Abstract
Taxanes are a class of anticancer agents with a broad spectrum and have been widely used to treat a variety of cancer. However, its long term use has been hampered by accumulating toxicity and development of drug resistance. The most extensively reported mechanism of resistance is the overexpression of P-glycoprotein (Pgp).
We have developed a PEGylated carboxymethylcellulose conjugate of docetaxel (Cellax), which condenses into ~120 nm nanoparticles. Here we demonstrated that Cellax therapy did not upregulate Pgp expression in MDA-MB-231 and EMT-6 breast tumor cells whereas a significant increase in Pgp expression was measured with native docetaxel (DTX) treatment. Treatment with DTX led to 4 to 7-fold higher Pgp mRNA expression and 2-fold higher Pgp protein expression compared to Cellax treatment in the in vitro and in vivo system respectively. Cellax also exhibited significantly increased efficacy compared to DTX in a taxane-resistant breast tumor model. Against the highly Pgp expressing EMT6/AR1 cells, Cellax exhibited a 6.5 times lower IC50 compared to native DTX, and in the in vivo model, Cellax exhibited 90% tumor growth inhibition, while native DTX had no significant antitumor activity.
Keywords: Carboxymethylcellulose, Drug Delivery, Docetaxel, Nanoparticles, P-glycoprotein, Multidrug resistance
1.Introduction
Taxanes are widely used anticancer drugs for the treatment of major cancers, including ovarian, breast, head and neck, lung, and prostate cancer.However, development of multidrug resistance (MDR) during taxane chemotherapy has been a major clinical challenge 1.A wide variety of mechanisms have been identified contributing to taxane resistance, including alterations of the expression pattern of tubulins and microtubulin associated proteins (overexpression β-III tubulin and tau), changes in apoptotic regulatory proteins (upregulation of Bcl-xL, clusterin an survivin), enhancement of cell proliferation (overexpression of Akt, PI3K) and overexpression of multidrug resistance protein (P-glycoprotein) 1. The most extensively studied mechanism of resistance to taxanes is the overexpression of P-glycoprotein (Pgp) and other multidrug transporters: these are membrane proteins belonging to the ATP-binging cassette (ABC) family of transporters 2, 3, and act as efflux pumps removing a large number of structurally diverse, mainly hydrophobic compounds from cells, thus keeping intracellular drug concentration below a cell-killing threshold and inducing cross-resistance to several chemically unrelated compounds. In many patients, Pgp over expression appears to be one of the major mechanisms for chemotherapy failure 4, 5. Several strategies have been investigated for the development of new chemotherapeutics or drug delivery systems to minimize the generation of resistance and for the therapy of the MDR tumors. Among them, nanoparticles appear to be an attractive approach to overcome MDR. Studies have shown that many nanoparticles, including carbon nanotubes 6 and nanocrystals 7, enhance cellular accumulation of anticancer drugs by bypassing Pgp via endocytosis. Pluronic block copolymers were found to hypersensitize MDR cancer cells by altering membrane microviscosity and depleting ATP to inhibit Pgp activity 8. It was also shown that excipients such as tocopheryl polyethylene glycol succinate (TPGS) and Brij78 which were used to stabilize the nanoparticles can inhibit Pgp function, improving cellular accumulation of the anticancer drugs 9, 10.
We have recently developed a polymeric conjugate of docetaxel (Cellax) composed of a PEGylated and acetylated carboxymethylcellulose backbone with docetaxel (DTX) attached via ester linkages 11. Cellax exhibits a drug loading of ~37 wt%, condenses into monodispersed ~120 nm particles in saline and releases drug at a controlled rate (5%/day) in serum 11. Compared to native DTX (Taxotare) and nab-paclitaxel (an albumin nano-formulation of paclitaxel, Abraxane), Cellax exhibits a 5 and 20 times longer circulating half life and 39 and 37 times higher plasma area under the curve, respectively 12, 13. Cellax also displays selective tumor accumulation with 5- and 10-fold increased uptake compared to native DTX and nab-paclitaxel, respectively 12, 13. These data suggest that Cellax can provide effective taxane chemotherapy. Here, we further analyzed the effect of Cellax treatment on the development of MDR during the therapy compared to native DTX. We also studied the antitumor efficacy of Cellax against MDR tumors compared to native DTX.
2. Materials and Methods
2.1. Reagents and Reference Drugs
Docetaxel (DTX) was obtained from LC Laboratories (Woburn, MA). Carboxymethylcellulose (CMC) sodium salt (CEKOL 30000-P) was purchased from CPKelco (Atlanta, GA). Poly(ethylene glycol) methyl ether (mPEG − OH, MW = 2000), N-hydroxysuccinimide (NHS), 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide HCl (EDC.HCl), and 4-dimethylaminopyridine (DMAP) were purchased from Sigma Aldrich (Oakville, ON). Hydrophobic fluorescent dye DiI (1,10-dioctadecyl-3,3,3030-tetramethylindocarbocyanine perchlorate, D-307) was purchased from Invitrogen (Burlington, ON).Taxotere (native DTX) were purchased from the University Health Network pharmacy (Toronto, ON). For all the in vitro studies, free DTX was first dissolved in DMSO and then diluted with DMEM medium.
2.2. Synthesis of Cellax polymer
Cellax polymer was synthesized in a two-step reaction protocol as described previously. Briefly, sodium CMC was first de-salted using 20% sulfuric acid solution and the free acid was then acetylated with acetic anhydride (CMC-Ac). The purified CMC-Ac polymer was subsequently conjugated to PEG and DTX via EDC/NHS coupling chemistry.The resulting polymer was purified by precipitation in ether and washing with water.Purity was measured by gel permeation chromatography analysis: polymers were dissolved in dimethyl sulfoxide (DMSO), and analyzed on a 250 mm × 10 mm Jordi Gel DVB Sulf MB column (Jordi, Bellingham MA) running a DMSO mobile phase at 1 mL/min on a Waters e2696 chromatography system equipped with a 2414 refractive index detector. The chemical composition of Cellax was determined by 1H-NMR (nuclear magnetic resonance), as reported previously 11, yielding a polymer conjugate containing 37.1 ± 1.5 wt% DTX and 4.7 ± 0.8 wt% PEG. Cellax nanoparticles were prepared as described previously 11. Particles were dialyzed in a Slide-A-Lyzer 10000 MWCO cartridge against 0.9% saline for 3 h to extract solvent. The particles were filtered through a 0.22 mm Millipore PVDF filter, and were concentrated using a Vivaspin unit (10 000MWCO). Particle size and zeta potential were measured with a Zetasizer (Nano-ZS, Malvern Instruments, Malvern, UK). DTX content of the Cellax nanoparticles was determined by 1H-NMR using a water pre-saturation method after dissolving 100 µL of the nanoparticle suspension into 900 µL of DMSO (D6). DiI (1,10-dioctadecyl -3,3,3030-tetramethylindocarbocyanine perchlorate) loaded nanoparticles were prepared by dissolving 10 mg of Cellax polymer in MeCN (1 mL) containing 0.1 mg/mL DiI and was precipitated into 0.9% saline with aggressive vortexing to form nanoparticles. The processing of Cellax-DiI was identical to the preparation of Cellax particles as described. DiI content of the Cellax-DiI nanoparticles was determined by dissolving Cellax-DiI in DMSO and assaying for fluorescence (Excitation filter: 535 nm; Emission Filter 590 nm) and comparing to a calibration curve of fluorescence versus DiI concentration, subtracting the background signal of un-loaded Cellax particle fluorescence.
2.3. Cell culture and animals
Human MDA-MB-231 and mouse EMT6 breast cancer cell lines were obtained from the American Type Culture Collection (ATCC). DTX resistant EMT6/AR1 cells were a gift from Ian Tannock, Princess Margaret Hospital, Toronto. EMT6/AR1 cells have previously been demonstrated to overexpress Pgp 14. DTX resistant MDA-MB-231 cells were generated from the sensitive phenotype by treating them with gradually increasing concentrations of DTX until the cells become fully resistant to 10 nM of DTX. The cells were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum. Female NOD-SCID and Balb/C mice were purchased from Jackson Laboratories (Bar Harbour, ME). All protocols were approved by the Animal Care Committee of the University Health Network.
2.4. Quantitative real-time RT-PCR
The expression levels of different genes like P-glycoprotein (Pgp) [human ATP-binding cassette, sub-family B (MDR/TAP), member 1 (ABCB1), NM_000927], β3 tubulin [human tubulin, beta 3 class III (TUBB3), transcript variant 1, NM_006086], tau [human microtubule-associated protein tau (MAPT), transcript variant 2, NM_005910], Bcl-2 [human Bcl2 binding component 3 (BBC3), transcript variant 1, NM_001127240], clusterin [human clusterin (CLU), transcript variant 1, NM_001831], survivin [human baculoviral IAP repeat containing 5 (BIRC5), transcript variant 2, NM_001012270], vegfa [human vascular endothelial growth factor A (VEGFA), transcript variant 1, NM_001025366], NFκB [human nuclear factor of kappa light polypeptide gene enhancer in B-cells 1 (NFKB1), transcript variant 1, NM_003998], PI3K [human phosphoinositide-3-kinase, regulatory subunit 1 (alpha) (PIK3R1), transcript variant 4, NM_001242466], Akt [human v-akt murine thymoma viral oncogene homolog 1 (AKT1), transcript variant 1, NM_005163] and Bcl-xL [human BCL2-like 11 (apoptosis facilitator) (BCL2L11), transcript variant 18, NM_001204113] were confirmed by quantitative real-time RT-PCR. For RNA extraction, the cells were washed three times with PBS and harvested. Total RNA was prepared with RNeasy Plus Mini Kit (Qiagen), and complementary DNA (cDNA) was reverse-transcribed with a QuantiTect reverse transcription kit (Qiagen). cDNA was amplified with specific primers with a SYBR Green Core Reagent Kit (Qiagen) and a real-time PCR instrument (Eppendorf). The sequences of the PCR primer used are listed in Table 1. Expression of each gene was normalized with endogenous actin as a control, and its relative levels were quantified by calculating 2−ΔΔCT, where ΔΔCT is the difference in CT (cycle number at which the amount of amplified target reaches a fixed threshold) between target and reference. A relative change in expression level of 2 or more is considered significant.
Table 1.
Sequences of primers used for the real time PCR analysis.
| Gene | Forward primer | Reverse primer |
|---|---|---|
| Pgp | 5’-GTGGGGCAAGTCAGTTCATT-3’ | 5’-TCTTCACCTCCAGGCTCAGT-3’ |
| β tubulin | 5’-ACCTCAACCACCTGGTATCG-3’ | 5’-TTCTTGGCATCGAACATCTG-3’ |
| tau | 5’-GCTGAGTCCCAGCAATTCTC-3’ | 5’-GAAGAGCAGGGCACAAGAAC-3’ |
| Bol-2 | 5’-GCCCAGACTGTGAATCCTGT-3’ | 5’-CTCCTCCCTCTTCCGAGATT-3’ |
| clusterin | 5’-ACATTTGGTGCCCAGAAGTC-3’ | 5’-CTGTGGTCCAGGGAAAGGTA-3’ |
| survivin | 5’-ACCTGAAAGCTTCCTCGACA-3’ | 5’-TAACCTGCCATTGGAACCTC-3’ |
| vegfa | 5’-GGGCAGAATCATCACGAAGT-3’ | 5’-ATCTGCATGGTGGTGATGTTGGA-3’ |
| NFκB | 5’-ACTGTGAGGATGGGATCTGC-3’ | 5’-CCTTCTGCTTGCAAATAGGC-3’ |
| pl3k | 5’-ACCCAGCAACAGAAAAATGG-3’ | 5’-GCGCTGTGAATTTAGCCTTC-3’ |
| Akt | 5’-CATCACACCACCTGACCAAG-3’ | 5’-CTCAAATGCACCCGAGAAAT-3’ |
| Bcl-xL | 5’-TCTGGTCCCTTGCAGCTAGT-3’ | 5’-CAGGGAGGCTAAGGGGTAAG-3’ |
| actin | 5’-AGATGTGGATCAGCAAGCAG-3’ | 5’-GCGCAAGTTAGGTTTTGTCA-3’ |
2.5. Effect of taxane treatment on Pgp up-regulation
To compare the effect of DTX and Cellax treatment on Pgp levels in chemo-sensitive MDA-MB-231 and EMT6 cells, the cells (1 × 106) were seeded in a 6 well plate. After 24 h, the cells were treated with DTX or Cellax (equivalent in DTX concentration, 15 nM) for 48 h. The cells were then harvested and Pgp mRNA level was analyzed by real time PCR using the above mentioned method.
To understand the kinetics of Pgp level during DTX and Cellax treatment, both MDA-MB-231 and EMT6 cells were treated with 5, 10, 15 nM DTX or Cellax for 24, 36 and 48 h. To simulate the slow releasing property of DTX from Cellax, one set of cells were treated with fractionated doses of DTX where ¼ of the dose of DTX was added in the culture every 6, 9 and 12 hours respectively for total incubation of 24, 36 and 48 h. Mixture of DTX and the carrier polymer [CMC(Ac)-PEG] was employed as a control to understand the role of the polymer.
2.6. Effect of taxane treatment on Pgp expression in MDA-MB-231 tumor in mammary fat pad
MDA-MB-231 cells were inoculated into the breast fad pad (2 × 106 cells/injection, n=10 mice per group) of female NOD-SCID mice. After 4 weeks when tumors attained a diameter of 7–8 mm, mice were treated by i.v. injection at the MTDs of Cellax (170 mg DTX/kg) and native DTX (10 mg DTX/kg). They were treated a second time one week later. Both treatment completely inhibited the tumor growth by 2 weeks, and then the tumors started to rebound. When the tumors reach 10 mm in diameter, the tumors were resected, and stained for Pgp by immunohistochemistry, scanned on a ScanScope XT microscope and the Pgp level was quantified by using Tissue Studio (Definiens Health Image Intelligence). Analyses were conducted on 5 tumors per group.
2.7. In vitro analysis of viability and cellular uptake study
Cell growth inhibition activity of Cellax was analyzed against EMT6 and EMT6/AR1 cells by measuring cell viability with the XTT assay. One thousand cells per well was plated in a 96 well plate. After 24 h of incubation, the parent (EMT6) and the resistant (EMT6/AR1) cells were treated with different concentrations of DTX and Cellax. In the fractionated DTX treatment, the DTX solutions added to the cultures were fractionated over two days, with addition of 1/4 the dose every 12 h for 2 days. After 72 h of treatment, viability was assayed by the XTT assay. Briefly, a 1 mg/mL solution of XTT reagent and 1.53 mg/mL solution of phenazine methylsulfate in water were prepared, and 5 µL of phenazine methylsulfate was added to each mL of the XTT solution. Twenty-five µL of the mixture solution was added to each well, the culture plates were incubated for 2 h at 37 °C, and absorbance at 480 nm was then measured. Wells treated with media (or 0.1% DMSO media) represent 100% viable cultures, and wells containing no cells represent background signal. For the nanoparticle uptake study, 2 × 105 EMT6 or EMT6/AR1 cells were plated on sterile glass coverslips placed in a 12 well plate. Cells were treated with DiI labelled Cellax particles at the concentration of 0.5 µg DiI/mL. After 4 h of incubation, the cells were washed with PBS and fixed with 10% formalin for 15 min. The fixed cells were washed again with PBS and glass coverslips were mounted on a glass slide with mounting media with DAPI. Slides were then scanned on the FluoView A confocal microscope (Olympus) at 40× objective + 5× zoom. The intensity of the DiI and DAPI signal was measured using ImageScope software with positive pixel count algorithm.
2.8. In vivo antitumor efficacy
Anticancer efficacy of Cellax was analyzed against a highly Pgp expressing tumor, EMT6/AR1. Female Balb/C mice were s.c. inoculated with EMT6/AR1 cells (2 × 105 cells/injection, n=5 mice per group). When tumors reached 4–5 mm in diameter, they were treated i.v. with MTD dose of DTX (10 mg/kg), matched dose of Cellax (10 mg DTX/kg) and increased dose of Cellax (30 mg DTX/kg) for 5 consecutive days. Vehicle control (Tween80/ethanol/saline) and polymer control (CMC(Ac)-PEG) groups were also included. Tumor volume and body weight of the treated animals were monitored.
2.9. Statistical analysis
All data are expressed as mean ± SD. Statistical analysis was conducted with the 2-tailed unpaired t test for 2-group comparison or one-way ANOVA, followed by Tukey multiple comparison test by using GraphPad Prism (for 3 or more groups). A difference with P < 0.05 was considered to be statistically significant.
3. Results and Discussion
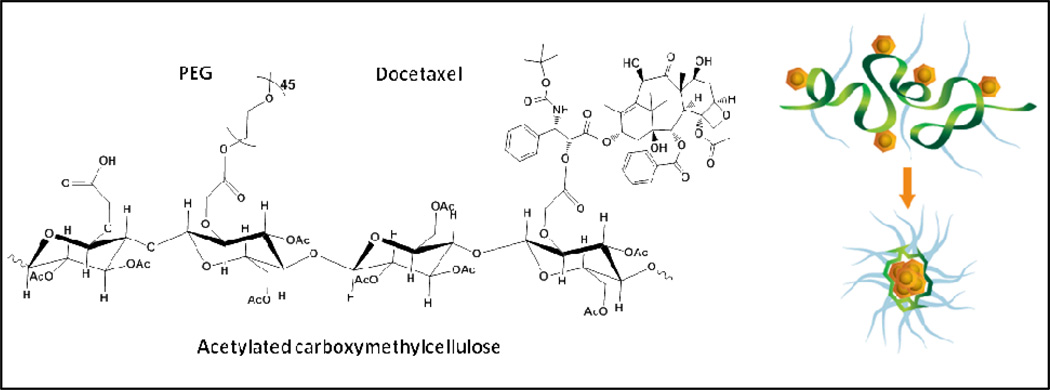
Taxanes are the most widely used class of chemotherapeutic drugs for cancer therapy, but its long term use in human patients has been limited by the accumulating toxicity and development of drug resistance 15. Patients with refractory tumors during or after taxane therapy are often out of effective treatment options, because these tumors usually develop a MDR phenotype that detoxicates a broad range of cytotoxic agents 1. The MDR mechanism induced by taxane chemotherapy can occur at different subcellular levels, including over-expression of a drug efflux transporter such as Pgp to effectively remove drugs from the cells 16, up-regulation of β-III tubulin and tau to attenuate taxane’s anti-tubulin activity 17, 18, and activation of pathways of proliferation (Akt, PI3K) and anti-apoptosis (clusterin, Bcl2, survivin, Bcl-xL) to reduce cell death 19. Our group has developed a nanoparticle formulation for DTX, aiming to address its clinical limitations, including non-specific drug delivery, significant side effects and induction of MDR in tumors 11. This DTX containing nanoparticle (Cellax) is prepared by covalently conjugating DTX onto a PEGylated and acetylated carboxymethyl cellulose, which self-assembles into 120-nm particles in normal saline (Fig 1) 11. We have previously shown that Cellax slowly released DTX at 5%/day in serum, and exhibited 40-fold prolonged blood circulation and 5-fold increased tumor delivery with reduced uptake in most normal tissues compared to native DTX 13. Due to this modification in pharmacokinetics, Cellax exhibited reduced side effects and enhanced therapeutic activity in multiple tumor models in mice 13. The current study focuses on whether Cellax can also address the second limitation of taxane therapy, which is development of MDR during the treatment. We are also interested in whether Cellax displays enhanced activity against MDR tumors because of improved drug delivery.
Figure 1.
Schematic of Cellax chemical composition and self assembly. The structure is a representation of the DTX, PEG, and free carboxylic acid elements which is randomly distributed throughout the polymer chain. In aqueous medium, the Cellax polymer condenses into nanoparticles with a mean diameter around 120 nm.
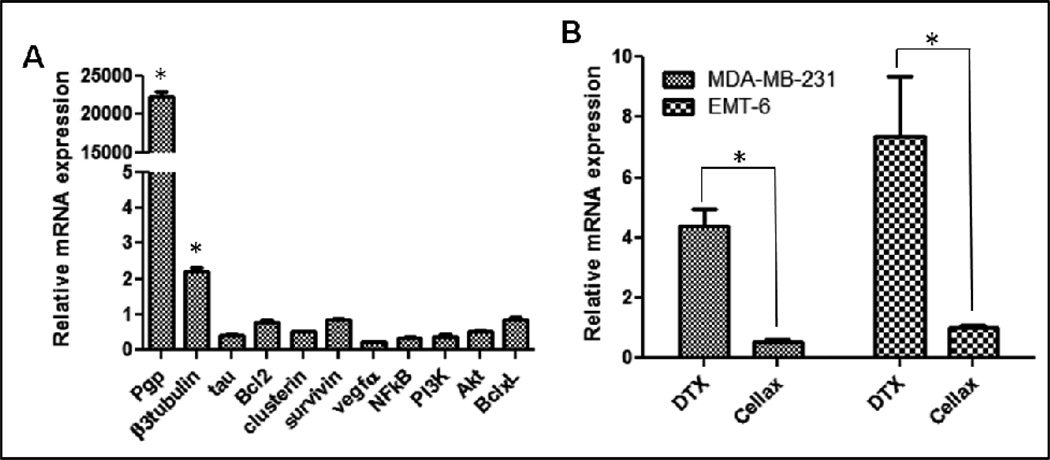
As discussed earlier, there are a variety of possible mechanisms for tumor cells to develop MDR to taxane, including Pgp, β-III tubulin, tau, Bcl-2, clusterin, survivin, VEGF-A, NFκB, PI3K, Akt and Bcl-xL 19. To identify a robust marker for DTX resistance in our model system, we have created a DTX-resistant MDA-MB-231 human breast cancer cell line by incubating the cells with DTX at gradually increasing concentrations until the cells developed complete resistance against 10 nM DTX. IC50 of the resistant line was increased by 25-fold, from 1.2 nM to 30 nM, compared to the parent line, and the resistant cells remained ~100% viable at 10 nM DTX (supplementary Fig 1). A panel of resistance markers in the resistant line compared to the parent line were then analyzed by qPCR, and Pgp was measured to be up-regulated by >20,000-fold in the DTX-resistant cells (Fig 2A). There was a slight elevation in β-III tubulin (~2-fold) in the resistant MDA-MB-231 cells, but not other tested markers (Fig 2A). The results were similar in PC3 human prostate cancer cells (Murakami et al. unpublished), and the data suggest that Pgp was the major contributor to DTX resistance in our model system and could be a robust biomarker for quantifying MDR phenotype during different taxane chemotherapies. Our data also correspond to clinical reports: it has been shown that tumor samples isolated from taxane non-responding patients expressed 28-fold higher level of Pgp compared to responding patients 5. We then compared native DTX and Cellax in up-regulating Pgp in MDA-MB-231 and EMT6 (murine breast tumor) cells, and as shown in Fig 2B, native DTX increased the mRNA level of Pgp by 4.5-and 7.5-fold in MDA-MB-231 and EMT6 cells in 48 h respectively, while Cellax did not up-regulate Pgp in either cell line (Fig 2B). The up-regulation of other resistance markers in these two cell lines was not significant by both treatments (data not shown). The results indicate that Cellax did not induce MDR phenotype in these two cell lines, while native DTX promoted tumor MDR mainly through the Pgp mechanism, which occurred within 48 h after treatment.
Figure 2. qPCR analysis of taxane resistance markers in cells after different treatments.
A. Relative mRNA level of different markers in DTX-resistant MDA-MB-231 cells compared to its parent line. B. Relative mRNA level of Pgp in two breast cancer cell lines, MDA-MB-231 (human) and EMT6 (mouse) after treatment with native DTX or Cellax for 48 h at equivalent DTX concentration 15 nM. Up-regulation by >2-fold was considered significant (*).
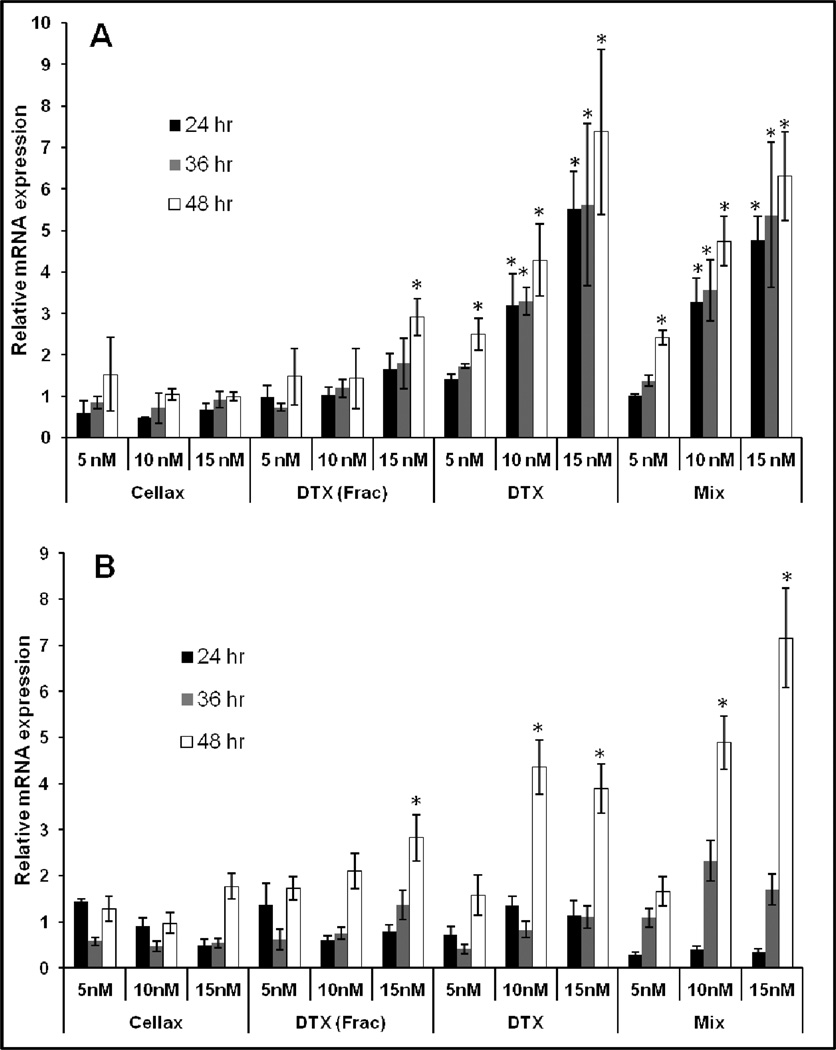
There could be two possible mechanisms by which Cellax did not up-regulate Pgp: first, DTX is slowly released from Cellax and the constantly low concentration of available DTX does not efficiently induce Pgp expression; second, the polymer carrier of Cellax decreases Pgp expression. To test the hypothesis, we analyzed Pgp mRNA level in the cells after treatment with bolus DTX, Cellax, fractionated DTX (DTX was divided in 4 doses and was given 6–12 h apart), and DTX mixed with the carrier polymer [CMC(Ac)-PEG]. As shown in Fig 3, over-expression of Pgp induced by native DTX was time and concentration dependent: DTX did not increase Pgp level significantly at low concentration (5 nM) and early timepoints (<36 h) in both MDA-MB-231 and EMT6 cells. However, as incubation time and DTX concentration increased, the Pgp levels increased correspondingly. At 15 nM DTX incubation for 48 h, the Pgp levels in MDA-MB-231 and EMT6 cells were enhanced by 4- and 7.5-fold, respectively. Cellax, on the other hand, did not up-regulate the Pgp levels in both cell lines at all the tested concentrations (5–15 nM) and incubation time (24–48 h). Fractionated DTX exhibited a comparable effect as Cellax; however, at the high concentration with a long incubation time (15 nM total for 48 h) a slight elevation in Pgp (~3-fold) induced by fractionated DTX was measured. A physical mixture of DTX with the polymer carrier showed similar results as the bolus DTX, and at 15 nM for 48 h, the Pgp levels in the MDA-MB-231 and EMT6 were increased by 7-fold. The results suggest that the carrier polymer of Cellax had little effect on Pgp expression, and Cellax circumvented Pgp up-regulation through a slow release mechanism. It has been demonstrated that slow and continuous infusion of DTX leads to significantly less Pgp induction in cancer cells compared to single bolus dose. For example, Ho et al. 20 has also shown that intraperitonial implantation of a slow releasing film of paclitaxel did not induce expression of Pgp in the human ovarian cancer cells residing in the peritoneal cavity. Our results mirrored the prior reports.
Figure 3.
Kinetics of Pgp mRNA level in the cells (A, EMT6; B, MDA-MB-231) treated with different concentrations of Cellax, fractionated DTX, bolus DTX and mixture of DTX with the carrier polymer. DTX (frac): fractionated dose of DTX, where ¼ of the dose of DTX was added in the culture every 6, 9 and 12 hours respectively for total incubation of 24, 36 and 48 h. Mix: DTX physically mixed with the carrier polymer [CMC(Ac)-PEG]. Up-regulation by >2-fold was considered significant (*).
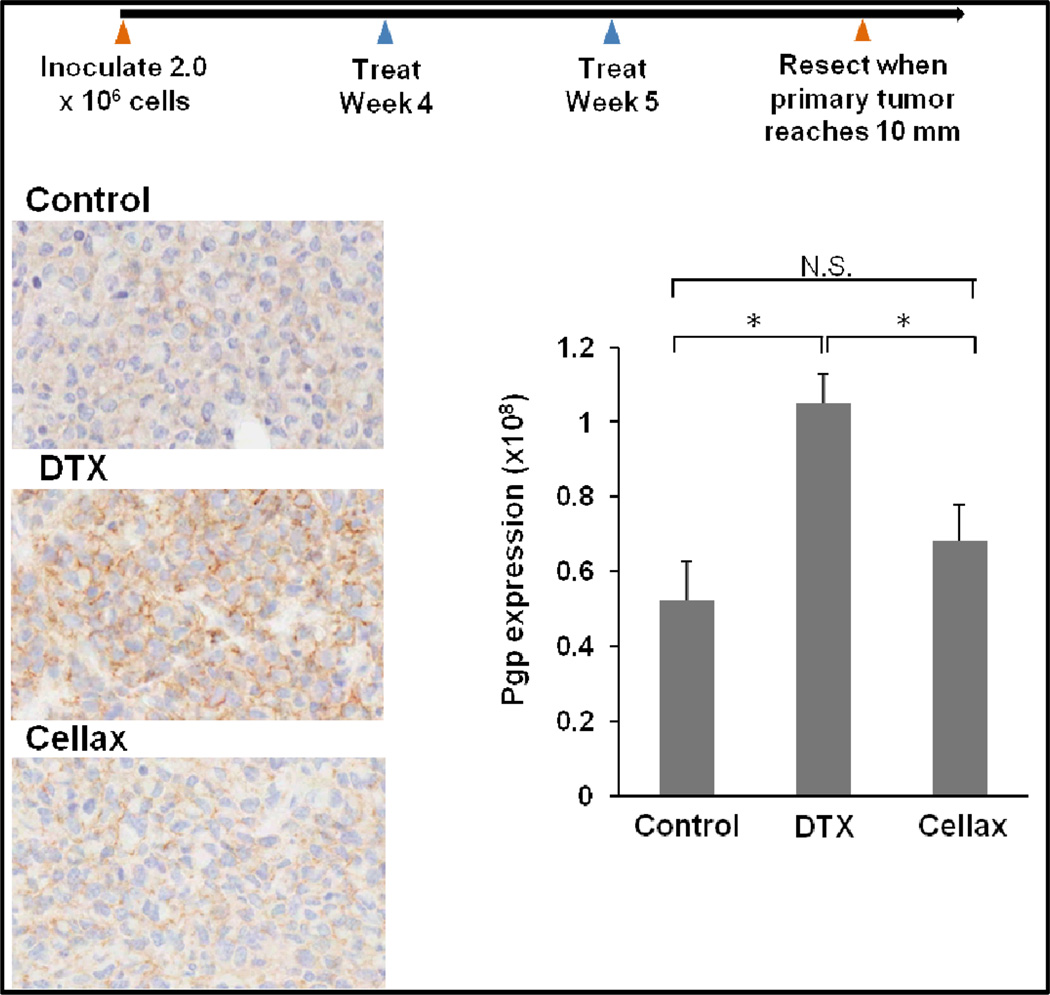
We next compared the effect of native DTX and Cellax on up-regulating Pgp in an in vivo tumor system. MDA-MB-231 cells were inoculated to the mammary fat pad of female NOD-SCID mice, and the mice were treated at their MTDs with two weekly doses of native DTX (10 mg DTX/kg) and Cellax (170 mg DTX/kg) when tumors grew to the size of 50–100 mm3. Both therapies were effective in inhibiting the tumor growth for 3 weeks, and no further treatment was given, allowing the tumors to rebound possibly due to the presence of residual tumor cells that survived the treatments. The tumors were surgically removed when the size reached ~500 mm3 in ~60 days post the final dose. Pgp was detected by immunohistochemistry and the images were quantified by Tissue Studio software. The saline treated tumor expressed a basal level of Pgp, and the DTX treatment increased the Pgp level by 2-fold, while Cellax therapy did not up-regulate Pgp in the tumor (Fig 4). The data confirm that Pgp up-regulation induced by native DTX was also measured in an in vivo system and in the protein level, while Cellax displayed little effect on up-regulating Pgp. The levels of other resistance markers were not significantly changed in both DTX and Cellax treated tumors compared to the naïve tumor (data not shown).
Figure 4.
In vivo expression of Pgp protein in an orthotopic breast tumor model of MDA-MB-231. Data = mean ± SD (n=5), * indicates p<0.05.
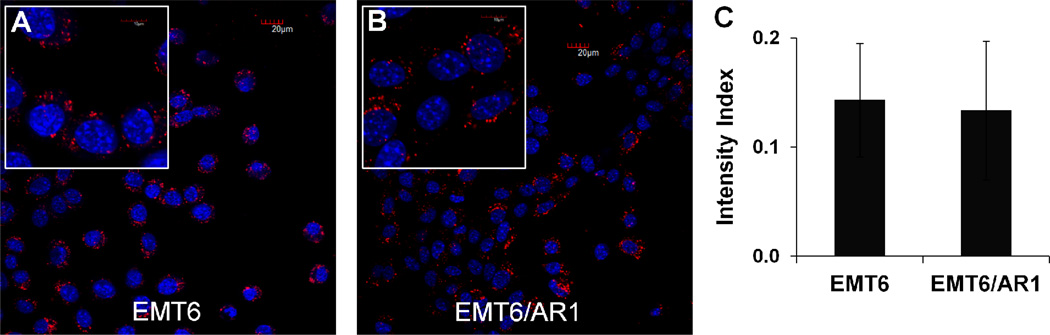
Whether Cellax could improve treatment of MDR tumors was also investigated, and there are a number of possible mechanisms that Cellax could be more efficacious against MDR tumors compared to native DTX. First, Cellax is a slow releasing formulation of DTX, and sustained drug delivery of taxane has been demonstrated to improve treatment of MDR tumors 20, 21. De Souza et al. 21 showed that continuous dosing with DTX resulted in superior antitumor efficacy and prevented drug resistance induction in chemosensitive and chemoresistant tumors compared to bolus DTX. Second, Cellax could be efficiently internalized by MDR tumor cells to bypass the Pgp efflux that is responsible for removing free DTX from the cells. Third, Cellax delivers >5-fold increased dose to tumors compared to native DTX at the equal dose, and Cellax can be administered at >4-fold increased dose relative to native DTX 13. Both factors could contribute to significantly increased concentrations of drug in the tumor by Cellax delivery compared to native DTX. We compared the in vitro cytotoxicity of bolus DTX, fractionated DTX and Cellax against EMT6 and EMT6/AR1 cells. EMT6/AR1 cells have been shown to expresses a high level of Pgp contributing to its significant drug resistance. Wong et al. 22 has shown that EMT6/AR1 cells express 2-fold higher level of Pgp compared to MDA435/LCC6/MDR1 cells whereas Pgp expression in both parent lines was undetectable. Over-expression of Pgp by EMT6/AR1 cells was also demonstrated by Patel and Tannock 14. As shown in Table 2, the IC50 for native DTX, fractionated DTX and Cellax against EMT6/AR1 cells were 407.9 ± 1.8, 160.8 ± 1.9, and 63.4 ± 2.3 nM, respectively.. This result showed that Cellax and fractionated DTX were 7 and 2.5-fold more potent than bolus DTX against the Pgp over-expressing resistant line. Cellax was found to be more potent than native DTX against the parent cell line as well: IC50 of Cellax was 12.7 ± 3.3 nM compared to 52.5 ± 2.6 nM for DTX (Table 2). These data are consistent with our previous report showing Cellax and fractionated DTX displayed comparably enhanced cytotoxicity against tumor cells compared with bolus DTX 11, possibly via continuous feeding of a cell cycle dependent cyctotoxic agent. Cellax exhibited the lowest resistance index (IC50 in resistant cells divided by IC50 in parent cells), indicating this agent was impacted to the least extent by Pgp over-expression among all. This phenomenon could be explained by efficient internalization of Cellax nanoparticles by both EMT6 and EMT6/AR1 cells (Fig 5A, B, C), bypassing the Pgp efflux. The confocal images were suggestive of sequestration of Cellax within intracellular compartment, such as endosomes or lysosomes. The slow release of the drug from these compartments often explains the reduced efflux through Pgp. To understand the actual localization of the nanoparticles inside the cells, we performed co-localization studies. Cellax-DiI nanoparticles were found to be located inside lysosomes after 4 h of incubation (Supplementary Figure 2). As lysosomes are acidic (pH 5–6) and enriched with hydrolytic enzymes such as carboxylesterase, the degradation of Cellax polymer and drug release are anticipated to occur in this environment.
Table 2.
IC50 for DTX and Cellax against EMT-6 and EMT-6 ADR cells.
| IC50 | DTX | Cellax | DTX frac |
|---|---|---|---|
| EMT6 | 52.5 ± 2.6 | 12.7 ± 3.3 | 7.8 ± 2.2 |
| EMT6/AR1 | 407.9 + 1.8 | 63.4 + 2.3 | 160.8 + 1.9 |
| Resistance index | 7.8 | 5 | 20.5 |
Figure 5.
In vitro cellular internalization of Cellax-DiI nanoparticles (A, B) in EMT6 (A) and EMT6/AR1 cells (B).blue = nuclei (DAPI) and red = Cellx nanoparticles (DiI), scale bar = 20 µm. Inset: 40 × 5 magnification. C. The relative intensity of the DiI signal in EMT6 and EMT6/AR1 cells treated with Cellax-DiI for 4 h (normalized against the nucleus stain). Average of 6 different samples with ± SD.
Previously, De Souza et al. 21 also demonstrated a significant antitumor activity of an intraperitonial implant of a slow releasing formulation of DTX against a multidrug resistant ovarian cancer, HeyA8-MDR compared to intermittent dosing of native DTX. Hence, continuous slow release of DTX from Cellax may also influence the activity against the resistant line.
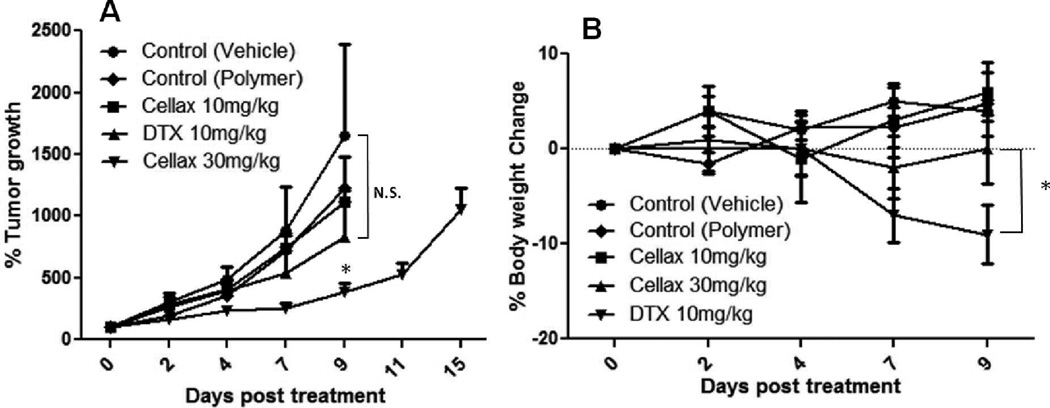
Finally, the in vivo efficacy of native DTX and Cellax against an MDR tumor model, EMT6/AR1, was examined. Native DTX was i.p. administered daily for 5 consecutive days at 10 mg DTX/kg, which was the MTD for this dosing regimen in this species of mouse, and Cellax was either dosed at the equal dose (10 mg DTX/kg) or increased dose (30 mg DTX/kg). Fig 6A shows that both DTX and Cellax at 10 mg DTX/kg displayed little antitumor activity against this aggressive MDR tumor, while Cellax at an increased dose (30 mg DTX/kg) exhibited significant antitumor efficacy, and on day 9, the tumor growth inhibition was 90%. Animals from all other groups had to be sacrificed on day 9 as they reached the end point (1500 mm3 in tumor volume or tumor ulceration) but Cellax treatment significantly controlled this aggressive growth of the tumor for 15 days. Additionally, the mice treated with native DTX suffered from significant body weight loss (10%, Fig 6B), and the mice showed visible signs of pain or distress including piloerection, lethargy, weight loss and weakness. On the other hand, the Cellax treated mice displayed no body weight loss or any significant sign of toxicity during the therapy. As discussed in the previous paragraph that Cellax exerted increased antitumor efficacy against the MDR tumor could be due to several different mechanisms, including sustained drug release, increased cellular internalization bypassing Pgp efflux and improved drug delivery to tumors. However, as the equal dose of Cellax did not show any significant antitumor efficacy, the mechanism of the enhanced activity of Cellax against the EMT6/AR1 tumor was largely because of increased dose that can be safely administered with Cellax.
Figure 6.
In vivo antitumor efficacy against murine EMT6/AR1 tumor. (A) tumor size; (B) body weight. Data = mean ± SD (n=4–5). * indicates p<0.05 compared to the saline control.
In the present study, we wanted to explore two distinct aspects of Cellax therapy: i) whether treatment with Cellax has any effect on the induction of resistance in the cancer cells and ii) whether Cellax can be used to enhance the treatment of tumors that have already acquired taxane resistance. Using the in vitro and in vivo systems, we have demonstrated that i) Pgp was the major molecule for induction of DTX resistance; ii) while native DTX therapy led to significant increase in Pgp expression in the tumor cells, Cellax did not up-regulate Pgp; iii) sustained drug release from Cellax might be a major contributing factor that Cellax did not significantly induce Pgp expression. However, apart from the slow drug releasing characteristics, it was also found that Cellax was rapidly internalized into tumor cells and localized in the endosomes/lysosomes, where efficient intracellular drug release could occur. This uptake pathway might also help Cellax avoid up-regulating Pgp, which requires further investigation. The detailed mechanistic study will be pursued and reported in the future.
For the treatment of DTX resistant tumors, we have shown that i) Cellax was efficiently internalized and exhibited increased potency against the resistant cells compared to the native drug. ii) due to the improved tolerability and the safety profile, a higher amount of dose can be injected to the (resistant-) tumor bearing mice, leading to significantly better control over tumor growth compared to native DTX.
4. Conclusion
Our data show that the primary mechanism of DTX resistance was via over-expression of Pgp in our model systems, and Cellax treatment did not up-regulate Pgp in breast tumor cells or orthotopic breast tumor possibly because of its sustained releasing mechanism. In the cultured cells, Cellax displayed enhanced potency compared to native DTX due to sustained drug release and efficient cellular internalization. The in vivo data with a MDR tumor demonstrated that Cellax could be safely given at an elevated dose to enhance the activity against the EMT6/AR1 tumor compared to native DTX. These encouraging data suggest that Cellax could be further developed as a DTX alternative to enhance taxane chemotherapy by circumventing MDR.
ACKNOWLEDGEMENTS
This work was funded by grants from the Ontario Institute for Cancer Research Intellectual Property Development and Commercialization Fund, the Canadian Institutes of Health Research (PPP-122898) and National Institutes of Health (CA17633901).S.D. Li is a recipient of a Coalition to Cure Prostate Cancer Young Investigator Award from the Prostate Cancer Foundation and a New Investigator Award from Canadian Institutes of Health Research (MSH-130195). The Ontario Institute for Cancer Research is financially supported by the Ontario Ministry of Economic Development and Innovation.
References
- 1.Yusuf RZ, Duan Z, Lamendola DE, Penson RT, Seiden MV. Paclitaxel resistance: molecular mechanisms and pharmacologic manipulation. Curr Cancer Drug Targets. 2003;3(1):1–19. doi: 10.2174/1568009033333754. [DOI] [PubMed] [Google Scholar]
- 2.Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer. 2002;2(1):48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- 3.Klein I, Sarkadi B, Varadi A. An inventory of the human ABC proteins. Biochim Biophys Acta. 1999;1461(2):237–262. doi: 10.1016/s0005-2736(99)00161-3. [DOI] [PubMed] [Google Scholar]
- 4.Baekelandt MM, Holm R, Nesland JM, Trope CG, Kristensen GB. P-glycoprotein expression is a marker for chemotherapy resistance and prognosis in advanced ovarian cancer. Anticancer Res. 2000;20(2B):1061–1067. [PubMed] [Google Scholar]
- 5.Kamazawa S, Kigawa J, Kanamori Y, Itamochi H, Sato S, Iba T, Terakawa N. Multidrug resistance gene-1 is a useful predictor of Paclitaxel-based chemotherapy for patients with ovarian cancer. Gynecol Oncol. 2002;86(2):171–176. doi: 10.1006/gyno.2002.6738. [DOI] [PubMed] [Google Scholar]
- 6.Li R, Zou H, Xiao H, Wu R. Carbon nanotubes as intracellular carriers for multidrug resistant cells studied by capillary electrophoresis-laser-induced fluorescence. Methods Mol Biol. 2010;625:153–168. doi: 10.1007/978-1-60761-579-8_13. [DOI] [PubMed] [Google Scholar]
- 7.Liu Y, Huang L, Liu F. Paclitaxel nanocrystals for overcoming multidrug resistance in cancer. Mol Pharm. 2010;7(3):863–869. doi: 10.1021/mp100012s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kabanov AV, Batrakova EV, Alakhov VY. Pluronic block copolymers for overcoming drug resistance in cancer. Adv Drug Deliv Rev. 2002;54(5):759–779. doi: 10.1016/s0169-409x(02)00047-9. [DOI] [PubMed] [Google Scholar]
- 9.Ma P, Dong X, Swadley CL, Gupte A, Leggas M, Ledebur HC, Mumper RJ. Development of idarubicin and doxorubicin solid lipid nanoparticles to overcome Pgp-mediated multiple drug resistance in leukemia. J Biomed Nanotechnol. 2009;5(2):151–161. doi: 10.1166/jbn.2009.1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang D, Tang J, Wang Y, Ramishetti S, Fu Q, Racette K, Liu F. Multifunctional nanoparticles based on a single-molecule modification for the treatment of drug-resistant cancer. Mol Pharm. 2013;10(4):1465–1469. doi: 10.1021/mp400022h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ernsting MJ, Tang WL, MacCallum N, Li SD. Synthetic modification of carboxymethylcellulose and use thereof to prepare a nanoparticle forming conjugate of docetaxel for enhanced cytotoxicity against cancer cells. Bioconjug Chem. 2011;22(12):2474–2486. doi: 10.1021/bc200284b. [DOI] [PubMed] [Google Scholar]
- 12.Ernsting MJ, Murakami M, Undzys E, Aman A, Press B, Li SD. A docetaxel-carboxymethylcellulose nanoparticle outperforms the approved taxane nanoformulation, Abraxane, in mouse tumor models with significant control of metastases. J Control Release. 2012;162(3):575–581. doi: 10.1016/j.jconrel.2012.07.043. [DOI] [PubMed] [Google Scholar]
- 13.Ernsting MJ, Tang WL, Maccallum NW, Li SD. Preclinical pharmacokinetic, biodistribution, and anti-cancer efficacy studies of a docetaxel-carboxymethylcellulose nanoparticle in mouse models. Biomaterials. 2012;33(5):1445–1454. doi: 10.1016/j.biomaterials.2011.10.061. [DOI] [PubMed] [Google Scholar]
- 14.Patel KJ, Tannock IF. The influence of P-glycoprotein expression and its inhibitors on the distribution of doxorubicin in breast tumors. BMC Cancer. 2009;9:356. doi: 10.1186/1471-2407-9-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Geney R, Ungureanu lM, Li D, Ojima I. Overcoming multidrug resistance in taxane chemotherapy. Clin Chem Lab Med. 2002;40(9):918–925. doi: 10.1515/CCLM.2002.161. [DOI] [PubMed] [Google Scholar]
- 16.Wang Y, Chen Q, Jin S, Deng W, Li S, Tong Q, Chen Y. Up-regulation of P-glycoprotein is involved in the increased paclitaxel resistance in human esophageal cancer radioresistant cells. Scand J Gastroenterol. 2012;47(7):802–808. doi: 10.3109/00365521.2012.683042. [DOI] [PubMed] [Google Scholar]
- 17.Smoter M, Bodnar L, Duchnowska R, Stec R, Grala B, Szczylik C. The role of Tau protein in resistance to paclitaxel. Cancer Chemother Pharmacol. 2011;68(3):553–557. doi: 10.1007/s00280-011-1696-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mozzetti S, Ferlini C, Concolino P, Filippetti F, Raspaglio G, Prislei S, Gallo D, Martinelli E, Ranelletti FO, Ferrandina G, Scambia G. Class III beta-tubulin overexpression is a prominent mechanism of paclitaxel resistance in ovarian cancer patients. Clin Cancer Res. 2005;11(1):298–305. [PubMed] [Google Scholar]
- 19.Castells M, Thibault B, Delord JP, Couderc B. Implication of tumor microenvironment in chemoresistance: tumor-associated stromal cells protect tumor cells from cell death. Int J Mol Sci. 2012;13(8):9545–9571. doi: 10.3390/ijms13089545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ho EA, Soo PL, Allen C, Piquette-Miller M. Impact of intraperitoneal, sustained delivery of paclitaxel on the expression of P-glycoprotein in ovarian tumors. J Control Release. 2007;117(1):20–27. doi: 10.1016/j.jconrel.2006.10.007. [DOI] [PubMed] [Google Scholar]
- 21.De Souza R, Zahedi P, Badame RM, Allen C, Piquette-Miller M. Chemotherapy dosing schedule influences drug resistance development in ovarian cancer. Mol Cancer Ther. 2011;10(7):1289–1299. doi: 10.1158/1535-7163.MCT-11-0058. [DOI] [PubMed] [Google Scholar]
- 22.Wong HL, Bendayan R, Rauth AM, Xue HY, Babakhanian K, Wu XY. A mechanistic study of enhanced doxorubicin uptake and retention in multidrug resistant breast cancer cells using a polymer-lipid hybrid nanoparticle system. J Pharmacol Exp Ther. 2006;317(3):1372–1381. doi: 10.1124/jpet.106.101154. [DOI] [PubMed] [Google Scholar]