Abstract
During cancer progression, bone marrow derived myeloid cells, including immature myeloid cells and macrophages, progressively accumulate at the primary tumour site where they contribute to the establishment of a tumour promoting microenvironment. A marked infiltration of macrophages into the stromal compartment and the generation of a desmoplastic stromal reaction is a particular characteristic of pancreatic ductal adenocarcinoma (PDA) and is thought to play a key role in disease progression and its response to therapy. Tumour associated macrophages (TAMs) foster PDA tumour progression by promoting angiogenesis, metastasis, and by suppressing an anti-tumourigenic immune response. Recent work also suggests that TAMs contribute to resistance to chemotherapy and to the emergence of cancer stem-like cells. Here we will review the current understanding of the biology and the pro-tumourigenic functions of TAMs in cancer and specifically in PDA, and highlight potential therapeutic strategies to target TAMs and to improve current therapies for pancreatic cancer. [BMB Reports 2013; 46(3): 131-138]
Keywords: Cancer, Macrophage, Myeloid cells, Pancreatic ductal adenocarcinoma, Tumour microenvironment
PANCREATIC CANCER
Pancreatic ductal adenocarcinoma (PDA) is an aggressive malignant disease of the exocrine pancreas with a 5-year survival rate of less than 5%. PDA is the fifth most common cause of cancer deaths worldwide, leading to estimated 227,000 deaths per year (1). Risk factors for this malignant disease include family history of chronic pancreatitis, smoking (1), advancing age (2), male sex, diabetes mellitus, obesity, non-O blood group (3), occupational exposures to chlorinated hydrocarbon solvents and nickel (4), African-American ethnic origin, and a high fat diet. Although the cause of pancreatic cancer is complex and multifactorial, family history and cigarette smoking are a dominant cause for this disease.
PDAs evolve through non-invasive precursor lesions, most typically pancreatic intraepithelial neoplasias (PanIN) (Fig. 1). Early-stage pancreatic cancer is usually clinically silent, and disease only becomes apparent after the tumour invades surrounding tissues or metastasizes to distant organs. Most people who present with symptoms attributed to pancreatic cancer have advanced disease. The biggest challenge faced by clinicians treating PDA is the poor response of the tumour to therapeutic intervention. Unfortunately, unlike in other cancer types, in PDA the survival has not improved substantially in the past 30 years despite advances in conventional therapies (chemotherapy and radiotherapy) and targeted therapies that have proved successful in other cancer types, including breast, lung, colorectal and melanoma (5). Gemcitabine is currently the standard chemotoxic drug used in the treatment of PDA, but produces only a modest increase in survival (6). Although there has been some limited progress, the newer treatments show only marginal improvement in survival (Abraxane; Masitinib) and may also be rather toxic (FOLFIRINOX) (7). Currently, surgery offers the only chance of cure for some patients, and improved quality of life for many others. The majority of patients initially present with advanced and metastatic disease and only 10-15% of the patients are candidates for surgical resection (8). Therefore, new strategies to treat pancreatic cancer are sorely needed for the vast majority of patients with PDA.
Fig. 1. Infiltration of macrophages promotes pancreatic cancer disease progression. The infiltration of leukocytes, including macrophages, occurs around the lowest grade of pre-invasive pancreatic cancer lesions (PanIN) and persists through the invasive phase (PDA). Macrophages are initially recruited to the site of tumour formation in response to chemotactic factors secreted by pancreatic cancer cells. Tumour educated macrophages promote tumour progression by suppressing an anti-tumour immune response, stimulating vascularization, invasion, metastasis, and increasing the tumorigenicity of cancer cells. Together with other stromal cells including fibroblasts, stellate cells, lymphocytes, and myeloid progenitor cells, macrophages generate an excessive tumour microenvironment during PDA. The inflammatory tumour microenvironment fosters the further recruitment of macrophages, facilitates PDA progression, and is thought to blunt the response to chemotherapy.
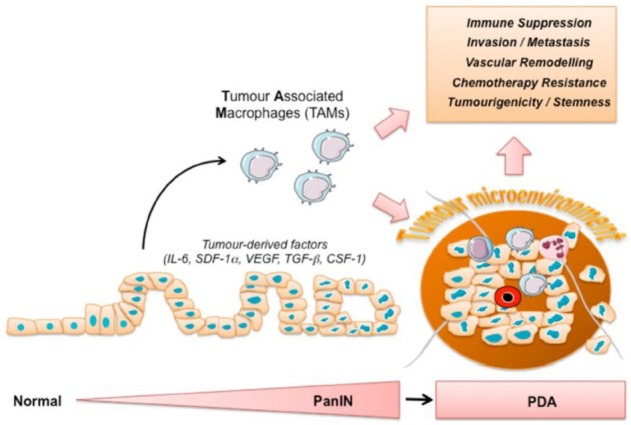
THE TUMOUR MICROENVIRONMENT IN PDA
A typical feature of PDA is the generation of a massive stromal tissue, which in some cases can make up to 80% of the tumour mass (9). It is now believed, that the excessive stromal compartment in PDA is in part responsible for the discrepancy observed between the relative success and effectiveness of therapies observed in pre-clinical experimental setting (using cell culture and xenograft mouse models) and the subsequent inefficiency observed in the clinical reality. The stromal compartment in PDA is very heterogeneous and consists of infiltrating immune cells, pancreatic stellate cells, vascular cells, fibroblasts, myofibroblasts and a dynamic assortment of extracellular matrix omponents. Although each component of the stromal compartment was described to promote pancreatic cancer disease progression (9-12), this review will discuss the recent findings of the tumour promoting functions of macrophages (Fig. 1).
MACROPHAGE CONVERSION IN PANCREATITIS AND PANCREATIC CANCER
Macrophages belong to the myeloid cell lineage and derive from myeloid progenitor cells. These precursor cells are located in the bone marrow; upon maturation, monocytes are released into the bloodstream. Circulating blood monocytes migrate into tissues where they differentiate into resident tissue macrophages. Macrophages are activated in response to environmental signals, including microbial products and cytokines. Activated macrophages can be divided into M1 (classical activated) and M2 (alternative activated) phenotype (13). Classical activation occurs in response to bacterial moieties such as lipopolysacharide (LPS) and immune stimuli such as interferon γ (IFNγ). M1 macrophages mediate resistance against intracellular parasites and tumours and elicit tissue disruptive reactions by secreting tumouricidal agents such as tumour necrosis factor α (TNF-α), interleukin-12 (IL-12), reactive nitrogen and oxygen intermediates (RNI, ROI). In addition, M1 macrophages promote T-helper-1 (Th1) responses. In contrast, M2 activated macrophages come in different varieties depending on the eliciting signals, which include IL-4, IL-13, IL-10, and glucocorticoid hormones. In general, M2 macrophages have an immune suppressive phenotype and release cytokines including IL-10 that promote a Th2 immune response (14,15).
Macrophages in tumours are usually termed tumour-associated macrophages (TAMs) and often express the M2 phenotype (16). However, recent evidence suggests that the phenotype of TAMs varies with the stage of tumour progression. M1 macrophages are mainly abundant in chronic inflammatory sites, where tumours are initiated and start to develop (17,18). During cancer progression, macrophages switch to an M2-like phenotype as the tumour begins to invade, vascularize and develop (19-21). In agreement with these findings, analysis of human patient samples using CD68 as a pan-macrophage marker and the macrophage scavenger receptor CD204 as a M2 macrophage marker revealed that more M2 converted macrophages were found in patients with pancreatic cancer compared to patients with chronic pancreatitis. High numbers of M2 macrophages were also associated with larger tumour size, early recurrence in the liver, local recurrence and shortened survival in patients with pancreatic cancer (22). These findings underline the plasticity of macrophages within the pancreas depending on their activation.
For example in acute pancreatitis, high levels of the pro-inflammatory cytokines TNF-α and IL-1β are characteristics for the onset of the disease (23,24). Analysis of human patient samples suggested that during the early inflammatory process of acute pancreatitis, TNF-α and IL-1β are not only produced by resident macrophages, but also by ascinar cells in the pancreas (25). High levels of pro-inflammatory cytokines in the pancreas induce the recruitment of monocytes and macrophages into the site of inflammation, and consequently trigger the generation of an inflammatory microenvironment. In addition, increased serum levels of TNF-α can systemically activate peritoneal and alveolar macrophages towards an M1 phenotype. However, the expression of TNF-α by infiltrating macrophages is also critical during acute pancreatitis. Using a genetic mouse model in which TNF-α was specifically depleted from the myeloid cell population markedly reduced the severity of pancreatitis (26). Similarly, neutralizing antibodies against TNF-α and genetic deletion of TNF-α prevented leukocyte-induced necrosis of acinar cells and reduced disease severity (27). Thus, for acute pancreatitis, the reprogramming of pro-inflammatory M1 macrophages towards immune suppressive and inflammation resolving M2 macrophages are emerging therapeutic strategies focused to restrain the inflammatory process. However, in the case of solid cancers, including PDA, tumour promoting TAMs rather resemble an M2-like phenotype and reprogramming of TAMs towards a M1 phenotype holds great promises in the treatment of cancers. One example of successful reactivation of macrophages in pancreatic cancer was shown by the use of and anti-CD40 antibody. In a spontaneous pancreatic tumour mouse model, administration of CD40 antibody resulted in the activation and recruitment of tumouricidal M1 macrophages to the site of pancreatic tumour formation and facilitated the depletion of tumour stroma, and consequently increased the efficacy of the chemotoxic agent gemcitabine (12).
The expression of secreted protein acidic and rich in cysteine (SPARC) has been reported to affect macrophage conversion in pancreatic cancer. SPARC is overexpressed by tumour associated fibroblasts in human and mouse pancreatic cancer and has been shown to inversely correlate with survival (28). Pancreatic tumours orthotopically grown in SPARC deficient mice were more invasive and metastatic and this was associated with reduced collagen deposition and increased macrophage infiltration. Macrophages isolated from pancreatic tumours grown in SPARC deficient mice displayed an increased M2 immune suppressive phenotype (29). However, whether a change in macrophage conversion is directly linked to SPARC expression, or whether it occurred in response to changes in the tumour microenvironment remains elusive. Of note, SPARC is also implicated in the accumulation of paclitaxel associated albumin (Abrazane) via binding of albumin to SPARC-positive tumour associated fibroblasts (30) and has provided a significant survival benefit in combination with gemcitabine in patients with elevated SPARC expression levels (31).
In general, combination of markers used to characterize macrophage conversion in mouse tumour models include F4/ 80+CD11c+CD206lowCD86+MHCIIhiiNOS+IL12+ for M1-like TAMs and F4/80+CD11cnegCD206highCD163+CD36+ARG1+IL10+IL4Rα+FIZZ1+YM+ for M2-like TAMs (32,33).
IMMUNE SUPPRESSIVE ACTIVITIES OF MACROPHAGES
In the vast majority of tumours, macrophage phenotypes are similar to the resolution phase of wound healing, wherein TAMs convey immunosuppressive activities. TAMs can convert to an immunosuppressive phenotype. Phenotyping of the transcriptome of TAMs has suggested that they express several genes with immunosuppressive potential, such as Arginase I, IL-10, TGF-β (13). Alternatively, in human ovarian and liver cancer, inhibition of cytotoxic CD8 T cells by human TAMs was associated with the expression of two members of the B7 family of co-signalling molecules (B7-H1 and B7-H4) (34,35). Moreover, alternatively activated (M2-like) macrophages express reduced levels of MHCII and CD11c, receptors involved in antigen presentation and markers for antigen presenting cells, respectively (16,36). The critical role of the immunosuppressive capacity of TAMs was recently also reported for pancreatic cancer. Indeed, reduction of TAM numbers in the pancreatic tumour microenvironment by the use of CCR2 or CSF1R inhibitors in combination with gemcitabine significantly increased CD8+ T cell, reduced FOXP3+ Treg infiltration and tumour progression compared to single agent gemcitabine, suggesting an elevated anti-tumour immune response in pancreatic cancer due to reduced macrophage numbers (37).
Beside macrophages, the immune suppressive capacity of myeloid derived suppressor cells (MDSCs) was recently reported to play a significantly role in pancreatic cancer progression (38). Using a genetically engineered mouse model of PDA, Vonderheide and colleagues reported that blockade of granulocyte-macrophage colony-stimulating factor (GM-CSF) inhibited the recruitment of MDSCs to the tumour microenvironment and reduced tumour development - a finding that was dependent on cytotoxic CD8+ T cell activation. MDSCs comprise of immature myeloid cells while macrophages are differentiated, mature myeloid cells. Since MDSCs and tumour educated macrophages both have the capacity to suppress a CTL immune response, it raises the possibility that immune suppressive TAMs are descendants of MDSCs. Although the relationship of these two cell populations is not fully understood, in the presence of tumour derived factors, MDSCs can differentiate in vitro, or after adoptive transfer into tumour bearing mice into immune suppressive macrophages (39,40).
MACROPHAGE PROMOTE TUMOUR VASCULARIZATION
In adult tissues, most blood vessels are quiescent, and angiogenesis (growth of new blood vessels from pre-existing ones) occurs only during the female reproduction cycle and under certain pathophysiological conditions, such as cancer. TAMs can secrete pro-angiogenic factors, including endothelial growth factors and extracellular matrix remodelling proteases (41-44). While the induction of new blood vessel formation during tumour growth, known as the angiogenic switch, was shown to be a necessary step for many solid cancer types, its role in pancreatic cancer is less clear. Specifically, the existence of the typical hypovascularized regions in PDA and the often impaired vascular perfusion of pancreatic tumours suggest that tumour angiogenesis might affect PDA progression to a lower extend than in other cancers. Indeed, while the anti-angiogenic drug Sunitinib, that targets VEGF and PDGF receptor signalling, showed temporal blockade of tumour growth in several tumour animal models, Sunitinib did not inhibit PDA progression in a mouse model (45). However, Sunitinib markedly reduced pancreatic tumour formation in a mouse model of endocrine origin, which is known to be highly vascularized, but which accounts in humans only for approx. 1% of pancreatic cancer (46).
However, also in pancreatic cancer macrophages can regulate tumour vascularization. Inhibition of macrophage recruitment to the tumour icroenvironment by targeting adhesion molecule integrin α4β1 or myeloid PI3Kγ resulted in a marked decrease of blood vessel formation in pancreatic cancer models and reduced tumour burden (47,48). In contrast, conversion of macrophages towards a more pronounced pro-angiogenic phenotype by depleting histidine rich glycoprotein (HRG) resulted in increased pancreatic tumour growth. Accelerated tumour formation in HRG deficient mice was in part associated with increased infiltration of M2 marker?expressingmacrophages and their increased pro-angiogenic gene expression profile resulting in excessive stimulation of tumour angiogenesis (49).
Using the endocrine pancreatic tumour model, the significance of the vascular remodelling functions of TAMs in pancreatic cancer has been recently shown in the context of anti-angiogenic therapy resistance. Since TAMs convey pro-angiogenic programs, they can counteract the efficacy of anti-angiogenic therapy. Blocking angiopoietin-2 (ANG2), a Tie2 ligand and angiogenic factor expressed by activated endothelial cells (ECs) and a subpopulation of tumour infiltrating macrophages with vascular remodelling function, regresses the tumour vasculature and inhibits progression of RIP1-Tag2 pancreatic neuroendocrine tumours. While ANG2 blockade did not inhibit recruitment of vascular remodelling macrophages, it impeded the upregulation of Tie2, their association with blood vessels, and their ability to restore angiogenesis in tumours in response to anti-antigenic therapy (50).
INVASION AND METASTASIS
Distant-site metastases are the leading cause of cancer-associated mortality. The most common places for pancreatic cancer to spread to is the liver, the peritoneum, the lungs, and the bones (46).
TAMs can promote metastasis at the primary tumour site by providing factors that enhance the invasion of malignant cells into ectopic tissues and by secreting ECM remodelling proteases and cathepsins (51).
In human pancreatic cancer, the macrophage inflammatory protein-3 alpha (MIP-3α) has been implicated as a regulator of tumour cell invasion. MIP-3α is expressed by pancreatic cancer cells as well as tumour-associated macrophages (52). MIP-3α induced MMP9 expression of pancreatic cells through its receptor CCR6 and consequently increased pancreatic cancer cell invasion in collagen Type IV (53,54). The role of TAMs in promoting pancreatic metastasis was also shown in tumour mouse models. Pharmacological inhibition of macrophage recruitment to the pancreatic tumour microenvironment using the CSF1R inhibitors reduced metastatic spreading to the liver (37), while increased macrophage conversing towards a M2 phenotype by the loss of HRG increased metastatic spreading (49). A reduction of macrophage numbers due to inhibition of their migration and reduced metastatic spreading of pancreatic cancer cells was also described in response to the focal adhesion kinase (FAK) inhibitor PF-562,271. However, PF-562,271 inhibited migration of a variety of cells present in the tumour microenvironment of pancreatic cancer, including cancer cells, fibroblasts, and immune cells, thus making the precise contribution of each cell to metastatic spreading elusive (55).
Endoneurial macrophages can foster the invasion of pancreatic cancer cells along the nerves. Specimens analysed from patients with PDA showed a significant increase in the number of endoneurial macrophages that lie around nerves invaded by cancer compared with normal nerves. These macrophages secreted high levels of glial-derived neurotrophic factor (GDNF), inducing the activation of the GDNF receptor GFRα1 and its co-receptor RET. Phosphorylation of RET induced ERK activation and the migration of pancreatic cancer cells (56).
Macrophages can promote metastatic spreading in pancreatic cancer also through their ability to promote lymph angiogenesis either by the secretion of the lymphatic endothelial growth factor VEGF-C (57) or their direct integration into tumour associated lymphatic vessels (58). In human patient samples, M2-polarized TAMs identified by CD163 and CD206 in the invasive front of pancreatic cancer were associated with a poor prognosis due to accelerated lymphatic metastasis (59). Interestingly, no correlation was found by using the pan-macrophage marker CD68, indicating that macrophage conversion can critically affect tumour progression, and not only absolute macrophage numbers. Similarly, M2 polarized TAM infiltration of regional lymphnodes was significantly associated with nodal lymphangiongenesis (60).
MACROPHAGES AND TUMOUR INITIATING CELLS/ CANCER STEM CELLS
There is increasing evidence suggesting that cancer stem cells (CSC) and/or tumour initiating cancer cells are affected by macrophages. For example, TAMs were described to increase cancer stem cell like properties of colon and lung cancer cells by promoting their tumourigenicity and resistance to chemotherapy (61). The authors described the TAM derived milk-fat globule-epidermal growth factor-VII (MFG-E8) as a major regulator of CSC activities. MFG-E8 activates STAT3 and Sonic Hedgehog pathways in CSCs. In pancreatic cancer, inhibition of macrophage recruitment to the site of tumour formation resulted in a reduction of pancreatic cancer cells expressing high levels of the CSC marker ALDH. Interestingly, in this model, tumour educated macrophage conditioned media was sufficient to enhance tumour initiating properties of murine pancreatic cancer cell lines (37).
CONCLUDING REMARKS
It has become evident in the past decade that the tumour infiltrating macrophages are critical components during cancer progression. Due to their regulatory roles, especially in vascular remodelling and tumour immunity, the presence of these cells and their state of differentiation can determine the course of the tumour progression, and the response of tumours to anti-cancer therapies (Table 1). Since TAMs are among the most frequent cells found in the tumour microenvironment and can promote tumour progression by different mechanisms, targeting macrophages emerges as an attractive and novel strategy to fight cancer, including pancreatic cancer. Indeed, studies have shown that inhibition of macrophage recruitment to the tumour microenvironment reduced pancreatic cancer progression (47) and in combination with chemotherapy markedly reduced tumour formation compared to chemotherapy alone (37,48). Besides targeting their recruitment, other studies have shown that re-activation of their cancer cell killing activities can improve the therapeutic outcome in pancreatic cancer in mouse and human (12). These findings indicate that targeting macrophages might represent an attractive novel approach to treat pancreatic cancer.
Table 1.
Tumour promoting functions of macrophages in pancreatic cancer
| Function | Mechanism | Ref. |
|---|---|---|
|
| ||
| Immune suppression | - Restored tumour immunity by CD40. | 12 |
| - Depletion of SPARC expression increases an immune suppressive TAM phenotype. | 29 | |
| Invasion/Metastasis | - Secretion of MIP3α promotes migration of cancer cells through CCR6. | 53,54 |
| - Secretion of GNDF promotes invasion of cancer cells through GDFR1α/RET. | 56 | |
| Vascular remodeling | - Host produced HRG promotes M1 TAM conversion resulting in vessel normalization. | 32,49 |
| - Blockade of ANG2 inhibits pro-angiogenic functions of TAMs. | 50 | |
| Chemoresistance/Tumourigenicity | - Inhibition of macrophage recruitment in combination with gemcitabine improves therapeutic benefits. | 37,48 |
| - Macrophage derived factors increase tumourigenicity of cancer cells through STAT3 activation. | 37 | |
Only a selection of published data are presented here.
Macrophages are part of the tumour microenvironment and can exert their tumour promoting functions also through their interaction with other stromal cells. Accumulating evidence suggests that the tumour microenvironment in pancreatic cancer may be partly responsible for the rather minor success of chemotherapy in pancreatic cancer, and might also be responsible for the discrepancy observed in xenograft tumour mouse models that do not accurately recapitulate the stromal component of the human disease. Thus, it becomes very important to test future therapeutic strategies in models reflecting the human disease in terms of both, the malignant cancer cells and the non-malignant microenvironment. To add more complexity, recent reports indicate that the cellular composition of the human microenvironment differs compared to the corresponding murine tumour microenvironment at least in breast cancer (62). Thus, it will be important to obtain a similar comparison of the murine and human tumour microenvironments in pancreatic cancer. In addition, the relationship of pancreatic cancer and pancreatitis is still poorly understood. Although only a minority of patients with pancreatitis develop pancreatic cancer, it is unknown whether patients with pancreatic cancer carried before a symptom free pancreatitis, or not. Thus, it remains elusive whether macrophages impact pancreatic cancer development differently, depending whether inflammation preceded PDA development (pancreatitis), or whether PanIN elicited an inflammatory response.
Taken together, targeting macrophages might open new avenues of research in the management of PDA.
Acknowledgments
We thank the Liverpool Cancer Research UK Centre, the National Institute for Health Research Pancreas Biomedical Research Unit, Department of Molecular and Clinical Cancer Medicine, University of Liverpool, UK, and the North West Cancer Research Fund.
References
- 1.Raimondi S., Maisonneuve P., Lowenfels A. B. Epidemiology of pancreatic cancer: an overview. Nat. Rev. Gastroenterol. Hepatol. (2009);6:699–708. doi: 10.1038/nrgastro.2009.177. [DOI] [PubMed] [Google Scholar]
- 2.Brune K. A., Lau B., Palmisano E., Canto M., Goggins M. G., Hruban R. H., Klein A. P. Importance of age of onset in pancreatic cancer kindreds. J. Natl. Cancer Inst. (2010);102:119–126. doi: 10.1093/jnci/djp466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wolpin B. M., Chan A. T., Hartge P., Chanock S. J., Kraft P., Hunter D. J., Giovannucci E. L., Fuchs C. S. ABO blood group and the risk of pancreatic cancer. J. Natl. Cancer Inst. (2009);101:424–431. doi: 10.1093/jnci/djp020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ojajarvi I. A., Partanen T. J., Ahlbom A., Boffetta P., Hakulinen T., Jourenkova N., Kauppinen T. P., Kogevinas M., Porta M., Vainio H. U., Weiderpass E., Wesseling C. H. Occupational exposures and pancreatic cancer: a meta-analysis. Occup. Environ. Med. (2000);57:316–324. doi: 10.1136/oem.57.5.316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Winter J. M., Brennan M. F., Tang L. H., D'Angelica M. I., Dematteo R. P., Fong Y., Klimstra D. S., Jarnagin W. R., Allen P. J. Survival after resection of pancreatic adenocarcinoma: results from a single institution over three decades. Ann. Surg. Oncol. (2012);19:169–175. doi: 10.1245/s10434-011-1900-3. [DOI] [PubMed] [Google Scholar]
- 6.Burris H. A. 3rd, Moore M. J., Andersen J., Green M. R., Rothenberg M. L., Modiano M. R., Cripps M. C., Portenoy R. K., Storniolo A. M., Tarassoff P., Nelson R., Dorr F. A., Stephens C. D., Von Hoff D. D. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J. Clin. Oncol. (1997);15:2403–2413. doi: 10.1200/JCO.1997.15.6.2403. [DOI] [PubMed] [Google Scholar]
- 7.Conroy T., Desseigne F., Ychou M., Bouche O., Guimbaud R., Becouarn Y., Adenis A., Raoul J. L., Gourgou-Bourgade S., de la Fouchardiere C., Bennouna J., Bachet J. B., Khemissa-Akouz F., Pere-Verge D., Delbaldo C., Assenat E., Chauffert B., Michel P., Montoto-Grillot C., Ducreux M. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. (2011);364:1817–1825. doi: 10.1056/NEJMoa1011923. [DOI] [PubMed] [Google Scholar]
- 8.Neoptolemos J. P. Adjuvant treatment of pancreatic cancer. Eur. J. Cancer. (2011);47(Suppl 3):S378–380. doi: 10.1016/S0959-8049(11)70210-6. [DOI] [PubMed] [Google Scholar]
- 9.Erkan M., Hausmann S., Michalski C. W., Fingerle A. A., Dobritz M., Kleeff J., Friess H. The role of stroma in pancreatic cancer: diagnostic and therapeutic implications. Nat. Rev. Gastroenterol. Hepatol. (2012);9:454–467. doi: 10.1038/nrgastro.2012.115. [DOI] [PubMed] [Google Scholar]
- 10.Olive K. P., Jacobetz M. A., Davidson C. J., Gopinathan A., McIntyre D., Honess D., Madhu B., Goldgraben M. A., Caldwell M. E., Allard D., Frese K. K., Denicola G., Feig C., Combs C., Winter S. P., Ireland-Zecchini H., Reichelt S., Howat W. J., Chang A., Dhara M., Wang L., Ruckert F., Grutzmann R., Pilarsky C., Izeradjene K., Hingorani S. R., Huang P., Davies S. E., Plunkett W., Egorin M., Hruban R. H., Whitebread N., McGovern K., Adams J., Iacobuzio-Donahue C., Griffiths J., Tuveson D. A. Ihibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. (2009);324:1457–1461. doi: 10.1126/science.1171362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Erez N., Truitt M., Olson P., Arron S. T., Hanahan D. Cancer-associated fibroblasts are activated in incipient neoplasia to orchestrate tumor-promoting inflammation in an NF-kappaB-Dependent manner. Cancer Cell. (2010);17:135–147. doi: 10.1016/j.ccr.2009.12.041. [DOI] [PubMed] [Google Scholar]
- 12.Beatty G. L., Chiorean E. G., Fishman M. P., Saboury B., Teitelbaum U. R., Sun W., Huhn R. D., Song W., Li D., Sharp L. L., Torigian D. A., O'Dwyer P. J., Vonderheide R. H. CD40 agonists alter tumor stroma and show efficacy against pancreatic carcinoma in mice and humans. Science. (2011);331:1612–1616. doi: 10.1126/science.1198443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Biswas S. K., Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat. Immunol. (2010);11:889–896. doi: 10.1038/ni.1937. [DOI] [PubMed] [Google Scholar]
- 14.Karp C. L, Murray P. J. Non-canonical alternatives: what a macrophage is 4. J. Exp. Med. (2012);209:427–431. doi: 10.1084/jem.20120295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gordon S., Martinez F. O. Alternative activation of macrophages: mechanism and functions. Immunity. (2010);32:593–604. doi: 10.1016/j.immuni.2010.05.007. [DOI] [PubMed] [Google Scholar]
- 16.Mantovani A., Sica A. Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr. Opin. Immunol. (2010);22:231–237. doi: 10.1016/j.coi.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 17.Greten F. R., Eckmann L., Greten T. F., Park J. M., Li Z. W., Egan L. J., Kagnoff M. F., Karin M. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. (2004);118:285–296. doi: 10.1016/j.cell.2004.07.013. [DOI] [PubMed] [Google Scholar]
- 18.Karin M., Greten F. R. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. (2005);5:749–759. doi: 10.1038/nri1703. [DOI] [PubMed] [Google Scholar]
- 19.Lin E. Y., Li J. F., Gnatovskiy L., Deng Y., Zhu L., Grzesik D. A., Qian H., Xue X. N., Pollard J. W. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. (2006);66:11238–11246. doi: 10.1158/0008-5472.CAN-06-1278. [DOI] [PubMed] [Google Scholar]
- 20.Qian B., Deng Y., Im J. H., Muschel R. J., Zou Y., Li J., Lang R. A., Pollard J. W. A distinct macrophage population mediates metastatic breast cancer cell extravasation, establishment and growth. PLoS One. (2009);4:e6562. doi: 10.1371/journal.pone.0006562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ruffell B., Affara N. I., Coussens L. M. Differential macrophage programming in the tumor microenvironment. Trends Immunol. (2012);33:119–126. doi: 10.1016/j.it.2011.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yoshikawa K., Mitsunaga S., Kinoshita T., Konishi M., Takahashi S., Gotohda N., Kato Y., Aizawa M., Ochiai A. Impact of tumor-associated macrophages on invasive ductal carcinoma of the pancreas head. Cancer Sci. (2012);103:2012–2020. doi: 10.1111/j.1349-7006.2012.02411.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sah R. P., Chari S. T. Autoimmune pancreatitis: an update on classification, diagnosis, natural history and management. Curr. Gastroenterol. Rep. (2012);14:95–105. doi: 10.1007/s11894-012-0246-8. [DOI] [PubMed] [Google Scholar]
- 24.Sah R. P., Garg P., Saluja A. K. Pathogenic mechanisms of acute pancreatitis. Curr. Opin. Gastroenterol. (2012);28:507–515. doi: 10.1097/MOG.0b013e3283567f52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dugernier T. L., Laterre P. F., Wittebole X., Roeseler J., Latinne D., Reynaert M. S., Pugin J. Compartmentalization of the inflammatory response during acute pancreatitis: correlation with local and systemic complications. Am. J. Respir. Crit. Care Med. (2003);168:148–157. doi: 10.1164/rccm.2204019. [DOI] [PubMed] [Google Scholar]
- 26.Perides G., Weiss E. R., Michael E. S., Laukkarinen J. M., Duffield J. S., Steer M. L. TNF-alpha-dependent regulation of acute pancreatitis severity by Ly-6C(hi) monocytes in mice. J. Biol. Chem. (2011);286:13327–13335. doi: 10.1074/jbc.M111.218388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sendler M., Dummer A., Weiss F. U., Kruger B., Wartmann T., Scharffetter-Kochanek K., van Rooijen N., Malla S. R., Aghdassi A., Halangk W., Lerch M. M., Mayerle J. Tumour necrosis factor alpha secretion induces protease activation and acinar cell necrosis in acute experimental pancreatitis in mice. Gut. (2013);62:430–439. doi: 10.1136/gutjnl-2011-300771. [DOI] [PubMed] [Google Scholar]
- 28.Infante J. R., Matsubayashi H., Sato N., Tonascia J., Klein A. P., Riall T. A., Yeo C., Iacobuzio-Donahue C., Goggins M. Peritumoral fibroblast SPARC expression and patient outcome with resectable pancreatic adenocarcinoma. J. Clin. Oncol. (2007);25:319–325. doi: 10.1200/JCO.2006.07.8824. [DOI] [PubMed] [Google Scholar]
- 29.Arnold S. A., Rivera L. B., Miller A. F., Carbon J. G., Dineen S. P., Xie Y., Castrillon D. H., Sage E. H., Puolakkainen P., Bradshaw A. D., Brekken R. A. Lack of host SPARC enhances vascular function and tumor spread in an orthotopic murine model of pancreatic carcinoma. Dis. Model Mech. (2010);3:57–72. doi: 10.1242/dmm.003228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Desai N., Trieu V., Yao Z., Louie L., Ci S., Yang A., Tao C., De T., Beals B., Dykes D., Noker P., Yao R., Labao E., Hawkins M., Soon-Shiong P. Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin. Cancer Res. (2006);12:1317–1324. doi: 10.1158/1078-0432.CCR-05-1634. [DOI] [PubMed] [Google Scholar]
- 31.Von Hoff D. D., Ramanathan R. K., Borad M. J., Laheru D. A., Smith L. S., Wood T. E., Korn R. L., Desai N., Trieu V., Iglesias J. L., Zhang H., Soon-Shiong P., Shi T., Rajeshkumar N. V., Maitra A., Hidalgo M. Gemcitabine plus nab-paclitaxel is an active regimen in patients with advanced pancreatic cancer: a phase I/II trial. J. Clin. Oncol. (2011);29:4548–4554. doi: 10.1200/JCO.2011.36.5742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rolny C., Mazzone M., Tugues S., Laoui D., Johansson I., Coulon C., Squadrito M. L., Segura I., Li X., Knevels E., Costa S., Vinckier S., Dresselaer T., Akerud P., De Mol M., Salomaki H., Phillipson M., Wyns S., Larsson E., Buysschaert I., Botling J., Himmelreich U., Van Ginderachter J. A., De Palma M., Dewerchin M., Claesson-Welsh L., Carmeliet P. HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PlGF. Cancer Cell. (2011);19:31–44. doi: 10.1016/j.ccr.2010.11.009. [DOI] [PubMed] [Google Scholar]
- 33.Gabrilovich D. I., Ostrand-Rosenberg S., Bronte V. Coordinated regulation of myeloid cells by tumours. Nat. Rev. Immunol. (2012);12:253–268. doi: 10.1038/nri3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuang D. M., Zhao Q., Peng C., Xu J., Zhang J. P., Wu C., Zheng L. Activated monocytes in peritumoral stroma of hepatocellular carcinoma foster immune privilege and disease progression through PD-L1. J. Exp. Med. (2009);206:1327–1337. doi: 10.1084/jem.20082173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kryczek I., Zou L., Rodriguez P., Zhu G., Wei S., Mottram P., Brumlik M., Cheng P., Curiel T., Myers L., Lackner A., Alvarez X., Ochoa A., Chen L., Zou W. B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J. Exp. Med. (2006);203:871–881. doi: 10.1084/jem.20050930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Squadrito M. L., Pucci F., Magri L., Moi D., Gilfillan G., Ranghetti A., Casazza A., Mazzone M., Lyle R., Naldini L., De Palma M. miR-511-3p Modulates Gentic Programs of Tumor-Associated Macrophages. Cell Rep. (2012);1:141–154. doi: 10.1016/j.celrep.2011.12.005. [DOI] [PubMed] [Google Scholar]
- 37.Mitchem J. B., Brennan D. J., Knolhoff B. L., Belt B. A., Zhu Y., Sanford D. E., Belaygorod L., Carpenter D., Collins L., Piwnica-Worms D., Hewitt S. M., Mallya Udupi G., Gallagher W. M., Wegner C., West B. L., Wang-Gillam A., Goedegebuure P., Linehan D. C., Denardo D. G. Targeting tumor-infiltrating macrophages decreases tumor-Initiating cells, relieves immunosuppression and improves chemotherapeutic response. Cancer Res. (2013);73:1128–1141. doi: 10.1158/0008-5472.CAN-12-2731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bayne L. J., Beatty G. L., Jhala N., Clark C. E., Rhim A. D., Stanger B. Z., Vonderheide R. H. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and T cell immunity in pancreatic cancer. Cancer Cell. (2012);21:822–835. doi: 10.1016/j.ccr.2012.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Narita Y., Wakita D., Ohkur T., Chamoto K, Nishimura T. Potential differentiation of tumor bearing mouse CD11b+Gr-1+ immature myeloid cells into both suppressor macrophages and immunostimulatory dendritic cells. Biomed. Res. (2009);30:7–15. doi: 10.2220/biomedres.30.7. [DOI] [PubMed] [Google Scholar]
- 40.Kusmartsev S., Nagaraj S., Gabrilovich D. I. Tumor-associated CD8+ T cell tolerance induced by bone marrow-derived immature myeloid cells. J. Immunol. (2005);175:4583–4592. doi: 10.4049/jimmunol.175.7.4583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lewis J. S., Landers R. J., Underwood J. C., Harris A. L., Lewis C. E. Expression of vascular endothelial growth factor by macrophages is up-regulated in poorly vascularized areas of breast carcinomas. J. Pathol. (2000);192:150–158. doi: 10.1002/1096-9896(2000)9999:9999<::AID-PATH687>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 42.Sunderkotter C., Steinbrink K., Goebeler M., Bhardwaj R., Sorg C. Macrophages and angiogenesis. J. Leukoc. Biol. (1994);55:410–422. doi: 10.1002/jlb.55.3.410. [DOI] [PubMed] [Google Scholar]
- 43.Giraudo E., Inoue M., Hanahan D. An amino-bisphosphonate targets MMP-9-expressing macrophages and angiogenesis to impair cervical carcinogenesis. J. Clin. Invest. (2004);114:623–633. doi: 10.1172/JCI200422087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hildenbrand R., Dilger I., Horlin A., Stutte H. J. Urokinase and macrophages in tumour angiogenesis. Br. J. Cancer. (1995);72:818–823. doi: 10.1038/bjc.1995.419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Olson P., Chu G. C., Perry S. R., Nolan-Stevaux O., Hanahan D. Imaging guided trials of the angiogenesis inhibitor sunitinib in mouse models predict efficacy in pancreatic neuroendocrine but not ductal carcinoma. Proc. Natl. Acad. Sci. U.S.A. (2011);108:E1275–1284. doi: 10.1073/pnas.1111079108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vincent A., Herman J., Schulick R., Hruban R. H., Goggins M. Pancreatic cancer. Lancet. (2011);378:607–620. doi: 10.1016/S0140-6736(10)62307-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Schmid M. C., Avraamides C. J., Dippold H. C., Franco I., Foubert P., Ellies L. G., Acevedo L. M., Manglicmot J. R., Song X., Wrasidlo W., Blair S. L., Ginsberg M. H., Cheresh D. A., Hirsch E., Field S. J., Varner J. A. Receptor tyrosine kinases and TLR/IL1Rs unexpectedly activate myeloid cell PI3kgamma, a single convergent point promoting tumor inflammation and progression. Cancer Cell. (2011);19:715–727. doi: 10.1016/j.ccr.2011.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schmid M. C., Avraamides C. J., Foubert P., Shaked Y., Kang S. W., Kerbel R. S., Varner J. A. Combined blockade of integrin-alpha4beta1 plus cytokines SDF-1alpha or IL-1beta potently inhibits tumor inflammation and growth. Cancer Res. (2011);71:6965–6975. doi: 10.1158/0008-5472.CAN-11-0588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tugues S., Honjo S., Konig C., Noguer O., Hedlund M., Botling J., Deschoemaeker S., Wenes M., Rolny C., Jahnen-Dechent W., Mazzone M., Claesson-Welsh L. Genetic deficiency in plasma protein HRG enhances tumor growth and metastasis by exacerbating immune escape and vessel abnormalization. Cancer Res. (2012);72:1953–1963. doi: 10.1158/0008-5472.CAN-11-2194. [DOI] [PubMed] [Google Scholar]
- 50.Mazzieri R., Pucci F., Moi D., Zonari E., Ranghetti A., Berti A., Politi L. S., Gentner B., Brown J. L., Naldini L., De Palma M. Targeting the ANG2/TIE2 axis inhibits tumor growth and metastasis by impairing angiogenesis and disabling rebounds of proangiogenic myeloid cells. Cancer Cell. (2011);19:512–526. doi: 10.1016/j.ccr.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 51.Joyce J. A., Pollard J. W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer. (2009);9:239–252. doi: 10.1038/nrc2618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kleeff J., Kusama T., Rossi D. L., Ishiwata T., Maruyama H., Friess H., Buchler M. W., Zlotnik A., Korc M. Detection and localization of Mip-3alpha/LARC/ Exodus, a macrophage proinflammatory chemokine, and its CCR6 receptor in human pancreatic cancer. Int. J. Cancer. (1999);81:650–657. doi: 10.1002/(sici)1097-0215(19990517)81:4<650::aid-ijc23>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 53.Campbell A. S., Albo D., Kimsey T. F., White S. L., Wang T. N. Macrophage inflammatory protein-3alpha promotes pancreatic cancer cell invasion. J. Surg. Res. (2005);123:96–101. doi: 10.1016/j.jss.2004.07.013. [DOI] [PubMed] [Google Scholar]
- 54.Kimsey T. F., Campbell A. S., Albo D., Wilson M., Wang T. N. Co-localization of macrophage inflammatory protein-3alpha (Mip-3alpha) and its receptor, CCR6, promotes pancreatic cancer cell invasion. Cancer J. (2004);10:374–380. doi: 10.1097/00130404-200411000-00007. [DOI] [PubMed] [Google Scholar]
- 55.Stokes J. B., Adair S. J., Slack-Davis J. K., Walters D. M., Tilghman R. W., Hershey E. D., Lowrey B., Thomas K. S., Bouton A. H., Hwang R. F., Stelow E. B., Parsons J. T., Bauer T. W. Inhibition of focal adhesion kinase by PF-562,271 inhibits the growth and metastasis of pancreatic cancer concomitant with altering the tumor microenvironment. Mol. Cancer Ther. (2011);10:2135–2145. doi: 10.1158/1535-7163.MCT-11-0261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cavel O., Shomron O., Shabtay A., Vital J., Trejo-Leider L., Weizman N., Krelin Y., Fong Y., Wong R. J., Amit M., Gil Z. Endoneurial macrophages induce perineural invasion of pancreatic cancer cells by secretion of GDNF and activation of RET tyrosine kinase receptor. Cancer Res. (2012);72:5733–5743. doi: 10.1158/0008-5472.CAN-12-0764. [DOI] [PubMed] [Google Scholar]
- 57.Schoppmann S. F., Fenzl A., Nagy K., Unger S., Bayer G., Geleff S., Gnant M., Horvat R., Jakesz R., Birner P. VEGF-C expressing tumor-associated macrophages in lymph node positive breast cancer: impact on lymphangiogenesis and survival. Surgery. (2006);139:839–846. doi: 10.1016/j.surg.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 58.Zumsteg A., Baeriswyl V., Imaizumi N., Schwendener R., Ruegg C., Christofori G. Myeloid cells contribute to tumor lymphangiogenesis. PLoS One. (2009);4:e7067. doi: 10.1371/journal.pone.0007067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kurahara H., Shinchi H., Mataki Y., Maemura K., Noma H., Kubo F., Sakoda M., Ueno S., Natsugoe S., Takao S. Significance of M2-polarized tumor- associated macrophage in pancreatic cancer. J. Surg. Res. (2011);167:e211–219. doi: 10.1016/j.jss.2009.05.026. [DOI] [PubMed] [Google Scholar]
- 60.Kurahara H., Takao S., Maemura K., Mataki Y., Kuwahata T., Maeda K., Sakoda M., Iino S., Ishigami S., Ueno S., Shinchi H., Natsugoe S. M2-Polarized tumor-associated macrophage infiltration of regional lymph nodes is associated with nodal lymphangiogenesis and occult nodal involvement in pN0 pancreatic cancer. Pancreas. (2013);42:155–159. doi: 10.1097/MPA.0b013e318254f2d1. [DOI] [PubMed] [Google Scholar]
- 61.Jinushi M., Chiba S., Yoshiyama H., Masutomi K., Kinoshita I., Dosaka-Akita H., Yagita H., Takaoka A., Tahara H. Tumor-associated macrophages regulate tumorigenicity and anticancer drug responses of cancer stem/initiating cells. Proc. Natl. Acad. Sci. U.S.A. (2011);108:12425–12430. doi: 10.1073/pnas.1106645108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ruffell B., Au A., Rugo H. S., Esserman L. J., Hwang E. S., Coussens L. M. Leukocyte composition of human breast cancer. Proc. Natl. Acad. Sci. U.S.A. (2012);109:2796–2801. doi: 10.1073/pnas.1104303108. [DOI] [PMC free article] [PubMed] [Google Scholar]


