Abstract
Nasopharyngeal carcinoma (NPC) is a common viral-associated neoplasm in which multiple signaling cascades are interfered with by Epstein-Bar virus (EBV) latent proteins and various genetic alterations. Aside from the previously reported PIK3CA amplification, we examined the role of INPP4B, a negative regulator of the PI3K/AKT signaling pathway in the development of NPC. By RT-PCR and Western blotting, we revealed that the expression of INPP4B was down-regulated in all five established EBV-positive tumor lines. While INPP4B was consistently expressed in normal nasopharyngeal epithelial cells, downregulation of INPP4B was found in 32/65 (49.2%) of primary tumors by immunohistochemistry. Furthermore, our study also demonstrated the hypermethylation of the 5′CpG island of INPP4B in the tumors in which INPP4B transcription was downregulated. Notably, the re-expression of INPP4B was detected in the NPC cells treated with the demethylation agent (5-aza-2′deoxycytidine). Our study showed that promoter hypermethylation was the major mechanism for transcriptional silencing of INPP4B in NPC. Furthermore, restoration of INPP4B expression significantly suppressed PI3K/AKT downstream signals in the NPC C666-1 cells. In vivo growth inhibition was clearly demonstrated in the tumor cells stably expressing INPP4B. The findings indicate that epigenetic inactivation of INPP4B is one of the key mechanisms in activating PI3K/AKT signaling cascade and playing a role in the tumorigenesis of NPC.
Introduction
The PI3K/AKT pathway regulates a number of cellular processes, such as cell growth, proliferation, apoptosis, migration, angiogenesis, and glucose metabolism [1]–[3]. PI3K is a major signaling component that transduces signals from various growth factors and cytokines into intracellular messages by generating the second messenger phosphatidylinositol 3,4,5-trisphosphate (Ptdlns(3,4,5)P3) from phosphatidylinositol 4,5-bisphosphate (Ptdlns(4,5)P2) [4]–[5]. This recruits AKT to the plasma membrane where it is subsequently phosphorylated at Threonine 308 and Serine 473 by PDK1 and PDK2 (mTOR2) respectively [6]. Activated AKT translocates to the cytoplasm and nucleus and phosphorylates many downstream targets (e.g. GSK3-beta, FKHRL1, BAD, mTOR, and 4E-BP1) that promote cell proliferation and inhibit apoptosis [1]–[3], [7]–[8]. The tumor suppressor PTEN functions as an antagonist of PI3K [9]. It is a 3-position lipid phosphatase that coverts Ptdlns(3,4,5)P3 back to Ptdlns(4,5)P2 and thus shuts off PI3K/AKT signaling [9]–[10]. Multiple studies have revealed altered expression or mutation of many components (e.g. PIK3CA, PTEN, AKT) of the PI3K/AKT pathway in a broad range of human cancers [1]–[3], [10]–[12]. These genetic and epigenetic changes promote proliferation and survival of tumor cells by activating AKT kinase activity.
Nasopharyngeal carcinoma (NPC) is a distinctive type of head and neck cancer that is closely associated with latent Epstein-Barr (EBV) infection and has a unique pattern of genomic changes [13]. We and others have demonstrated that the regulation of multiple signaling pathways (e.g. NF-kappaB) is disrupted by either viral factors or somatic alterations [14]–[20]. For the PI3K/AKT signaling cascade, Morrison et al (2004) first demonstrated that activated AKT in majority of primary NPC [16]. The constitutive activation of PI3K/AKT signalling pathway may contribute to cell proliferation, survival, migration and genomic instability of this epithelial cancer. In the NPC cases with LMP1 or LMP2A expression, the PI3K/AKT signalling pathway is thought to be activated by these viral latent proteins [17]–[18], [21]. Notably, amplification and mutation of PIK3CA were also reported in 10–20% of this EBV-associated epithelial cancer [22]–[23]. The findings suggest that both viral oncoproteins and genetic alterations contribute to the dysregulation of PI3K/AKT signaling. Recently, Gewinner et al. showed that inositol polyphosphate 4-phosphatase type II (INPP4B) plays a critical role in suppressing PI3K/AKT pathway [5]. INPP4B hydrolyzes phosphatidylinositol-3,4-bisphosphate (Ptdlns(3,4)P2) which directs AKT plasma membrane engagement. Thus, the loss of INPP4B expression in human epithelial cells leads to constitutive association of AKT-PH domain with the plasma membrane, increased AKT activation and enhances tumor formation [4]–[5]. In this study, we explored whether inactivation of INPP4B play a critical role in activation of this oncogenic signaling pathway in NPC. Our findings demonstrated that INPP4B was frequently silenced in NPC via promoter methylation and its inactivation contributed to activated PI3K/AKT signaling in this EBV-associated cancer.
Materials and Methods
All primary NPC specimens were recruited from Department of Anatomical and Cellular Pathology at Prince of Wales Hospital with patients' written consent. The study protocol was approved by the Joint CUHK-NTE Clinical Research Ethics Committee, Hong Kong (CREC Ref. No. 2010.102). The procedures for in vivo tumorigenicity assay in nude mice were approved by the Animal Experimentation Ethics Committee (AEEC) of The Chinese University of Hong Kong, Hong Kong SAR.
Cell lines, xenografts and primary tumors
NPC cell lines (C666-1 and HK1) and xenografts (C15, C17, xeno-1915 and xeno-99186) were included in this study and maintained as described previously [19], [24]-[25]. A SV40 large T-immortalized normal nasopharyngeal epithelial cell line NP69 was used as control [26]. For immunohistochemistry (IHC) study, a total of 65 primary NPC patients were recruited with written consent and their archival formalin-fixed paraffin-embedded tumor specimens were collected from the tissue bank of the Department of Anatomical and Cellular Pathology at Prince of Wales Hospital. The study protocol was approved by the Joint CUHK-NTE Clinical Research Ethics Committee, Hong Kong. All specimens were taken before treatment and were histologically evaluated to be EBV-positive undifferentiated or poorly differentiated carcinomas as demonstrated by EBER in-situ hybridization. The clinical parameters are listed in Table 1.
Table 1. Characteristics of the EBV-positive NPC Patients (n = 65).
| Sex (No. of Male/Female) | 50/15 |
| Age (Yrs, Mean ± SE) | 50.42±1.29 |
| Clinical Stage | No. of patients |
| 1 | 7 |
| 2 | 17 |
| 3 | 22 |
| 4 | 19 |
| Disease-free Survival Time (Months, Mean ±SE) | 36.19±2.12 |
| Total Score of INPP4B expression | % of patients |
| Absence/Weak (Score = 0–3) | 49.32 |
| Intermediate/High (Score>3) | 50.77 |
RT-PCR
The transcription of INPP4B was determined by RT-PCR analysis as described previously [27]. Expression of β-actin was used as a control for each sample. The primers used are listed in Table S1.
Western blotting and IHC analysis
Expression or phosphorylation of INPP4B, AKT, PTEN, mTOR, GSK3α/βand ACTIN in the tumor samples were determined by Western blotting. The primary antibodies used are shown in the Table S2. The intensities of protein expression were quantified by densitometric scanning using ImageJ software. The expression of INPP4B was also assessed in 65 paraffin-embedded primary tumors using a semi-quantitative method [19]. The INPP4B expressing cells were counted and scored according to their prevalence and intensity among the tumor cells. The INPP4B expression score was the product of proportion and intensity scores, ranging from 0 to 12. The INPP4B expression was categorized into absence (score 0), low (score 1–3), intermediate (score 4–6), and high (score 7–12) and then correlated with respective clinical parameters.
Bisulfite sequencing and methylation specific PCR (MSP)
Bisulfite sequencing and MSP were performed as described previously [28]. The DNA samples were subjected to bisulfite modification using the EZ DNA Methylation-Gold Kit (Zymo Research). A total of 74 CpG sites spanning approximately 717-bp on the 5'CpG island of INPP4B were analyzed by bisulfite sequencing. This region covered the critical transcriptional regulatory domains sufficient for INPP4B expression in epithelial cells [29]. The PCR primers for bisulfite sequencing and MSP assay are listed in Table S1.
Restoration of INPP4B expression in C666-1 cells
Full length INPP4B was amplified from a TrueORF cDNA clone (Origene, Rockville, MD) and inserted into pCMV6 (OriGene) to produce the INPP4B expression vector. pCMV6-INPP4B and the vector only were transfected into C666-1 cells using LipofectAMINE 2000 (Invitrogen, Carlsbad, CA, USA) as described previously [28]. The restoration of INPP4B expression was confirmed by Western blot analysis.
Cell proliferation and in vivo tumorigenicity assays
The cell viability and proliferation of the INPP4B-expressing C666 cells was detected by WST-1 assays (Roche) [19]. For the in vivo tumorigenicity assay, four nude mice (6 to 8 week old) were injected subcutaneously with 2×106 INPP4B-expressing or control C666-1 cells. Subcutaneous tumor growth was monitored for 28 days by caliper measurements of the tumor size [28].
Results
Downregulation of INPP4B in NPC
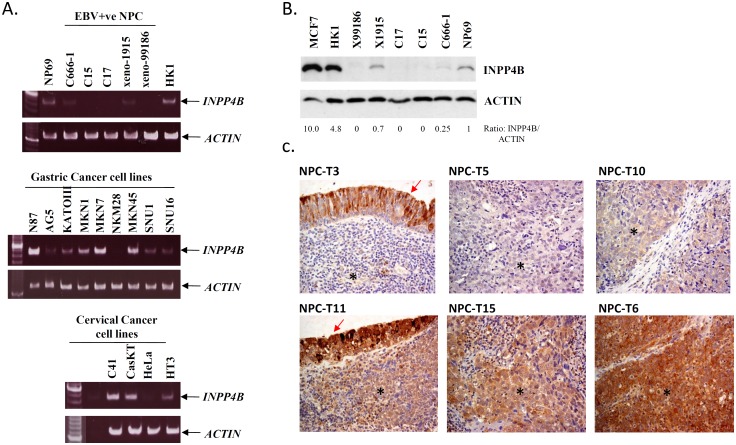
To assess the role of INPP4B in the activation of PI3K/AKT pathway in NPC, we conducted RT-PCR and Western blotting to detect its expression in a panel of NPC cell lines and patient derived xenografts (PDXs). As shown in Figure 1A, the transcription of INPP4B was greatly reduced or completely lost in five tumor lines, while its expression was detected in the immortalized nasopharyngeal epithelial cells NP69 and an EBV-ve NPC cell line, HK-1. A similar INPP4B protein expression pattern was also detected in these samples by Western blotting (Figure 1B). In addition to NPC, we also examined the INPP4B expression in other epithelial cancers. However, the downregulation of INPP4B was only found in 1/9 (11.1%) gastric cancer cell lines and 1/4 (25%) cervical cancer cell lines. This finding implies that the inactivation of INPP4B is a common event in EBV-associated NPC. Using immunohistochemistry, we further assessed the INPP4B expression in the primary tumors. INPP4B was consistently expressed in the normal nasopharyngeal epithelia. Reduction or loss of INPP4B expression (score 0–3) was found in 32/65 (49.2%) of primary tumors (Figure 1C). As shown in Table 2 and Figure S1, INPP4B expression was not correlated with staging and clinical outcome of the patients.
Figure 1. Expression of INPP4B in NPC. (A) RT-PCR analysis of INPP4B transcription in NPC, gastric and cervical cancer cell lines.
The reduced expression of INPP4B was detected in the EBV+ve NPC tumor lines when compared with the EBV-negative NPC cell line HK1 and immortalized nasopharyngeal epithelial cells NP69. Loss of INPP4B transcription was also detected in a gastric cancer cell line NKM28 and a cervical cancer cell line HeLa. The RT-PCR experiments were performed in duplicate. (B) The reduced INPP4B protein expression of EBV-positive NPC tumor lines was detected by Western blotting. A high level of INPP4B expression was found in the EBV-negative NPC cell line HK1. The experiment were carried out in duplicate (C) By IHC staining, a reduction in, or loss of, INPP4B expression was detected in the NPC tumor T3, T5 and T10. In tumor T3, intensive staining of INPP4B was demonstrated in the normal nasopharyngeal epithelial cells (red arrow). Representative NPC cases with medium (T11 and T15) and high expression levels of INPP4B (T6) are shown. NPC tumor cells are indicated (*).
Table 2. Correlation between INPP4B expression and clinicopathological features in NPC.
| Variables | No. of patients (n = 65) | INPP4B expression (no. of patients) | |
| Absence/Weak (Score = 0–3) | Intermediate/High (Score>3) | ||
| Age (years) | |||
| ≤50 | 34 | 17 | 17 |
| >50 | 31 | 15 | 16 |
| Gender | |||
| Male | 50 | 25 | 25 |
| Female | 15 | 7 | 8 |
| Clinical stage | |||
| Early (stage 1 and 2) | 24 | 9 | 15 |
| Late (stage 3 and 4) | 41 | 23 | 18 |
| Loco-regional recurrence or distant metastasis | |||
| Absent | 45 | 23 | 22 |
| Present | 20 | 9 | 11 |
Promoter hypermethylation of INPP4B in NPC
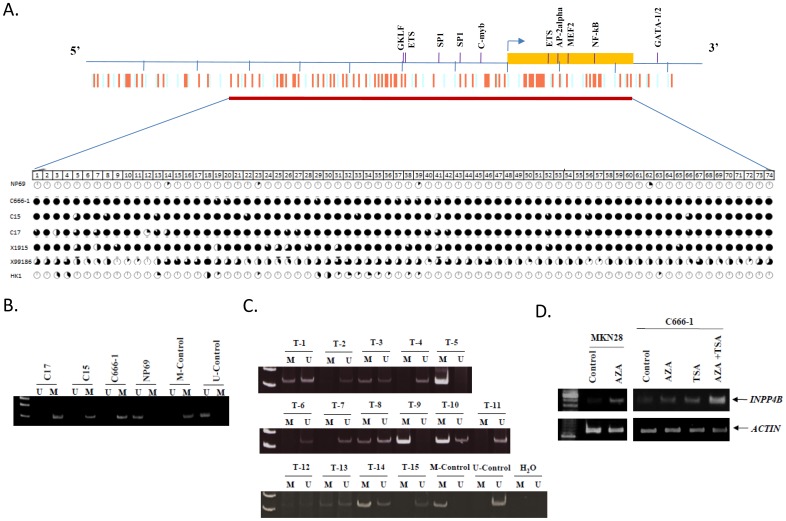
Promoter hypermethylation is one of the main mechanisms for the inactivation of cancer-related genes in EBV-associated NPC [13]–[14], [28], [30]–[32]. In the INPP4B gene, a 717 bp CpG island that contains 74 CpG sites spanning the promoter region and exon 1 was detected (Figure 2A). We explored the methylation status of this region in the NPC tumor lines by genomic bisulfite sequencing. As shown in Figure 2a, dense methylation of the 5′CpG island was detected in all 5 EBV-positive NPC tumor lines. DNA methylation in this region was rarely detected in the INPP4B-expressing NP69 and HK-1 cell lines. A MSP analysis was established for detecting INPP4B methylation in both NPC tumor lines and primary tumors (Figure 2B). The hypermethylation of INPP4B was detected in 9/15 (60%) of primary NPCs (Figure 2C). To further confirm that the downregulation of INPP4B in NPC was due to DNA methylation, we have treated the C666-1 cells with a demethylation agent (5-aza-2′deoxycytidine) or a histone deacetylase (HDAC) inhibitor (trichostatin A). The transcription of INPP4B was highly upregulated in the cells treated with both 5-aza-2′deoxycytidine and tricostatin A (Figure 2D). The findings indicate that INPP4B transcription in NPC cells is frequently silenced by epigenetic alterations.
Figure 2. Promoter hypermethylation of INPP4B in EBV-positive NPC.
(A) Genomic bisufite sequencing of the 5′CpG island of INPP4B in NPC cell line and xenografts. Dense methylation was detected in the EBV+ve tumor lines, C666-1, C15, C17, xeno-1915, and xeno-99186. In NP69 (immortalized normal NP cells) and HK-1 (EBV-ve NPC cell line), almost all of the CpG sites were unmethylated. Each circle represents a CpG site and the percentage of methylated alleles is shown as a proportion of the dark area. Eight to ten clones were sequenced for each sample. Yellow box: exon 1; arrow: transcription start site; orange bars: CpG sites. The raw data was available in the MethDB database (http://www.methdb.de/). The link of the data is: http://methdb.univ-perp.fr/cgi/methdbcrossref.cgi?author_id=79. (B) MSP analysis of NPC tumor lines. Methylated alleles were shown in C17, C15 and C666-1. In NP69, only unmethylated alleles were detected. U: unmethylated allele; M: methylated alleles; M-control: methylated control; U-control: unmethylated control. (C) Detection of hypermethylation of INPP4B in 8/15 primary tumors (T-1, T-3, T-5, T-8, T-9, T-10, T-12, T-13, T-14). (D) RT-PCR showed the restoration of INPP4B expression in the C666-1 cell line after treatment with the DNA methyltransferase inhibitor 5-aza-dC (5-Aza-2′-deoxycytidine) or HDAC inhibitor TSA (Trichostatin-A). Restoration of INPP4B gene expression by 5-aza-dC treatment was also shown in the gastric cancer cell line MKN28. The experiments were carried out in triplicate.
Loss of INPP4B contributes to activated PI3K/AKT signaling in NPC cells
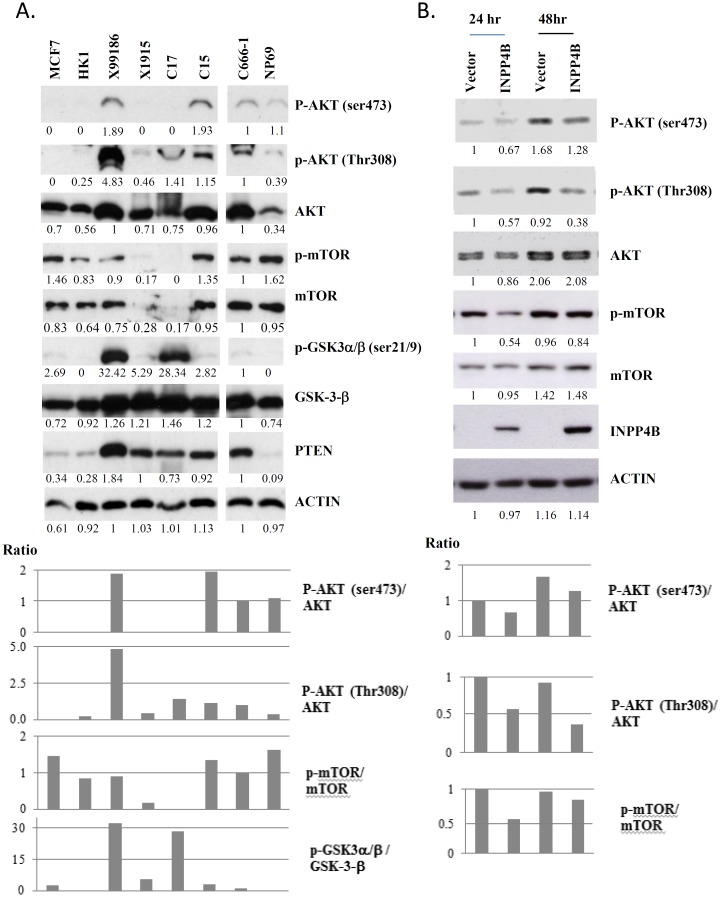
Since INPP4B is a negative regulator of PI3K/AKT signalling, the depletion of INPP4B may result in activation of PI3K/AKT downstream signals [4]–[5]. By Western blotting, we confirmed the increase of p-AKT (Thr308) in the NPC tumor lines (C666-1, C15, C17 and X99186) in which the expression of INPP4B is reduced or absent (Figure 3A). An increase in p-AKT (Ser-473) was also found in C666-1, C15 and X99186. Like INPP4B, PTEN is a negative regulator of the PI3K/AKT pathway. However, a high level of PTEN expression was detected in all 5 EBV-positive NPC tumor lines (Figure 3A). The phosphorylation of mTOR and GSK3-β was also detected in these tumors. The results suggest that INPP4B inactivation contributes to the constitutive activation of the PI3K/AKT signaling pathway in these EBV-positive NPC tumors. Ectopic expression of wild type INPP4B in the C666-1 cells significantly suppressed the phosphorylation of AKT and mTOR (Figure 3B). This finding confirms that epigenetic inactivation of INPP4B plays a role in activation of the PI3K/AKT signaling pathway in NPC cells.
Figure 3. Activation of the PI3K/AKT pathway in EBV+ve NPC tumor cell line and xengrafts.
(A) By Western blotting, p-AKT (Thr308) was detected in all EBV-positive NPC tumor lines and p-AKT (Ser473) was found in C666-1, C15 and xeno-99186. The weak expression of p-AKT was shown in the NP69 cells. No AKT activation was observed in the EBV-ve NPC cell line HK-1 and the breast cancer cell line MCF7. p-mTOR was found in all of the cell lines except C17 and xeno-1915 which also shown absence of mTOR expression. The phosphorylation of GSK-3β (Ser9) was detected in xeno-99186 and C17. A high level of PTEN expression was found in the EBV+ve NPC tumor lines. Relative protein expression was calculated using densitometry with C666-1 at 1. The ratios of the phosphorylation and total protein of AKT, mTOR and GSK-3β were also indicated. (B) Suppression of AKT and mTOR phosphorylation was detected in NPC C666-1 cells transient transfected with INPP4B. Relative protein expression was calculated using densitometry with vector control (24hr) at 1. The ratios of the phosphorylation and total protein of AKT and mTOR were indicated.
Restoration of INPP4B suppresses in vivo tumor growth in NPC cells
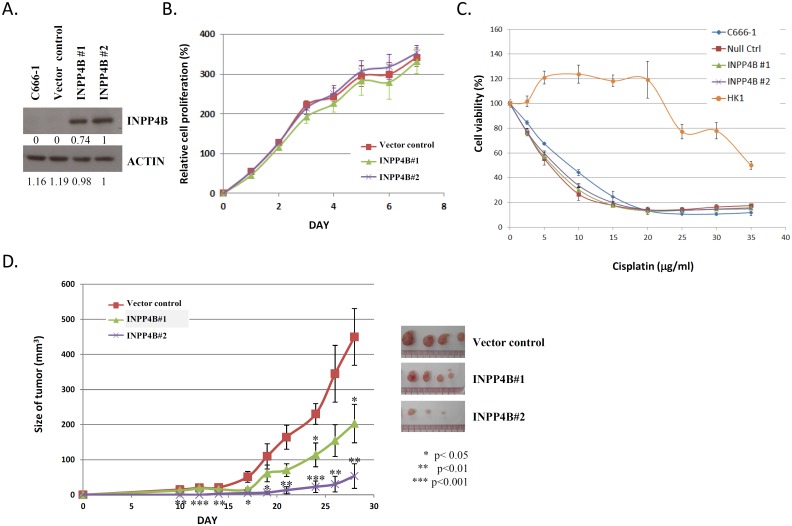
To explore whether INPP4B modulate the in vivo and in vitro growth of NPC cells, two stable INPP4B-expressing C666-1 cell clones, INPP4B#1 and INPP4B#2, were generated (Figure 4A). As shown in Figure 4B, INPP4B expression did not suppress the in vitro proliferation of C666-1 cells. The stable INPP4B-expressing cells also demonstrated a similar sensitivity to the cisptain treatment compared with the controls (Figure 4C). Similar finding was also observed in the cisplatin-resistant C666-1 cells transiently transfected with INPP4B (Figure S2). Despite the limited in vitro effect on cell proliferation and survival, significant suppression was observed in in vivo tumor growth of the cells stably expressing INPP4B (Figure 4D). This finding indicates that the inactivation of INPP4B contributes to in vivo tumor growth in NPC.
Figure 4. Effect of INPP4B expression on the in vitro and in vivo growth of NPC cells.
(A) The expression of INPP4B was detected in the stable INPP4B-C666-1 clones, INPP4B#1 and INPP4B#2, by Western blotting. Relative protein expression was calculated using densitometry with INPP4B#2 set at 1. (B) No significant inhibition of in vitro cell proliferation was detected in the stable INPP4B-expressing C666-1 cells. The WST-1 assay for the detection of the proliferation of INPP4B-expressing and control C666-1 cells was performed in triplicate. (C) Similar sensitivity to the cisplatin treatment was found in the stable INPP4B-expressing C666-1 cells and vector control. The cell viability assays were carried out in triplicate. A cisplatin resistant EBV-negative NPC cell line HK1 was also shown as reference. (D) Significant in vivo growth inhibition was observed in the stable INPP4B-expressing cells compared with the control. Four nude mice were injected subcutaneously with stably INPP4B-expressing or control C666-1 cells and the tumor sizes were measured from day 10 to 28 post inoculation. The data are shown as the mean±SEM. A student t-test was used to assess statistical significance, with a p-value of less than 0.05 considered significant (*p<0.05).
Discussion
The activation of PI3K/AKT signaling pathway promotes tumor development and resistance to anticancer therapies in human cancers [2]–[3], [11]–[12]. In EBV-associated NPC, activated AKT was demonstrated in 42 to 85% of primary tumors by immunohistochemistry. The phosphorylation of the AKT downstream targets, GSK-3β, FKHR, and BAD was also detected in the majority of cases with activated AKT [2]–[3], [11]–[12]. In this study, we also confirmed the consistent activation of PI3K/AKT signaling in all of the EBV-positive tumor lines examined. Notably, the downregulation of INPP4B was commonly found in both the tumor lines and primary tumors. We proved that the restoration of INPP4B expression inhibited the phosphorylation of AKT and mTOR in the NPC C666-1 cells. Furthermore, the NPC cells stably expressing INPP4B showed reduced in vivo tumorigenicity. A similar effect of INPP4B depletion on the PI3K/AKT signaling axis was previously demonstrated in melanoma, breast and prostate cancers [4]–[5], [33]–[35]. In agreement with observations in melanoma, our functional study revealed no obvious effect of INPP4B expression on the proliferation and survival of NPC cells in vitro [35]. In a recent study of tumor suppressor function of INPP4B in melanoma, Perez-Lorenzo et al. showed that the growth rate in BRAF/NRAS wild type melanoma cells on INPP4B knockdown was significantly increased. However, the effect was not observed in melanoma cells with oncogenic BRAF or NRAS mutations [35]. The finding indicates that the effect of INPP4B on cell proliferation is depended on the presence of other genetic changes. Nevertheless, a significant inhibitory effect of INPP4B expression on in vivo NPC tumor growth in nude mice was observed, similar to that previously reported in melanoma and breast cancers [5], [33], [35]. The reduced in vivo tumorigenicity in the INPP4B-expressing cells strongly supports the tumor suppressor role of INPP4B in NPC. Our findings indicate the importance of activating the PI3K/AKT signaling pathway by INPP4B depletion in NPC pathogenesis. Up to now, a number of studies have explored the mechanisms that are responsible for activating PI3K/AKT signaling in this EBV-associated epithelial cancer. Studies of EBV-encoded LMP1 and LMP2A have revealed that these latent proteins mediate the transformation of epithelial cells through the activation of PI3K/AKT pathway [17]–[18], [21]. In our earlier study, we also detected the high-level amplification of PIK3CA in 20% of primary tumors [22]. In addition to the viral oncoproteins and genetic changes, Zhang also demonstrated that the upregulation of microRNA-144 is able to activate the PI3K/AKT pathway by repressing PTEN expression in NPC cells [36]. Interestingly, we found a high level PTEN expression in the EBV-positive NPC tumor lines which also showed increased phosphorylation of AKT. The expression of PTEN in C666-1 has also been reported in previous studies [36], [37]. In our recent whole genome sequencing study of EBV-positive NPC tumor lines, we did not identify any genetic alterations including indels and mutations of PTEN genes (unpublished findings). It is likely that INPP4B is a major target for inactivation in these tumors.
Here, we have shown, for the first time, that INPP4B was transcriptionally silenced by promoter hypermethylation. We have demonstrated that the epigenetic inactivation of INPP4B is one of the key mechanisms in activating the PI3K/AKT signaling cascade and thereby contributes to the oncogenesis of EBV-associated NPC.
Supporting Information
INPP4B expression and clinicopathological features in NPC patients. (A) No correlation of INPP4B expression with disease stages was found in the NPC patients. Archival formalin-fixed paraffin-embedded EBV-positive specimens were processed for IHC staining of INPP4B expression. The number and intensity of positive reactions were recorded and correlations were analysed against different clinicopathological features. Archival formalin-fixed paraffin-embedded EBV-positive specimens were processed for IHC staining on INPP4B expression. The number and intensity of positive reactions were recorded and correlations were analysed against different clinicopathological features. (B) INPP4B expression was not associated with recurrence or metastatic diseases. The bar chart shows the number of patients with or without metastasis/recurrence in groups of high/intermediate and low/absent INPP4B expression. Fischer's exact test was used to determine any statistical significance. (C) No significant correlation of INPP4B expression with the overall and disease-free survival of NPC patients was found. The graphs show the survival curves of the NPC patients against INPP4B expression. The survival time of the NPC patients was correlated witht high/intermediate and low/absent INPP4B expression. No significant correlation of INPP4B expression with the overall and disease-free survival of the NPC patients was found. Statistical analysis was performed using the log-rank (Mantel-Cox) test.
(TIF)
Effect of INPP4B expression on the sensitivity to the cisplatin treatment in the selected cisplatin resistant C666-1 cells. A cisplatin resistant C666-1 cell line was established and transiently transfected with INPP4B. No significant change of sensitivity was observed in the INPP4B-transfected cells. The cell viability assays were carried out in triplicate.
(TIF)
Primer sequences for RT-PCR, MSP and bisulfite sequencing.
(DOCX)
Primary Antibodies used in Western blotting.
(DOCX)
Funding Statement
This work was supported by Li Ka Shing Institute of Health Science and Hong Kong Research Grant Council (471610 and T12-401/13-R). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Manning BD, Cantley BD (2007) AKT/PKB signaling: navigating downstream. Cell 129: 1261–1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Engelman JA (2009) Targeting PI3K signalling in cancer: opportunities, challenges and limitation. Nat Rev Cancer 9: 550–562. [DOI] [PubMed] [Google Scholar]
- 3. Liu P, Cheng H, Roberts TM, Zhao JJ (2009) Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov 8: 627–644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bertucci MC, Mitchell CA (2013) Phosphoinositide 3-kinase and INPP4B in human breast cancer. Ann N Y Acad Sci 1280: 1–5. [DOI] [PubMed] [Google Scholar]
- 5. Gewinner C, Wang ZC, Richardson A, Teruya-Feldstein J, Etemadmoghadam D, et al. (2009) Evidence that inositol polyphosphate 4-phosphatase type II is a tumor suppressor that inhibits PI3K signaling. Cancer Cell 16: 115–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ma K, Cheung SM, Marshall AJ, Duronio V (2008) PI(3,4,5)P3 and PI(3,4)P2 levels correlate with PKB/akt phosphorylation at Thr308 and Ser473, respectively; PI(3,4)P2 levels determine PKB activity. Cell Signal 20: 684–94. [DOI] [PubMed] [Google Scholar]
- 8. Aoki M, Blazek E, Vogt PK (2001) A role of the kinase mTOR in cellular transformation induced by the oncoproteins P3k and Akt. Proc Natl Acad Sci U S A 98: 136–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Vazquez F, Sellers WR (2000) The PTEN tumor suppressor protein: an antagonist of phosphoinositide 3-kinase signaling. Biochim Biophys Acta 1470: M21–35. [DOI] [PubMed] [Google Scholar]
- 10. Song MS, Salmena L, Pandolfi PP (2012) The functions and regulation of the PTEN tumour suppressor. Nat Rev Mol Cell Biol 13: 283–296. [DOI] [PubMed] [Google Scholar]
- 11. Chalhoub N, Baker SJ (2009) PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol 4: 127–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yuen TL, Cantley LC (2008) PI3K pathway alterations in cancer: variation on a theme. Oncogene 27: 5497–5510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Lo KW, To KF, Huang DP (2004) Focus on nasopharyngeal Carcinoma. Cancer Cell 5: 423–428. [DOI] [PubMed] [Google Scholar]
- 14. Lo KW, Chung GT, To KF (2012) Deciphering the molecular genetic basis of NPC through molecular, cytogenetic, and epigenetic approaches. Semin Cancer Biol 22: 79–86. [DOI] [PubMed] [Google Scholar]
- 15. Tsao SW, Tsang CM, Pang PS, Zhang G, Chen H, et al. (2012) The biology of EBV infection in human epithelial cells. Semin Cancer Biol 22: 137–143. [DOI] [PubMed] [Google Scholar]
- 16. Morrison JA, Gulley ML, Pathmanathan R, Raab-Traub N (2004) Differential signaling pathways are activated in the Epstein-Barr virus-associated malignancies nasopharyngeal carcinoma and Hodgkin lymphoma. Cancer Res 64: 5251–5260. [DOI] [PubMed] [Google Scholar]
- 17. Dawson CW, Tramountanis G, Eliopoulos AG, Young LS (2003) Epstein-Barr virus latent membrane protein 1 (LMP1) activates the phosphatidylinositol 3-kinase/Akt pathway to promote cell survival and induce actin filament remodeling. J Biol Chem 278: 3694–3704. [DOI] [PubMed] [Google Scholar]
- 18. Fukuda M, Longnecker R (2007) Epstein-Barr virus latent membrane protein 2A mediates transformation through constitutive activation of the Ras/PI3-K/Akt Pathway. J Virol 81: 9299–9306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chung GT, Lou WP, Chow C, To KF, Choy KW, et al. (2013) Constitutive activation of distinct NF-kB signals in EBV-associated nasopharyngeal carcinoma. J Pathol 231: 311–322. [DOI] [PubMed] [Google Scholar]
- 20. Or YY, Chung GT, To KF, Chow C, Choy KW, et al. (2010) Identification of a novel 12p13.3 amplicon in nasopharyngeal carcinoma. J Pathol 220: 97–107. [DOI] [PubMed] [Google Scholar]
- 21. Dawson CW, Port RJ, Young LS (2012) The role of the EBV-encoded latent membrane proteins LMP1 and LMP2 in the pathogenesis of nasopharyngeal carcinoma (NPC). Semin Cancer Biol 22: 144–153. [DOI] [PubMed] [Google Scholar]
- 22. Or YY, Hui AB, To KF, Lam CN, Lo KW (2006) PIK3CA mutations in nasopharyngeal carcinoma. Int J Cancer 118: 1065–1067. [DOI] [PubMed] [Google Scholar]
- 23. Chou CC, Chou MJ, Tzen CY (2009) PIK3CA mutation occurs in nasopharyngeal carcinoma but does not significantly influence the disease-specific survival. Med Oncol 26: 322–326. [DOI] [PubMed] [Google Scholar]
- 24. Cheung ST, Huang DP, Hui AB, Lo KW, Ko CW, et al. (1999) Nasopharyngeal carcinoma cell line (C666-1) consistently harbouring Epstein-Barr virus. Int J Cancer 83: 121–126. [DOI] [PubMed] [Google Scholar]
- 25. Huang DP, Ho JH, Poon YF, Chew EC, Saw D, et al. (1980) Establishment of a cell line (NPC/HK1) from a differentiated squamous carcinoma of the nasopharynx. Int J cancer 26: 127–132. [DOI] [PubMed] [Google Scholar]
- 26. Tsao SW, Wang X, Liu Y, Cheung YC, Feng H, et al. (2002) Establishment of two immortalized nasopharyngeal epithelial cell lines using SV40 large T and HPV16E6/E7 viral oncogenes. Biochim Biophys Acta 1590: 150–158. [DOI] [PubMed] [Google Scholar]
- 27. Chow LSN, Lo KW, Kwong J, Lam EC, To KF, et al. (2004) RASSF1A is a target tumor suppressor at chromosome region 3p21.3 in nasopharyngeal carcinoma. Int J Cancer 109: 839–847. [DOI] [PubMed] [Google Scholar]
- 28. Kwong J. Chow LSN, Wong AYH, Hung WK, Chung GTY, et al. (2007) Epigenetic inactivation of deleted in lung and esophageal cancer 1 (DLEC1) gene in nasopharyngeal carcinoma. Genes Chromosomes Cancer 46: 171–180. [DOI] [PubMed] [Google Scholar]
- 29. Ferron M, Vacher J (2006) Characterization of the murine Inpp4b gene and identification of a novel isoform. Gene 376: 152–161. [DOI] [PubMed] [Google Scholar]
- 30. Lo KW, Kwong J, Hui ABY, Chan SYY, To KF, et al. (2001) High frequency of promoter hypermethylation of RASSF1A in nasopharyngeal carcinoma. Cancer Res 61: 3877–3881. [PubMed] [Google Scholar]
- 31. Kwong J, Lo KW, To KF, Teo PML, Johnson PJ, et al. (2002) Promoter hypermethylation of multiple genes in nasopharyngeal carcinoma. Clin Cancer Res 8: 467–473. [PubMed] [Google Scholar]
- 32. Shu XS, Li L, Ji M, Cheng Y, Ying J, et al. (2013) FEZF2, a novel 3p14 tumor suppressor gene, represses oncogene EZH2 and MDM2 expression and is frequently methylated in nasopharyngeal carcinoma. Carcinogenesis 34: 1984–1993. [DOI] [PubMed] [Google Scholar]
- 33. Fedele CG, Ooms LM, Ho M, Vieusseux J, O'Toole SA, et al. (2010) Inositol polyphosphate 4-phosphatase II regulates PI3K/Akt signaling and is lost in human basal-like breast cancers. Proc Natl Acad Sci USA 107: 22231–22236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Hodgson MC, Shao LJ, Frolov A, Li R, Peterson LE, et al. (2011) Decreased expression and androgen regulation of the tumor suppressor gene INPP4B in prostate cancer. Cancer Res 71: 572–582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Perez-Lorenzo R, Gill KZ, Shen CH, Zhao FX, Zheng B, et al. (2014) A tumor suppressor function for the lipid phosphatase INPP4B in melanocytic neoplasms. J Invest Dermatol 134: 1359–1368. [DOI] [PubMed] [Google Scholar]
- 36. Zhang LY, Ho-Fun Lee V, Wong AM, Kwong DL, Zhu YH, et al. (2013) MicroRNA-144 promotes cell proliferation, migration and invasion in nasopharyngeal carcinoma through repression of PTEN. Carcinogenesis 34: 454–463. [DOI] [PubMed] [Google Scholar]
- 37. Song LB, Li J, Liao WT, Feng Y, Yu CP, et al. (2009) The polycomb group protein Bmi-1 represses the tumor suppressor PTEN and induces epithelial-mesenchymal transition in human nasopharyngeal epithelial cells. J Clin Invest 119: 3626–3636. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
INPP4B expression and clinicopathological features in NPC patients. (A) No correlation of INPP4B expression with disease stages was found in the NPC patients. Archival formalin-fixed paraffin-embedded EBV-positive specimens were processed for IHC staining of INPP4B expression. The number and intensity of positive reactions were recorded and correlations were analysed against different clinicopathological features. Archival formalin-fixed paraffin-embedded EBV-positive specimens were processed for IHC staining on INPP4B expression. The number and intensity of positive reactions were recorded and correlations were analysed against different clinicopathological features. (B) INPP4B expression was not associated with recurrence or metastatic diseases. The bar chart shows the number of patients with or without metastasis/recurrence in groups of high/intermediate and low/absent INPP4B expression. Fischer's exact test was used to determine any statistical significance. (C) No significant correlation of INPP4B expression with the overall and disease-free survival of NPC patients was found. The graphs show the survival curves of the NPC patients against INPP4B expression. The survival time of the NPC patients was correlated witht high/intermediate and low/absent INPP4B expression. No significant correlation of INPP4B expression with the overall and disease-free survival of the NPC patients was found. Statistical analysis was performed using the log-rank (Mantel-Cox) test.
(TIF)
Effect of INPP4B expression on the sensitivity to the cisplatin treatment in the selected cisplatin resistant C666-1 cells. A cisplatin resistant C666-1 cell line was established and transiently transfected with INPP4B. No significant change of sensitivity was observed in the INPP4B-transfected cells. The cell viability assays were carried out in triplicate.
(TIF)
Primer sequences for RT-PCR, MSP and bisulfite sequencing.
(DOCX)
Primary Antibodies used in Western blotting.
(DOCX)