Abstract
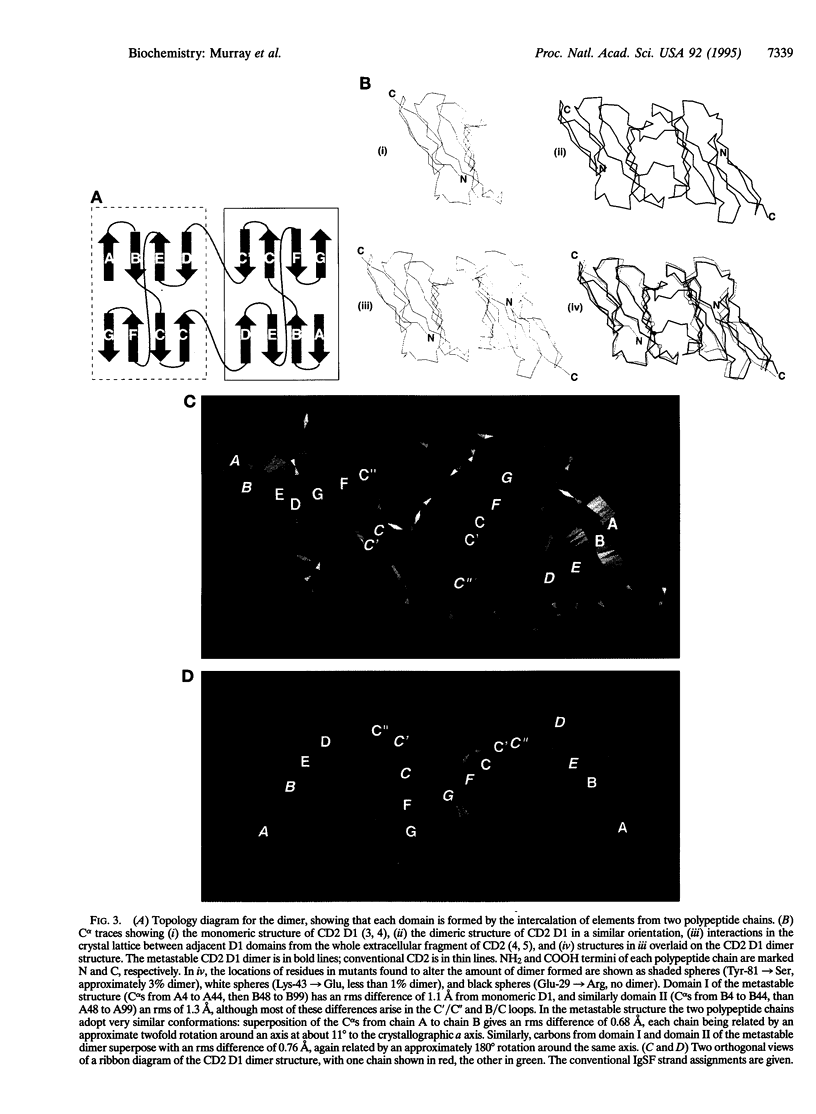
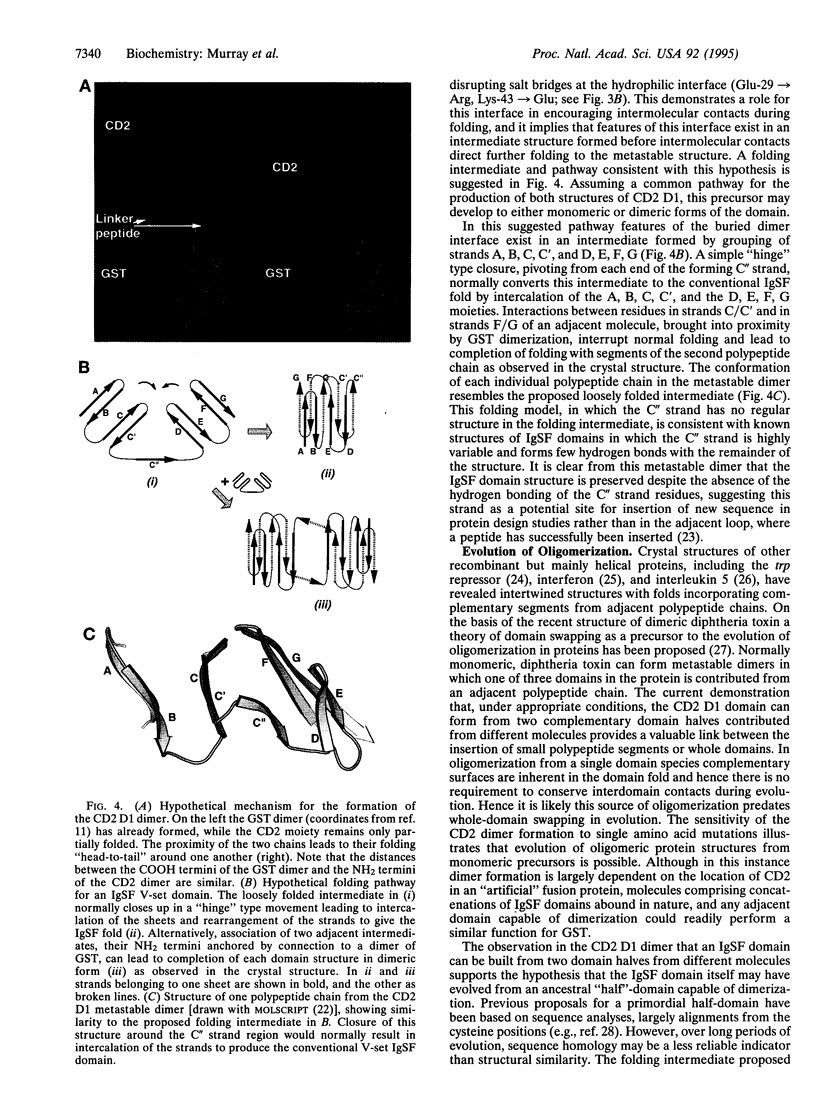
When expressed as part of a glutathione S-transferase fusion protein the NH2-terminal domain of the lymphocyte cell adhesion molecule CD2 is shown to adopt two different folds. The immunoglobulin superfamily structure of the major (85%) monomeric component has previously been determined by both x-ray crystallography and NMR spectroscopy. We now describe the structure of a second, dimeric, form present in about 15% of recombinant CD2 molecules. After denaturation and refolding in the absence of the fusion partner, dimeric CD2 is converted to monomer, illustrating that the dimeric form represents a metastable folded state. The crystal structure of this dimeric form, refined to 2.0-A resolution, reveals two domains with overall similarity to the IgSF fold found in the monomer. However, in the dimer each domain is formed by the intercalation of two polypeptide chains. Hence each domain represents a distinct folding unit that can assemble in two different ways. In the dimer the two domains fold around a hydrophilic interface believed to mimic the cell adhesion interaction at the cell surface, and the formation of dimer can be regulated by mutating single residues at this interface. This unusual misfolded form of the protein, which appears to result from inter- rather than intramolecular interactions being favored by an intermediate structure formed during the folding process, illustrates that evolution of protein oligomers is possible from the sequence for a single protein domain.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arulanandam A. R., Withka J. M., Wyss D. F., Wagner G., Kister A., Pallai P., Recny M. A., Reinherz E. L. The CD58 (LFA-3) binding site is a localized and highly charged surface area on the AGFCC'C" face of the human CD2 adhesion domain. Proc Natl Acad Sci U S A. 1993 Dec 15;90(24):11613–11617. doi: 10.1073/pnas.90.24.11613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett M. J., Choe S., Eisenberg D. Domain swapping: entangling alliances between proteins. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):3127–3131. doi: 10.1073/pnas.91.8.3127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bierer B. E., Sleckman B. P., Ratnofsky S. E., Burakoff S. J. The biologic roles of CD2, CD4, and CD8 in T-cell activation. Annu Rev Immunol. 1989;7:579–599. doi: 10.1146/annurev.iy.07.040189.003051. [DOI] [PubMed] [Google Scholar]
- Blechman J. M., Lev S., Barg J., Eisenstein M., Vaks B., Vogel Z., Givol D., Yarden Y. The fourth immunoglobulin domain of the stem cell factor receptor couples ligand binding to signal transduction. Cell. 1995 Jan 13;80(1):103–113. doi: 10.1016/0092-8674(95)90455-7. [DOI] [PubMed] [Google Scholar]
- Bodian D. L., Jones E. Y., Harlos K., Stuart D. I., Davis S. J. Crystal structure of the extracellular region of the human cell adhesion molecule CD2 at 2.5 A resolution. Structure. 1994 Aug 15;2(8):755–766. doi: 10.1016/s0969-2126(94)00076-x. [DOI] [PubMed] [Google Scholar]
- Clark S. J., Law D. A., Paterson D. J., Puklavec M., Williams A. F. Activation of rat T lymphocytes by anti-CD2 monoclonal antibodies. J Exp Med. 1988 Jun 1;167(6):1861–1872. doi: 10.1084/jem.167.6.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis S. J., Davies E. A., van der Merwe P. A. Mutational analysis of the epitopes recognized by anti-(rat CD2) and anti-(rat CD48) monoclonal antibodies. Biochem Soc Trans. 1995 Feb;23(1):188–194. doi: 10.1042/bst0230188. [DOI] [PubMed] [Google Scholar]
- Driscoll P. C., Cyster J. G., Campbell I. D., Williams A. F. Structure of domain 1 of rat T lymphocyte CD2 antigen. Nature. 1991 Oct 24;353(6346):762–765. doi: 10.1038/353762a0. [DOI] [PubMed] [Google Scholar]
- Ealick S. E., Cook W. J., Vijay-Kumar S., Carson M., Nagabhushan T. L., Trotta P. P., Bugg C. E. Three-dimensional structure of recombinant human interferon-gamma. Science. 1991 May 3;252(5006):698–702. doi: 10.1126/science.1902591. [DOI] [PubMed] [Google Scholar]
- Erickson H. P. Reversible unfolding of fibronectin type III and immunoglobulin domains provides the structural basis for stretch and elasticity of titin and fibronectin. Proc Natl Acad Sci U S A. 1994 Oct 11;91(21):10114–10118. doi: 10.1073/pnas.91.21.10114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holden H. M., Ito M., Hartshorne D. J., Rayment I. X-ray structure determination of telokin, the C-terminal domain of myosin light chain kinase, at 2.8 A resolution. J Mol Biol. 1992 Oct 5;227(3):840–851. doi: 10.1016/0022-2836(92)90226-a. [DOI] [PubMed] [Google Scholar]
- Hünig T. The cell surface molecule recognized by the erythrocyte receptor of T lymphocytes. Identification and partial characterization using a monoclonal antibody. J Exp Med. 1985 Sep 1;162(3):890–901. doi: 10.1084/jem.162.3.890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji X., Zhang P., Armstrong R. N., Gilliland G. L. The three-dimensional structure of a glutathione S-transferase from the mu gene class. Structural analysis of the binary complex of isoenzyme 3-3 and glutathione at 2.2-A resolution. Biochemistry. 1992 Oct 27;31(42):10169–10184. doi: 10.1021/bi00157a004. [DOI] [PubMed] [Google Scholar]
- Jones E. Y., Davis S. J., Williams A. F., Harlos K., Stuart D. I. Crystal structure at 2.8 A resolution of a soluble form of the cell adhesion molecule CD2. Nature. 1992 Nov 19;360(6401):232–239. doi: 10.1038/360232a0. [DOI] [PubMed] [Google Scholar]
- Jones T. A., Zou J. Y., Cowan S. W., Kjeldgaard M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr A. 1991 Mar 1;47(Pt 2):110–119. doi: 10.1107/s0108767390010224. [DOI] [PubMed] [Google Scholar]
- Kato K., Koyanagi M., Okada H., Takanashi T., Wong Y. W., Williams A. F., Okumura K., Yagita H. CD48 is a counter-receptor for mouse CD2 and is involved in T cell activation. J Exp Med. 1992 Nov 1;176(5):1241–1249. doi: 10.1084/jem.176.5.1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milburn M. V., Hassell A. M., Lambert M. H., Jordan S. R., Proudfoot A. E., Graber P., Wells T. N. A novel dimer configuration revealed by the crystal structure at 2.4 A resolution of human interleukin-5. Nature. 1993 May 13;363(6425):172–176. doi: 10.1038/363172a0. [DOI] [PubMed] [Google Scholar]
- Peterson A., Seed B. Monoclonal antibody and ligand binding sites of the T cell erythrocyte receptor (CD2). 1987 Oct 29-Nov 4Nature. 329(6142):842–846. doi: 10.1038/329842a0. [DOI] [PubMed] [Google Scholar]
- Schevitz R. W., Otwinowski Z., Joachimiak A., Lawson C. L., Sigler P. B. The three-dimensional structure of trp repressor. 1985 Oct 31-Nov 6Nature. 317(6040):782–786. doi: 10.1038/317782a0. [DOI] [PubMed] [Google Scholar]
- Selvaraj P., Plunkett M. L., Dustin M., Sanders M. E., Shaw S., Springer T. A. The T lymphocyte glycoprotein CD2 binds the cell surface ligand LFA-3. 1987 Mar 26-Apr 1Nature. 326(6111):400–403. doi: 10.1038/326400a0. [DOI] [PubMed] [Google Scholar]
- Sollazzo M., Billetta R., Zanetti M. Expression of an exogenous peptide epitope genetically engineered in the variable domain of an immunoglobulin: implications for antibody and peptide folding. Protein Eng. 1990 Dec;4(2):215–220. doi: 10.1093/protein/4.2.215. [DOI] [PubMed] [Google Scholar]
- Somoza C., Driscoll P. C., Cyster J. G., Williams A. F. Mutational analysis of the CD2/CD58 interaction: the binding site for CD58 lies on one face of the first domain of human CD2. J Exp Med. 1993 Aug 1;178(2):549–558. doi: 10.1084/jem.178.2.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soteriou A., Clarke A., Martin S., Trinick J. Titin folding energy and elasticity. Proc Biol Sci. 1993 Nov 22;254(1340):83–86. doi: 10.1098/rspb.1993.0130. [DOI] [PubMed] [Google Scholar]
- van der Merwe P. A., Barclay A. N., Mason D. W., Davies E. A., Morgan B. P., Tone M., Krishnam A. K., Ianelli C., Davis S. J. Human cell-adhesion molecule CD2 binds CD58 (LFA-3) with a very low affinity and an extremely fast dissociation rate but does not bind CD48 or CD59. Biochemistry. 1994 Aug 23;33(33):10149–10160. doi: 10.1021/bi00199a043. [DOI] [PubMed] [Google Scholar]
- van der Merwe P. A., Brown M. H., Davis S. J., Barclay A. N. Affinity and kinetic analysis of the interaction of the cell adhesion molecules rat CD2 and CD48. EMBO J. 1993 Dec 15;12(13):4945–4954. doi: 10.1002/j.1460-2075.1993.tb06188.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Merwe P. A., McNamee P. N., Davies E. A., Barclay A. N., Davis S. J. Topology of the CD2-CD48 cell-adhesion molecule complex: implications for antigen recognition by T cells. Curr Biol. 1995 Jan 1;5(1):74–84. doi: 10.1016/s0960-9822(95)00019-4. [DOI] [PubMed] [Google Scholar]
- van der Merwe P. A., McPherson D. C., Brown M. H., Barclay A. N., Cyster J. G., Williams A. F., Davis S. J. The NH2-terminal domain of rat CD2 binds rat CD48 with a low affinity and binding does not require glycosylation of CD2. Eur J Immunol. 1993 Jun;23(6):1373–1377. doi: 10.1002/eji.1830230628. [DOI] [PubMed] [Google Scholar]