Abstract
BACKGROUND:
Research has been carried out to look for safe and effective anti-inflammation drugs from traditional Chinese herbal medicine. As a powerful research technology of life science, molecular biology has entered many areas of traditional Chinese medicine. This study aimed to investigate the effect of triptolide on tumor necrosis factor-a (TNF-α) and interleukin-8 (IL-8) of peritoneal macrophages activated by lipopolysaccharide (LPS) in mice.
METHODS:
Peritoneal elicited macrophages were separated, purified and activated by LPS in mice, then cultured in vitro with triptolide at different concentrations. The activity of TNF-α and the level of IL-8 of cellular supernatants were determined by MTT colorimetric assay and ELISA, respectively.
RESULTS:
The activity of TNF-α in macrophages was significantly inhibited (P<0.01) by triptolide (10-1-101μg/ml) during 4-24 hours in a time- and dose-dependent manner. The level of IL-8 in macrophages was significantly inhibited (P<0.01) by triptolide (10-1-101μg/ml) in 12 hours in a dose-dependent manner.
CONCLUSION:
Triptolide could inhibit the activity of TNF-α and the level of IL-8 in macrophages activated by LPS.
KEY WORDS: Triptolide, Lipopolysaccharide, Macrophage, Tumor necrosis factor-α, Interleukin-8
INTRODUCTION
Tripterygium wilfordii, one of the famous Chinese medical material, has been used to treat some autoimmune diseases such as dermatitis eczema. Many components have been extracted from Tripterygium wilfordii, with activities such as anti-inflammation, anti-fertility, anti-oncosis, antibiosis, immunosuppression, etc. Among them the extracted components include alkaloid, diterpenes, triterpenes, sesquiterpene, glycoside, etc.[1]
Triptolide is one of the over 70 effective components extracted from Tripterygium wilfordii. The effect on the secretion of inflammatory cellular factors TNF-α and IL-8 in the peritoneal macrophages of mice activated by
triptolide has been rarely reported.
If there are serious trauma and early stage disease, the organism releases cytokine as one of the stress reactions to them, which is part of the compensation mechanism. Because of a “secondary strike”, there are infection and other factors. Such reactions are harmful to the organism at later stage.[2] Endotoxin, which is also called lipopolysaccharide, is the most effective activator of macrophages. The present study aimed to activate the peritoneal macrophages of mice by LPS, and to explore the effect of triptolide on the activated macrophages during an inflammatory reaction, so that we could look for the effective target cells and to regulate homo-inflammatory disease.
METHODS
Reagents and medicine
PRMI-1640 substratum (Gibco), calf serum (Lingfei, Wuhan), lipopolysaccharide (Sigma), MTT (Duchefa), DMSO (Ameresco), L-glutamine (Zhengxiang, Shanghai), IL-8 detect kit (Zhongshan, Beijing), triptolide monomer (Fujian Traditional Chinese Medical College) with a purity of 99% were compounded into a necessary density with normal sodium after dissolving with a small amount of DMSO, then degerminged by millipore filter (0.22 μm) and kept for use. Other reagents were mixed into a necessary density with normal sodium, then degerminged by the millipore filter (0.22 μm) and reserved for use.
Experimental animals and cell strain
Kunming mice of both sexes, 8-12 weeks old, weighing 22-25 g, were provided by the Department of Experimental Animals of Tongji Medical College. L929 cell strain was provided by the Department of Biochemistry and Molecular Biology of Tongji Medical College. The cells of the strain were conventionally cultured in an incubator at 37°C, with a saturation humidity of 5% CO2. Nutrient was RPMI-1640 substratum containing 10% deactivation calf serum, 2 mmol/ L-glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin.
Separation, purification and cultivation of mice peritoneal macrophages[3]
Four ml PBS was injected into the abdominal cavity of the mice, while rubbing the abdomen gently to make the liquid flow thoroughly. The mice were sacrificed at 5 minutes later. The abdomen was sterilized with 75% alcohol, then abdominal liquid was collected and put into an aseptic tube for centrifugation at 1500r/min for 10 minutes. RPMI-1640 culture medium containing 10% heat-inactivated fetal calf serum was used for cell suspension. When the density was adjusted to a concentration of 1×106/ml, the tissue culture was put into a 96-well plate, with 100μl cellular suspension per port, and cultured in an incubator at 37°C, with a 5% CO2 saturation humidity for 4 hours. Finally the supernatants were abandoned and the non-adherent cells were washed out by phosphate-buffered saline to get macrophages.
The macrophages were activated by adding LPS into each port with a final concentration of 10μg/ml, and different dosages of triptolide were used at the concentrations of 10-3μg/ml, 10-2μg/ml, 10-2μg/ml, 1μg/ml and 10μg/ml separately, each for 6 re-holes. Normal saline served as a blank control. The macrophages were cultured at 37°C, with a 5% CO2 saturation humidity for 12 hours. The supernatants were obtained to determine the activity of TNF-α and the content of IL- 8, respectively. At the same time, triptolide with a final concentration of 10μg/ml was added and cultured for 1, 4, 8, 12, and 24 hours respectively to determine the activity of TNF-α.
Determination of TNF-α activity
The activity of TNF-α was determined using the MTT colorimetric assay.[4] L929 cells in the logarithmic growth phase were collected and adjusted to the density of 2×105/ ml. They were put into 96-well plates, with 100μl cellular suspension in each hole. The L929 cells as a control were cultured in an incubator at 37°C with a 5% CO2 saturation humidity for 24 hours. Later, 20μl of 5 mg/ml MTT was added into each port for continuous culture for 4 hours. The supernatants of the culture were abandoned, while 150 μl dissolved DMSO was added. Fifteen minutes later, the absorbance (A) of the solution at 570nm was determined using an enzyme marking instrument and the formula:
Activity of TNF-α (%) = [1 - (A of research/A of control)]×100%
Determination of IL-8[5]
The density of IL-8 was measured by ELISA with Zhongsan Jinqiao IL-8 test kit according to the instructions of the manufacturer. Standard preparation was diluted into 6 different densities: 1000pg/ml, 500pg/ ml, 250pg/ml, 125 pg/ml, 62.5pg/ml and 31.25pg/ml. Its A was assayed at A490 and the Concn-Abs standard curve was made; again the A of cell cultural supernatants was assayed at A490, and the density of IL-8 was counted by the Concn-Abs standard curve. Cultured macrophages with different densities of triptolide for 12 hours, then the supernatants were collected in different test tubes for measurement. The plate was taken from a sealing bag which was neutralized to room temperature. By mixed different dilution the supernatants were subjected to an assay together with IL-8 standard preparation, then 100μg mixed liquor was injected into a 96 ports plate coated by IL-8 mono-antibodies of mice, cultured at 37°C for 90 minutes. Washed out for 4 times, 100μg biotinylate antibody fluid was injected into each port except blank ones. The reaction port was sealed by bakelized paper, and the fluid was cultured in an incubator for 60 minutes, the liquid of the port was abandoned, washed for 4 times, and removed. Combined fluid 100μg was injected into each port except blank ports, and it was protected away from light. It was cultured in an incubator for 10 to 25 minutes, 100μg stop buffer was injected into each port, then the absorbance (A) of the solution was assayed at 490nm by an enzyme marking instrument.
Determination of cell survival rate
When single cellular suspension was prepared, the density of cells was adjusted to be 1×106/ml, mixed by 9 drops of cellular suspension with 1 drop of 0.4% Congo blue solution. The number of viable and dead cells was counted in 3 minutes and the activity of cells was determined by the following formula:
Cell survival rate(%)=[total number of viable cells/ (total number of viable cells+total number of dead cells)]×100%
Statistical analysis
Data were expressed as mean±SD. Student's t test was used to analyze the difference between the groups.
RESULTS
Effect of effective dose of triptolide on secretion of TNF-α in active peritoneal macrophages
Triptolide inhibited macrophage to secrete TNF-α at a density of 10-1μg/ml (P<0.01), and its inhibition effect was strengthened with increased concentration (Table 1).
Table 1.
Effect of triptolide on TNF-α production in activated peritoneal macrophages of mice (mean ±SD)
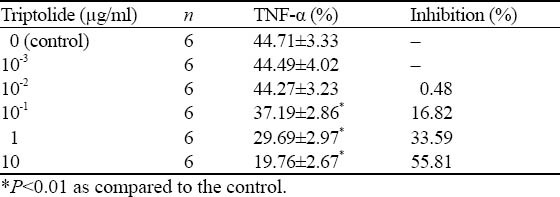
Effect of triptolide on secretion of TNF-α in active peritoneal macrophages
Triptolide10μg/ml inhibited macrophages to secrete TNF-α when it was used for 4 hours (P<0.01), and the effect of inhibition was time-dependent (Figure 1).
Figure. 1.
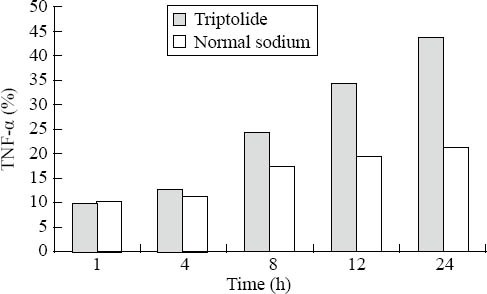
The effect of triptolide on TNF-α production in activated peritoneal macrophages of mice.
Effect of triptolide on secretion of IL-8 in active peritoneal macrophages at an effective dose
Triptolide inhibited macrophage to secrete TNF-α at a density of 10-1μg/ml (P <0.01), and the effect of inhibition was strengthened with the increasing concentration (Table 2), as illustrated by the Concn-Abs standard curve of IL-8 (Figure 2).
Table 2.
Effect of triptolide on IL-8 production in activated peritoneal macrophages of mice (mean ±SD)
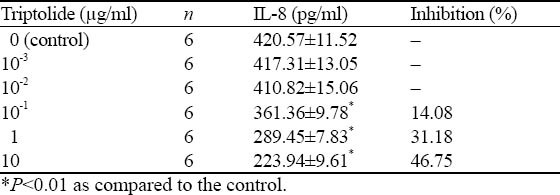
Figure. 2.
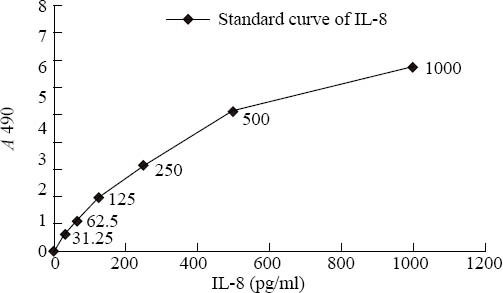
The concn-Abs standard curve of IL-8.
DISCUSSION
The process of urbanization and industrialization speeds up in China, but trauma gradually has been a prominent health care problem. The number of trauma patients has been increasing in recent years. Most of these patients have serious injuries which are easy to develop systemic inflammatory response syndrome (SIRS) and multiple organ dysfunction syndrome (MODS). Since the mortality rates of SIRS and MODS are relatively high, clinical research focuses on how to treat them effectively as soon as possible. It is known that inflammatory cellular factors such as TNF-α and IL-8 play an important role in the process of MODS, and the problem at present is how to regulate or control the relevant cytokines and to predict the prognosis.[6] The network of cytokines is made up of a dozen of cytokines which play an important role in systematic inflammatory reaction. TNF-α and IL-8 among them are “leaders” and major effectors inducing the activation of macrophages. They are also the important mediators of inflammation, which affect the development and turnover of inflammatory reaction.[7, 8]
TNF-α is secreted by active macrophage, and endotoxin is the most effective activator of macrophage. Gramble et al[9] reported that TNF-α is one of the key factors leading to inflammatory reaction. TNF-α can activate cascade reaction as an initiator of inflammation, produce “waterfall “ effect, and release other cytokines like IL-8. Moreover, it mediates cellular inflammatory reaction after trauma, and causes damage to multiple organs. The increased levels of TNF-α and IL-8 and injury severity are correlated with the shock of patients.[9]
IL-8 secretes rapidly after activation of mononuclear macrophages for 6-12 hours by LPS and TNF-α, and it is maintained continuously for 24 hours. The main biological effect of IL-8 is to activate neutrophils which will make morphologic changes after their contact with IL-8. IL-8 also transmigrates to the reactive site and releases a series of active products. These effects can cause regional inflammatory reaction for sterilization and cellular damage. Thus IL-8 as one of the neutrophil activating agents plays an important role in inflammatory reaction of the organism.[10]
Immune mediated inflammation is prevalent in many diseases that threaten human health. In recent years we have discovered many immune factors with powerful biological activities mediating inflammation such as TNF-α and IL-8. We have already explored their structure as well as the biological and deleterious effects on the organism. However, it is still a challenge how the high-level mediators of inflammation can be regulated or controlled.
In the present study we cultured triptolide with LPS activated mouse peritoneal macrophages at 37°C and 5% CO2 and then observed its effect on secretion of the inflammatory cytokines TNF-α and IL-8. A traditional Chinese herbal medicine was used as an immunomodulator to revise the function of active macrophages, thereby providing a way to regulate and control the high-level mediators of inflammation.
Triptolide in a dose-dependent manner significantly inhibited TNF-α secretion of LPS-activated macrophages at concentrations between 10-1μg/ml to 101μg/ml after 12 hours (P<0.01; Table 1). When 10μg/ml of triptolide was cultured with mouse peritoneal macrophages activated with LPS for 1, 4, 8, 12 and 24 hours respectively, TNF-α secretion was obviously inhibited (P<0.01). This effect was also time-dependant (Figure 1). Triptolide inhibited TNF-α secretion of macrophages at its concentration was as low as 10-1μg/ml (P<0.01), but the inhibition increased in a dose-dependent manner (Table 2). Figure 2 shows the standard curve of IL-8 concentration-absorbance. Triptolide in a dose-dependent manner also inhibited secretion of IL-8 in LPS-activated macrophages at concentrations from 10-1μg/ml to 101μg/ ml after 12 hours (P<0.01;Table 2). It is not cytotoxic to the macrophages in this range of concentrations, with a viability being greater than 95% as determined by Congo blue staining.
Triptolide is the active component of Tripterygium wilfordii, and it is known to cause problems with normal functioning of the liver, kidney, genitals and other systems in clinical practice.[11] Because the therapeutic dose is very close to the toxic dose, toxicity is significantly influenced by dose and time. Some researchers suggest clinical toxicity be minimized by control of these factors.[12]
The results of the present study show that triptolide inhibits the secretion of inflammatory cytokines TNF-α and IL-8 from LPS-activated peritoneal macrophages of mice by regulating and controlling the levels of TNF-α and IL-8. It is suggested that triptolide may limit inflammation and protect against excessive inflammation. However, the molecular mechanism underlying the inhibition of TNF-α and IL-8 secretion in macrophages is still under investigation.
Footnotes
Funding: None
Ethical approval: Not needed.
Competing interest: None.
Contributors: Yang F wrote the first draft of this paper. All authors contributed to the intellectual content and approved the final versions.
REFERENCES
- 1.Yin XQ, Zhang DS. The progress of clinical effect on tripterygium wilfordii. Medical Recapitulate. 2001;7:438–439. [Google Scholar]
- 2.Liu DH, Su YP, Zhang W, Lou SF. Role of macrophages in causing hypercytokinemia in scalded rats and its functional changes. Chin J Crit Care Med. 2001;11(21):630–632. [Google Scholar]
- 3.Er Z. Beijing: The People's Medical Publishing House; 1997. Tissue culture and molecular cytology technology; pp. 127–128. [Google Scholar]
- 4.Flick DA, Gifford GE. Comparison of in vitro cell cytotoxic assays for tumor necrosis factor. Immunol Assays. 1984;68:167–169. doi: 10.1016/0022-1759(84)90147-9. [DOI] [PubMed] [Google Scholar]
- 5.Wang LL. Beijing: The People's Medical Publishing House; 2003. Clinical immunology and Immunology examination; pp. 146–155. [Google Scholar]
- 6.Sheng ZY. Prevent and treatment of sepsis and MODS after serious traumatic burning injury. Chin J Trauma. 2005;21:11. [Google Scholar]
- 7.Spielmann S, Kerner T, Ahlers O, Keh D, Gerlach M, Gerlach H. Early detection of increased tumuor necrosis factor alpha (TNF-α) and soluble TNF receptor protein plasma levels after trauma reveals associations with the clinical course. Acta Anaesthesiol. 2001;45:364–370. doi: 10.1034/j.1399-6576.2001.045003364.x. [DOI] [PubMed] [Google Scholar]
- 8.Liu GX, Li L, Li XF, Li JX, Li JF, Jiang QH, et al. Effect of proinflammatory cytokine expression on the prognosis of patients with severe multiple trauma. J Traumatic Surg. 2008;24:103–105. [Google Scholar]
- 9.Gamble L, Bagby GJ, Quinton LJ, Happel KI, Mizgerd JP, Zhang P, et al. The systemic and pulmonary LPS binding protein response to intratracheal lipopolysaccharide. Shock. 2009;31:212–217. doi: 10.1097/SHK.0b013e31817c0d7d. [DOI] [PubMed] [Google Scholar]
- 10.Iwabe T, Harada T, Tsudo T, Nagano Y, Yoshida S, Tanikawa M, et al. Tumor necrosis factor alpha promotes proliferation of endometriotic stromal cells by inducing interleukin-8 gene and protein expression. J Clin Endocrinol Metab. 2000;85:824–829. doi: 10.1210/jcem.85.2.6335. [DOI] [PubMed] [Google Scholar]
- 11.Yang XH, Zhang WC. Application and adverse reaction of tripterygium wilfordii preparation. Clin J TC Med. 2004;16:511–512. [Google Scholar]
- 12.Huang Z, Mao QQ. Clinical practice, adverse effect and prevention of tripterygium glycosides. Drug Evaluation. 2005;2:125–128. [Google Scholar]


