Abstract
BACKGROUND/OBJECTIVES
Ultraviolet B (UVB) irradiation on skin can induce production of reactive oxygen species (ROS), which cause expression of matrix metalloproteinases (MMPs) and collagen degradation. Thus, chronic exposure of skin to UVB irradiation leads to histological changes consistent with aging, such as wrinkling, abnormal pigmentation, and loss of elasticity. We investigated the protective effect of the standardized green tea seed extract (GSE) on UVB-induced skin photoaging in hairless mice.
MATERIALS/METHODS
Skin photoaging was induced by UVB irradiation on the back of Skh-1 hairless mice three times per week and UVB irradiation was performed for 10 weeks. Mice were divided into six groups; normal control, UVB irradiated control group, positive control (UVB + dietary supplement of vitamin C 100 mg/kg), GSE 10 mg/kg (UVB + dietary supplement of GSE 10 mg/kg), GSE 100 mg/kg (UVB + dietary supplement of GSE 100 mg/kg), and GSE 200 mg/kg (UVB + dietary supplement of GSE 200 mg/kg).
RESULTS
The dietary supplement GSE attenuated UVB irradiation-induced wrinkle formation and the decrease in density of dermal collagen fiber. In addition, results of the antioxidant analysis showed that GSE induced a significant increase in antioxidant enzyme activity compared with the UVB irradiation control group. Dietary supplementation with GSE 200 mg/kg resulted in a significant decrease in expression of MMP-1, MMP-3, and MMP-9 and an increase in expression of TIMP and type-1 collagen.
CONCLUSIONS
Findings of this study suggest that dietary supplement GSE could be useful in attenuation of UVB irradiation-induced skin photoaging and wrinkle formation due to regulation of antioxidant defense systems and MMPs expression.
Keywords: Green tea seed, photoaging, MMPs, antioxidant, UVB irradiation
INTRODUCTION
In recent years, consumption of skin care products for maintenance of healthy skin is on the increase. Accordingly, studies of material development for prevention of skin aging and for maintenance of healthy skin have been actively conducted. Studies of anti-skin aging have focused on development of functional materials, from natural herbs, with antioxidant for anti-wrinkle and skin whitening. Several studies have reported that antioxidant materials isolated from natural herbs attenuate the risk of skin damage induced by UVB irradiation [1,2]. These reports have mainly studied the effects of topical application for development of functional cosmetics. However, recent studies have indicated that effects of oral administration can be enhanced by combination with topical application in the antioxidant systems for anti-skin aging [3].
Overexposure to UV irradiation, particularly to UVB (290-320 nm), leads to damage of the skin, called photoaging, which is characterized by wrinkle formation, dry and rough skin, irregular pigmentation, and poor elastic recoil [4]. Chronic exposure to UV radiation is known to induce overproduction of reactive oxygen species (ROS) in the epidermis, which destroy the antioxidant defense systems in the body, and finally cause oxidative stress [5,6,7]. Oxidative stress induces formation of lipid peroxidation, through lipid-derived radical, which leads to disruption of the cell membrane, and, consequently, cell death [8]. In addition, overproduction of ROS triggers the release of proinflammatory cytokines such as interleukin (IL)-1, -6, and -8, and tumor necrosis factor-alpha (TNF-α) from keratinocytes of the epidermis [9,10]. In the fibroblasts of dermis, these cytokines induce expression of matrix metalloproteinases (MMPs), which play key roles in degradation of collagen, elastin, and other proteins in connective tissue and bone [11]. In addition, collagen synthesis was decreased by reduced expression of type-1 procollagen. Collagen, the most abundant protein in the dermis, provides structure of skin, and more than 90% of collagen in the body is type-1 collagen. Thus, UV-induced expression of MMPs and impaired procollagen synthesis result in activation of damage to skin structure and wrinkle formation on skin photoaging [12,13]. These reports demonstrate that antioxidant materials can prevent photoaging mediated by reduction of UV-induced ROS.
Green tea, a concentrate of polyphenols, is a representative herb of the natural antioxidant herbs. Several studies have reported on the effects of green tea, including antioxidant, antiobesity, antiinflammation, and anticancer [14,15]. Despite the increased demand for green tea, studies and practical utilization of other parts are insufficient, except the leaf. A few studies have reported that oil of green tea seed has antioxidant, antiobesity, and antibiotic effects [16,17,18]. The chemical constituents of green tea seed include saponin, flavonoid, and vitamins [19], which suggest that it might have the effect of green tea seed extract (GSE) on UV-induced photoaging. In the current study, we investigated the protective effect of GSE on UVB-induced photoaging in hairless mice as measured by antioxidant enzyme activity and expression of type-1 collagen, tissue inhibitor of metalloproteinase (TIMP) and MMPs.
MATERIALS AND METHODS
Plant material and preparation of the extracts
The green tea seed was boiled with 70% ethanol for 2h in a reflux apparatus. The ethanol of the extracts was removed in vacuo and filtered. The extracts were concentrated to 50° Brix in vacuo and the concentration of 87.5% was mixed with dextrin 17.5%. The mixture was lyophilized and kept in a tight and light-protected container at -20℃ until use.
Standardization of GSE
Standardization of GSE was performed by high performance liquid chromatography (HPLC system, Agilent 1260 Infinity, Waldbronn, Germany). GSE was extracted with methanol and filtered using a 0.45 µm syringe filter before injection to HPLC. A reference standard stock solution containing epicatechingallate (ECG) (Sigma, MO, USA) was used. A SP column C18, MG (4.6 × 250 mm, 5 µm) was used for the separation and detected by UV detection at 280 nm. The mobile phase consisted of A (acetonitrile) and B (0.1% acetic acid) at a flow rate of 1.0 mL/min. After injection of a 20 µL sample, the system was increased from 85% A to 50% A in 20 min, and maintained at 85% A for another 10 min. The amount of ECG in the GSE was determined from a calibration curve obtained by the concentration of ECG standard against the peak area. Average ECG contents in GSE were 19.72 ± 1.81 mg/g.
Experimental animals and supplement
The experimental protocol was approved by the Institutional Animal Care and Use Review Committee of Kyung Hee University (KHUASP(SU)-12-14). Female hairless mice (Skh-1) (six weeks old) were purchased from Daehan Bio Link (Eumseong, Korea). The animals were placed in wire mesh bottomed individual cages and housed in climate-controlled quarters (24 ± 1℃ at 50% relative humidity) with a 12-h light:12-h dark cycle. All animals were acclimatized for seven days before the experiment, fed standard pellet chow, and given fresh water ad libitum.
A total of 48 mice were randomly divided into six groups (eight animals per group): normal control (-UVB), UVB irradiated control group (+UVB), positive control (UVB + dietary supplement of vitamin C 100 mg/kg), GSE 100 mg/kg (UVB + dietary supplement of GSE 100 mg/kg), and GSE 200 mg/kg (UVB + dietary supplement of GSE 200 mg/kg). Animals in each group were fed their experimental diets, which were based on the AIN93G diet, and UVB irradiation was performed for 10 weeks.
Wrinkle formation induced by UVB irradiation
Skin wrinkles were induced using a UVB lamp (five Sankyo Denki G5T5 lamps, Sankyo Denki Co., Yokohama, Japan with application to the dorsal skin of Skh-1 hairless mice three times per week for 10 weeks. Minimal erythematous dose (MED) was set at 80 mJ/cm2. Each time intensity of UVB irradiation was 1MED (80 mJ/cm2) at week 1; 2MED (160 mJ/cm2) at week 2; 3MED (240 mJ/cm2) at week 3; and 4MED (320 mJ/cm2) at week 4~10. At the end of the 10 weeks, all animals were sacrificed by cervical dislocation and the dorsal skin and blood was collected by orbital venipuncture for analysis.
Morphological and histopathological observation
Morphologic changes of mouse skin surface were observed using a Digital microscope (DG-3x, Scalar Co., Tokyo, Japan). For histopathological assessment, the dorsal skin tissue was fixed in 10% neutral buffered formaldehyde solution, embedded in paraffin, and stained with hematoxylin and eosin and observed under a light microscope.
Determination of antioxidant enzymes activity
Serum was collected from whole blood by centrifugation at 3,000 g at 4℃ for 20 minutes. The levels of serum SOD activity were measured using the SOD assay kit-WST (Dojindo, Kumamoto, Japan), according to the manufacturer's instructions. A 20 µL sample solution was mixed with 200 µL of WST-1 (2-(4-iodophenyl)-3-(4-nitrophenyl)-5-(2.4-disulfophenyl)-2H-tetr azolium, monosodium salt) and 20 µL of xanthine oxidase in a 96-well plate, followed by incubation at 37℃ for 20 min, and the absorbance was measured at 450 nm. Levels of serum catalase activity were measured using the catalase assay kit (BioVision Inc., Milpitas, CA, USA). Catalase was first reacted with H2O2 to produce water and oxygen, the unconverted H2O2 was reacted with the OxiRedTM probe and the absorbance was measured at 570 nm. GPx activity was determined using the Glutathione peroxidase activity assay kit (BioVision Inc., Milpitas, CA, USA) according to the manufacturer's protocol, and the absorbance was measured at 340 nm.
RNA isolation and real-time PCR
Dorsal skin tissue was homogenized using rotor-stator homogenizers in the presence of buffer RLT (lysis buffer, Qiagen, Valencia, CA, USA), including β-mercaptoethanol. Total RNA was extracted from dorsal skin tissue lysate using the RNeasy Mini kit (Qiagen, Valencia, CA, USA). Complementary DNA was synthesized from 1 µL purified RNA in 20 µL of reaction buffer using the iScript™ cDNA Synthesis Kit (Biorad, Hercules, CA, USA). Real-Time PCR (Applied Biosystems, Foster City, CA, USA) was performed using the cDNA (1 µL) with the SYBR Green PCR Master Mix (iQ SYBR Green Supermix, BIORAD, Hercules, CA, USA). The cDNA was amplified for 45 cycles of denaturation (95℃ for 30 s), annealing (58℃ for 30 s), and extension (72℃ for 45 s) using the following primers: GAPDH forward primer 5'-CAT GGC CTT CCG TGT TCC TA-3', reverse primer 5'-GCG GCA CGT CAG ATC CA-3'; MMP-1 forward primer 5'-TTG CCC AGA GAA AAG CTT CAG-3', reverse primer 5'-TAG CAG CCC AGA GAA GCA ACA-3'; MMP-3 forward primer 5'-CCC CTG ATG TCC TCG TGG TA-3' reverse primer 5'-GCA CAT TGG TGA TGT CTC AGG TT-3'; MMP-9 forward primer 5'-AGT GGG ACC ATC ATA ACA TCA CAT-3' reverse primer 5'-TCT CGC GGC AAG TCT TCA G-3'; TIMP-1 forward primer 5'-GCC CTT CGC ATG GAC ATT TA-3' reverse primer 5'-CCC CGA TCT GCG ATG ATG-3'; Type 1 collagen forward primer 5'-GCA CGA GTC ACA CCG GAA CT-3' reverse primer 5'-AAG GGA GCC ACA TCG ATG AT-3'. Data analysis of real-time RT-PCR results and calculation of the relative quantitation were performed using the 7500 System SDS software version 1.3.1 (Applied Biosystems, Foster City, CA, USA)
Statistical analysis
Data are expressed as mean ± SD. All data were analyzed by One-way ANOVA using SPSS statistical procedures for Windows (SPSS PASW Statistic 20.0, SPSS Inc. Chicago, IL, USA) and Duncan's multiple range test was used for examination of differences among groups. Statistical significance was considered at P < 0.05.
RESULTS
Morphological and histopathological observation of wrinkle formation
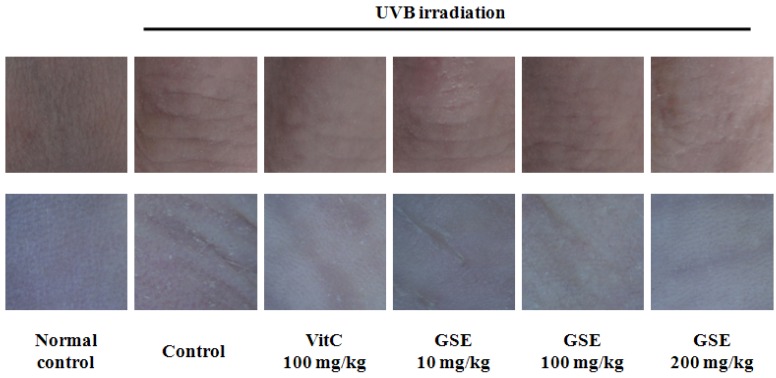
The UVB irradiation control group was observed for wrinkle formation, which shows patterns of furrows and thick crests. We used vitamin C for comparison with GSE. Vitamin C is well known in treatment of skin disorders for enhancement of epidermal differentiation and skin photoprotection [20]. Dietary supplement of vitamin C 100 mg/kg, positive control, showed attenuation of wrinkle formation induced by UVB irradiation. In the case of the GSE groups, wrinkles were formed as a pattern of shallow furrows and thin crests in a dose-dependent manner (Fig. 1).
Fig. 1.
Representative images of macroscopic morphological observation on skin of UVB-induced hairless mice after a 10-week experiment to evaluate the inhibitory effect of green tea seed extract (GSE). Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg.
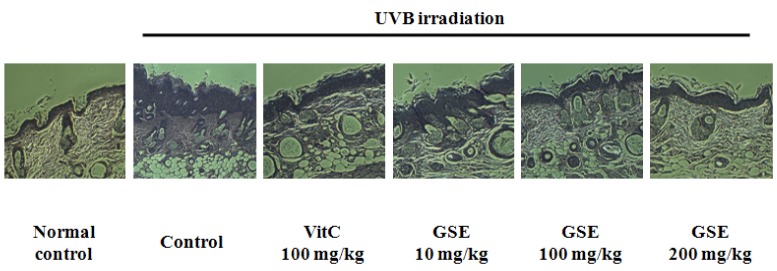
Results of histopathological observation on UVB-induced hairless mice skin are shown Fig. 2. When compared with the control group, the UVB irradiation control group showed a marked increase in epidermal thickness of dorsal skin and epidermal cells developed an irregular shape. In addition, density of collagen fiber in dermis showed a remarkable reduction. Dietary supplementation of vitamin C 100 mg/kg resulted in attenuation of histopathological changes induced by UVB irradiation. Dietary supplement of GSE 10 mg/kg had negligible effects, however, 100 mg/kg and 200 mg/kg tended to suppress the UVB-associated increase in epidermal thickness and decrease in density of dermal collagen fiber.
Fig. 2.
Representative images of histological observation by H&E staining on skin of UVB-induced hairless mice after a 10-week experiment to evaluate the inhibitory effect of green tea seed extract (GSE). Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg.
Effects of green tea seed extract on antioxidant enzyme activities
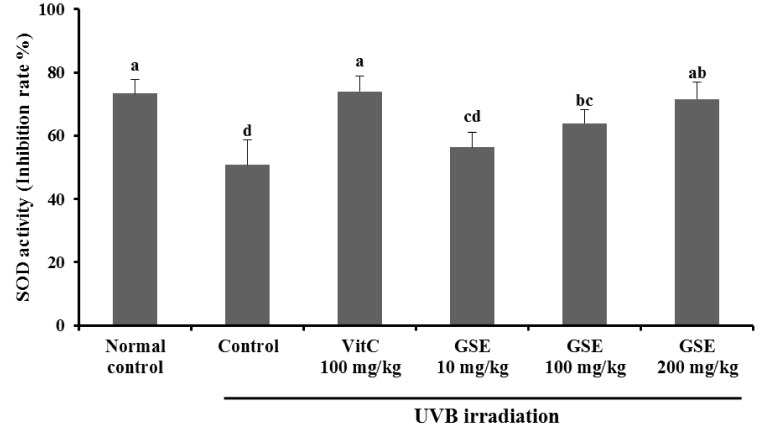
SOD activity of the UVB irradiation control group (50.70 ± 70.89%) was significantly reduced when compared with the normal control (73.34 ± 4.35%) (P < 0.05), whereas the dietary supplementation of vitamin C 100 mg/kg (73.97 ± 4.79%) resulted in a significant increase in SOD activity compared with the UVB irradiation control group, and no significant difference was observed in comparison with the normal control group (P < 0.05). No significant difference was observed between the UVB irradiation control group and GSE 10 mg/kg. However, SOD activity of GSE 200 mg/kg showed a significant increase, compared with the UVB irradiation control group, which was similar to the level of vitamin C 100 mg/kg (P < 0.05) (Fig. 3).
Fig. 3.
Effects of green tea seed extract (GSE) on serum superoxide dismutase activity of UVB-induced photoaging in hairless mice. Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg. Values are expressed as mean ± SD of eight mice. Different letters show a significant difference at P < 0.05 as determined by Duncan's multiple range test.
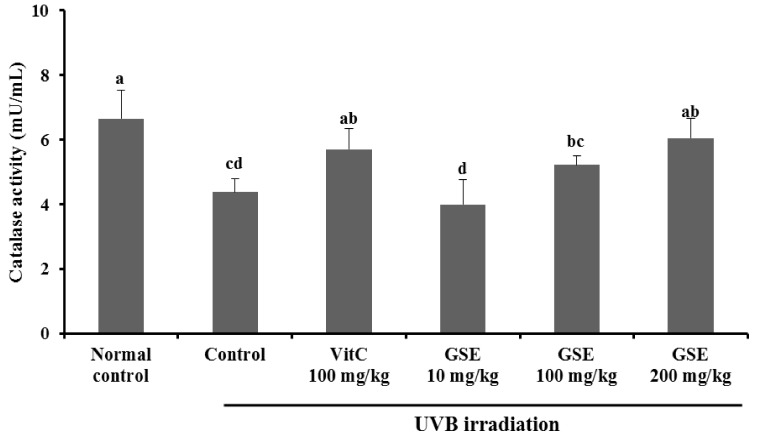
We found that UVB irradiation caused in a marked decrease in serum catalase activity compared with the normal control, whereas catalase activity of dietary supplement of vitamin C 100 mg/kg showed a significant increase, compared with the UVB irradiation control group (P < 0.05). Catalase activities of dietary supplement of GSE 10 mg/kg, 100 mg/kg, and 200 mg/kg were 3.97 ± 0.78 mU/mL, 5.21 ± 0.29 mU/mL, and 6.04 ± 0.62 mU/mL, respectively. No significant difference in serum catalase activity was observed between the dietary supplement of vitamin C 100 mg/kg and GSE 200 mg/kg (Fig. 4).
Fig. 4.
Effects of green tea seed extract (GSE) on catalase activity of UVB-induced photoaging in hairless mice. Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg. Values are expressed as mean ± SD of eight mice. Different letters show a significant difference at P < 0.05 as determined by Duncan's multiple range test.
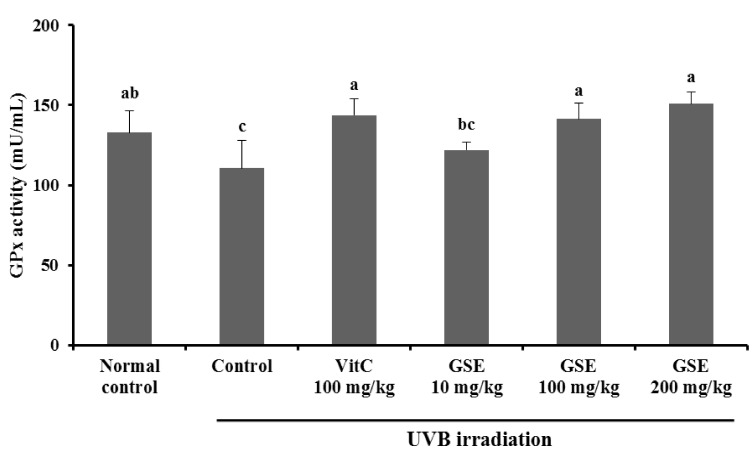
Results of the current study showed a significant decrease in serum GPx activity in the UVB irradiation control group when compared with the normal control (P < 0.05), whereas dietary supplement vitamin C 100 mg/kg exerted protection against the decrease in serum catalase activity induced by UVB irradiation. In the case of dietary supplement GSE, 100 mg/kg and 200 mg/kg, not 10 mg/kg, induced a significant increase when compared with the UVB irradiation control group (P < 0.05) (Fig. 5).
Fig. 5.
Effects of green tea seed extract (GSE) on glutathione peroxidase (GPx) activity of UVB-induced photoaging in hairless mice. Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg. Values are expressed as mean ± SD of eight mice. Different letters show a significant difference at P < 0.05 as determined by Duncan's multiple range test.
Effects of green tea seed extract on mRNA expression of MMPs, TIMP, and type-1 collagen
We investigated the effect of GSE on expression of MMP-1, MMP-3, and MMP-9, TIMP, and type-1 collagen (Table 1). Results of the current study showed that UVB irradiation induced an increase in expression of MMP-1, MMP-3, and MMP-9 and a decrease in expression of TIMP-1. In addition, type-1 collagen expression was significantly reduced by UVB irradiation compared with the normal group (P < 0.05). The dietary supplement vitamin C 100 mg/kg was found to cause a significant decrease in expression of MMP-1, MMP-3, and MMP-9 and an increase in expression of TIMP-1 and type-1 collagen (P < 0.05). No significant differences in mRNA expression were observed between the GSE 10 mg/kg and UVB irradiation control group. Supplementation with GSE 200 mg/kg resulted in a significant decrease in mRNA expression of MMP-1, MMP-3, and MMP-9 and an increase in expression of TIMP-1 and type-1 collagen (P < 0.05).
Table 1.
Effects of green tea seed extract (GSE) on mRNA expression of MMP-1, MMP-3, MMP-9, TIMP, and type-1 collagen of UVB-induced photoaging in hairless mice.
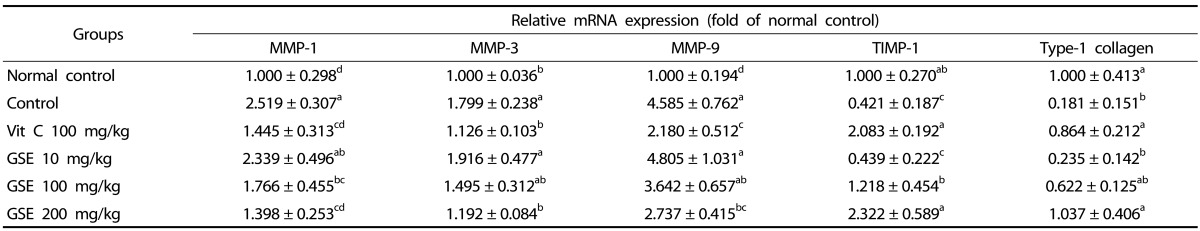
Normal control, No treatment group; Control, UVB-irradiated control group; Vit C, UVB irradiation with dietary supplement of vitamin C 100 mg/kg; GSE 10 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 10 mg/kg; GSE 100 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 100 mg/kg; GSE 200 mg/kg, UVB irradiation with dietary supplement of green tea seed extract 200 mg/kg. Values are expressed as mean ± SD of eight mice. Different letters show a significant difference at P < 0.05 as determined by Duncan's multiple range test.
DISCUSSION
Ultraviolet, a risk factor of photoaging, is classified according to three types, depending on its wavelength: UVA (320-400 nm), UVB (280-320 nm), and UVC (200-280 nm). UVC is absorbed completely in the atmosphere; however, UVA and UVB can reach the earth's surface and have harmful physiological effects on premature skin aging. UVB is absorbed in the epidermis, which is important to generation of skin photoaging [21]. Thus, in this study, we evaluated the effects of GSE on UVB irradiation-induced photoaging in hairless mice.
Skin, the largest organ of the body, has several functions as a physical barrier to environment and consists of three layers: the epidermis, the dermis, and the subcutis. The outer layer is the epidermis, which is composed of keratinocytes, melanocytes, and langerhans cell. This layer serves as a barrier to protect the body by synthesis of the protein keratin. The dermis is made up of collagen fiber, elastin fiber, and derma matrix, which are produced by fibroblasts [22]. Epidermis exposed to UVB irradiation has an irregular shape, large follicles, excessive deposition of keratin, and an increase in epidermal thickness for protection of dermis from UV damage. In the dermal areas, UV radiation induces collagen fiber loss, degradation of matrix macromolecules, and an amorphous mass of abnormal elastic fibers, which lead to formation of wrinkles and loss of elasticity in skin [23]. In the current study, we found that UVB irradiation caused deep wrinkle formation, rough skin surface, and histopathological changes characterized by irregular shape of the epidermis, collagen fiber loss, and an amorphous mass of abnormal elastic fibers. However, we showed that dietary supplement of GSE attenuated macroscopic and histopathological changes induced by UVB irradiation in a dose-dependent manner. These results, from macroscopic and histopathological observation, indicated that GSE has a photoprotective effect and anti-wrinkle activity.
To cope with ROS production, the normal cell is equipped with the antioxidative defense system, consisting of enzymatic and nonenzymatic antioxidants. The enzymatic antioxidants of the antioxidative defense system include superoxide dismutase (SOD), catalase, and glutathione peroxidase (GPx) [24]. However, when ROS production level reaches above threshold, overproduction of ROS can disturb the balance between ROS production and antioxidant defenses, which aggravate a decrease in activities of antioxidant enzymes. This condition is known as oxidative stress [25]. Several studies have reported that ROS overproduction, a factor of oxidative stress, can induce skin damage and photoaging during chronic UV radiation [9,10]. Okada et al. [26] demonstrated that repeated exposure to UVB suppressed activity of SOD, which catalyzes the dismutation of superoxide into oxygen and hydrogen peroxide. Hydrogen peroxide, produced by SOD activity, is decomposed to water and oxygen by catalase activity. Low catalase activity has been observed in the skin of photoaging induced by UVB. In addition, GPx protects the cell from oxidative damage during UVB irradiation at high hydrogen peroxide concentration. Acute UVB exposure induces activation of GPx for detoxification of hydrogen peroxide [27]. However, chronic UVB exposure reduces GPx activity due to disturbance of antioxidant defense [25]. In the current study, we found that dietary supplement of GSE 100 mg/kg and GSE 200 mg/kg imparted protection against the UVB irradiation-induced reduction of antioxidant enzyme activity. Park et al. [16] reported that green tea seed contained a reasonable amount of kaempferol, a type of flavonoid and a strong antioxidant. Our data suggested that the antioxidant properties of GSE defended against a decrease in antioxidant enzyme activity induced by UVB irradiation.
When UVB is absorbed in the epidermis, release of cytokines in epidermal keratinocytes is generated by overproduction of ROS, which stimulates the expression of MMPs and inhibits the expression of TIMP [10]. MMPs expression, suppressed by TIMP, induces degradation of collagen and elastin, thereby impairing the skin structure and formation of wrinkles [28]. Reduction of collagen is caused mainly by the action of MMP-1 expression and down-regulation of type-1 procollagen expression [29]. In this study, we showed that UVB irradiation resulted in an increase in expression of MMP-1, MMP-3, and MMP-9 and a decrease in expression of TIMP-1 and type-1 collagen. Dietary supplement GSE 200 mg/kg was found to cause a significant decrease in mRNA expression of MMP-1, MMP-3, and MMP-9 and an increase in expression of TIMP-1 and type-1 collagen (P < 0.05). These results indicate that GSE attenuates the change of expression of MMPs, TIMP, and type-1 collagen induced by UVB irradiation, and thereby inhibition of wrinkle formation. Green tea and green tea polyphenol have been reported to attenuate a decrease in antioxidant enzyme activities, as well as the skin damage and wrinkle formation induced by UVB irradiation [30]. According to the results of this study, green tea seed also had an antioxidant effect and an anti-wrinkle formation effect. We suggest that green tea seed may be a useful agent in protection against UVB irradiation-induced skin photoaging and wrinkle formation.
In this study, we showed that UVB irradiation induced wrinkle formation as a result of the decreases of antioxidant enzyme activity and TIMP expression and the increase of MMPs expression. However, the dietary supplement of GSE attenuated UVB irradiation-induced wrinkle formation, production of MMPs, and inhibition of antioxidant enzyme activities. In conclusion, the overall results of the current study demonstrated that the dietary supplement of GSE prevented wrinkle formation in the skin of UVB irradiation-induced hairless mice by regulation of antioxidant defense systems and expression of MMPs and TIMP. Therefore, green tea seed may be considered as a potential agent for prevention of skin photoaging and wrinkle formation.
References
- 1.Katiyar SK, Korman NJ, Mukhtar H, Agarwal R. Protective effects of silymarin against photocarcinogenesis in a mouse skin model. J Natl Cancer Inst. 1997;89:556–566. doi: 10.1093/jnci/89.8.556. [DOI] [PubMed] [Google Scholar]
- 2.Limtrakul P, Suttajit M, Semura R, Shimada K, Yamamoto S. Suppressive effect of soybean milk protein on experimentally induced skin tumor in mice. Life Sci. 1993;53:1591–1596. doi: 10.1016/0024-3205(93)90182-3. [DOI] [PubMed] [Google Scholar]
- 3.Passi S, De Pità O, Grandinetti M, Simotti C, Littarru GP. The combined use of oral and topical lipophilic antioxidants increases their levels both in sebum and stratum corneum. Biofactors. 2003;18:289–297. doi: 10.1002/biof.5520180233. [DOI] [PubMed] [Google Scholar]
- 4.Fisher GJ, Wang ZQ, Datta SC, Varani J, Kang S, Voorhees JJ. Pathophysiology of premature skin aging induced by ultraviolet light. N Engl J Med. 1997;337:1419–1428. doi: 10.1056/NEJM199711133372003. [DOI] [PubMed] [Google Scholar]
- 5.Zhang X, Rosenstein BS, Wang Y, Lebwohl M, Wei H. Identification of possible reactive oxygen species involved in ultraviolet radiation-induced oxidative DNA damage. Free Radic Biol Med. 1997;23:980–985. doi: 10.1016/s0891-5849(97)00126-3. [DOI] [PubMed] [Google Scholar]
- 6.Kovacs D, Raffa S, Flori E, Aspite N, Briganti S, Cardinali G, Torrisi MR, Picardo M. Keratinocyte growth factor down-regulates intracellular ROS production induced by UVB. J Dermatol Sci. 2009;54:106–113. doi: 10.1016/j.jdermsci.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 7.Aquino R, Morelli S, Tomaino A, Pellegrino M, Saija A, Grumetto L, Puglia C, Ventura D, Bonina F. Antioxidant and photoprotective activity of a crude extract of Culcitium reflexum H.B.K. leaves and their major flavonoids. J Ethnopharmacol. 2002;79:183–191. doi: 10.1016/s0378-8741(01)00379-8. [DOI] [PubMed] [Google Scholar]
- 8.Katiyar SK, Afaq F, Azizuddin K, Mukhtar H. Inhibition of UVB-induced oxidative stress-mediated phosphorylation of mitogen-activated protein kinase signaling pathways in cultured human epidermal keratinocytes by green tea polyphenol (-)-epigallocatechin-3-gallate. Toxicol Appl Pharmacol. 2001;176:110–117. doi: 10.1006/taap.2001.9276. [DOI] [PubMed] [Google Scholar]
- 9.Kawada S, Ishii N. Suppression of UVB-induced HIF-1α up-regulation by hyperoxia does not prevent wrinkle formation associated with increased MMPs activity in mouse skin. Biomed Res. 2011;32:363–372. doi: 10.2220/biomedres.32.363. [DOI] [PubMed] [Google Scholar]
- 10.Shephard P, Martin G, Smola-Hess S, Brunner G, Krieg T, Smola H. Myofibroblast differentiation is induced in keratinocyte-fibroblast co-cultures and is antagonistically regulated by endogenous transforming growth factor-beta and interleukin-1. Am J Pathol. 2004;164:2055–2066. doi: 10.1016/s0002-9440(10)63764-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Inomata S, Matsunaga Y, Amano S, Takada K, Kobayashi K, Tsunenaga M, Nishiyama T, Kohno Y, Fukuda M. Possible involvement of gelatinases in basement membrane damage and wrinkle formation in chronically ultraviolet B-exposed hairless mouse. J Invest Dermatol. 2003;120:128–134. doi: 10.1046/j.1523-1747.2003.12021.x. [DOI] [PubMed] [Google Scholar]
- 12.Talwar HS, Griffiths CE, Fisher GJ, Hamilton TA, Voorhees JJ. Reduced type I and type III procollagens in photodamaged adult human skin. J Invest Dermatol. 1995;105:285–290. doi: 10.1111/1523-1747.ep12318471. [DOI] [PubMed] [Google Scholar]
- 13.Fisher GJ, Voorhees JJ. Molecular mechanisms of photoaging and its prevention by retinoic acid: ultraviolet irradiation induces MAP kinase signal transduction cascades that induce Ap-1-regulated matrix metalloproteinases that degrade human skin in vivo. J Investig Dermatol Symp Proc. 1998;3:61–68. [PubMed] [Google Scholar]
- 14.Ruch RJ, Cheng SJ, Klaunig JE. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis. 1989;10:1003–1008. doi: 10.1093/carcin/10.6.1003. [DOI] [PubMed] [Google Scholar]
- 15.Zheng G, Sayama K, Okubo T, Juneja LR, Oguni I. Anti-obesity effects of three major components of green tea, catechins, caffeine and theanine, in mice. In Vivo. 2004;18:55–62. [PubMed] [Google Scholar]
- 16.Park JS, Rho HS, Kim DH, Chang IS. Enzymatic preparation of kaempferol from green tea seed and its antioxidant activity. J Agric Food Chem. 2006;54:2951–2956. doi: 10.1021/jf052900a. [DOI] [PubMed] [Google Scholar]
- 17.Kim NH, Choi SK, Kim SJ, Moon PD, Lim HS, Choi IY, Na HJ, An HJ, Myung NY, Jeong HJ, Um JY, Hong SH, Kim HM. Green tea seed oil reduces weight gain in C57BL/6J mice and influences adipocyte differentiation by suppressing peroxisome proliferator-activated receptor-gamma. Pflugers Arch. 2008;457:293–302. doi: 10.1007/s00424-008-0537-y. [DOI] [PubMed] [Google Scholar]
- 18.Yoon WH, Choi JH, Lee KH, Kim CH. Antimicrobial and antitumor activities of seed extracts of Camellia sinensis L. Korean J Food Sci Technol. 2005;37:108–112. [Google Scholar]
- 19.Rah HH, Baik SO, Han SB, Bock JY. Chemical compositions of the seed of Korean green tea plant (Camellia sinecis L.) Agric Chem Biotechnol. 1992;35:272–275. [Google Scholar]
- 20.McArdle F, Rhodes LE, Parslew R, Jack CI, Friedmann PS, Jackson MJ. UVR-induced oxidative stress in human skin in vivo: effects of oral vitamin C supplementation. Free Radic Biol Med. 2002;33:1355–1362. doi: 10.1016/s0891-5849(02)01042-0. [DOI] [PubMed] [Google Scholar]
- 21.Bowden GT. Prevention of non-melanoma skin cancer by targeting ultraviolet-B-light signalling. Nat Rev Cancer. 2004;4:23–35. doi: 10.1038/nrc1253. [DOI] [PubMed] [Google Scholar]
- 22.Rittié L, Fisher GJ. UV-light-induced signal cascades and skin aging. Ageing Res Rev. 2002;1:705–720. doi: 10.1016/s1568-1637(02)00024-7. [DOI] [PubMed] [Google Scholar]
- 23.Yaar M, Gilchrest BA. Skin aging: postulated mechanisms and consequent changes in structure and function. Clin Geriatr Med. 2001;17:617–630. v. doi: 10.1016/s0749-0690(05)70089-6. [DOI] [PubMed] [Google Scholar]
- 24.Morganti P, Bruno C, Guarneri F, Cardillo A, Del Ciotto P, Valenzano F. Role of topical and nutritional supplement to modify the oxidative stress. Int J Cosmet Sci. 2002;24:331–339. doi: 10.1046/j.1467-2494.2002.00159.x. [DOI] [PubMed] [Google Scholar]
- 25.Yamamoto Y. Role of active oxygen species and antioxidants in photoaging. J Dermatol Sci. 2001;27(Suppl 1):S1–S4. doi: 10.1016/s0923-1811(01)00120-7. [DOI] [PubMed] [Google Scholar]
- 26.Okada K, Takahashi Y, Ohnishi K, Ishikawa O, Miyachi Y. Time-dependent effect of chronic UV irradiation on superoxide dismutase and catalase activity in hairless mice skin. J Dermatol Sci. 1994;8:183–186. doi: 10.1016/0923-1811(94)90052-3. [DOI] [PubMed] [Google Scholar]
- 27.Filip A, Daicoviciu D, Clichici S, Mocan T, Muresan A, Postescu ID. Photoprotective effects of two natural products on ultraviolet B-induced oxidative stress and apoptosis in SKH-1 mouse skin. J Med Food. 2011;14:761–766. doi: 10.1089/jmf.2010.0142. [DOI] [PubMed] [Google Scholar]
- 28.Brennan M, Bhatti H, Nerusu KC, Bhagavathula N, Kang S, Fisher GJ, Varani J, Voorhees JJ. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem Photobiol. 2003;78:43–48. doi: 10.1562/0031-8655(2003)078<0043:mmitmc>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 29.Philips N, Conte J, Chen YJ, Natrajan P, Taw M, Keller T, Givant J, Tuason M, Dulaj L, Leonardi D, Gonzalez S. Beneficial regulation of matrixmetalloproteinases and their inhibitors, fibrillar collagens and transforming growth factor-beta by Polypodium leucotomos, directly or in dermal fibroblasts, ultraviolet radiated fibroblasts, and melanoma cells. Arch Dermatol Res. 2009;301:487–495. doi: 10.1007/s00403-009-0950-x. [DOI] [PubMed] [Google Scholar]
- 30.Vayalil PK, Mittal A, Hara Y, Elmets CA, Katiyar SK. Green tea polyphenols prevent ultraviolet light-induced oxidative damage and matrix metalloproteinases expression in mouse skin. J Invest Dermatol. 2004;122:1480–1487. doi: 10.1111/j.0022-202X.2004.22622.x. [DOI] [PubMed] [Google Scholar]