Abstract
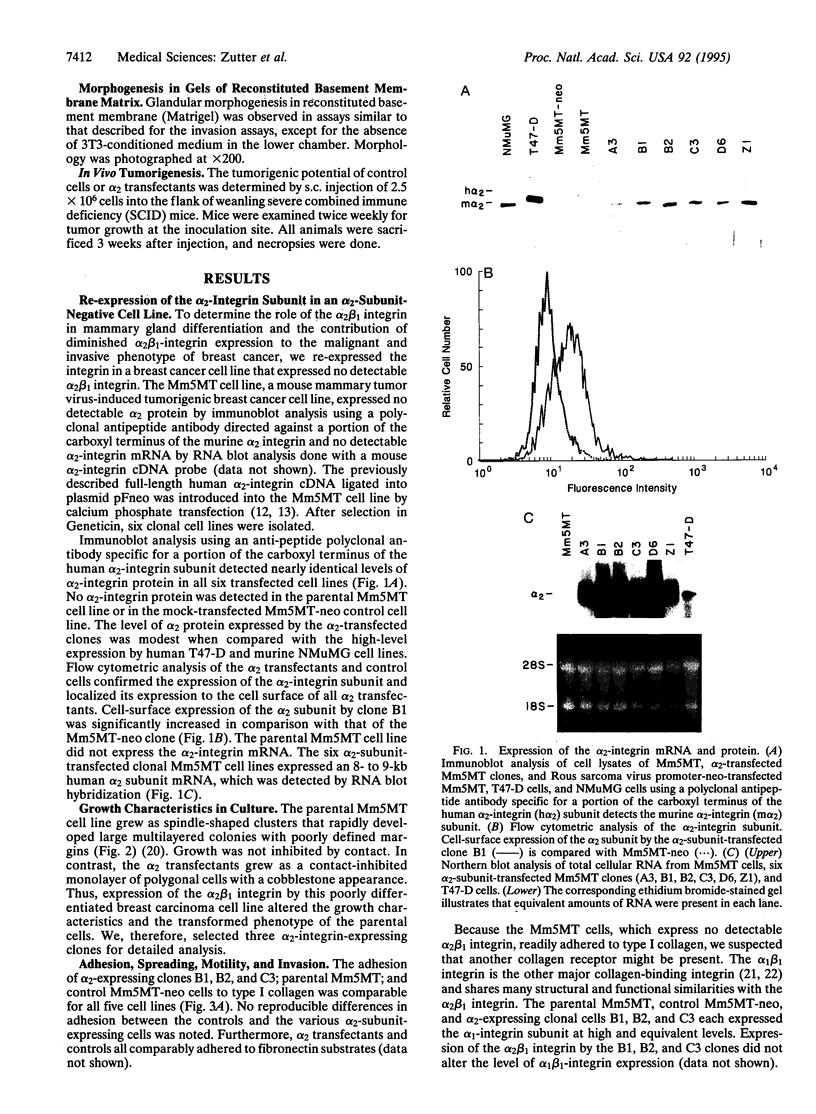
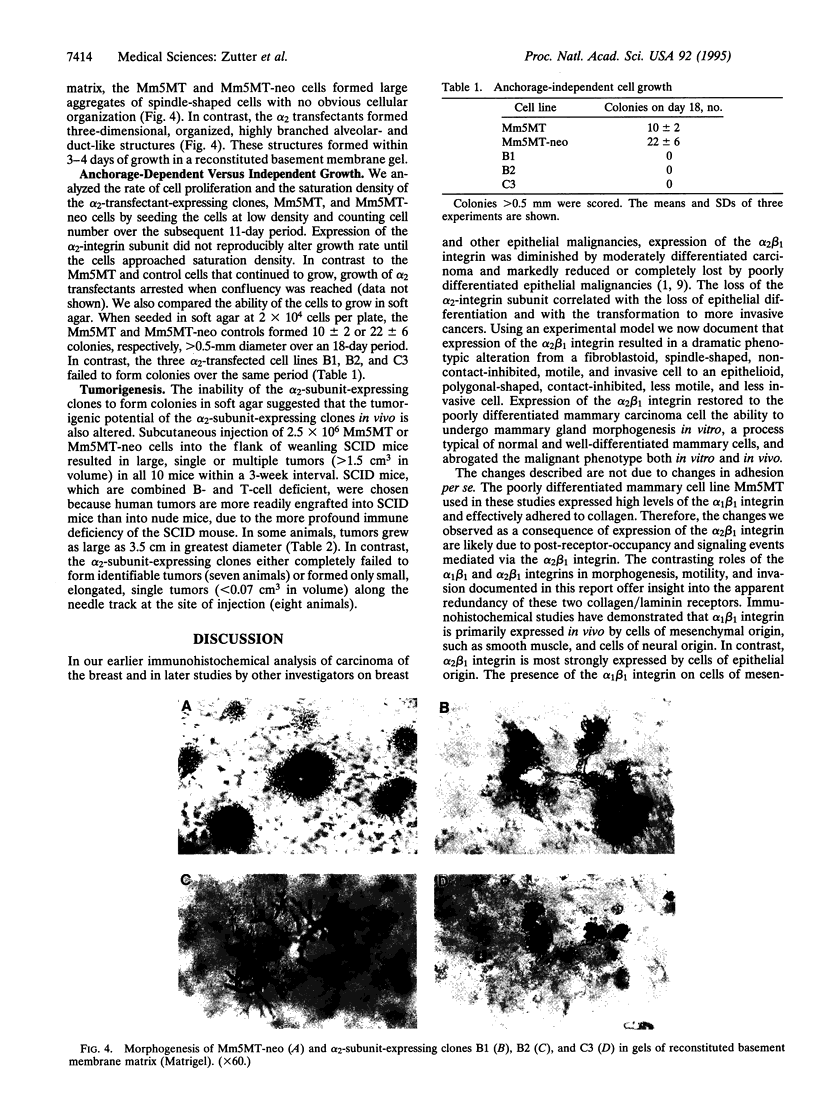
To assess the role of altered alpha 2 beta 1 integrin expression in breast cancer, we expressed the alpha 2 beta 1 integrin de novo in a poorly differentiated mammary carcinoma that expressed no detectable alpha 2-integrin subunit. Expression of the alpha 2 beta 1 integrin resulted in a dramatic phenotypic alteration from a fibroblastoid, spindle-shaped, non-contact-inhibited, motile, and invasive cell to an epithelioid, polygonal-shaped, contact-inhibited, less motile, and less invasive cell. Although expression of the alpha 2 subunit did not alter adhesion to collagen, it profoundly altered cell spreading. Re-expression of the alpha 2 beta 1 integrin restored the ability to differentiate into gland-like structures in three-dimensional matrices and markedly reduced the in vivo tumorigenicity of the cells. These results indicate that the consequences of diminished alpha 2 beta 1-integrin expression in the development of breast cancer and, presumably, of other epithelial malignancies are increased tumorigenicity and loss of the differentiated epithelial phenotype.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albelda S. M. Role of integrins and other cell adhesion molecules in tumor progression and metastasis. Lab Invest. 1993 Jan;68(1):4–17. [PubMed] [Google Scholar]
- Albini A., Iwamoto Y., Kleinman H. K., Martin G. R., Aaronson S. A., Kozlowski J. M., McEwan R. N. A rapid in vitro assay for quantitating the invasive potential of tumor cells. Cancer Res. 1987 Jun 15;47(12):3239–3245. [PubMed] [Google Scholar]
- Aznavoorian S., Stracke M. L., Krutzsch H., Schiffmann E., Liotta L. A. Signal transduction for chemotaxis and haptotaxis by matrix molecules in tumor cells. J Cell Biol. 1990 Apr;110(4):1427–1438. doi: 10.1083/jcb.110.4.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan B. M., Kassner P. D., Schiro J. A., Byers H. R., Kupper T. S., Hemler M. E. Distinct cellular functions mediated by different VLA integrin alpha subunit cytoplasmic domains. Cell. 1992 Mar 20;68(6):1051–1060. doi: 10.1016/0092-8674(92)90077-p. [DOI] [PubMed] [Google Scholar]
- Chan B. M., Matsuura N., Takada Y., Zetter B. R., Hemler M. E. In vitro and in vivo consequences of VLA-2 expression on rhabdomyosarcoma cells. Science. 1991 Mar 29;251(5001):1600–1602. doi: 10.1126/science.2011740. [DOI] [PubMed] [Google Scholar]
- Dedhar S., Saulnier R. Alterations in integrin receptor expression on chemically transformed human cells: specific enhancement of laminin and collagen receptor complexes. J Cell Biol. 1990 Feb;110(2):481–489. doi: 10.1083/jcb.110.2.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elices M. J., Hemler M. E. The human integrin VLA-2 is a collagen receptor on some cells and a collagen/laminin receptor on others. Proc Natl Acad Sci U S A. 1989 Dec;86(24):9906–9910. doi: 10.1073/pnas.86.24.9906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emerman J. T., Pitelka D. R. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro. 1977 May;13(5):316–328. doi: 10.1007/BF02616178. [DOI] [PubMed] [Google Scholar]
- Giancotti F. G., Ruoslahti E. Elevated levels of the alpha 5 beta 1 fibronectin receptor suppress the transformed phenotype of Chinese hamster ovary cells. Cell. 1990 Mar 9;60(5):849–859. doi: 10.1016/0092-8674(90)90098-y. [DOI] [PubMed] [Google Scholar]
- Hynes R. O. Integrins: a family of cell surface receptors. Cell. 1987 Feb 27;48(4):549–554. doi: 10.1016/0092-8674(87)90233-9. [DOI] [PubMed] [Google Scholar]
- Ignatius M. J., Large T. H., Houde M., Tawil J. W., Barton A., Esch F., Carbonetto S., Reichardt L. F. Molecular cloning of the rat integrin alpha 1-subunit: a receptor for laminin and collagen. J Cell Biol. 1990 Aug;111(2):709–720. doi: 10.1083/jcb.111.2.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keely P. J., Fong A. M., Zutter M. M., Santoro S. A. Alteration of collagen-dependent adhesion, motility, and morphogenesis by the expression of antisense alpha 2 integrin mRNA in mammary cells. J Cell Sci. 1995 Feb;108(Pt 2):595–607. doi: 10.1242/jcs.108.2.595. [DOI] [PubMed] [Google Scholar]
- Kirchhofer D., Languino L. R., Ruoslahti E., Pierschbacher M. D. Alpha 2 beta 1 integrins from different cell types show different binding specificities. J Biol Chem. 1990 Jan 15;265(2):615–618. [PubMed] [Google Scholar]
- Liotta L. A., Steeg P. S., Stetler-Stevenson W. G. Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell. 1991 Jan 25;64(2):327–336. doi: 10.1016/0092-8674(91)90642-c. [DOI] [PubMed] [Google Scholar]
- Liotta L. A. Tumor invasion and metastases--role of the extracellular matrix: Rhoads Memorial Award lecture. Cancer Res. 1986 Jan;46(1):1–7. [PubMed] [Google Scholar]
- Marcantonio E. E., Hynes R. O. Antibodies to the conserved cytoplasmic domain of the integrin beta 1 subunit react with proteins in vertebrates, invertebrates, and fungi. J Cell Biol. 1988 May;106(5):1765–1772. doi: 10.1083/jcb.106.5.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mechtersheimer G., Barth T., Quentmeier A., Möller P. Differential expression of beta 1 integrins in nonneoplastic smooth and striated muscle cells and in tumors derived from these cells. Am J Pathol. 1994 Jun;144(6):1172–1182. [PMC free article] [PubMed] [Google Scholar]
- Menko A. S., Philip N. J. Beta 1 integrins in epithelial tissues: a unique distribution in the lens. Exp Cell Res. 1995 Jun;218(2):516–521. doi: 10.1006/excr.1995.1186. [DOI] [PubMed] [Google Scholar]
- Owens R. B., Hackett A. J. Tissue culture studies of mouse mammary tumor cells and associated viruses. J Natl Cancer Inst. 1972 Nov;49(5):1321–1332. [PubMed] [Google Scholar]
- Plantefaber L. C., Hynes R. O. Changes in integrin receptors on oncogenically transformed cells. Cell. 1989 Jan 27;56(2):281–290. doi: 10.1016/0092-8674(89)90902-1. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E. Integrins. J Clin Invest. 1991 Jan;87(1):1–5. doi: 10.1172/JCI114957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santoro S. A., Zutter M. M., Wu J. E., Staatz W. D., Saelman E. U., Keely P. J. Analysis of collagen receptors. Methods Enzymol. 1994;245:147–183. doi: 10.1016/0076-6879(94)45010-2. [DOI] [PubMed] [Google Scholar]
- Staatz W. D., Rajpara S. M., Wayner E. A., Carter W. G., Santoro S. A. The membrane glycoprotein Ia-IIa (VLA-2) complex mediates the Mg++-dependent adhesion of platelets to collagen. J Cell Biol. 1989 May;108(5):1917–1924. doi: 10.1083/jcb.108.5.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tawil N. J., Houde M., Blacher R., Esch F., Reichardt L. F., Turner D. C., Carbonetto S. Alpha 1 beta 1 integrin heterodimer functions as a dual laminin/collagen receptor in neural cells. Biochemistry. 1990 Jul 10;29(27):6540–6544. doi: 10.1021/bi00479a028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Papadimitriou J., Berdichevsky F., D'Souza B., Burchell J. Human models of breast cancer. Cancer Surv. 1993;16:59–78. [PubMed] [Google Scholar]
- Terranova V. P., Hujanen E. S., Loeb D. M., Martin G. R., Thornburg L., Glushko V. Use of a reconstituted basement membrane to measure cell invasiveness and select for highly invasive tumor cells. Proc Natl Acad Sci U S A. 1986 Jan;83(2):465–469. doi: 10.1073/pnas.83.2.465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vleminckx K., Vakaet L., Jr, Mareel M., Fiers W., van Roy F. Genetic manipulation of E-cadherin expression by epithelial tumor cells reveals an invasion suppressor role. Cell. 1991 Jul 12;66(1):107–119. doi: 10.1016/0092-8674(91)90143-m. [DOI] [PubMed] [Google Scholar]
- Wu J. E., Santoro S. A. Complex patterns of expression suggest extensive roles for the alpha 2 beta 1 integrin in murine development. Dev Dyn. 1994 Apr;199(4):292–314. doi: 10.1002/aja.1001990405. [DOI] [PubMed] [Google Scholar]
- Zutter M. M., Krigman H. R., Santoro S. A. Altered integrin expression in adenocarcinoma of the breast. Analysis by in situ hybridization. Am J Pathol. 1993 May;142(5):1439–1448. [PMC free article] [PubMed] [Google Scholar]
- Zutter M. M., Mazoujian G., Santoro S. A. Decreased expression of integrin adhesive protein receptors in adenocarcinoma of the breast. Am J Pathol. 1990 Oct;137(4):863–870. [PMC free article] [PubMed] [Google Scholar]
- Zutter M. M., Santoro S. A. Widespread histologic distribution of the alpha 2 beta 1 integrin cell-surface collagen receptor. Am J Pathol. 1990 Jul;137(1):113–120. [PMC free article] [PubMed] [Google Scholar]







