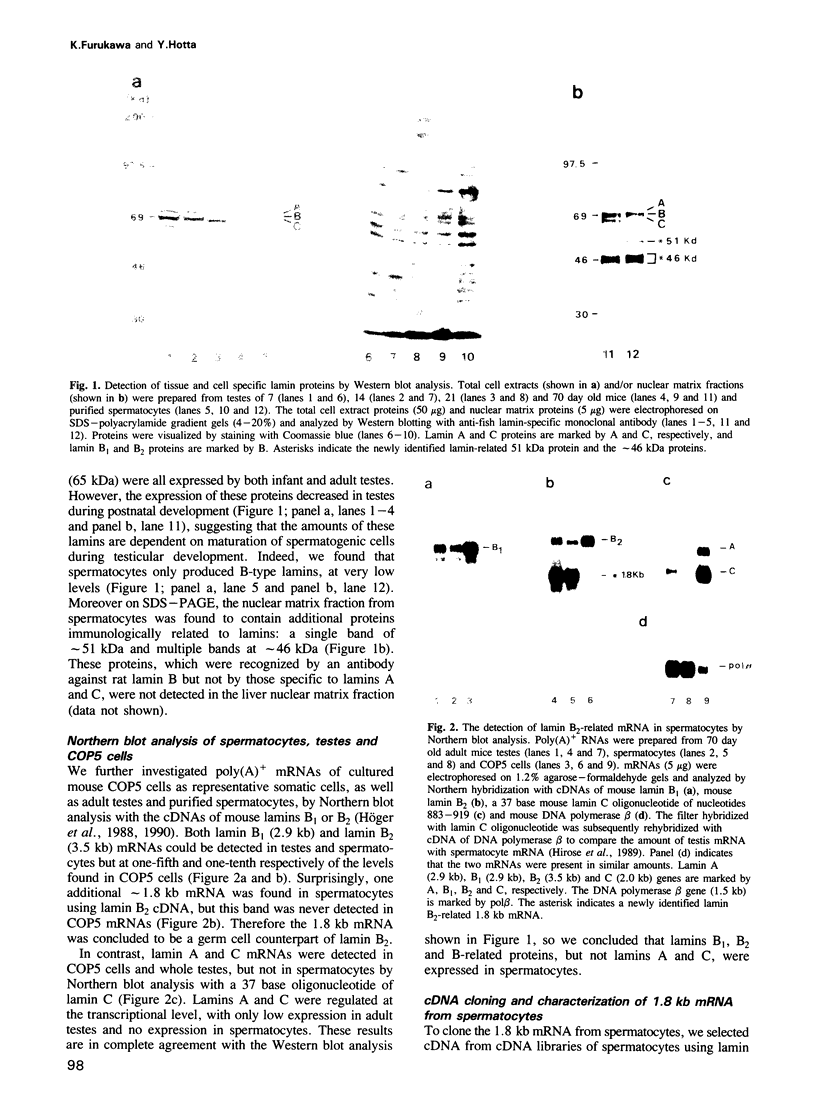
Abstract
The nuclear lamina is a fundamental component involved in the assembly of the nuclear envelope and higher order chromosomal structures in eukaryotes. In mammals, it is composed of four major lamin proteins, termed lamins A, B1, B2 and C. Here we first report cDNA cloning of a new 53 kDa lamin protein from mouse spermatocytes, termed lamin B3, the expression of which appears restricted to spermatogenic cells. Its gene structure indicates that lamin B3 is generated by differential splicing and alternative polyadenylation from lamin B2. When lamin B3 is introduced into somatic cells in culture, their nuclear morphology is transformed from spherical to hook-shaped. On the basis of the results obtained, we suggest that the germ cell specific lamin B3 is involved in the reorganization of nuclear and chromosomal structures during meiotic division.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aebi U., Cohn J., Buhle L., Gerace L. The nuclear lamina is a meshwork of intermediate-type filaments. Nature. 1986 Oct 9;323(6088):560–564. doi: 10.1038/323560a0. [DOI] [PubMed] [Google Scholar]
- Bellvé A. R., Cavicchia J. C., Millette C. F., O'Brien D. A., Bhatnagar Y. M., Dym M. Spermatogenic cells of the prepuberal mouse. Isolation and morphological characterization. J Cell Biol. 1977 Jul;74(1):68–85. doi: 10.1083/jcb.74.1.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benavente R., KrohneG Change of karyoskeleton during spermatogenesis of Xenopus: expression of lamin LIV, a nuclear lamina protein specific for the male germ line. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6176–6180. doi: 10.1073/pnas.82.18.6176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke B., Gerace L. A cell free system to study reassembly of the nuclear envelope at the end of mitosis. Cell. 1986 Feb 28;44(4):639–652. doi: 10.1016/0092-8674(86)90273-4. [DOI] [PubMed] [Google Scholar]
- Burke B. On the cell-free association of lamins A and C with metaphase chromosomes. Exp Cell Res. 1990 Jan;186(1):169–176. doi: 10.1016/0014-4827(90)90223-w. [DOI] [PubMed] [Google Scholar]
- Chandley A. C., Hotta Y., Stern H. Biochemical analysis of meiosis in the male mouse. I. Separation of DNA labelling of specific spermatogenic stages. Chromosoma. 1977 Jul 8;62(3):243–253. doi: 10.1007/BF00286046. [DOI] [PubMed] [Google Scholar]
- Counce S. J., Meyer G. F. Differentiation of the synaptonemal complex and the kinetochore in Locusta spermatocytes studied by whole mount electron microscopy. Chromosoma. 1973 Nov 21;44(2):231–253. doi: 10.1007/BF00329119. [DOI] [PubMed] [Google Scholar]
- Djabali K., Portier M. M., Gros F., Blobel G., Georgatos S. D. Network antibodies identify nuclear lamin B as a physiological attachment site for peripherin intermediate filaments. Cell. 1991 Jan 11;64(1):109–121. doi: 10.1016/0092-8674(91)90213-i. [DOI] [PubMed] [Google Scholar]
- Esponda P., Giménez-Martín G. The attachment of the synaptonemal complex to the nuclear envelope. An ultrastructural and cytochemical analysis. Chromosoma. 1972;38(4):405–417. doi: 10.1007/BF00320159. [DOI] [PubMed] [Google Scholar]
- Fawcett D. W., Chemes H. E. Changes in distribution of nuclear pores during differentiation of the male germ cells. Tissue Cell. 1979;11(1):147–162. doi: 10.1016/0040-8166(79)90015-6. [DOI] [PubMed] [Google Scholar]
- Fisher D. Z., Chaudhary N., Blobel G. cDNA sequencing of nuclear lamins A and C reveals primary and secondary structural homology to intermediate filament proteins. Proc Natl Acad Sci U S A. 1986 Sep;83(17):6450–6454. doi: 10.1073/pnas.83.17.6450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furukawa K., Yamaguchi Y., Ogawa E., Shigesada K., Satake M., Ito Y. A ubiquitous repressor interacting with an F9 cell-specific silencer and its functional suppression by differentiated cell-specific positive factors. Cell Growth Differ. 1990 Mar;1(3):135–147. [PubMed] [Google Scholar]
- Georgatos S. D., Blobel G. Lamin B constitutes an intermediate filament attachment site at the nuclear envelope. J Cell Biol. 1987 Jul;105(1):117–125. doi: 10.1083/jcb.105.1.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgatos S. D., Stournaras C., Blobel G. Heterotypic and homotypic associations between the nuclear lamins: site-specificity and control by phosphorylation. Proc Natl Acad Sci U S A. 1988 Jun;85(12):4325–4329. doi: 10.1073/pnas.85.12.4325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgatos S. D., Weber K., Geisler N., Blobel G. Binding of two desmin derivatives to the plasma membrane and the nuclear envelope of avian erythrocytes: evidence for a conserved site-specificity in intermediate filament-membrane interactions. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6780–6784. doi: 10.1073/pnas.84.19.6780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerace L., Blobel G. The nuclear envelope lamina is reversibly depolymerized during mitosis. Cell. 1980 Jan;19(1):277–287. doi: 10.1016/0092-8674(80)90409-2. [DOI] [PubMed] [Google Scholar]
- Gerace L., Burke B. Functional organization of the nuclear envelope. Annu Rev Cell Biol. 1988;4:335–374. doi: 10.1146/annurev.cb.04.110188.002003. [DOI] [PubMed] [Google Scholar]
- Glass J. R., Gerace L. Lamins A and C bind and assemble at the surface of mitotic chromosomes. J Cell Biol. 1990 Sep;111(3):1047–1057. doi: 10.1083/jcb.111.3.1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heald R., McKeon F. Mutations of phosphorylation sites in lamin A that prevent nuclear lamina disassembly in mitosis. Cell. 1990 May 18;61(4):579–589. doi: 10.1016/0092-8674(90)90470-y. [DOI] [PubMed] [Google Scholar]
- Heyting C., Dietrich A. J., Redeker E. J., Vink A. C. Structure and composition of synaptonemal complexes, isolated from rat spermatocytes. Eur J Cell Biol. 1985 Mar;36(2):307–314. [PubMed] [Google Scholar]
- Heyting C., Moens P. B., van Raamsdonk W., Dietrich A. J., Vink A. C., Redeker E. J. Identification of two major components of the lateral elements of synaptonemal complexes of the rat. Eur J Cell Biol. 1987 Feb;43(1):148–154. [PubMed] [Google Scholar]
- Hirose F., Hotta Y., Yamaguchi M., Matsukage A. Difference in the expression level of DNA polymerase beta among mouse tissues: high expression in the pachytene spermatocyte. Exp Cell Res. 1989 Mar;181(1):169–180. doi: 10.1016/0014-4827(89)90191-2. [DOI] [PubMed] [Google Scholar]
- Holtz D., Tanaka R. A., Hartwig J., McKeon F. The CaaX motif of lamin A functions in conjunction with the nuclear localization signal to target assembly to the nuclear envelope. Cell. 1989 Dec 22;59(6):969–977. doi: 10.1016/0092-8674(89)90753-8. [DOI] [PubMed] [Google Scholar]
- Höger T. H., Krohne G., Franke W. W. Amino acid sequence and molecular characterization of murine lamin B as deduced from cDNA clones. Eur J Cell Biol. 1988 Dec;47(2):283–290. [PubMed] [Google Scholar]
- Höger T. H., Zatloukal K., Waizenegger I., Krohne G. Characterization of a second highly conserved B-type lamin present in cells previously thought to contain only a single B-type lamin. Chromosoma. 1990 Oct;99(6):379–390. doi: 10.1007/BF01726689. [DOI] [PubMed] [Google Scholar]
- Kaufmann S. H. Additional members of the rat liver lamin polypeptide family. Structural and immunological characterization. J Biol Chem. 1989 Aug 15;264(23):13946–13955. [PubMed] [Google Scholar]
- Kitten G. T., Nigg E. A. The CaaX motif is required for isoprenylation, carboxyl methylation, and nuclear membrane association of lamin B2. J Cell Biol. 1991 Apr;113(1):13–23. doi: 10.1083/jcb.113.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolodziej P., Young R. A. RNA polymerase II subunit RPB3 is an essential component of the mRNA transcription apparatus. Mol Cell Biol. 1989 Dec;9(12):5387–5394. doi: 10.1128/mcb.9.12.5387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krohne G., Benavente R. The nuclear lamins. A multigene family of proteins in evolution and differentiation. Exp Cell Res. 1986 Jan;162(1):1–10. doi: 10.1016/0014-4827(86)90421-0. [DOI] [PubMed] [Google Scholar]
- Krohne G., Waizenegger I., Höger T. H. The conserved carboxy-terminal cysteine of nuclear lamins is essential for lamin association with the nuclear envelope. J Cell Biol. 1989 Nov;109(5):2003–2011. doi: 10.1083/jcb.109.5.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lebel S., Lampron C., Royal A., Raymond Y. Lamins A and C appear during retinoic acid-induced differentiation of mouse embryonal carcinoma cells. J Cell Biol. 1987 Sep;105(3):1099–1104. doi: 10.1083/jcb.105.3.1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehner C. F., Stick R., Eppenberger H. M., Nigg E. A. Differential expression of nuclear lamin proteins during chicken development. J Cell Biol. 1987 Jul;105(1):577–587. doi: 10.1083/jcb.105.1.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loewinger L., McKeon F. Mutations in the nuclear lamin proteins resulting in their aberrant assembly in the cytoplasm. EMBO J. 1988 Aug;7(8):2301–2309. doi: 10.1002/j.1460-2075.1988.tb03073.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo F. J., Krohne G., Franke W. W. Basic proteins of the perinuclear theca of mammalian spermatozoa and spermatids: a novel class of cytoskeletal elements. J Cell Biol. 1987 Sep;105(3):1105–1120. doi: 10.1083/jcb.105.3.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maul G. G., French B. T., Bechtol K. B. Identification and redistribution of lamins during nuclear differentiation in mouse spermatogenesis. Dev Biol. 1986 May;115(1):68–77. doi: 10.1016/0012-1606(86)90228-9. [DOI] [PubMed] [Google Scholar]
- McKeon F. D., Kirschner M. W., Caput D. Homologies in both primary and secondary structure between nuclear envelope and intermediate filament proteins. Nature. 1986 Feb 6;319(6053):463–468. doi: 10.1038/319463a0. [DOI] [PubMed] [Google Scholar]
- McKeon F. Nuclear lamin proteins: domains required for nuclear targeting, assembly, and cell-cycle-regulated dynamics. Curr Opin Cell Biol. 1991 Feb;3(1):82–86. doi: 10.1016/0955-0674(91)90169-y. [DOI] [PubMed] [Google Scholar]
- Moss S. B., Donovan M. J., Bellvé A. R. The occurrence and distribution of lamin proteins during mammalian spermatogenesis and early embryonic development. Ann N Y Acad Sci. 1987;513:74–89. doi: 10.1111/j.1749-6632.1987.tb24999.x. [DOI] [PubMed] [Google Scholar]
- Mukohata Y., Ihara K., Yoshida M., Konishi J., Sugiyama Y., Yoshida M. The halobacterial H+-translocating ATP synthase relates to the eukaryotic anion-sensitive H+-ATPase. Arch Biochem Biophys. 1987 Dec;259(2):650–653. doi: 10.1016/0003-9861(87)90532-7. [DOI] [PubMed] [Google Scholar]
- Nigg E. A. The nuclear envelope. Curr Opin Cell Biol. 1989 Jun;1(3):435–440. doi: 10.1016/0955-0674(89)90002-1. [DOI] [PubMed] [Google Scholar]
- Niman H. L., Houghten R. A., Walker L. E., Reisfeld R. A., Wilson I. A., Hogle J. M., Lerner R. A. Generation of protein-reactive antibodies by short peptides is an event of high frequency: implications for the structural basis of immune recognition. Proc Natl Acad Sci U S A. 1983 Aug;80(16):4949–4953. doi: 10.1073/pnas.80.16.4949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peter M., Heitlinger E., Häner M., Aebi U., Nigg E. A. Disassembly of in vitro formed lamin head-to-tail polymers by CDC2 kinase. EMBO J. 1991 Jun;10(6):1535–1544. doi: 10.1002/j.1460-2075.1991.tb07673.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinlan R. A., Stewart M. Molecular interactions in intermediate filaments. Bioessays. 1991 Nov;13(11):597–600. doi: 10.1002/bies.950131110. [DOI] [PubMed] [Google Scholar]
- Riedel W., Werner D. Nucleotide sequence of the full-length mouse lamin C cDNA and its deduced amino-acid sequence. Biochim Biophys Acta. 1989 Jun 1;1008(1):119–122. doi: 10.1016/0167-4781(89)90179-6. [DOI] [PubMed] [Google Scholar]
- Röber R. A., Weber K., Osborn M. Differential timing of nuclear lamin A/C expression in the various organs of the mouse embryo and the young animal: a developmental study. Development. 1989 Feb;105(2):365–378. doi: 10.1242/dev.105.2.365. [DOI] [PubMed] [Google Scholar]
- Satake M., Luftig R. B. Comparative immunofluorescence of murine leukemia virus-derived membrane-associated antigens. Virology. 1983 Jan 30;124(2):259–273. doi: 10.1016/0042-6822(83)90343-4. [DOI] [PubMed] [Google Scholar]
- Schatten G., Maul G. G., Schatten H., Chaly N., Simerly C., Balczon R., Brown D. L. Nuclear lamins and peripheral nuclear antigens during fertilization and embryogenesis in mice and sea urchins. Proc Natl Acad Sci U S A. 1985 Jul;82(14):4727–4731. doi: 10.1073/pnas.82.14.4727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seed B. An LFA-3 cDNA encodes a phospholipid-linked membrane protein homologous to its receptor CD2. 1987 Oct 29-Nov 4Nature. 329(6142):840–842. doi: 10.1038/329840a0. [DOI] [PubMed] [Google Scholar]
- Smith A., Benavente R. Identification of a structural protein component of rat synaptonemal complexes. Exp Cell Res. 1992 Feb;198(2):291–297. doi: 10.1016/0014-4827(92)90382-i. [DOI] [PubMed] [Google Scholar]
- Smith D. B., Johnson K. S. Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene. 1988 Jul 15;67(1):31–40. doi: 10.1016/0378-1119(88)90005-4. [DOI] [PubMed] [Google Scholar]
- Spiegelman B. M., Frank M., Green H. Molecular cloning of mRNA from 3T3 adipocytes. Regulation of mRNA content for glycerophosphate dehydrogenase and other differentiation-dependent proteins during adipocyte development. J Biol Chem. 1983 Aug 25;258(16):10083–10089. [PubMed] [Google Scholar]
- Steinert P. M., Roop D. R. Molecular and cellular biology of intermediate filaments. Annu Rev Biochem. 1988;57:593–625. doi: 10.1146/annurev.bi.57.070188.003113. [DOI] [PubMed] [Google Scholar]
- Stewart C., Burke B. Teratocarcinoma stem cells and early mouse embryos contain only a single major lamin polypeptide closely resembling lamin B. Cell. 1987 Nov 6;51(3):383–392. doi: 10.1016/0092-8674(87)90634-9. [DOI] [PubMed] [Google Scholar]
- Stick R., Angres B., Lehner C. F., Nigg E. A. The fates of chicken nuclear lamin proteins during mitosis: evidence for a reversible redistribution of lamin B2 between inner nuclear membrane and elements of the endoplasmic reticulum. J Cell Biol. 1988 Aug;107(2):397–406. doi: 10.1083/jcb.107.2.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stick R., Schwarz H. Disappearance and reformation of the nuclear lamina structure during specific stages of meiosis in oocytes. Cell. 1983 Jul;33(3):949–958. doi: 10.1016/0092-8674(83)90038-7. [DOI] [PubMed] [Google Scholar]
- Stick R., Schwarz H. The disappearance of the nuclear lamina during spermatogenesis: an electron microscopic and immunofluorescence study. Cell Differ. 1982 Jun;11(4):235–243. doi: 10.1016/0045-6039(82)90071-9. [DOI] [PubMed] [Google Scholar]
- Stick R. cDNA cloning of the developmentally regulated lamin LIII of Xenopus laevis. EMBO J. 1988 Oct;7(10):3189–3197. doi: 10.1002/j.1460-2075.1988.tb03186.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sudhakar L., Rao M. R. Stage-dependent changes in localization of a germ cell-specific lamin during mammalian spermatogenesis. J Biol Chem. 1990 Dec 25;265(36):22526–22532. [PubMed] [Google Scholar]
- Sudhakar L., Sivakumar N., Behal A., Rao M. R. Evolutionary conservation of a germ cell-specific lamin persisting through mammalian spermiogenesis. Exp Cell Res. 1992 Jan;198(1):78–84. doi: 10.1016/0014-4827(92)90151-w. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyndall C., La Mantia G., Thacker C. M., Favaloro J., Kamen R. A region of the polyoma virus genome between the replication origin and late protein coding sequences is required in cis for both early gene expression and viral DNA replication. Nucleic Acids Res. 1981 Dec 11;9(23):6231–6250. doi: 10.1093/nar/9.23.6231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woollam D. H., Ford E. H., Millen J. W. The attachment of pachytene chromosomes to the nuclear membrane in mammalian spermatocytes. Exp Cell Res. 1966 Jun;42(3):657–661. doi: 10.1016/0014-4827(66)90278-3. [DOI] [PubMed] [Google Scholar]
- Yuan J., Simos G., Blobel G., Georgatos S. D. Binding of lamin A to polynucleosomes. J Biol Chem. 1991 May 15;266(14):9211–9215. [PubMed] [Google Scholar]
- Zewe M., Höger T. H., Fink T., Lichter P., Krohne G., Franke W. W. Gene structure and chromosomal localization of the murine lamin B2 gene. Eur J Cell Biol. 1991 Dec;56(2):342–350. [PubMed] [Google Scholar]