Abstract
The ATP-inhibited Plant Mitochondrial K+ Channel (PmitoKATP) was discovered about fifteen years ago in Durum Wheat Mitochondria (DWM). PmitoKATP catalyses the electrophoretic K+ uniport through the inner mitochondrial membrane; moreover, the co-operation between PmitoKATP and +/H+ antiporter allows such a great operation of a K+ cycle to collapse mitochondrial membrane potential (ΔΨ) and ΔpH, thus impairing protonmotive force (Δp). A possible physiological role of such ΔΨ control is the restriction of harmful reactive oxygen species (ROS) production under environmental/oxidative stress conditions. Interestingly, DWM lacking Δp were found to be nevertheless fully coupled and able to regularly accomplish ATP synthesis; this unexpected behaviour makes necessary to recast in some way the classical chemiosmotic model. In the whole, PmitoKATP may oppose to large scale ROS production by lowering ΔΨ under environmental/oxidative stress, but, when stress is moderate, this occurs without impairing ATP synthesis in a crucial moment for cell and mitochondrial bioenergetics. [BMB Reports 2013; 46(8): 391-397]
Keywords: Chemiosmosis, Durum wheat, Mitochondrial potassium channel, Oxidative stress, Protonmotive force
INTRODUCTION
The existence of an ATP-inhibited mitochondrial potassium channel in plants was firstly shown in Durum Wheat Mitochondria (DWM) (1); it was named Plant mitoKATP (PmitoKATP) in analogy with the possible animal counterpart [mitoKATP; (2)]. Although the molecular nature of the channel is yet unknown (see 3 for some hypotheses about this point), we have recently confirmed the existence of a DWM cation channel inhibited by ATP referable to the original PmitoKATP by using patch clamp technique, for the first time successfully applied to plant mitochondria (3). By using swelling technique and/or by following channel-dependent membrane potential changes, other mitochondrial potassium channels were characterized in several plant species such as pea, soybean, three coniferous species, Arum, potato, maize and tomato, while less characterized potassium pathways were reported in mitochondria from bread wheat, spelt, rye, barley, spinach, topinambur, triticale, lentil and Arabidopsis (see 4-6 and refs therein). These channels may display characteristics different from that of the original PmitoKATP; for example, as reported by Ruy et al. (7), in potato, maize and tomato mitochondria, they may show ATP-insensitivity. Moreover, by using electrophysiological measurements in a reconstituted system, three different potassium channels were identified in potato tuber mitochondria: a large conductance Ca2+-activated K+ channel, the mitoBKCa (8), an ATP-sensitive mitoK, and a large-conductance Ca2+-insensitive and iberiotoxin-sensitive channel (9). So, mitochondrial K+ channels are widely present in plants and are characterized by different modulation, specificity, conductance and possible physiological role [for recent reviews see Jarmuszkiewicz et al. (10) and Pastore et al. (11)].
As for DWM, the PmitoKATP displays a conductance of 150 pS in 150 mM K+, a strong voltage dependence, relatively low selectivity and ATP inhibition with an IC50 of 0.5 mM, as calculated by De Marchi et al. (3), or Ki of about 0.3 mM, as obtained by Pastore et al. (1). As for modulators, PmitoKATP activity is mainly increased by superoxide anion, diazoxide and mersalyl, as well as by free fatty acids (FFAs) and their acyl-CoA ester derivatives (12), these latters also modulating the connected activity of the mitochondrial anion channel (13).
THE EFFECT OF PmitoKATP ON THE MITOCHONDRIAL PROTONMOTIVE FORCE IN DWM
The PmitoKATP is very active being able to completely collapse electrical membrane potential (ΔΨ) in DWM isolated in vitro in a KCl medium mimicking cell condition; 25 mM KCl are often already sufficient to do this in succinate oxidizing DWM (1,14). Moreover, the channel may act together with the K+/H+ antiporter, very active in plant mitochondria (15), generating a potassium cycle (1) that collapses also ΔpH (6) (Fig. 1).
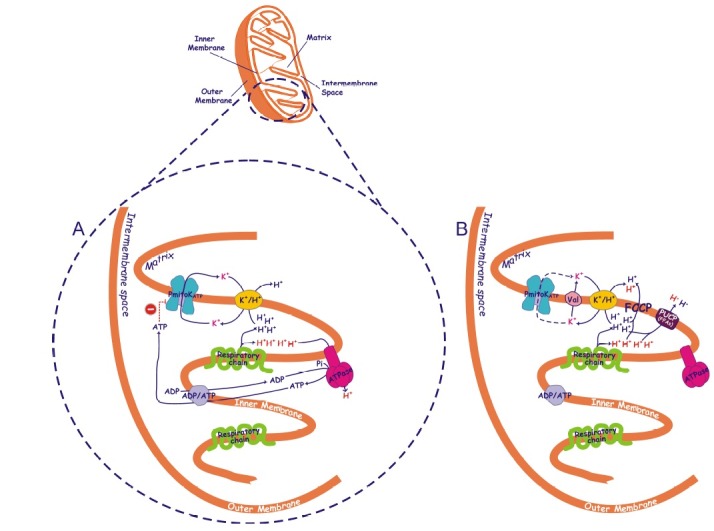
Fig. 1. Possible mechanism of coupling in the absence of measurable protonmotive force mediated by the plant mitochondrial potassium channel. A simplified picture of a durum wheat mitochondrion is reported, with mitochondrial cristae enlarged to schematize the presence in the inner mitochondrial membrane of respiratory chain, ATP synthase (ATPase), ADP/ATP and K+/H+ antiporters and ATP-inhibited plant mitochondrial potassium channel (PmitoKATP) (A) as well as of K+ ionophore valinomycin (Val), plant uncoupling protein (PUCP) activated by free fatty acids (FFAs) and chemical uncoupler carbonyl cyanide p-(trifluoromethoxy)phenylhydrazone (FCCP) (B). Partial inhibition of PmitoKATP by ATP occurring at the cytosolic face of the channel (1), may control the extent of channel activity. In the presence of high K+ concentration, the potassium cycle deriving from the cooperation of the PmitoKATP (A) or valinomycin (B) with K+/H+ antiporter may partly or fully uncouple mitochondria, respectively. Protons ejected by the respiratory chain in the course of substrate oxidation are reported in blue and in red; the first ones contribute to the measurable bulk phase ΔΨ and ΔpH, while the second ones are assumed to represent a latent non-classically measurable, localized, protonmotive force. The controlled cooperation of PmitoKATP with K+/H+ antiporter may collapse measurable bulk phase ΔΨ and ΔpH without excluding the ATP synthase pathway (A). When uncontrolled K+ uptake by valinomycin bypasses ATP brake, classical uncoupling is observed involving all protons and excluding ATP synthase (B). As expected, uncoupling is also observed when FFAs or FCCP are used (B). The scheme does not consider topology of proteins and interaction sites. For detailed explanation see the text.
In this regard, the PmitoKATP is very different from other mitochondrial potassium channels. In a recent study it was reported that the effect of potato mitoKATP on mitochondrial ΔΨ is very limited (up to few mV) (9) with respect to that of durum wheat channel. In heart mitochondria the increased K+ influx associated to potassium channel opening was small and it was able to depolarize by only 1-2 mV (16).
In rat liver mitochondria some ΔΨ decrease was observed depending on KCl concentration (up to about 20 mV at 100 mM KCl), but it was compensated by ΔpH increase so that the protonmotive force (Δp) remained almost constant (17); in the same mitochondria Devin et al. (18) showed that, for the same respiration rate, Δp was lesser in a KCl medium than in sucrose medium, but the ratio between the amount of phosphorylated ADP and oxygen consumed (ADP/O) did not vary. In respiring yeast mitochondria, addition of KCl in the presence of 4-5 mM phosphate generated some potassium cycle through electrophoretic K+ entry and electroneutral K+/H+ exchange without promoting any uncoupling between respiration and ATP synthesis, but even increasing ATP synthesis on the basis of a compensatory ΔpH increase that drives the activation of phosphate/H+ cotransporter (19,20). On the other hand, Castrejón et al. (20) and Manon and Guérin (21) showed that at low phosphate concentration (0.4-0.5 mM), mitochondrial uncoupling by KCl occurred with collapse of Δp and dramatic reduction of ADP/O and respiratory control (RC) ratios, as well as of ATP synthesis rate (about -65% than in high phosphate).
THE UNEXPECTED EFFECT OF PmitoKATP ON THE ATP SYNTHESIS IN DWM
According to Mitchell’s chemiosmotic theory the energy-rich intermediate of mitochondrial oxidative phosphorylation (OXPHOS) is the proton gradient across the inner mitochondrial membrane. Its driving force was defined by Mitchell (22,23) as the protonmotive force (Δp), consisting of an electrical (ΔΨ) and a chemical (ΔpH) part (24). A major prediction of the chemiosmotic model is that the phosphorylation potential and the rate of ATP synthesis should depend on the magnitude of the bulk Δp. As reported above, OXPHOS-dependent ATP synthesis by mitochondria suspended in a KCl medium is actually related to Δp, but, once again, the effect of PmitoKATP operation is unexpected.
As stated, fully functional DWM that oxidize succinate show negligible bulk phase ΔΨ and ΔpH in high KCl media. Anyway, DWM are equally fully coupled since they preserve ADP/O ratio and are able to regularly accomplish ATP synthesis under conditions that exclude adenylate kinase activity (6). ATP synthesis via OXPHOS has been observed in three different and independent ways, i) by following in continuous ATP synthesis and efflux from mitochondria by using an enzymatic ATP detecting system, ii) by measuring the synthesized ATP at the end of a phosphorylation cycle and iii) by oxygraphic measurements of the RC and ADP/O ratios and of the ATP synthesis rate calculated by multiplying state 3 oxygen uptake rate by ADP/O (6). KCl-treated DWM always showed ATP synthesis statistically equal to control DWM, although showing very low (60-120 mV) ΔΨ and no measurable ΔpH. On the contrary, as expected, classical uncouplers, carbonyl cyanide p-(trifluoromethoxy)phenylhydrazone (FCCP), FFAs (activating the plant uncoupling protein, PUCP) and valinomycin plus KCl are able to completely collapse both Δp and ATP synthesis.
The paradoxical behaviour of DWM may be connected with the ATP and, to a lesser extent, ADP sensitivity of the potassium channel, that we hypothesize may induce a “controlled collapse” of Δp. At this regard, it should be underlined a notable quantitative difference between PmitoKATP and its mammalian counterpart mitoKATP regarding ATP inhibition. The mitoKATP is strongly inhibited by very low ATP concentration (K0.5 = 22-40 μM) and by Mg2+ in the presence of ATP (25,26). Since the physiological ATP concentration in mammalian cells is in the millimolar range, this suggests that ATP can hardly modulate the degree of channel opening in vivo (26). On the contrary, PmitoKATP has lower affinity towards ATP with half inhibition of 0.3-0.5 mM (10- to 15-fold lower than in mitoKATP) and is insensitive to Mg2+ (1,3); these properties may allow DWM channel regulation by ATP in vivo. Indeed, on the basis of NMR analysis, it was reported that in plant cells the nucleotide triphosphate concentration ranged between 0.9 and 1.2 mM (27) with about 70% of this content represented by ATP (28). Moreover, under severe stress conditions, even a 40% drop of ATP content may be measured (6), thus reaching concentrations able to significantly mitigate channel brake.
The mechanism that induces the “controlled collapse” of Δp is summarized in Fig. 1. It is based on the ability of ATP at physiological concentration to only partially inhibit the channel, being the level of channel opening a function of the changes of cytosolic ATP concentration in the balance with the effect of different activators (see below). Since PmitoKATP represents the rate-limiting step of the potassium cycle (1), the inhibition by ATP, although partial, lowers the potassium cycle rate, thus preventing complete uncoupling. As a result of the controlled activity of the channel, the potassium cycle may strongly lower measurable bulk phase Δp, but it appears unable to compete with ATP synthase for protons, so the existence of a latent proton movement non-classically detectable may be preserved (Fig. 1A). Consistently, the addition of valinomycin, that overcomes ATP inhibition and maximize potassium uptake and cycle, causes a complete uncoupling. Similarly, classical uncouplers such as FFAs, that activate the PUCP, or FCCP completely uncouple mitochondria (Fig. 1B). In practice, while the classical uncouplers are unable to distinguish among different proton pools, somehow the PmitoKATP/ATP system appears to be able to distinguish the bulk phase Δp from a non-classically detectable driving force for ATP synthesis. The magnitude of this phenomenon will depend on channel opening, i.e. on the balance among channel activators and inhibitors. This mechanism is in accordance with that probably operating in other similar biological systems.
Other biological systems troubling classical chemiosmosis
Some other energy-transducing membranes were shown to trouble the statement of classical chemiosmosis. ATP synthesis was detected even in the presence of an inverted ΔpH, alkaline outside, in extreme alkaliphilic bacteria (29). The attenuation of the rate of succinate oxidation resulted in a parallel decrease in the rate of ATP synthesis with little or no change in Δp in bovine hearth submitochondrial particles (30). Light-induced ATP synthesis was found to occur in the absence of an apparent ΔΨ or ΔpH in both Halobacterium halobium (31) and thylakoid vesicles (32). As a result of the accumulation of these kind of observations it was speculated that the delocalized transmembrane Δp cannot be the principal driving force for ATP synthesis (33); so, it was hypothesized that a localized rather than delocalized energy transfer between the electron transfer complexes and the ATP synthase may occur (34-37). These findings may be explained by assuming that a proton transfer through direct protein-protein interaction exists (38,39). Alternatively, protons generated at the surface of the membrane may diffuse laterally through the surface polar groups of phospholipids or the organized water at the surface (40-42). In any case, it has been proposed that the coupling between proton donor and acceptor sites can be direct, without involving the bulk phase, also at a considerable distance of either nanometers or even micrometers (43-46), so, the localized protons could be directly coupled to ATP synthesis when the protons are channeled through the ATP synthase or alternatively exchanged with ions at symports or antiports (46).
THE POSSIBLE PHYSIOLOGICAL ROLE OF PmitoKATP
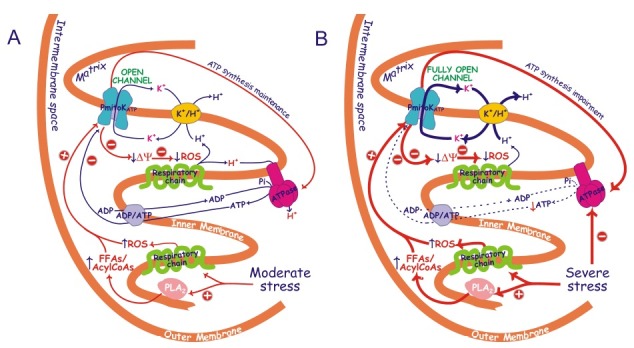
The control of ΔΨ may allow the control of reactive oxygen species (ROS) production (47), so this property of the PmitoKATP sets out its possible physiological role. Really, opening/closure of the channel in a 100 mM KCl medium may vary up to about 35-fold superoxide anion production (6). It is well known that cellular ROS production can be increased as a result of plant exposure to various environmental stresses, thus inducing oxidative stress (48-50). Mitochondria, in particular, were reported to increase ROS generation under drought and salt stress (51). The deriving hypothesis that PmitoKATP may operate as defense against these stresses in DWM together with alternative oxidase (52) and PUCP (53) was demonstrated: an increase in channel activity up to fourfold in mitochondria purified from osmotic- and salt-stressed durum wheat seedlings and a concurrent decrease (about 60%) of mitochondrial ROS generation was observed (53). Under conditions of hyperosmotic stress an increase of channel activators such as ROS and FFAs/AcylCoA esters (53,12), the latters deriving from the activation (up to about two times) of a mitochondrial PLA2 (54), is observed. Under moderate hyperosmotic stress conditions inducing a starting cellular oxidative stress, but not a damage on substrate oxidation, on ATP synthesis and mitochondria intactness (55,56), activation of the channel may induce ΔΨ decrease and control of ROS production, but, according to the mechanism of Fig. 1A, may preserve ATP synthesis just when the cell has much more need of ATP to overcome the insult (Fig. 2A).
Fig. 2. Possible mechanism of PmitoKATP modulation by FFAs/acylCoAs, ROS and ATP under moderate (A) and severe (B) hyperosmotic stress conditions. Schematization is as in Fig. 1, PLA2 is a mitochondrial phospholipase A2. Under moderate stress conditions (A) an increase in FFAs due to PLA2 activity and probably of their acylCoA derivatives is observed in DWM, as well as an increased ROS production by respiratory chain, thus leading to PmitoKATP activation. At the same time, channel inhibition by ATP is able to carefully regulate the rate of K+ cycle due to the PmitoKATP - K+/H+ combined function, so that the measurable bulk phase ΔΨ/ΔpH (blue protons, see also Fig. 1) can be lowered, thus dampening excess harmful ROS generation; under these conditions, however, ATP synthesis can be maintained using the latent localized protonmotive force (red protons) as proposed in Fig. 1A. Under severe stress (B), ATP synthesis is inhibited; therefore, PmitoKATP activation by ROS and by PLA2/FFAs/acylCoAs pathway may greatly overcome ATP inhibition. Functioning of fully open PmitoKATP may actively prevent large scale ROS production, but it may be so high to collapse both bulk phase and localized ΔΨ to such an extent to impair ATP synthesis. The scheme does not consider topology of proteins and interaction sites. The large arrows refer to a more active pathway. Abbreviations are as in Fig. 1. ROS, reactive oxygen species; ↑, increase; ↓, decrease; ⊕, activation; ⊖, inhibition.
One can argue that ATP synthase cannot work under low force condition. Really, ΔΨ and ΔpH are not kinetically equivalent driving forces for ATP synthase. ΔΨ represents the essential driving force for rotation of the “rotor” γεcn of the synthase; one turn of rotation of the γεcn part yields three ATP driven by the translocation of protons through c subunits (57 and refs therein). The ΔΨ required is a function of H+/ATP stoichiometry that depends, in turn, on the number of the c subunits in Fo rotating ring. For example, in Escherichia coli, and probably in mammalian mitochondria, 100-120 mV are assumed to be necessary for maximal ATP synthesis by the ATP synthase that has probably 9-10 c subunits, so giving calculated H+/ATP equal to 3-3.3; anyway, about 70-80 mV are sufficient to obtain midpoint potential (58). Unfortunately, the number of c subunits of ATP synthase in DWM is so far unknown thus preventing H+/ATP calculation; moreover, possible alternative calculation of thermodynamic H+/ATP stoichiometry as ΔGp/Δp is unlikely due to an unspecific proton leak of the inner membrane typical of mitochondria, preventing a thermodynamic equilibrium (59). However, it should be noted that in plants, for ATP synthesis by chloroplast ATP synthase, saturation is already obtained at only 50-60 mV, this enzyme having 14 c subunits, so giving calculated H+/ATP equal to 4.7 (58). This shows that ATP synthases may be able to synthesize ATP at rather low membrane potential. Consistently, in vivo, low mitochondrial ΔΨ has been often measured. In plant cells, mitochondrial ΔΨ was estimated on the basis of the subcellular ATP/ADP ratio measured by means of rapid subcellular fractionation of barley leaf protoplasts; interestingly, ΔΨs ranging from 70 to 95 mV under different physiological conditions were calculated (60). As for mammalian cells, mitochondrial ΔΨs of about 105 mV in fibroblasts and 81 mV in neuroblastoma cells were measured (61), these results were obtained by applying a novel method using the combination of conventional fluorescence microscopy and three-dimensional deconvolution by exhaustive photon reassignment. ΔΨs ranging from about 100 to 115 mV were measured under different metabolic conditions in perfused rat hearts under high cardiac work (62). Really, in vitro values of ΔΨ measured in DWM under PmitoKATP operation in a KCl medium ranged from 60 to 120 mV in different experiments (14, 6, and unpublished data). These ΔΨ values fit well with the above measurements in vivo, thus suggesting that in DWM under stress, ATP synthesis at suboptimal ΔΨ may be possible by an energetic point of view. This may keep in balance mitochondrial/cellular bioenergetics and ROS control under controlled stress conditions as depicted in Fig. 2A.
On the other hand, if the stress becomes so severe as to induce a drop of substrate oxidation (53,63) and of ATP synthesis (55) inducing remarkable ATP content decrease (6,56), a substantial decrease of channel inhibition by ATP may be observed. Under these extreme conditions up to about 25 times increase of FFAs has been also observed (12). So, the decrease of the inhibitor along with the notable increase of an activator may strongly activate the channel and the potassium cycle, thus controlling large scale ROS production; anyway, under these conditions the fully open channel may impair ATP synthesis (6) (Fig. 2B).
In conclusion, the uniqueness of the plant PmitoKATP regarding effects on protonmotive force, ATP synthesis and ROS control may be considered as a complex basic mechanism to adapt mitochondrial and cellular bioenergetics to changing environmental conditions and to oppose oxidative stress.
Acknowledgments
This work was supported by grants from the Italian Ministry of Education, University and Research (MIUR) project ‘AGROGEN’.
References
- 1.Pastore D., Stoppelli M. C., Di Fonzo N., Passarella S. The existence of the K+ channel in plant mitochondria. J. Biol. Chem. (1999);274:26683–26690. doi: 10.1074/jbc.274.38.26683. [DOI] [PubMed] [Google Scholar]
- 2.Paucek P., Mironova G., Mahdi F., Beavis A. D., Woldegiorgis G., Garlid K. D. Reconstitution and partial purification of the glibenclamide-sensitive, ATP-dependent K+ channel from rat liver and beef heart mitochondria. J. Biol. Chem. (1992);267:26062–26069. [PubMed] [Google Scholar]
- 3.De Marchi U., Checchetto V., Zanetti M., Teardo E., Soccio M., Formentin E., Giacometti G. M., Pastore D., Zoratti M., Szabò I. ATP-sensitive cation-channel in wheat (Triticum durum Desf.): identification and characterization of a plant mitochondrial channel by patch-clamp. Cell. Physiol. Biochem. (2010);26:975–982. doi: 10.1159/000324010. [DOI] [PubMed] [Google Scholar]
- 4.Petrussa E., Bertolini A., Krajnáková J., Casolo V., Macrì F., Vianello A. Isolation of mitochondria from embryogenic cultures of Picea abies (L.) Karst. and Abies cephalonica Loud.: characterization of a K+ATP channel. Plant Cell Rep. (2008);27:137–146. doi: 10.1007/s00299-007-0436-2. [DOI] [PubMed] [Google Scholar]
- 5.Petrussa E., Casolo V., Peresson C., Krajnáková J., Macrì F., Vianello A. Activity of a K+ATP channel in Arum spadix mitochondria during thermogenesis. J. Plant Physiol. (2008);165:1360–1369. doi: 10.1016/j.jplph.2007.10.012. [DOI] [PubMed] [Google Scholar]
- 6.Trono D., Soccio M., Laus M. N., Pastore D. Potassium channel-oxidative phosphorylation relationship in durum wheat mitochondria from control and hyperosmotic-stressed seedlings. Plant Cell Environ. (2011);34:2093–2108. doi: 10.1111/j.1365-3040.2011.02407.x. [DOI] [PubMed] [Google Scholar]
- 7.Ruy F., Vercesi A. E., Andrade P. B. M., Bianconi M. L., Chaimovich H., Kowaltowski A. J. A highly active ATP-insensitive K+ import pathway in plant mitochondria. J. Bioenerg. Biomembr. (2004);36:195–202. doi: 10.1023/B:JOBB.0000023623.70126.04. [DOI] [PubMed] [Google Scholar]
- 8.Koszela-Piotrowska I., Matkovic K., Szewczyk A., Jarmuszkiewicz W. A large-conductance calcium-activated potassium channel in potato (Solanum tuberosum) tuber mitochondria. Biochem. J. (2009);424:307–316. doi: 10.1042/BJ20090991. [DOI] [PubMed] [Google Scholar]
- 9.Matkovic K., Koszela-Piotrowska I., Jarmuszkiewicz W., Szewczyk A. Ion conductance pathways in potato tuber (Solanum tuberosum) inner mitochondrial membrane. Biochim. Biophy. Acta. (2011);1807:275–285. doi: 10.1016/j.bbabio.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 10.Jarmuszkiewicz W., Matkovic K., Koszela-Piotrowska I. Potassium channels in the mitochondria of unicellular eukaryotes and plants. FEBS Lett. (2010);584:2057–2062. doi: 10.1016/j.febslet.2010.01.023. [DOI] [PubMed] [Google Scholar]
- 11.Pastore D., Laus M. N., Soccio M. Plant mitochondrial potassium channel or channels? In Mitochondria: Structure, Function and Dysfunction. Nova Science Publishers,Inc.; Hauppauge, NY: (2010). pp. 883–898. [Google Scholar]
- 12.Laus M. N., Soccio M., Trono D., Liberatore M. T., Pastore D. Activation of the plant mitochondrial potassium channel by free fatty acids and acyl-CoA esters: a possible defence mechanism in the response to hyperosmotic stress. J. Exp. Bot. (2011);62:141–154. doi: 10.1093/jxb/erq256. [DOI] [PubMed] [Google Scholar]
- 13.Laus M. N., Soccio M., Trono D., Cattivelli L., Pastore D. Plant Inner Membrane Anion Channel (PIMAC) function in plant mitochondria. Plant Cell Physiol. (2008);49:1039–1055. doi: 10.1093/pcp/pcn082. [DOI] [PubMed] [Google Scholar]
- 14.Pastore D., Trono D., Laus M. N., Di Fonzo N., Flagella Z. Possible plant mitochondria involvement in cell adaptation to drought stress. A case study: durum wheat mitochondria. J. Exp. Bot. (2007);58:195–210. doi: 10.1093/jxb/erl273. [DOI] [PubMed] [Google Scholar]
- 15.Diolez P., Moreau F. Correlations between ATP synthesis, membrane potential and oxidation rate in plant mitochondria. Biochim. Biophys. Acta. (1985);806:56–63. doi: 10.1016/0005-2728(85)90081-7. [DOI] [Google Scholar]
- 16.Kowaltowski A. J., Seetharama S., Paucek P., Garlid K. D. Bioenergetic consequences of opening the ATP-sensitive K+ channel of heart mitochondria. Am. J. Physiol. Heart Circ. Physiol. (2001);280:H649–H657. doi: 10.1152/ajpheart.2001.280.2.H649. [DOI] [PubMed] [Google Scholar]
- 17.Czyz A., Szewczyk A., Nalecz M. J., Wojtczak L. The role of mitochondrial potassium fluxes in controlling the protonmotive force in energized mitochondria. Biochem. Biophys. Res. Co. (1995);210:98–104. doi: 10.1006/bbrc.1995.1632. [DOI] [PubMed] [Google Scholar]
- 18.Devin A., Guérin B., Rigoulet M. Response of isolated rat liver mitochondria to variation of external osmolarity in KCl medium: regulation of matrix volume and oxidative phosphorylation. J. Bioenerg. Biomembr. (1997);29:579–590. doi: 10.1023/A:1022435102552. [DOI] [PubMed] [Google Scholar]
- 19.Manon S., Roucou X., Rigoulet M., Guérin M. Stimulation of oxidative phosphorylation by electrophoretic K+ entry associated to electroneutral K+/H+ exchange in yeast mitochondria. Biochim. Biophys. Acta. (1995);1231:282–288. doi: 10.1016/0005-2728(95)00088-Z. [DOI] [PubMed] [Google Scholar]
- 20.Castrejón V., Parra C., Moreno R., Pena A., Uribe S. Potassium collapses the ΔP in yeast mitochondria while the rate of ATP synthesis is inhibited only partially: modulation by phosphate. Arch. Biochem. Biophys. (1997);346:37–44. doi: 10.1006/abbi.1997.0273. [DOI] [PubMed] [Google Scholar]
- 21.Manon S., Guérin M. The ATP-induced K+-transport pathway of yeast mitochondria may function as an uncoupling pathway. Biochim. Biophys. Acta. (1997);1318:317–321. doi: 10.1016/S0005-2728(96)00160-0. [DOI] [PubMed] [Google Scholar]
- 22.Mitchell P. Coupling of phosphorylation to electron and hydrogen transfer by a chemiosmotic type of mechanism. Nature. (1961);191:144–148. doi: 10.1038/191144a0.. [DOI] [PubMed] [Google Scholar]
- 23.Mitchell P. Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol. Rev. (1966);41:445–502. doi: 10.1111/j.1469-185X.1966.tb01501.x. [DOI] [PubMed] [Google Scholar]
- 24.Nicholls D. G., Ferguson S. J. The chemiosmotic proton circuit. In Bioenergetics. 2nd ed. Academic press limited; London: (1992). pp. 82–87. [Google Scholar]
- 25.Garlid K. D. Cation transport in mitochondria - the potassium cycle. Biochim. Biophys. Acta. (1996);1275:123–126. doi: 10.1016/0005-2728(96)00061-8. [DOI] [PubMed] [Google Scholar]
- 26.Garlid K. D., Paucek P. Mitochondrial potassium transport: the K+ cycle. Biochim. Biophys. Acta. (2003);1606:23–41. doi: 10.1016/S0005-2728(03)00108-7. [DOI] [PubMed] [Google Scholar]
- 27.Gout E., Bligny R., Douce R. Regulation of intracellular pH values in higher plant cells. Carbon-13 and phosphorous-31 nuclear magnetic resonance studies. J. Biol. Chem. (1992);267:13903–13909. [PubMed] [Google Scholar]
- 28.Roby C., Martin J.-B., Bligny R., Douce R. Biochemical changes during sucrose deprivation in higher plant cells. Phosphorous-31 nuclear magnetic resonance studies. J. Biol. Chem. (1987);262:5000–5007. [PubMed] [Google Scholar]
- 29.Krulwich T. A. Alkaliphiles: 'basic' molecular problems of pH tolerance and bioenergetics. Mol. Microbiol. (1995);15:403–410. doi: 10.1111/j.1365-2958.1995.tb02253.x. [DOI] [PubMed] [Google Scholar]
- 30.Sorgato M. C., Branca D., Ferguson S. J. The rate of ATP synthesis by submitochondrial particles can be independent of the magnitude of the protonmotive force. Biochem. J. (1980);188:945–948. doi: 10.1042/bj1880945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Michel H., Oesterhelt D. Electrochemical proton gradient across the cell membrane of Halobacterium halobium: Comparison of the light-induced increase with the increase of intracellular adenosine triphosphate under steady-state illumination. Biochemistry. (1980);19:4615–4619. doi: 10.1021/bi00561a012. [DOI] [PubMed] [Google Scholar]
- 32.Ort D. R., Dilley R. A., Good N. E. Photophosphorylation as a function of illumination time. II. Effects of permeant buffers. Biochim. Biophys. Acta. (1976);449:108–124. doi: 10.1016/0005-2728(76)90011-6. [DOI] [PubMed] [Google Scholar]
- 33.Matsuno-Yagi A., Hatefi Y. Kinetic modalities of ATP synthesis. Regulation by the mitochondrial respiratory chain. J. Biol. Chem. (1986);261:14031–14038. [PubMed] [Google Scholar]
- 34.Hitchens G. D., Kell D. B. On the extent of localization of the energized membrane state in chromatophores from Rhodopseudomonas capsulata N22. Biochem. J. (1982);206:351–357. doi: 10.1042/bj2060351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rottenberg H. Uncoupling of oxidative phosphorylation in rat liver mitochondria by general anesthetics. Proc. Natl. Acad. Sci. U.S.A. (1983);80:3313–3317. doi: 10.1073/pnas.80.11.3313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Westerhoff H. V., Melandri B. A., Venturoli G., Azzone G. F., Kell D. B. A minimal hypothesis for membrane-linked free-energy transduction. The role of independent, small coupling units. Biochim. Biophys. Acta. (1984);768:257–292. doi: 10.1016/0304-4173(84)90019-3. [DOI] [PubMed] [Google Scholar]
- 37.Westerhoff H. V., Melandri B. A., Venturoli G., Azzone G. F., Kell D. B. Mosaic protonic coupling hypothesis for free energy transduction. FEBS Lett. (1984);165:1–5. doi: 10.1016/0014-5793(84)80002-2. [DOI] [PubMed] [Google Scholar]
- 38.Qui Z. H., Yu L., Yu C. A. Spin-label electron paramagnetic resonance and differential scanning calorimetry studies of the interaction between mitochondrial cytochrome c oxidase and adenosine triphosphate synthase complex. Biochemistry. (1992);31:3297–3302. doi: 10.1021/bi00127a036. [DOI] [PubMed] [Google Scholar]
- 39.Gupte S. S., Chazotte B., Leesnitzer M. A., Hackenbrock C. R. Two-dimensional diffusion of F1F-ATP synthase and ADP/ATP translocator. Testing a hypothesis for ATP synthesis in the mitochondrial inner membrane. Biochim. Biophys. Acta. (1991);1069:131–138. doi: 10.1016/0005-2736(91)90114-N. [DOI] [PubMed] [Google Scholar]
- 40.Heberle J., Riesle J., Thiedemann G., Oesterhelt D., Dencher N. A. Proton migration along the membrane surface and retarded surface to bulk transfer. Nature. (1994);370:379–382. doi: 10.1038/370379a0. [DOI] [PubMed] [Google Scholar]
- 41.Scherrer P., Alexiev U., Marti T., Khorana H. G., Heyn M. P. Covalently bound pH-indicator dyes at selected extracellular or cytoplasmic sites in bacteriorhodopsin. 1. Proton migration along the surface of bacteriorhodopsin micelles and delayed transfer from surface to bulk. Biochemistry. (1994);33:13684–13692. doi: 10.1021/bi00250a019. [DOI] [PubMed] [Google Scholar]
- 42.Alexiev U., Mollaaghababa R., Scherrer P., Khorana H. G., Heyn M. P. Rapid long-range proton diffusion along the surface of the purple membrane and delayed proton transfer into the bulk. Proc. Natl. Acad. Sci. U.S.A. (1995);92:372–376. doi: 10.1073/pnas.92.2.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.de Grey A. D. Incorporation of transmembrane hydroxide transport into the chemiosmotic theory. Bioelectrochem. Bioenerg. (1999);49:43–50. doi: 10.1016/S0302-4598(99)00064-1. [DOI] [PubMed] [Google Scholar]
- 44.Georgievskii Y., Medvedev E. S., Stuchebrukhov A. A. Proton transport via the membrane surface. Biophys. J. (2002);82:2833–2846. doi: 10.1016/S0006-3495(02)75626-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Serowy S., Saparov S. M., Antonenko Y. N., Kozlovsky W., Hagen V., Pohl P. Structural proton diffusion along lipid bilayers. Biophys. J. (2003);84:1031–1037. doi: 10.1016/S0006-3495(03)74919-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tedeschi H. Old and new data, new issues: the mitochondrial ΔΨ. Biochim. Biophys. Acta. (2005);1709:195–202. doi: 10.1016/j.bbabio.2005.07.008. [DOI] [PubMed] [Google Scholar]
- 47.Skulachev V. P. The latest news of the sodium world. Biochim. Biophys. Acta. (1994);1187:216–221. doi: 10.1016/0005-2728(94)90114-7. [DOI] [Google Scholar]
- 48.Scandalios J. G. Oxygen stress and superoxide dismutases. Plant Physiol. (1993);101:7–12. doi: 10.1104/pp.101.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Foyer C. H., Lescure J. C., Lefebvre C., Morot-Gaudry J. F., Vincentz M., Vaucheret H. Adaptations of photosynthetic electron transport, carbon assimilation, and carbon partitioning in transgenic Nicotiana plumbaginifolia plants to changes in nitrate reductase activity. Plant Physiol. (1994);104:171–178. doi: 10.1104/pp.104.1.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Møller I. M. Plant mitochondria and oxidative stress: electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu. Rev. Plant Physiol. Plant Mol. Biol. (2001);52:561–591. doi: 10.1146/annurev.arplant.52.1.561. [DOI] [PubMed] [Google Scholar]
- 51.Alscher R. G., Donahue J. L., Cramer C. L. Reactive oxygen species and antioxidants: relationships in green cells. Physiol. Plantarum. (1997);100:224–233. doi: 10.1111/j.1399-3054.1997.tb04778.x. [DOI] [Google Scholar]
- 52.Pastore D., Trono D., Laus M. N., Di Fonzo N., Passarella S. Alternative oxidase in durum wheat mitochondria. Activation by pyruvate, hydroxypyruvate and glyoxylate and physiological role. Plant Cell Physiol. (2001);42:1373–1382. doi: 10.1093/pcp/pce174. [DOI] [PubMed] [Google Scholar]
- 53.Trono D., Flagella Z., Laus M. N., Di Fonzo N., Pastore D. The uncoupling protein and the potassium channel are activated by hyperosmotic stress in mitochondria from durum wheat seedlings. Plant Cell Environ. (2004);27:437–448. doi: 10.1111/j.1365-3040.2003.01162.x. [DOI] [Google Scholar]
- 54.Trono D., Soccio M., Laus M. N., Pastore D. The existence of phospholipase A2 activity in plant mitochondria and its activation by hyperosmotic stress in durum wheat (Triticum durum Desf.). Plant Sci. (2013);199-200:91–102. doi: 10.1016/j.plantsci.2012.11.002. [DOI] [PubMed] [Google Scholar]
- 55.Flagella Z., Trono D., Pompa M., Di Fonzo N., Pastore D. Seawater stress applied at germination affects mitochondrial function in durum wheat (Triticum durum) early seedlings. Funct. Plant Biol. (2006);33:357–366. doi: 10.1071/FP05244. [DOI] [PubMed] [Google Scholar]
- 56.Soccio M., Laus M. N., Trono D., Pastore D. A new simple fluorimetric method to assay cytosolic ATP content. Application to durum wheat seedlings to assess modulation of mitochondrial potassium channel and uncoupling protein activity under hyperosmotic stress. Biologia. (2013);68:421–432. doi: 10.2478/s11756-013-0176-4. [DOI] [Google Scholar]
- 57.Dimroth P., Kaim G., Matthey U. Crucial role of the membrane potential for ATP synthesis by F1Fo ATP synthases. J. Exp. Biol. (2000);203:51–59. doi: 10.1242/jeb.203.1.51. [DOI] [PubMed] [Google Scholar]
- 58.Kaim G., Dimroth P. ATP synthesis by F-type ATP synthase is obligatorily dependent on the transmembrane voltage. EMBO J. (1999);18:4118–4127. doi: 10.1093/emboj/18.15.4118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kadenbach B. Intrinsic and extrinsic uncoupling of oxidative phosphorylation. Biochim. Biophys. Acta. (2003);1604:77–94. doi: 10.1016/S0005-2728(03)00027-6. [DOI] [PubMed] [Google Scholar]
- 60.Igamberdiev A. U., Kleczkowski L. A. Membrane potential, adenylate levels and Mg2+ are interconnected via adenylate kinase equilibrium in plant cells. Biochim. Biophys. Acta. (2003);1607:111–119. doi: 10.1016/j.bbabio.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 61.Zhang H., Huang H. M., Carson R. C., Mahmood J., Thomas H. M., Gibson G. E. Assessment of membrane potentials of mitochondrial populations in living cells. Anal. Biochem. (2001);298:170–180. doi: 10.1006/abio.2001.5348. [DOI] [PubMed] [Google Scholar]
- 62.Wan B., Doumen C., Duszynski J., Salama G., Vary T. C., LaNoue K. F. Effects of cardiac work on electrical potential gradient across mitochondrial membrane in perfused rat hearts. Am. J. Physiol. (1993);265:H453–H460. doi: 10.1152/ajpheart.1993.265.2.H453. [DOI] [PubMed] [Google Scholar]
- 63.Soccio M., Laus M. N., Spera G. P., Trono D., Pompa M., Flagella Z., Pastore D. Mitochondrial proline oxidation is affected by hyperosmotic stress in durum wheat seedlings. Ann. Appl. Biol. (2010);157:1–11. doi: 10.1111/j.1744-7348.2010.00392.x. [DOI] [Google Scholar]


