. Reproductive toxicity of vanadyl sulphate in male rats. J Health Sci. 2007;53:137–141. [Google Scholar]
- 12.Bulikowski W, Wozniak F, Borzecki Z, Radomska K, Kaliszuk K, Swies Z. Effect of potassium dichromate on histopathologic changes in testicles of white rats and results of atomic pilograms on fur. Annales Universitatis Mariae Curie-Sklodowska Med. 1991;46:69–73. [PubMed] [Google Scholar]
- 13.Ohkawa H, Ohishi N, Yagi K. Assay for lipid per oxides in animal tissues by thiobarbituric acid reaction. J Anal Biochem. 1979;95:351–358. doi: 10.1016/0003-2697(79)90738-3. [DOI] [PubMed] [Google Scholar]
- 14.Kakkar P, Das B, Viswanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biol. 1984;21:130–132. [PubMed] [Google Scholar]
- 15.Aebi H. Catalase. Method in enzymatic analysis. Vol. 3. New York: Academic Press; 1974. pp. 276–286. [Google Scholar]
- 16.Bancroft JD. Histopathological stains and their diagnostic uses. New York: Churchill Livingstone; 1975. [Google Scholar]
- 17.Goulart M, Batoreu MC, Rodrigues AS, Laires A, Rueff J. Lipoperoxidation products and thiol antioxidants in chromium exposed workers. Mutagenesis. 2005;20:311–315. doi: 10.1093/mutage/gei043. DOI: 10.1093/mutage/gei043. [DOI] [PubMed] [Google Scholar]
- 18.Ernst E, Bonde JP. Sex hormones and epididymal sperm parameters in rats following sub-chronic treatment with hexavalent chromium. Human and Experimental Toxicology. 1992;11:255–258. doi: 10.1177/096032719201100403. [DOI] [PubMed] [Google Scholar]