Abstract
BACKGROUND:
This study was undertaken to observe the concentration of SP-A/B and the pulmonary surfactant in the lung tissue of rats with acute lung injury/acute respiratory distress syndrome caused by paraquat poisoning after the treatment of metabolic antioxidant-lipoic acid and whether its influence was related to TNF-α.
METHODS:
Sixty-six male Sprage-Dawley rats were randomly divided into three groups: normal control group (NS group), 6 rats; paraquat poisoning group (PQ group), 30 rats; and paraquat+lipoic acid treatment group (LA group), 30 rats. The rats in the PQ and LA groups were subdivided into 3-, 6-, 12-, 24-, 48-hour subgroups, with 6 rats in each group. After the rats were sacrificed, lung tissue from the same part was taken from the rats. After HE staining, histological changes were observed in the tissue under a light microscope. Lung tissue was also taken to test the levels of superoxide dismutase (SOD) and malondialdehyde (MDA). Whole blood (0.8 mL) without anticoagulant was drawn from the tail vein of rats for the determination of the TNF-α level. The total RNA of the lung tissue was collected, and the Rt-PCR method was used to measure the levels of SP-A and SP-B mRNA.
RESULTS:
HE staining showed that histopathological changes were milder in the LA group than in the PQ group. There were significant differences in MDA and SOD levels between different intervals both in intergroups and intragroups except the 3-hour subgroup (P<0.01). Likewise, the significant differences in the levels of TNF-α were also present between the three groups and between different intervals (P<0.01). The significant differences in SP-A mRNA and SP-B mRNA amplification ratio were seen between the three groups at the same intervals (P<0.01), but the differences between different intervals in the PQ group were statistically significant (P<0.05). The differences between different intervals in the LA group were statistically significant (P<0.01).
CONCLUSION:
Lipoic acid in acute paraquat poisoning could diminish lung tissue damage by regulating directly tumor necrosis factor and indirectly the content of pulmonary surfactant so as to reduce pulmonary edema, improve lung compliance, and finally protect lung tissues.
KEYWORDS: Lipoic acid, Paraquat, Acute lung injury, Surfactant protein, Tumor necrosis factor-α, Oxygen free radical, Superoxide dimutase, Malonaldehyde
INTRODUCTION
Paraquat (PQ) or gramoxone is a worldwide used non-selective contact herbicide. The death rate of patients with paraquat poisoning is quite high, and there is still lack of special treatment for paraquat poisoning.[1]
The lung is one of the biggest target organs that PQ reacted to the human toxicity. The injury of the lung was caused by PQ-induced oxidative stress. PQ concentrated in the lung tissue and stimulated the immune activation. With the effects of inflammatory mediators, PQ stimulated body macrophages and granulocytes to generate a large number of effect factors, such as cytokines, chemokines, and inflammatory mediators, to induce inflammatory reaction. Of these factors, tumor necrosis factor, TNF-α, played a very important role.
Lipoic acid (LA) belongs to the compounds of vitamin B, and can enter the cell after intestinal absorption in vivo. Exogenous LA has antioxidant and anti-inflammatory effects. Studies showed that LA also could reduce the level of TNF-α. And the reduction of TNF-α could reduce the inhibition of pulmonary surfactant (PS), so that alveolar surfactants increased relatively, especially surfactant protein (SP) A and B.
In this experiment, LA was used to treat pulmonary tissue injury in rats caused by paraquat poisoning. The levels of superoxide dismutase (SOD), malondialdehyde (MDA), SP-A mRNA, SP-B mRNA and TNF-α from the venous blood of the pulmonary tissue were observed. The effect of LA on acute paraquat poisoning PS and related mechanisms were studied.
METHODS
Main reagents
LA (item T1395) and PQ (purity of 98%, number 36541) were purchased from Sigma Company, USA. TNF-α kit was bought from Wuhan Boster Company, China. Malonaldehyde (MDA) kit (item A003-1) and superoxide dimutase (SOD) kit (item A001) were from Nanjing Research Institute of Biological Engineering, China.
Grouping of experimental animals and establishment of models
Sixty-six healthy male Sprage-Dawley rats, weighing 200±15 g, were purchased from the Experimental Animal Center of Tongji Medical College, Huazhong University of Science and Technology. These rats were randomly divided into three groups: normal control group (NS group), 6 rats; paraquat poisoning group (PQ group), 30 rats; paraquat + lipoic acid treatment group (LA group), 30 rats. After the models were established, the rats in the PQ and LA groups were subdivided into 5 groups, with 6 rats in each group. The NS group was given a same volume of physiological saline by celiac injection; the PQ group was given PQ 25 mg/kg[2,3] one time by celiac injection; and the LA group was given PQ 25 mg/kg one time by celiac injection, and 100 mg/kg medicine through the caudal vein.
Histological examination
After the rats were sacrificed, lung tissue was taken from the same lobe. After conventional dehydration, embedding, slicing, and HE staining, histological changes in the tissue were observed under a light microscope (Olympus CX31-72302).
Determination of MDA levels and SOD activity
Lung tissue was also taken from another lobe of the rats, and added with 0.4 mol/L PCA solution (Beijing Beihua Fine Chemicals Co., Ltd, Beijing). The mixture was centrifuged immediately, and the supernatant fluid was put into an EP tube. The SOD and MDA levels were determined according to the kit instructions (Nanjing Jiancheng Bioengineering Institute, China).
The whole blood (0.8 mL) without anticoagulant was drawn from the tail vein of rats for the determination of TNF-α level. The blood was centrifuged immediately, and the supernatant fluid was placed in an EP tube. The SOD and MDA levels were determined in accordance with the kit instructions (Wuhan Boshide Company, China).
Determination of SP-A/B mRNA level
The total RNA of lung tissue was collected, and RT-PCR (SLAN, Shanghai HongShi Medical Technology Co., LTD.) analysis was performed to measure the levels of SP-A and SP-B mRNA. Internal reference β-actin mRNA primer, raw: 5’-CGTTGACATCCGTAAAGACCTC-3’, downstream: 5’-TAGGAGCCAGGGCAGTAATCT-3’, product 110 bp. SP-A mRNA primer, raw: 5’-CTTCA CCCTCTTCTTGACTGTTG-3’, downstream: 5’-TCTCCC TTGTCTCCACGTCCT-3’, product 249 bp. SP-B mRNA primer, raw: 5’-TGGCTACTGCTCCTTCCTACACT-3’, downstream: 5’-GCGTCTTCCTTGGTCATCTTTG-3’, product 233 bp. Amplification conditions of PCR: 95 °C initial denaturation for 2 minutes, 95 °C denaturation for 15 seconds, annealing for 15 seconds, 72 °C denaturation for 45 seconds, cycling 40 times, extending for 10 minutes after 72 °C.
∆CT=CTtarget genes–CTβ-actin; ∆∆CT=∆CTexperimental–∆CTcontrol; amplification factor=2–∆∆CT; with 2–∆∆CT method for calculation.
Statistical analysis
The results of analysis were expressed as mean ± standard deviation. The experiment belonged to the comparison between multiple samples of single factor continuous variables, so ANOVA analysis was used with SPSS17.0 statistical software. P<0.05 was considered statistically significant.
RESULTS
HE dyeing histology
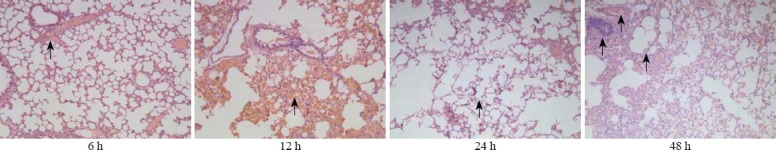
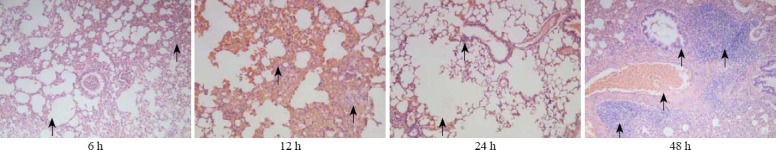
In the NS group, the alveolar structure was complete, alveolar cavity didn’t bleed, and there was no neutrophil infiltration (Figure 1). In the PQ group, lung interval damage increased with time, alveolar cavity bleeded, and edema and neutrophil infiltration increased (Figure 2). There were less changes in the LA group compared with the PQ group, and the damage of the lung tissue was mild (Figure 3).
Figure 1.
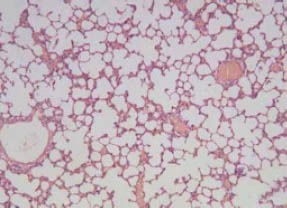
The results of HE staining in the NS group (original magnification×100).
Figure 2.
The results of HE staining in the LA group (original magnification×100).
Figure 3.
The results of HE staining in the PQ group (original magnification×100).
MDA level, SOD level, and TNF-α
There were significant differences in MDA and SOD levels between different intervals both in the intergroups and intragroups except the 3-hour group (P<0.01). Likewise, significant differences in the levels of TNF-α were also seen between the three groups and between different intervals (P<0.01) (Tables 1–3).
Table 1.
Comparison of MDA levels between the NS, PQ, and LA groups
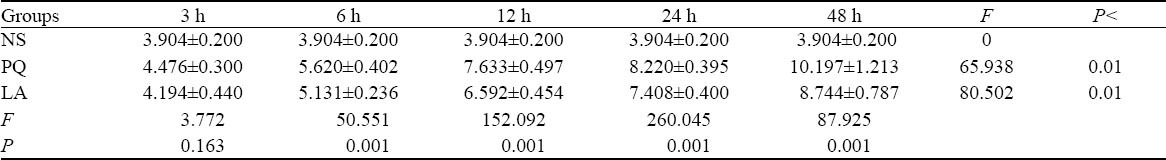
Table 2.
Comparison of SOD levels between the NS, PQ, and LA groups
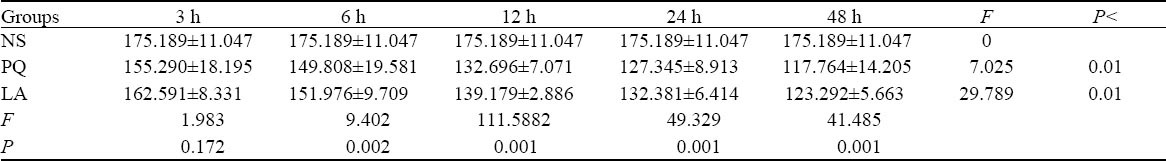
Table 3.
Comparison of TNF-α levels between the NS, PQ, and LA groups
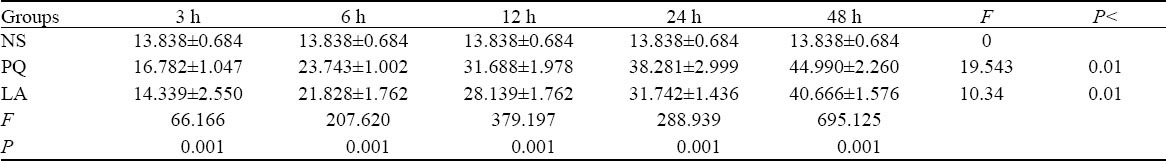
SP-A mRNA level and SP-B mRNA level
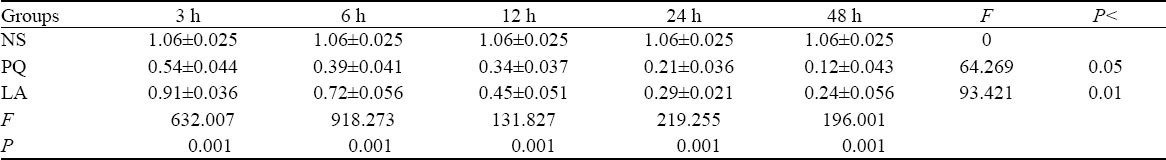
The significant differences in SP-A mRNA and SP-B mRNA amplification ratio existed between the three groups at the same intervals (P<0.01), but those differences between different intervals in the PQ group were statistically significant (P<0.05). And those differences between different intervals in the LA group were statistically significant (P<0.01) (Tables 4, 5).
Table 4.
Comparison of SP-A mRNA levels between the NS, PQ, and LA groups
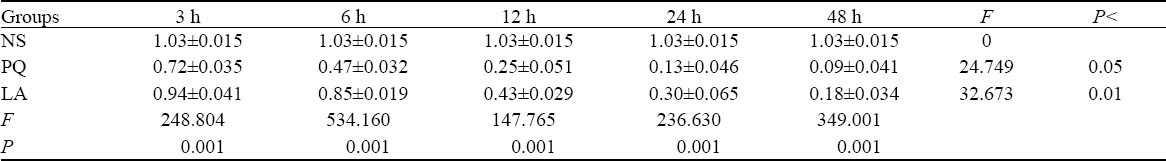
Table 5.
Comparison of SP-B mRNA levels between the NS, PQ, and LA groups
DISCUSSION
Under the normal condition, the production and removal of oxygen free radicals keep a dynamic balance, namely a certain ratio. PQ itself causes an oxidation-reduction reaction. The reaction can constantly produce ultra oxygen free radicals, which would consume more SOD. With the reduced SOD level, the ability of scavenging free radicals is reduced. Similar to this kind of cascading effect produced by hydroxyl free radicals, these radicals in PQ poisoning after lipid peroxidation could cause damage to cell membrane.[4–6] MDA quantity could reflect the degree of lipid peroxidation, and indirectly reflect the extent of the damage to cells. The determination of SOD and MDA could reflect the ability of scavenging oxygen free radicals and the anti-lipid peroxidation damage, respectively.[7]
Studies[5,6] have reported that PQ might exert its cytotoxicity through mitochondrial dysfunction. Mitochondrial dysfunction is one of the manifestations that free radicals attack the body. The unsmooth metabolism of the respiratory chain causes accumulation of MDA. Whereas MDA can cause damage to the mitochondrial respiratory chain, pyruvate dehydrogenase, α-ketoglutarate dehydrogenase, and malate dehydrogenase.
In this study, the SOD activity of the LA group at each time point was lower than that of the NS group, but it was higher than that of the PQ group. It was also shown that PQ reduced the ability of scavenging free radicals, but after adding with LA, the ability was obviously enhanced. The MDA levels in the LA group at each time point were higher than those in the NS group, but were lower than those of the PQ group. This indicated that after adding with LA, the ability of anti-lipid oxidation reaction to injury was enhanced.
LA serves as an auxiliary factor in some multienzyme systems, such as in the pyruvate dehydrogenase multienzyme complex.[8] LA could be used as a co-factor of the mitochondrial α-pyruvate dehydrogenase complex. Besides, LA could covalently bound with multi-enzyme complexes via lipid-protein ligase, and this showed that LA could promote mitochondrial respiratory chain metabolism.[9,10] The changes of MDA and the metabolism of the mitochondrial respiratory chain was closely related to each other. This study showed that the changes of MDA were related to the antioxidant of LA.
TNF-α is a pleiotropic inflammatory cytokine. It was first isolated by Carswell et al[11] in 1975 in an attempt to identify tumor necrosis factors responsible for necrosis of the sarcoma Meth A. TNF-α and IL-6 participated in the process of PQ poisoning. They could serve as a reference indicator for the severity of the disease, and as an early warning indicator of poor prognosis in patients with PQ poisoning. There was no significant correlation between the progressively increased levels of serum TNF-α, IL-6 and PQ concentration. In patients with severe PQ poisoning, the serum concentrations of TNF-α and IL-6 progressively increased, and this may cause systemic inflammatory response syndrome, and ultimately lead to multiple organ failure.[12] TNF-α is an important inflammatory cytokine in the body, and is involved in the pathological damage in certain diseases, reflecting the development of inflammation.
LA could regulate the level of TNF-α through many ways. The decreased expression of platelet endothelial cell adhesion molecule-1 played an important role in PQ-induced lung injury.[13] LA could inhibit the generation of vascular cell adhesion molecules (VCAM-1) and intercellular adhesion molecule (ICAM-1),[14,15] alleviate the inflammation and vascular permeability, and reduce the level of TNF-α. LA could effectively suppress reactive oxygen species (ROS), and play an antioxidant effect.[18] CD40 molecule is a member of the superfamily of tumor necrosis factor receptors, and could promote the secretion of TNF-α and mononuclear cell chemotaxis protein. LA could reduce the CD40 expression of monocytes in the blood, thus inhibiting the secretion of TNF-α and mononuclear cell chemotaxis protein.[17] LA could produce antioxidation by suppressing the family of ROS. The present study showed that after adding with LA, the TNF-α level was significantly decreased as compared with the PQ group (P<0.01), suggesting that LA is closely related to the process of inhibiting inflammation.
The protection mechanism of LA in the treatment of acute lung injury has been confirmed. Pulmonary surfactant includes surface active proteins (surfactant protein, SP) A, B, C, D. SP-A and SP-B are synthesized by alveolar type II cells. They reduce the alveolar and air interface surface tension, improve the stability of the lung, and at last prevent atelectasis and pulmonary edema. Changes in pulmonary surfactant structure and biological activity play an important role in acute lung injury. LA is able to reduce the generation of TNF-α induced by LPS, while TNF-α regulates the active substance of alveolar surface.[21] The increase of TNF-α could restrain the generation of active substance of the alveolar surface, reduce the synthesis of phosphatidy choline (PtdCho), and promote the degradation of active substance PtdCho of the alveolar surface.[20] LA could reduce the inhibition of TNF-α via the water channel protein-5 and the degree of pulmonary edema in addition to the improvement of pulmonary compliance. Surface active substance PtdCho was degraded increasingly, whereas alveolar surface active substance decreased. Added with LA, the TNF-α level was decreased, and thus blocking the inhibition. This increased the lung surface active substance relatively, ie the increase of SP-A, SP-B levels and the reduction of lung injury. In this study, we found after added with LA, the levels of SP-A and SP-B increased significantly as compared to the PQ group.
In this study, HE staining showed that the lung interval of rats was damaged more seriously in the PQ group than in the LA group. Moreover, their alveolar cavity bled, and edema and neutrophil infiltration increased. These findings also proved that LA effectively reduced the pathological damage of lung tissues. The TNF-α expression of the PQ group increased with time, and was obviously higher than that of the NS group. This indicated that the inflammatory reaction of the body was marked after paraquat poisoning. Added with LA, TNF-α expression was significantly decreased compared to the PQ group, indicating the improvement of inflammation. Compared to the PQ group, the SOD activity was increased and MDA reduced in the LA group. This indicated the increased abilities of LA scavenging free radicals and antioxidant.
In conclusion, in the treatment of PQ poisoning, LA is related to its resistance to oxidation and the inhibition of accumulated inflammatory factors. It could increase the active protein content of A/B in pulmonary surfactant by regulating the TNF-α level so as to protect the lung.
Footnotes
Funding: This study was supported by a grant from the National Natural Science Foundation project of China (30671783).
Ethical approval: The study was approved by the Animal Care and Use Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
Conflicts of interest: The authors declare that there is no conflict of interest.
Contributors: Li SP designed the research, analyzed the data, and wrote the paper. All authors read and approved the final version.
REFERENCES
- 1.Suntres ZE. Role of antioxidants in paraquat toxicity. Toxicology. 2002;180:65–77. doi: 10.1016/s0300-483x(02)00382-7. [DOI] [PubMed] [Google Scholar]
- 2.Dinis-Oliveira RJ, Remião F, Duarte JA, Ferreira R, Sánchez Navarro A, Bastos ML, et al. P-glycoprotein induction:an antidotal pathway for paraquat-induced lung toxicity. Free Radic Biol Med. 2006;41:1213–1224. doi: 10.1016/j.freeradbiomed.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 3.Dinis-Oliveira RJ, Duarte JA, Remião F, Sánchez-Navarro A, Bastos ML, Carvalho F. Single high dose dexamethasone treatment decreases the pathological score and increases the survival rate of paraquat-inroxicated rats. Toxicoligy. 2006;227:73–85. doi: 10.1016/j.tox.2006.07.025. [DOI] [PubMed] [Google Scholar]
- 4.Bus JS, Aust SD, Gibson JE. Superoxide and singlet oxygen catalyzed lipid peroxidation as a possible mechanism for paraquat (methyl viologen) toxicity. Biochem Biophys Res Commun. 1974;58:749–755. doi: 10.1016/s0006-291x(74)80481-x. [DOI] [PubMed] [Google Scholar]
- 5.Chen CM, Lua AC. Lung toxicity of paraquat in the rat. J Toxicol Environ Health A. 2000;60:477–487. doi: 10.1080/00984100050079548. [DOI] [PubMed] [Google Scholar]
- 6.Youngman RJ, Elstner EF. Oxygen species in paraquat toxicity: the crypto-OH radical. FEBS Letters. 1981;129:265–268. doi: 10.1016/0014-5793(81)80180-9. [DOI] [PubMed] [Google Scholar]
- 7.Meng XX, Wang RL, Gao S, Xie H, Tan JT, Qian YB. Effect of ulinastatin on paraquat-induced-oxidative stress in human type II alveolar epithelial cells. World J Emerg Med. 2013;4:133–137. doi: 10.5847/wjem.j.issn.1920-8642.2013.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sommer D, Fakata KL, Swanson SA, Stemmer PM. Modulation of the phosphatase activity of calcineurin by oxidants and antioxidantsin vitro. Eur J Biochem. 2000;267:2312–2322. doi: 10.1046/j.1432-1327.2000.01240.x. [DOI] [PubMed] [Google Scholar]
- 9.Yi X, Maeda N. Endogenous production of lipoic acid is essential for mouse development. Mol Cell Biol. 2005;25:8387–8392. doi: 10.1128/MCB.25.18.8387-8392.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim DJ, Kim KH, Lee HH, Lee SJ, Ha JY, Yoon HJ, et al. Crystal structure of lipoate-protein ligase A bound with the activated intermediate: insights into interaction with lipoyl domains. Biol Chem. 2005;280:38081–38089. doi: 10.1074/jbc.M507284200. [DOI] [PubMed] [Google Scholar]
- 11.Carswell EA, Old LJ, Kassel RL, Green S, Fiore N, Williamson B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc Natl Acad Sci U S A. 1975;72:3666–3670. doi: 10.1073/pnas.72.9.3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cheng ZQ, Han JY, Sun P, Weng YY, Chen J, Wu GY, et al. Edaravone attenuates paraquat-induced lung injury by inhibiting oxidative stress in human type II alveolar epithelial cells. World J Emerg Med. 2012;3:55–59. doi: 10.5847/wjem.j.issn.1920-8642.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Graczyk M, Przybyszewski M, Kuźmiński A, Tlappa J, Mućka J, Napiórkowska K, et al. Role of E-selectin and platelet endothelial cell adhesion molecule 1 in gastritis in food allergy patients. Postepy Dermatol Alergol. 2013;30:271–276. doi: 10.5114/pdia.2013.38354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chaudhary P, Marracci GH, Bourdette DN. Lipoic acid inhibits expression of ICAM-1 and VCAM-1 by CNS endothelial cells and T cell migration into the spinal cord in experimental autoimmune encephalomyelitis. J Neuroimmunol. 2006;175:87–96. doi: 10.1016/j.jneuroim.2006.03.007. Epub 2006 Apr 27. [DOI] [PubMed] [Google Scholar]
- 15.Kunt T, Forst T, Wilhelm A, Tritschler H, Pfuetzner A, Harzer O, et al. Alpha-lipoic acid reduces expression of vascular cell adhesion molecule-1 and endothelial adhesion of human monocytes after stimulation with advanced glycation end products. Clin Sci (Lond) 1999;96:75–82. [PubMed] [Google Scholar]
- 16.Zhang WJ, Wei H, Hagen T, Frei B. Alpha-lipoic acid attenuates LPS-induced inflammatory responses by activating the phosphoinositide 3-kinase/Akt signaling pathway. Proc Natl Acad Sci USA. 2007;104:4077–4082. doi: 10.1073/pnas.0700305104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Salinthone S, Schillace RV, Marracci GH, Bourdette DN, Carr DW. Lipoic acid stimulates cAMP production via the EP2 and EP4 prostanoid receptors and inhibits IFN gamma synthesis and cellular cytotoxicity in NK cells. Neuroimmunol. 2008;199:46–55. doi: 10.1016/j.jneuroim.2008.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wollin SD, Jones PJ. Alpha-lipoic acid and cardiovascular disease. Nutr. 2003;133:3327–3330. doi: 10.1093/jn/133.11.3327. [DOI] [PubMed] [Google Scholar]
- 19.Pearson FG. Liaoning Education Press; 1999. Zhao Fengrui [translation] Thoracic Surgery; p. 42. [Google Scholar]
- 20.Shi J, Hu CL, Gao YF, Liao XX, Xu H. The relationship between platelet endothelial cell adhesion molecule-1 and paraquat-induced lung injury in rabbits. World J Emerg Med. 2012;3:60–64. doi: 10.5847/wjem.j.issn.1920-8642.2012.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xie LX, Liu YN, Zhao XW, Chen LA, Zhao XM, Liu YY. The changes in effective local blood perfusion and compensatory ventilation in different lung areas of acute respiratory distress syndrome rabbits model. Zhonghua Nei Ke Za Zhi. 2004;43:522–526. [PubMed] [Google Scholar]


