Abstract
Purpose
To identify trends in embryo catheter loading and embryo culture techniques performed worldwide.
Methods
A retrospective evaluation using the results of a web-based survey, (IVFWorldwide (www.IVF-worldwide.com), was performed.
Results
Responses from 265 centers in 71 countries were obtained. Most centers (97 %) prefered a catheter with its orifice on top, with only 3 % preferring a catheter with the orifice on its side; 41 % preferred a catheter marked for clear ultrasound view. The most commonly-reported methods of embryo loading were medium-air-embryo-airmedium (42 %), medium in catheter with embryo at end (20 %) and medium-air-embryo (15 %). In 68 % of centers the final volume of the catheter was up to 0.3 ml, with only 19 % using 0.3-0.5 ml and 1 % using 0.5-0.7 ml. Using reduced oxygen concentrations for embryo culture was divided between those who used it in combination with the twogas system (34 %) and those who did not use it at all (39 %); 24 % reported using a three-gas system. Most clinics using reduced oxygen concentrations used it throughout the entire culture period. Half of centers (51 %) reported using reduced oxygen concentrations for the entire IVF population while 6 % reserved it only for blastocyst transfer. The use of sequential media was highly dominant with 40 % reporting its use.
Keywords: Embryo culture, Embryo transfer, In vitro fertilization, Infertility, Assisted reproductive techniques
Introduction
Over the past three decades, major advances in assisted reproductive technologies (ART) have benefited couples worldwide, with over 4 million babies born as a result [1]. Despite these technological advances, in vitro fertilization (IVF) remains a highly complex process with fertility centers internationally all having the same common goal to achieve a live birth. While many aspects of IVF have been tested via evidence-based research, other facets have been less investigated with much variation in practice. Two areas of focus in maximizing IVF success lie in the areas of embryo transfer loading and embryo culture techniques.
Embryo catheter loading is a critical step in the embryo transfer process. While research has evaluated areas such as ultrasound guidance, uterine cavity blood, mock transfer and myometrial contractions, less is known about the benefits of specific embryo catheter loading techniques [2–6]. Embryo culture conditions, particularly oxygen tension parameters, have been a major focus of research. The two most common incubator systems utilize, either, carbon dioxide and air or carbon dioxide, air and nitrogen. In a three gas system, nitrogen is used to displace oxygen to allow a low oxygen tension of 5 %. Low oxygen tension has been of interest due to the potential benefits of protecting an embryo from the harmful effects of reactive oxygen species (ROS) [7, 8]. Additionally, it is hypothesized that induction of gene expression by hypoxia may result in cellular mechanisms that improve embryonic development [9–11]. During the 1990’s the concept of low oxygen tension embryo incubation was reexamined when culturing embryos to the blastocyst stage was introduced. Advances in incubator technology such as managing the incubator environment through triple gas incubation and the introduction of sequential media also contributed to the culture of human embryos under low oxygen tension [12, 13]. Sequential media involves a combination of two or three different media designed to meet the nutritional needs of the embryo as it advances in time, while nonsequential media is designed to meet an embryo’s nutritional needs at all stages [14].
IVF-Worldwide (www.IVF-Worldwide.com) is a comprehensive IVF-focused website for physicians, embryologists, nurses and social workers. The website allows members to locate IVF centers anywhere in the world to communicate directly, facilitating the sharing of ideas and discussion of treatments and medications. The non-commercial, non-profit website with an advisory panel of 52 leaders in the fertility field routinely performs surveys focusing on various aspects of ART.
The purpose of this web-based survey was to identify trends in embryo catheter loading and embryo culture techniques performed worldwide and correlate these results with current evidence-based literature. This information may prove useful for programs struggling with poor outcomes or undecided on a particular practice related to embryo transfer or culture techniques.
Materials and methods
A 19-item survey entitled “Embryo Culture and Catheter Loading” was developed by two embryologists and an IVF lab director and approved by the IVF-Worldwide.com advisory board. The questionnaire was composed of two sections: one section focusing on embryo catheter loading and transfer methods (12 questions) and a second section focusing on embryo culture techniques (7 questions). Catheter loading questions addressed: preferred catheter, who loads the catheter, embryo transfer media, catheter washing prior to loading, method for loading embryos into catheter, number of drops used when transferring more than two embryos, and the final volume of medium in the catheter. Regarding the transfer, topics included time the embryos are in catheter from loading to transfer, rate at which embryos are injected, whether catheter is checked after transfer for embryos, and how the catheter is checked. Five of the seven embryo culture technique questions focused specifically on utilization of reduced (5 %) oxygen systems: whether a reduced oxygen system is used, stages of embryo development it is used for, type of media used with reduced oxygen, whether reduced oxygen is used for specific populations and whether reduced oxygen is used for a patient’s entire cohort of embryos or if they are divided between two culture systems. The two final questions addressed blastocyst culture: whether a single media is used and if they are cultured in groups or individually.
The survey was posted on the IVF-Worldwide website from February 19, 2011 to March 19, 2011. In addition to the questions described above, the survey also gathered demographic questions including the name of the IVF unit and its medical director, e-mail address, country and number of IVF cycles performed by the unit in the past year. This study was determined to be exempt from institutional review board approval by Johns Hopkins University School of Medicine.
Quality assurance methods
To complete the survey, the respondent’s name, IVF center name and country, e-mail address, and estimated total number of IVF cycles performed annually was collected. This demographic information was used to verify that only one IVF provider per center completed the survey. In order to minimize duplicate reports from a unit and possible false data, a computerized software program assessed the consistency of the four parameters in the self-reported data of the unit surveyed with existing data of units registered on the IVF-worldwide website. If at least three of these parameters from the survey matched the website archive data, the reporting site’s data was included in the statistical analyses. Discrimination between ‘professional’ and ‘non-professional’ entries was not possible.
Statistical analysis
Statistical analysis was based on the number of IVF cycles reported by the unit and not by the number of units responding to the survey. For each question, the survey provided multiple choices from which only a single answer could be chosen (‘radio buttons’). For example, for a question with four answers (a, b, c, d), the following results were calculated:
% ‘a’ = Ʃ Number of cycles of units who answered ‘a’ × 100.
Ʃ Number of cycles of all the units.
% ‘b’ = Ʃ Number of cycles of units who answered ‘b’ × 100.
Ʃ Number of cycles of all the units.
% ‘c’ = Ʃ Number of cycles of units who answered ‘c’ × 100.
Ʃ Number of cycles of all the units.
% ‘d’ = Ʃ Number of cycles of units who answered ‘d’ × 100.
Ʃ Number of cycles of all the units.
After approximately 4 weeks, data corresponding to 161,300 cycles/year was received and this sample size was considered sufficient. Proportions were compared using the chi-squared test with significance defined as P < 0.05 using the Stata 12 (College Station, TX) statistical software package.
Results
During the time period the survey was open, 265 units from 71 countries responded to the survey and passed the computerized system’s quality assurance test. In total, this report analyzed 161,300 IVF cycles. Distribution by continent is as follows: Europe, 37.1 % (59, 700 cycles); North America 29.0 % (46, 700 cycles); Asia 19.5 % (31, 400 cycles); Africa 5.5 % (8,900 cycles); Australia 5.3 % (8,700 cycles) and South America 3.6 % (5,900 cycles). Table 1 summarizes distribution by country.
Table 1.
Survey responders by country, number of centers and number of IVF cycles per year
| Country (n = 54) | Number of centers | Number of cycles/year |
|---|---|---|
| Argentina | 3 | 800 |
| Australia | 10 | 7,300 |
| Austria | 2 | 800 |
| Belgium | 1 | 900 |
| Brazil | 6 | 1,300 |
| Canada | 9 | 3,800 |
| Colombia | 1 | 400 |
| Croatia | 1 | 400 |
| Czech Republic | 3 | 4,500 |
| Egypt | 5 | 5,600 |
| France | 3 | 3,200 |
| Germany | 7 | 5,300 |
| Ghana | 1 | 200 |
| Greece | 8 | 4,900 |
| Honduras | 1 | 100 |
| Hong Kong | 1 | 500 |
| Hungary | 5 | 2,700 |
| India | 14 | 5,300 |
| Indonesia | 1 | 200 |
| Iran | 3 | 1,400 |
| Israel | 5 | 8,600 |
| Italy | 7 | 4,400 |
| Jordan | 1 | 900 |
| Kazakhstan | 1 | 200 |
| Kosova | 1 | 200 |
| Lebanon | 1 | 500 |
| Macedonia | 1 | 400 |
| Malaysia | 1 | 200 |
| Mexico | 2 | 500 |
| Montenegro | 1 | 300 |
| Netherlands | 6 | 9,700 |
| New Zealand | 1 | 1,400 |
| Norway | 1 | 600 |
| Peru | 1 | 1,600 |
| Poland | 1 | 900 |
| Portugal | 1 | 300 |
| Romania | 1 | 100 |
| Russia | 3 | 1,800 |
| Saudi Arabia | 2 | 1,600 |
| Serbia | 2 | 700 |
| South Africa | 2 | 900 |
| South Korea | 1 | 500 |
| Spain | 13 | 5,800 |
| Sri Lanka | 1 | 200 |
| Sweden | 4 | 4,400 |
| Switzerland | 1 | 200 |
| Taiwan | 3 | 300 |
| Tunisia | 3 | 2,200 |
| Turkey | 7 | 6,800 |
| United Arab Emirates | 3 | 2,600 |
| United Kingdom | 13 | 7,000 |
| United States | 85 | 42,900 |
| Venezuela | 3 | 1,200 |
| Vietnam | 1 | 1,800 |
| Grand Total | 265 | 161,300 |
Embryo catheter-loading and techniques
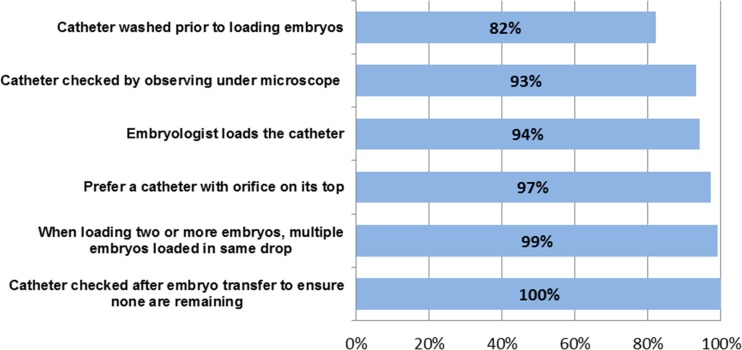
There were several common practices in the area of embryo catheter-loading and transfer techniques that the vast majority of centers employed (Fig. 1). For instance, the embryologist loads the embryo transfer catheter in 94 % of practices. In a small number of centers, it is the physician (2 %) or both (4 %) that load the catheter. Almost all units (97 %) prefer a catheter with the orifice on its top, with only 3 % of units preferring a catheter with the orifice on its side. Almost half of centers, 41 %, prefer a catheter marked for clear ultrasound view. The majority of units, 82 %, wash the catheter prior to loading the embryos. Regarding the transfer media, 42 % of units use a different medium depending on embryo stage while 32 % of units use a blastocyst medium in all cases, 9 % use a HEPES buffer and 17 % use a different medium.
Fig. 1.
Common catheter loading practices among 80 % or more of respondents
There is a large variety of methods in which embryos are loaded into the catheter. The most commonly-reported method was medium-air-embryo-air-medium (42 %), followed by medium in catheter with embryo at end (20 %), medium-air-embryo (15 %), other (16 %) and medium with embryo with no air in between (7 %). In most centers (68 %), the final volume of the catheter is up to 0.3 ml, with only 19 % using 0.3-0.5 ml and 1 % using 0.5-0.7 ml. When transferring two or more embryos, almost all (99 %) units load more than one embryo in the same drop.
Once embryos are loaded into the catheter, the time embryos remain in the catheter does not exceed 60 s in most cases. 49 % of centers reported embryos were in the catheter for 30–60 s, for 41 % they remain in the catheter 15–30 s and 9 % report the embryos stay in the catheter more than 60 s. During the actual transfer, the majority of centers (61 %) inject embryo (s) at a very slow rate while 28 % report injecting embryos at a very fast rate to avoid embryos attaching to the wall of the catheter and 11 % reporting a special volume is not needed. Every center, 100 %, reported checking the catheter after the transfer to make sure there is not a remaining embryo. 93 % describe examining the catheter under a microscope and then washing the catheter while 7 % only observe under the microscope. For the centers that wash the catheter, 42 % report disconnecting the syringe, aspirating air, reconnecting the syringe followed by aspirating media and then expelling contents of the catheter. The remaining centers simply aspirate media and then expel (33 %) or use another method (24 %).
Embryo culture techniques
Several questions focused on reduced oxygen concentration (5 %) in the embryo culture system. Table 2 summarizes the number of cycles/year utilizing various oxygen concentrations and gas systems. The practice of using reduced oxygen concentrations was almost equally divided between those who use it in combination with the two-gas system (34 %) and those who do not use it at all (39 %). 24 % of respondents use a three-gas system. Most clinics using reduced oxygen concentrations use it throughout the entire culture period with 42 % of units reporting using it for days 1-5/6 followed by day 5 or 6 transfers. A smaller amount of units report using it days 1–3 followed by day 2 or 3 transfers (8 %), days 3–5 followed by day 5 or 6 transfers (6 %) and days 1–3 followed by day 5 or 6 transfers (2 %).
Table 2.
In your IVF laboratory, is reduced (5 %) oxygen concentration used in your embryo culture system? (Number of cycles/year)
| Only a two-gas system (6 % CO2 in air) is used N (%) | Both three- (6 % CO2, 5 % O2, 89 % N2) and two-gas systems are used N (%) | Only three-gas system is used N (%) | Other N (%) | Total N | P-value | |
|---|---|---|---|---|---|---|
| United States and Canada | 14,600 (31.26) | 20,800 (44.54) | 11,200 (23.98) | 100 (0.21) | 46,700 | <0.001 |
| South America and Mexico | 3,500 (59.32) | 1,800 (30.51) | 300 (30.51) | 300 (5.08) | 5,900 | |
| Europe | 28,300 (48.88) | 17,500 (27.13) | 15,800 (24.50) | 2,900 (4.50) | 64,500 | |
| Africa | 2,700 (30.34) | 900 (10.11) | 5,300 (59.55) | 0 | 8,900 | |
| Asia | 12,900 (48.50) | 11,100 (41.73) | 1,200 (4.51) | 1,400 (5.26) | 26,600 | |
| Australia | 600 (6.90) | 3,300 (37.93) | 4,800 (55.17) | 0 | 8,700 |
Table 3 summarizes special populations or situations in which reduced oxygen concentrations is used in cycles/year. The majority of units (51 %) report using reduced oxygen concentrations for the entire IVF population while 6 % of units reserve it only for patients intended for blastocyst transfer (the remainder reported not using reduced oxygen). Regarding media used, the use of sequential media is highly dominant with 40 % of units reporting its use. Less often utilized are one-step media (12 %) and both types of media (6 %). When culturing under conditions of reduced oxygen concentration, most units who use it report using the system for the entire cohort of patients chosen (51 %). Usually, no attempt is made to divide the embryos between the two culture systems with only 3 % reporting that the embryos are equally divided between the two oxygen concentrations (remaining centers reported not using reduced oxygen systems).
Table 3.
Reported patient populations that reduced oxygen concentration is used for: (Number of cycles/year)
| The entire IVF population N (%) | Poor responder patients N (%) | Patients with repeated implantation failures N (%) | Patients intended for blastocyst transfer N (%) | Not used at all N (%) | Total Cycles N | P-value | |
|---|---|---|---|---|---|---|---|
| USA & Canada | 30,100 (64.45) | 300 (0.64) | 300 (0.64) | 2,200 (4.71) | 13,800 (29.55) | 46,700 | <0.001 |
| South America | 1,900 (32.20) | 0 | 0 | 200 (3.39) | 3,800 (64.41) | 5,900 | |
| Europe | 28,300 (43.88) | 200 (0.31) | 0 | 3,200 (4.96) | 32,800 (50.85) | 64,500 | |
| Africa | 3,100 (34.83) | 0 | 0 | 3,000 (33.71) | 2,800 (31.46) | 8,900 | |
| Asia | 9,000 (33.83) | 0 | 500 (1.88) | 1,300 (4.89) | 15,800 (59.40) | 26,600 | |
| Australia | 8,100 (93.10) | 0 | 0 | 0 | 600 (6.90) | 8,700 |
For those units not using sequential media (58 %), units using single media were asked how often they change media during blastocyst culture. Most units using single media change media on day 3 (21 %) with less changing media on days 1, 3 and 5 (10 %) and changing media on day 3 and 5 (5 %). Finally, units were asked when growing embryos to the blastocyst stage, do they grow them in groups or singly. The results were almost evenly divided with 39 % of units growing embryos in individual drops days 0-5/6 and 35 % of units growing them in groups from days 0-5/6. 5 % of units reported growing embryos singly from day 0–3 and then in groups for the duration of culture, 11 % reported other practices and 10 % reported not growing embryos to blastocyst stage.
Discussion
To our knowledge, this is the largest study to date addressing specifically embryo catheter loading techniques and embryo culture media conditions among an international group of IVF centers. Our survey has identified many common practices among IVF centers worldwide in the area of embryo catheter loading and embryo culture as well as many practices with significant variation.
Regarding embryo catheter loading and transfer, several factors stood out as practiced by the majority of IVF programs. These included having the embryologist load the catheter, using a catheter with an orifice on top and loading more than one embryo in the same drop. However, the international nature of our study reveals differences that may be specific to the United States. Van Voorhis et al. (2010) recently reported on common practices of high-performing IVF centers in the United States, and their results show differences compared to our worldwide survey results. For example, they found that 80 % of programs load air bubbles into the catheter along with embryos to aid in visualization during transfer and to prevent embryo migration in the fluid column. This is slightly increased from our finding that 57 % of centers utilize air bubbles in the embryo catheter. Additionally, their study noted that 50 % of centers report expelling embryos at a moderate rate, 20 % expel quickly and 30 % report expelling the embryos slowly [15]. This is a bit different from our results in which the majority of centers (61 %) inject embryo (s) at a very slow rate while 28 % report injecting embryos at a very fast rate. While our survey addressed preferred catheters with respect to the location of the orifice or whether the catheter was marked for ultrasound view, we did not explore more specific preferences such as hard versus soft catheters. This is of clinical interest as two separate meta-analyses/systematic reviews show a significantly higher pregnancy rate with soft versus firm catheters [16, 17].
Our survey results also showed a wide variation in embryo transfer media used. This is consistent with lack of evidence of a superior transfer media. Protein concentration and viscosity has not been demonstrated to impact pregnancy outcomes, whereas the benefits of fibrin sealant and hyaluronanic acid in the transfer media is still unclear [18–21]. A recent Cochrane Library review examined adherence compounds in embryo transfer media and found no evidence that fibrin sealant increased pregnancy rates. For hyaluronic acid, evidence of a positive treatment effect was identified in six trials that reported live birth rates (odds ratio (OR) 1.41, 95 % CI 1.17 to 1.69) and in 14 trials reporting clinical pregnancy rates (OR 1.39, 95 % CI 1.21 to 1.60) [22]. Finally, 90 % of centers reported the embryos were in the catheter from loading to transfer for 15–60 s. This is supported by one study that demonstrated longer time intervals result in lower pregnancy rates with an interval of more than 120 s carrying a poor prognosis [23].
One aspect of embryo catheter loading that can be correlated to evidence based research is the presence of air bubbles in the catheter. Our study did reveal that over half of programs load air bubbles in the embryo catheter. Supporters of this practice argue that the air bubbles can protect an embryo from trauma prior to entering the endometrial cavity. Others, however, believe that the introduction of a small amount of air into the uterine cavity could have a detrimental effect on implantation rates. Ebner et al. (2001) demonstrated that the presence of air bubbles and extremely low volumes of culture media in catheter (<10 μL) was associated with lower pregnancy rates [24]. Our study did not inquire about culture media volumes that low to assess if this was a widespread practice. Likewise, one study showed that transfer volumes of more than 60 μL may result in failed transfer with expulsion of embryos into the vagina [25]. Only 1 % of survey respondents reported using volumes in this range. A systematic review by Abou-Setta et al. (2007) identified two randomized control trials comparing fluid-only to air-fluid filled catheters and found no significant differences in pregnancy rates between the two methods [26]. Our survey did not address the location of the air bubbles with respect to the uterine fundus during ultrasound-guided embryo transfer, but there is evidence that pregnancy rates are higher when the bubble visualized is less than 10 mm from the uterine fundus [27, 28].
While our study showed consistent practices among many embryo catheter loading techniques, there was much variability when it came to embryo culture practices. The use of low oxygen tension systems varied widely among the respondents, demonstrating this remains an area of controversy among IVF providers. Our study demonstrated that 34 % of respondents use both a two-gas system combined with a low oxygen tension system during a cycle while 24 % use only a low-oxygen tension system. Only a two-gas system is used by 39 %. These mixed results seem consistent with the current evidence regarding the benefit of low-oxygen systems and the fact that there is currently not a consensus regarding which oxygen concentration provides the best success rates of IVF procedures. However, it appears that the low oxygen environment may demonstrate over time to be the most beneficial system with regards to live birth rates, especially for blastocyst culture. This correlates with our survey results in that 42 % of units report using reduced oxygen culture for days 1-5/6 followed by day 5 or 6 transfers. Kirkegaard et al. (2013) demonstrated using time-lapse monitoring that culture in 20 % oxygen results in a delay in the third cleavage cycle for embryos and overall development rate compared to embryos cultured in low (5 %) oxygen [29].
Gomes Sobrhino et al. (2012) conducted a meta-analysis of studies evaluating the effects of a low-oxygen environment on IVF/ICSI outcomes. Their results showed that for all cycles of the 7 randomized control trials (RCTs) identified, the implantation and ongoing pregnancy rates were not different between embryos cultures a low oxygen environment and those cultured in atmospheric oxygen levels. When looking at embryos transferred on day 2/3, implantation and ongoing pregnancy rates were not different between the groups. However, when embryos were transferred on days 5/6, groups with transferred sets of embryos cultured in low-oxygen conditions had significantly higher implantation rates than those cultured in the conventional system. Ongoing pregnancy rates were not different between the two groups [30]. Potential benefits of low oxygen systems have also been demonstrated by a recent Cochrane Library systematic review and meta-analysis. The systematic review included 7 studies with a total of 2,422 participants while meta-analysis could be performed on four included studies with a total of 1,382 participants. A beneficial effect of low oxygen concentration culture was found for live birth rate (OR 1.39; 95 % CI 1.11 to 1.76). The authors used the example that a typical clinic could increase a 30 % live birth rate using atmospheric oxygen concentration to as high as 32 % to 43 % using a low oxygen concentration. The results were similar for ongoing and clinical pregnancy rates. Culturing embryos under low oxygen concentrations did not demonstrate an increase in adverse events such as multiple pregnancies, spontaneous abortion or congenital abnormalities [31].
It is also very important to mention the new trend that is increasing worldwide, the use of time lapse systems in IVF labs. While this modality was not addressed in our survey, there is accumulating evidence that shows a significant increase in the success rate when time lapse systems are used and in the future it may play a larger role in the IVF lab during embryo culture [32, 33]. The main advantage of this system is that it is not necessary to remove the embryos from the incubator for observation or media change and they are thus kept in constant conditions throughout their development. The use of “single step” media has made it possible to grow the embryos singly up to the blastocyst stage in a small chamber that keeps the conditions stable with a triple gas mixture. The clinical benefits of this technology are still under active investigation. A prospective study by Kirkegaard et al. (2013) showed that using time lapse monitoring development to high-quality blastocysts could be predicted within the first 48 h of culture but the time-lapse parameters could not predict pregnancy [34].
Strengths of this study include the comprehensive nature of the survey focusing on two important areas: embryo catheter loading and techniques and embryo culture. An additional strength is the sheer number of IVF centers and cycles represented, as well as the great diversity in the number of countries represented. Our study exhibits weaknesses as well. For example, there may be a selection bias in that units that chose to participate in a worldwide survey may have different practice techniques than other centers worldwide that chose not to participate. Additionally, questions on aspects of embryo transfer such as embryo placement in the uterus and use of mock embryo transfers would have elucidated additional insight into IVF practices worldwide.
In conclusion, contemporary IVF is performed worldwide via various methodologies but with comparable outcomes. High success rates require a close cooperation between the clinician and the laboratory. This survey confirms that while some embryo transfer and embryo culture techniques are widely established among centers internationally, other methodologies are utilized that achieve the same desired endpoint of high live birth rates for patients. Perhaps our most salient finding is the great variability and lack of agreement regarding many aspects of these areas. Practices common to most programs likely play a major role in contributing to clinical success, while those practices with a variety of differences may not be as important in the overall success of an IVF program. This reflects the fact that IVF is a multifaceted medical treatment with numerous factors playing a role in achieving the ultimate outcome of a successful pregnancy.
Footnotes
Capsule A worldwide survey of fertility centers demonstrates many common trends in embryo catheter loading and embryo culture techniques as well as several areas of variation.
References
- 1.Rosenbluth EM, Van Voorhis BJ. Evolving role of assisted reproductive technologies. Clin Obstet Gynecol. 2011;54:734–45. doi: 10.1097/GRF.0b013e318236021d. [DOI] [PubMed] [Google Scholar]
- 2.Goudas VT, Hammitt DG, Damario MA, Session DR, Singh AP, Dumesic DA. Blood on the embryo transfer catheter is associated with decreased rates of embryo implantation and clinical pregnancy with the use of in vitro fertilization-embryo transfer. Fertil. Steril. 1998;70:878–82. doi: 10.1016/S0015-0282(98)00315-X. [DOI] [PubMed] [Google Scholar]
- 3.Lesny P, Killick SR, Tetlow RL, Robinson J, Maguiness SD. Embryo transfer–can we learn anything new from the observation of junctional zone contractions? Hum. Reprod. 1998;13:1540–6. doi: 10.1093/humrep/13.6.1540. [DOI] [PubMed] [Google Scholar]
- 4.Buckett WM. A meta-analysis of ultrasound-guided versus clinical touch embryo transfer. Fertil. Steril. 2003;80:1037–41. doi: 10.1016/S0015-0282(03)01015-X. [DOI] [PubMed] [Google Scholar]
- 5.Mirkin S, Jones EL, Mayer JF, Stadtmauer L, Gibbons WE, Oehninger S. Impact of transabdominal ultrasound guidance on performance and outcome of transcervical uterine embryo transfer. J. Assist. Reprod. Genet. 2003;20:318–22. doi: 10.1023/A:1024809607966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schoolcraft WB, Surrey ES, Gardner DK. Embryo transfer: techniques and variables affecting success. Fertil. Steril. 2001;76:863–70. doi: 10.1016/S0015-0282(01)02731-5. [DOI] [PubMed] [Google Scholar]
- 7.Guerin P, El Mouatassim S, Menezo Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum. Reprod. Update. 2001;7:175–89. doi: 10.1093/humupd/7.2.175. [DOI] [PubMed] [Google Scholar]
- 8.Catt JW, Henman M. Toxic effects of oxygen on human embryo development. Hum. Reprod. 2000;15(Suppl 2):199–206. doi: 10.1093/humrep/15.suppl_2.199. [DOI] [PubMed] [Google Scholar]
- 9.Katz-Jaffe MG, Linck DW, Schoolcraft WB, Gardner DK. A proteomic analysis of mammalian preimplantation embryonic development. Reproduction. 2005;130:899–905. doi: 10.1530/rep.1.00854. [DOI] [PubMed] [Google Scholar]
- 10.Harvey AJ, Kind KL, Pantaleon M, Armstrong DT, Thompson JG. Oxygen-regulated gene expression in bovine blastocysts. Biol. Reprod. 2004;71:1108–19. doi: 10.1095/biolreprod.104.028639. [DOI] [PubMed] [Google Scholar]
- 11.Kind KL, Collett RA, Harvey AJ, Thompson JG. Oxygen-regulated expression of GLUT-1, GLUT-3, and VEGF in the mouse blastocyst. Mol. Reprod. Dev. 2005;70:37–44. doi: 10.1002/mrd.20183. [DOI] [PubMed] [Google Scholar]
- 12.Biggers JD. Thoughts on embryo culture conditions. Reprod Biomed Online. 2002;4(Suppl 1):30–8. doi: 10.1016/S1472-6483(12)60009-1. [DOI] [PubMed] [Google Scholar]
- 13.Summers MC, Biggers JD. Chemically defined media and the culture of mammalian preimplantation embryos: historical perspective and current issues. Hum. Reprod. Update. 2003;9:557–82. doi: 10.1093/humupd/dmg039. [DOI] [PubMed] [Google Scholar]
- 14.Pool TB. Development of culture media for human assisted reproductive technology. Fertil. Steril. 2004;81:287–9. doi: 10.1016/j.fertnstert.2003.10.012. [DOI] [PubMed] [Google Scholar]
- 15.Van Voorhis BJ, Thomas M, Surrey ES, Sparks A. What do consistently high-performing in vitro fertilization programs in the U.S. do? Fertil. Steril. 2010;94:1346–9. doi: 10.1016/j.fertnstert.2010.06.048. [DOI] [PubMed] [Google Scholar]
- 16.Abou-Setta AM, Al-Inany HG, Mansour RT, Serour GI, Aboulghar MA. Soft versus firm embryo transfer catheters for assisted reproduction: a systematic review and meta-analysis. Hum. Reprod. 2005;20:3114–3121. doi: 10.1093/humrep/dei198. [DOI] [PubMed] [Google Scholar]
- 17.Buckett W. A review and meta-analysis of prospective trials comparing different catheters used for embryo transfer. Fertil. Steril. 2006;85:728–734. doi: 10.1016/j.fertnstert.2005.08.031. [DOI] [PubMed] [Google Scholar]
- 18.Friedler S, Schacter M, Strassburger D, Esther K, El Ron R, Raziel A. A randomized clinical trial comparing recombinant hyaluronan/recombinant albumin versus human tubal fluid for cleavage stage embryo transfer in patients with multiple IVF-embryo transfer failure. Hum. Reprod. 2007;22:2444–2448. doi: 10.1093/humrep/dem220. [DOI] [PubMed] [Google Scholar]
- 19.Khan I. C. Staessen, P. Devroey, A.C. Van Steirteghem. Human serum albumin versus serum: a comparative study on embryo transfer medium. Fertil. Steril. 1991;56:98–101. [PubMed] [Google Scholar]
- 20.Menezo Y, Arnal F, Humeau C, Ducret L, Nicollet B. Increased viscosity in transfer medium does not improve the pregnancy rates after embryo transfer. Fertil. Steril. 1989;52:680–682. doi: 10.1016/s0015-0282(16)60987-1. [DOI] [PubMed] [Google Scholar]
- 21.Karimian L, Rezazadeh VM, Baghestani AR, Moeini A. A prospective randomized comparison of two commercial embryo transfer medium in IVF/ICSI cycles. Hum. Reprod. 2004;19(Suppl 1):i52. [Google Scholar]
- 22.Bontekoe S, Heineman MJ, Johnson N, Blake D. Adherence compounds in embryo transfer media for assisted reproductive technologies. Cochrane Database Syst. Rev. 2014;2 doi: 10.1002/14651858.CD007421.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matorras R, Mendosa R, Exposito A, Rodriguez-Escudero FJ. Influence of the time interval between embryo catheter loading and discharging on the success of IVF. Hum. Reprod. 2004;19:2027–2030. doi: 10.1093/humrep/deh366. [DOI] [PubMed] [Google Scholar]
- 24.Ebner T, Yaman C, Moser M, Sommergruber M, Polz W, Tews G. The ineffective loading process of the embryo transfer catheter alters implantation and pregnancy rates. Fertil. Steril. 2001;76:630–2. doi: 10.1016/S0015-0282(01)01980-X. [DOI] [PubMed] [Google Scholar]
- 25.Poindexter AN, Thompson DJ, Gibbons WE, Findley WE, Dodson MG, Young RL. Residual embryos in failed embryo transfer. Fertil. Steril. 1986;46:262–267. [PubMed] [Google Scholar]
- 26.Abou-Setta AM. Air fluid versus fluid-only models of embryo catheter loading: a systematic review and meta-analysis. Reprod Biomed Online. 2007;14:80–4. doi: 10.1016/S1472-6483(10)60767-5. [DOI] [PubMed] [Google Scholar]
- 27.Cenksoy PO, Fıcıcıoglu C, Yesiladali M, Akcin OA, Kaspar C. The importance of the length of uterine cavity, the position of the tip of the inner catheter and the distance between the fundal endometrial surface and the air bubbles as determinants of the pregnancy rate in IVF cycles. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014;172:46–50. doi: 10.1016/j.ejogrb.2013.09.023. [DOI] [PubMed] [Google Scholar]
- 28.Friedman BE, Lathi RB, Henne MB, Fisher SL, Milki AA. The effect of air bubble position after blastocyst transfer on pregnancy rates in IVF cycles. Fertil. Steril. 2011;95(3):944–7. doi: 10.1016/j.fertnstert.2010.07.1063. [DOI] [PubMed] [Google Scholar]
- 29.Kirkegaard K, Hindkjaer JJ, Ingerslev HJ. Effect of oxygen concentration on human embryo development evaluated by time-lapse monitoring. Fertil Steril. 2013 Mar 1;99 (3):738–744.e4. [DOI] [PubMed]
- 30.Gomes Sobrinho DB, Oliveira JB, Petersen CG, Mauri AL, Silva LF, Massaro FC, et al. IVF/ICSI outcomes after culture of human embryos at low oxygen tension: a meta-analysis. Reprod. Biol. Endocrinol. 2011;9:143. doi: 10.1186/1477-7827-9-143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bontekoe S, Mantikou E, van Wely M, Seshadri S, Repping S, Mastenbroek S. Low oxygen concentrations for embryo culture in assisted reproductive Technologies. Cochrane Database Syst. Rev. 2012;7 doi: 10.1002/14651858.CD008950.pub2. [DOI] [PubMed] [Google Scholar]
- 32.Meseguer M, Kruhne U, Laursen S. Full in vitro fertilization laboratory mechanization: toward robotic assisted reproduction? Fertil. Steril. 2012;97(6):1277–86. doi: 10.1016/j.fertnstert.2012.03.013. [DOI] [PubMed] [Google Scholar]
- 33.Kirkegaard K, Agerholm IE, Ingerslev HJ. Time-lapse monitoring as a tool for clinical embryo assessment. Hum. Reprod. 2012;27(5):1277–85. doi: 10.1093/humrep/des079. [DOI] [PubMed] [Google Scholar]
- 34.Kirkegaard K, Kesmodel US, Hindkjær JJ, Ingerslev HJ. Time-lapse parameters as predictors of blastocyst development and pregnancy outcome in embryos from good prognosis patients: a prospective cohort study. Hum. Reprod. 2013;28(10):2643–51. doi: 10.1093/humrep/det300. [DOI] [PubMed] [Google Scholar]