Abstract
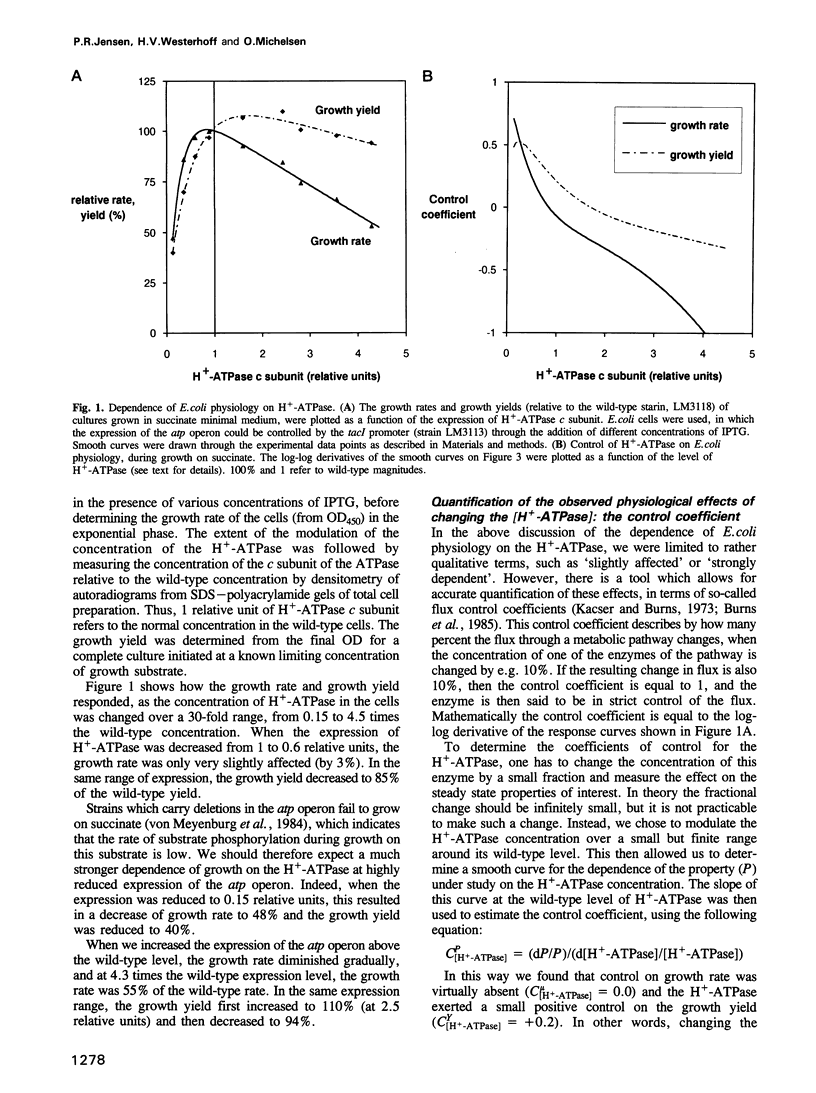
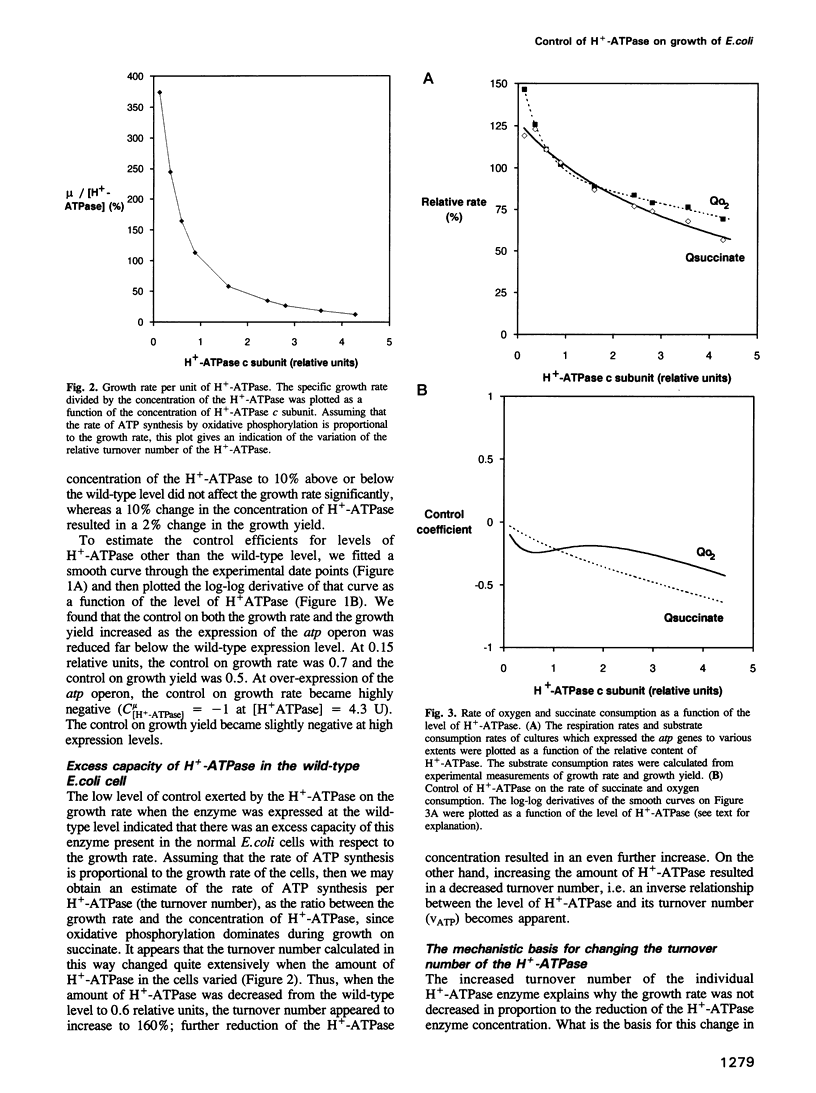
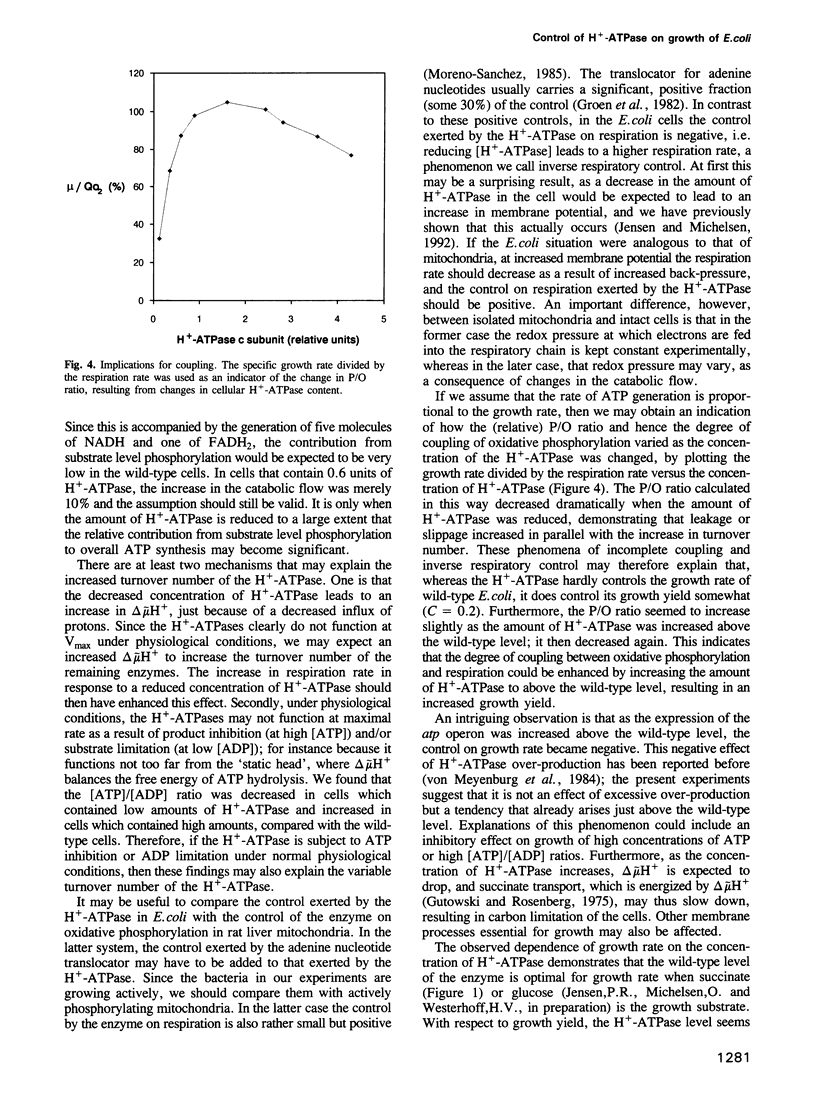
With succinate as free-energy source, Escherichia coli generating virtually all ATP by oxidative phosphorylation might be expected heavily to tax its ATP generating capacity. To examine this the H(+)-ATPase (ATP synthase) was modulated over a 30-fold range. Decreasing the amount of H(+)-ATPase reduced the growth rate much less than proportionally; the H(+)-ATPase controlled growth rate by < 10%. This lack of control reflected excess capacity: the rate of ATP synthesis per H(+)-ATPase (the turnover number) increased by 60% when the number of enzymes was decreased by 40%. At 15% H(+)-ATPase, the enzyme became limiting and its turnover was increased even further, due to an increased driving force caused by a reduction in the total flux through the enzymes. At smaller reductions of [H(+)-ATPase] the total flux was not reduced, revealing a second cause for increased turnover number through increased membrane potential: respiration was increased, showing that in E.coli, respiration and ATP synthesis are, in part, inversely coupled. Indeed, growth yield per O2 decreased, suggesting significant leakage or slip at the high respiration rates and membrane potential found at low H(+)-ATPase concentrations, and explaining that growth yield may be increased by activating the H(+)-ATPase.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dykhuizen D. E., Dean A. M., Hartl D. L. Metabolic flux and fitness. Genetics. 1987 Jan;115(1):25–31. doi: 10.1093/genetics/115.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groen A. K., Wanders R. J., Westerhoff H. V., van der Meer R., Tager J. M. Quantification of the contribution of various steps to the control of mitochondrial respiration. J Biol Chem. 1982 Mar 25;257(6):2754–2757. [PubMed] [Google Scholar]
- Gutowski S. J., Rosenberg H. Succinate uptake and related proton movements in Escherichia coli K12. Biochem J. 1975 Dec;152(3):647–654. doi: 10.1042/bj1520647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen P. R., Michelsen O. Carbon and energy metabolism of atp mutants of Escherichia coli. J Bacteriol. 1992 Dec;174(23):7635–7641. doi: 10.1128/jb.174.23.7635-7641.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen P. R., Westerhoff H. V., Michelsen O. The use of lac-type promoters in control analysis. Eur J Biochem. 1993 Jan 15;211(1-2):181–191. doi: 10.1111/j.1432-1033.1993.tb19885.x. [DOI] [PubMed] [Google Scholar]
- Moreno-Sánchez R. Contribution of the translocator of adenine nucleotides and the ATP synthase to the control of oxidative phosphorylation and arsenylation in liver mitochondria. J Biol Chem. 1985 Oct 15;260(23):12554–12560. [PubMed] [Google Scholar]
- Neidhardt F. C., Bloch P. L., Smith D. F. Culture medium for enterobacteria. J Bacteriol. 1974 Sep;119(3):736–747. doi: 10.1128/jb.119.3.736-747.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruyter G. J., Postma P. W., van Dam K. Control of glucose metabolism by enzyme IIGlc of the phosphoenolpyruvate-dependent phosphotransferase system in Escherichia coli. J Bacteriol. 1991 Oct;173(19):6184–6191. doi: 10.1128/jb.173.19.6184-6191.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh K., Koshland D. E., Jr Characterization of rate-controlling steps in vivo by use of an adjustable expression vector. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3577–3581. doi: 10.1073/pnas.82.11.3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Meyenburg K., Jørgensen B. B., Nielsen J., Hansen F. G. Promoters of the atp operon coding for the membrane-bound ATP synthase of Escherichia coli mapped by Tn10 insertion mutations. Mol Gen Genet. 1982;188(2):240–248. doi: 10.1007/BF00332682. [DOI] [PubMed] [Google Scholar]
- von Meyenburg K., Jørgensen B. B., van Deurs B. Physiological and morphological effects of overproduction of membrane-bound ATP synthase in Escherichia coli K-12. EMBO J. 1984 Aug;3(8):1791–1797. doi: 10.1002/j.1460-2075.1984.tb02047.x. [DOI] [PMC free article] [PubMed] [Google Scholar]



