Abstract
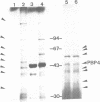
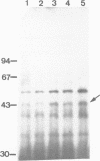
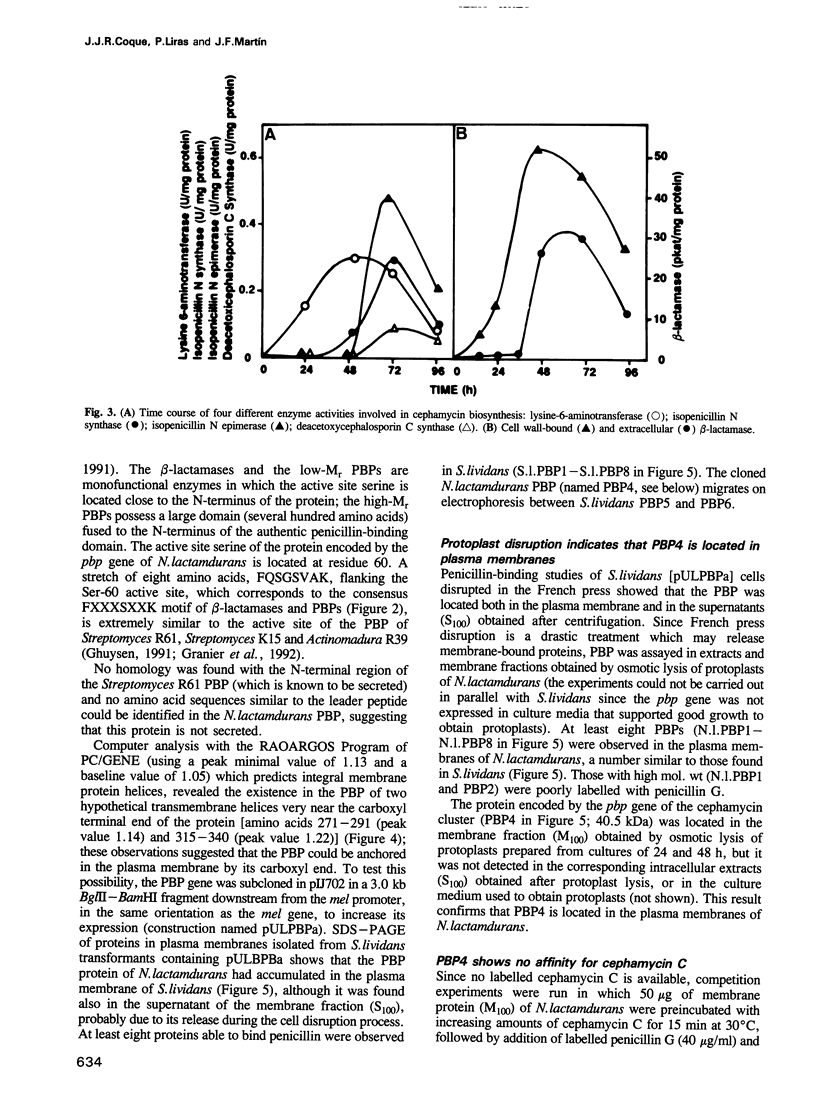
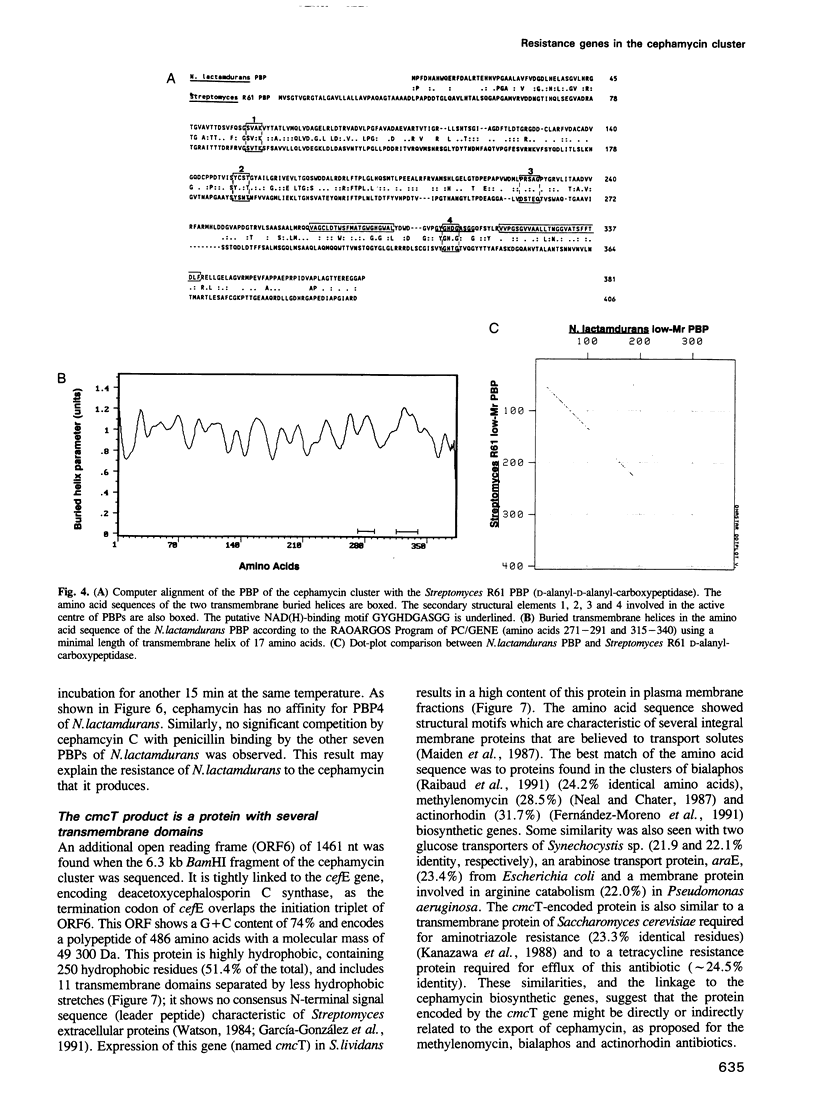
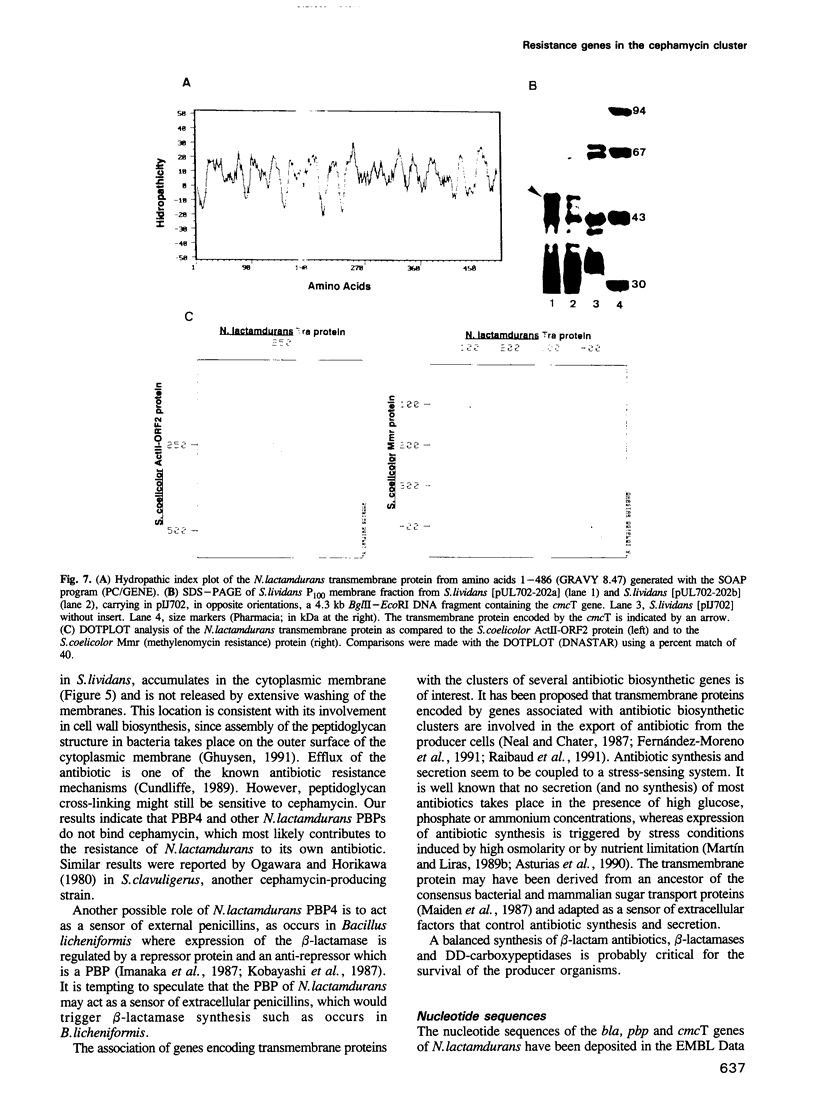
Three genes encoding a typical beta-lactamase, a penicillin-binding protein (PBP4) and a transmembrane protein are located in the cluster of cephamycin biosynthetic genes in Nocardia lactamdurans. The similarity of the N. lactamdurans beta-lactamase to class A beta-lactamases from clinical isolates supports the hypothesis that antibiotic resistance genes in pathogenic bacteria are derived from antibiotic-producing organisms. The beta-lactamase is secreted and is active against penicillins (including the biosynthetic intermediates penicillin N and isopenicillin N), but not against cephamycin C. The beta-lactamase is synthesized during the active growth phase, prior to the formation of three cephamycin biosynthetic enzymes. The PBP of N. lactamdurans is a low-M(r) protein that is very similar to DD-carboxypeptidases of Streptomyces and Actinomadura. The pbp gene product expressed in Streptomyces lividans accumulates in the membrane fraction. By disruption of N. lactamdurans protoplasts, the PBP4 was shown to be located in the plasma membrane. Eight PBPs were found in the membranes of N. lactamdurans, none of which bind cephamycin C, which explains the resistance of this strain to its own antibiotic. A transmembrane protein encoded by the cmcT gene of the cluster also accumulates in the membrane fraction and is probably related to the control of synthesis and secretion of the antibiotic. A balanced synthesis of beta-lactam antibiotics, beta-lactamase and PBP is postulated to be critical for the survival of beta-lactam-producing actinomycetes.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aharonowitz Y., Cohen G., Martin J. F. Penicillin and cephalosporin biosynthetic genes: structure, organization, regulation, and evolution. Annu Rev Microbiol. 1992;46:461–495. doi: 10.1146/annurev.mi.46.100192.002333. [DOI] [PubMed] [Google Scholar]
- Asturias J. A., Liras P., Martín J. F. Phosphate control of pabS gene transcription during candicidin biosynthesis. Gene. 1990 Sep 1;93(1):79–84. doi: 10.1016/0378-1119(90)90139-i. [DOI] [PubMed] [Google Scholar]
- Benveniste R., Davies J. Aminoglycoside antibiotic-inactivating enzymes in actinomycetes similar to those present in clinical isolates of antibiotic-resistant bacteria. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2276–2280. doi: 10.1073/pnas.70.8.2276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coque J. J., Liras P., Laiz L., Martín J. F. A gene encoding lysine 6-aminotransferase, which forms the beta-lactam precursor alpha-aminoadipic acid, is located in the cluster of cephamycin biosynthetic genes in Nocardia lactamdurans. J Bacteriol. 1991 Oct;173(19):6258–6264. doi: 10.1128/jb.173.19.6258-6264.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coque J. J., Martín J. F., Calzada J. G., Liras P. The cephamycin biosynthetic genes pcbAB, encoding a large multidomain peptide synthetase, and pcbC of Nocardia lactamdurans are clustered together in an organization different from the same genes in Acremonium chrysogenum and Penicillium chrysogenum. Mol Microbiol. 1991 May;5(5):1125–1133. doi: 10.1111/j.1365-2958.1991.tb01885.x. [DOI] [PubMed] [Google Scholar]
- Cortés J., Martín J. F., Castro J. M., Láiz L., Liras P. Purification and characterization of a 2-oxoglutarate-linked ATP-independent deacetoxycephalosporin C synthase of Streptomyces lactamdurans. J Gen Microbiol. 1987 Nov;133(11):3165–3174. doi: 10.1099/00221287-133-11-3165. [DOI] [PubMed] [Google Scholar]
- Cundliffe E. How antibiotic-producing organisms avoid suicide. Annu Rev Microbiol. 1989;43:207–233. doi: 10.1146/annurev.mi.43.100189.001231. [DOI] [PubMed] [Google Scholar]
- Doran J. L., Leskiw B. K., Aippersbach S., Jensen S. E. Isolation and characterization of a beta-lactamase-inhibitory protein from Streptomyces clavuligerus and cloning and analysis of the corresponding gene. J Bacteriol. 1990 Sep;172(9):4909–4918. doi: 10.1128/jb.172.9.4909-4918.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duez C., Piron-Fraipont C., Joris B., Dusart J., Urdea M. S., Martial J. A., Frère J. M., Ghuysen J. M. Primary structure of the Streptomyces R61 extracellular DD-peptidase. 1. Cloning into Streptomyces lividans and nucleotide sequence of the gene. Eur J Biochem. 1987 Feb 2;162(3):509–518. doi: 10.1111/j.1432-1033.1987.tb10669.x. [DOI] [PubMed] [Google Scholar]
- Fernández-Moreno M. A., Caballero J. L., Hopwood D. A., Malpartida F. The act cluster contains regulatory and antibiotic export genes, direct targets for translational control by the bldA tRNA gene of Streptomyces. Cell. 1991 Aug 23;66(4):769–780. doi: 10.1016/0092-8674(91)90120-n. [DOI] [PubMed] [Google Scholar]
- Forsman M., Häggström B., Lindgren L., Jaurin B. Molecular analysis of beta-lactamases from four species of Streptomyces: comparison of amino acid sequences with those of other beta-lactamases. J Gen Microbiol. 1990 Mar;136(3):589–598. doi: 10.1099/00221287-136-3-589. [DOI] [PubMed] [Google Scholar]
- Garcia-Gonzalez M. D., Martin J. F., Vigal T., Liras P. Characterization, expression in Streptomyces lividans, and processing of the amylase of Streptomyces griseus IMRU 3570: two different amylases are derived from the same gene by an intracellular processing mechanism. J Bacteriol. 1991 Apr;173(8):2451–2458. doi: 10.1128/jb.173.8.2451-2458.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Domínguez M., Liras P., Martín J. F. Cloning and characterization of the isopenicillin N synthase gene of Streptomyces griseus NRRL 3851 and studies of expression and complementation of the cephamycin pathway in Streptomyces clavuligerus. Antimicrob Agents Chemother. 1991 Jan;35(1):44–52. doi: 10.1128/aac.35.1.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghuysen J. M. Serine beta-lactamases and penicillin-binding proteins. Annu Rev Microbiol. 1991;45:37–67. doi: 10.1146/annurev.mi.45.100191.000345. [DOI] [PubMed] [Google Scholar]
- Granier B., Duez C., Lepage S., Englebert S., Dusart J., Dideberg O., Van Beeumen J., Frère J. M., Ghuysen J. M. Primary and predicted secondary structures of the Actinomadura R39 extracellular DD-peptidase, a penicillin-binding protein (PBP) related to the Escherichia coli PBP4. Biochem J. 1992 Mar 15;282(Pt 3):781–788. doi: 10.1042/bj2820781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imanaka T., Himeno T., Aiba S. Cloning and nucleotide sequence of the penicillinase antirepressor gene penJ of Bacillus licheniformis. J Bacteriol. 1987 Sep;169(9):3867–3872. doi: 10.1128/jb.169.9.3867-3872.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joris B., Ledent P., Dideberg O., Fonzé E., Lamotte-Brasseur J., Kelly J. A., Ghuysen J. M., Frère J. M. Comparison of the sequences of class A beta-lactamases and of the secondary structure elements of penicillin-recognizing proteins. Antimicrob Agents Chemother. 1991 Nov;35(11):2294–2301. doi: 10.1128/aac.35.11.2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanazawa S., Driscoll M., Struhl K. ATR1, a Saccharomyces cerevisiae gene encoding a transmembrane protein required for aminotriazole resistance. Mol Cell Biol. 1988 Feb;8(2):664–673. doi: 10.1128/mcb.8.2.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz E., Thompson C. J., Hopwood D. A. Cloning and expression of the tyrosinase gene from Streptomyces antibioticus in Streptomyces lividans. J Gen Microbiol. 1983 Sep;129(9):2703–2714. doi: 10.1099/00221287-129-9-2703. [DOI] [PubMed] [Google Scholar]
- Kieser T., Melton R. E. Plasmid pIJ699, a multi-copy positive-selection vector for Streptomyces. Gene. 1988 May 15;65(1):83–91. doi: 10.1016/0378-1119(88)90419-2. [DOI] [PubMed] [Google Scholar]
- Kobayashi T., Zhu Y. F., Nicholls N. J., Lampen J. O. A second regulatory gene, blaR1, encoding a potential penicillin-binding protein required for induction of beta-lactamase in Bacillus licheniformis. J Bacteriol. 1987 Sep;169(9):3873–3878. doi: 10.1128/jb.169.9.3873-3878.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maiden M. C., Davis E. O., Baldwin S. A., Moore D. C., Henderson P. J. Mammalian and bacterial sugar transport proteins are homologous. Nature. 1987 Feb 12;325(6105):641–643. doi: 10.1038/325641a0. [DOI] [PubMed] [Google Scholar]
- Martin J. F. Clusters of genes for the biosynthesis of antibiotics: regulatory genes and overproduction of pharmaceuticals. J Ind Microbiol. 1992 Feb-Mar;9(2):73–90. doi: 10.1007/BF01569737. [DOI] [PubMed] [Google Scholar]
- Martín M. F., Liras P. Organization and expression of genes involved in the biosynthesis of antibiotics and other secondary metabolites. Annu Rev Microbiol. 1989;43:173–206. doi: 10.1146/annurev.mi.43.100189.001133. [DOI] [PubMed] [Google Scholar]
- Neal R. J., Chater K. F. Nucleotide sequence analysis reveals similarities between proteins determining methylenomycin A resistance in Streptomyces and tetracycline resistance in eubacteria. Gene. 1987;58(2-3):229–241. doi: 10.1016/0378-1119(87)90378-7. [DOI] [PubMed] [Google Scholar]
- Ogawara H. Antibiotic resistance in pathogenic and producing bacteria, with special reference to beta-lactam antibiotics. Microbiol Rev. 1981 Dec;45(4):591–619. doi: 10.1128/mr.45.4.591-619.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawara H., Horikawa S. Penicillin-binding proteins of Streptomyces cacaoi, Streptomyces olivaceus, and Streptomyces clavuligerus. Antimicrob Agents Chemother. 1980 Jan;17(1):1–7. doi: 10.1128/aac.17.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawara H., Horikawa S., Shimada-Miyoshi S., Yasuzawa K. Production and property of beta-lactamases in Streptomyces: comparison of the strains isolated newly and thirty years ago. Antimicrob Agents Chemother. 1978 May;13(5):865–870. doi: 10.1128/aac.13.5.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raibaud A., Zalacain M., Holt T. G., Tizard R., Thompson C. J. Nucleotide sequence analysis reveals linked N-acetyl hydrolase, thioesterase, transport, and regulatory genes encoded by the bialaphos biosynthetic gene cluster of Streptomyces hygroscopicus. J Bacteriol. 1991 Jul;173(14):4454–4463. doi: 10.1128/jb.173.14.4454-4463.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scrutton N. S., Berry A., Perham R. N. Redesign of the coenzyme specificity of a dehydrogenase by protein engineering. Nature. 1990 Jan 4;343(6253):38–43. doi: 10.1038/343038a0. [DOI] [PubMed] [Google Scholar]
- Tobin M. B., Kovacevic S., Madduri K., Hoskins J. A., Skatrud P. L., Vining L. C., Stuttard C., Miller J. R. Localization of the lysine epsilon-aminotransferase (lat) and delta-(L-alpha-aminoadipyl)-L-cysteinyl-D-valine synthetase (pcbAB) genes from Streptomyces clavuligerus and production of lysine epsilon-aminotransferase activity in Escherichia coli. J Bacteriol. 1991 Oct;173(19):6223–6229. doi: 10.1128/jb.173.19.6223-6229.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson M. E. Compilation of published signal sequences. Nucleic Acids Res. 1984 Jul 11;12(13):5145–5164. doi: 10.1093/nar/12.13.5145. [DOI] [PMC free article] [PubMed] [Google Scholar]