Abstract
The treatment of endophthalmitis is becoming very challenging due to the emergence of multidrug-resistant bacteria. Hence, the development of novel therapeutic alternatives for ocular use is essential. Here, we evaluated the therapeutic potential of Ply187AN-KSH3b, a chimeric phage endolysin derived from the Ply187 prophage, in a mouse model of Staphylococcus aureus endophthalmitis. Our data showed that the chimeric Ply187 endolysin exhibited strong antimicrobial activity against both methicillin-sensitive S. aureus and methicillin-resistant S. aureus (MRSA) strains, as evidenced by MIC determinations, reductions in turbidity, and disruption of biofilms. Moreover, exposure of S. aureus to Ply187 for up to 10 generations did not lead to resistance development. The intravitreal injection of chimeric Ply187 (at 6 or 12 h postinfection) significantly improved the outcome of endophthalmitis, preserved retinal structural integrity, and maintained visual function as assessed by electroretinogram analysis. Furthermore, phage lysin treatment significantly reduced the bacterial burden and the levels of inflammatory cytokines and neutrophil infiltration in the eyes. These results indicate that the intravitreal administration of a phage lytic enzyme attenuates the development of bacterial endophthalmitis in mice. To the best of our knowledge, this is the first study demonstrating the therapeutic use of phage-based antimicrobials in ocular infections.
INTRODUCTION
Bacterial endophthalmitis is a vision-threatening complication of ocular trauma and surgeries, particularly cataract surgery, a common procedure performed on the aging population worldwide (1). The increased use of intravitreal (IVT) injections (2) for the management of retinal diseases has also been implicated as a contributing factor to the increased incidence of endophthalmitis (3). The reported incidence of endophthalmitis per eye in multicenter clinical trials with anti-vascular endothelial growth factor (VEGF) therapy ranged from 0.7% to 1.6% (4). As most cases of postoperative endophthalmitis are caused by bacteria from the ocular surface (5, 6), prophylactic measures include the use of topical antibiotics to reduce the density of the ocular flora (7, 8). Recent studies have demonstrated that the use of topical antibiotics, specifically after IVT, does not reduce the risk of endophthalmitis. Rather, there is a trend toward a higher incidence of endophthalmitis (9). This could be in part because the repeated use of topical antibiotics increases the presence of antibiotic-resistant bacterial strains on the ocular surface (10).
In the recent past, fluoroquinolones, such as moxifloxacin, were reported to be effective in preventing Staphylococcus aureus endophthalmitis (11). Although this class of antibiotics is active against a broad spectrum of organisms, the antibiotics are largely ineffective against multidrug-resistant (MDR) strains of S. aureus, the leading cause of severe endophthalmitis (12, 13). Furthermore, there is increasing evidence suggesting that ocular surface microflora are becoming more resistant to “fourth-generation” (broad-spectrum) fluoroquinolones, with up to 30% of cultured ocular isolates being resistant (14–16). These findings support the necessity to search for new alternative prophylactic/therapeutic modalities against resistant bacteria in general and S. aureus in particular to prevent postoperative endophthalmitis.
Bacteriophages (phages) are viruses that infect bacteria. The phage endolysin is a peptidoglycan hydrolase that is produced near the end of the phage lytic cycle to degrade the cell wall and allow the nascent phage particle to escape in order to infect new host cells (17). The endolysins are highly specific to the host bacterium (genus) and have evolved to bind to unique and essential bacterial cell wall targets. In recent years, phage endolysins have attracted considerable interest as novel antibacterial agents (18) and have been used to treat a variety of bacterial infections, as demonstrated by the increasing number of experimental animal studies (19–22). Similarly, human studies have documented the use of live antistaphylococcal phages for the treatment of conjunctivitis and blepharitis in 28 patients, with no side effects (23, 24).
The alarming increase in antibiotic-resistant ocular pathogens, along with a complete lack of studies examining the ocular use of phage lytic enzymes, led us to test the therapeutic potential of a chimeric endolysin, Ply187AN-KSH3b, in an experimental model of bacterial endophthalmitis. Our data demonstrated that a single intravitreal injection of a chimeric staphylolytic phage endolysin cleared S. aureus from eyes and markedly diminished the progression of endophthalmitis in mice.
MATERIALS AND METHODS
Bacterial strains and chimeric Ply187 endolysin.
For in vivo studies, a methicillin-sensitive S. aureus (MSSA) strain, RN6390, was used to induce endophthalmitis, whereas the in vitro studies were performed using antibiotic-resistant strains (see Table 1, below) that included USA300, a community-associated methicillin-resistant S. aureus (CA-MRSA) strain, and three clinical isolates, R2932 (CA-MRSA) and R2952 and R3000 (both hospital-associated MRSA [HA-MRSA] strains), kindly provided by Michael J. Rybak (Department of Pharmaceutical Sciences, Wayne State University, Detroit, MI). All bacteria were routinely cultured in TSB (tryptic soy broth; Sigma, St. Louis, MO) or on TSB agar plates.
TABLE 1.
Antibacterial activity of chimeric Ply187 against S. aureus strains
| Isolate ID | Phenotype | MICa (μg/ml) |
|---|---|---|
| RN6390 | MSSA | 4.16 ± 1.04 |
| USA300 | CA-MRSA | 4.16 ± 1.04 |
| R2932 | CA-MRSA | 4.16 ± 1.04 |
| R2952 | HA-MRSA | 8.33 ± 2.08 |
| R3000 | HA-MRSA | 4.16 ± 1.04 |
Data are mean values from three experiments ± SEM.
The candidate therapeutic agent, Ply187AN-KSH3b (here, chimeric Ply187), is a staphylococcal peptidoglycan hydrolase fusion protein that contains the endopeptidase domain from a staphylococcal prophage endolysin, Ply187, the SH3b cell wall-binding domain of the staphylococcal phage K endolysin, LysK, and a C-terminal 6×His tag for nickel column purification. A detailed description of the cloning, expression, and purification of this agent was previously reported by Mao et al. (25). For intravitreal injection, the nickel column-purified chimeric Ply187 protein was dialyzed gradually against phosphate-buffered saline (PBS) containing 30% glycerol (EB) and filter sterilized.
Turbidity reduction assay.
The turbidity reduction assay with chimeric Ply187 was performed in a 96-well plate as described previously by Becker et al. (26, 27). Briefly, all S. aureus strains/isolates were grown to logarithmic phase (optical density at 600 nm, 0.4 to 0.6) at 37°C in TSB. The culture was harvested by centrifugation, and the pellet was resuspended in an assay buffer (400 mM NaCl, 20 mM Tris-HCl, 1% glycerol; pH 7.5). The bacterial culture (100 μl/well) was mixed with 100 μl (as per the MICs in Table 1) of chimeric Ply187 protein diluted in the same assay buffer. The reduction in the turbidity was measured every 5 min for up to 1 h by using a microplate reader. EB diluted in the same assay buffer (without chimeric Ply187 protein) was used as a control (EB C).
MIC and resistance development determinations.
A microdilution broth method was used to determine MICs, as described by Becker et al. (27), with slight modifications. The MICs were determined in TSB corrected for EB addition (50% EB with or without protein and 50% 2× TSB). Resistance development against Ply187 was tested by using repeated exposures in a MIC assay as described by Rodríguez-Rubio and coworkers (28). In brief, bacterial cultures (105 CFU/well) were exposed overnight to a 2-fold serial dilution of Ply187 or lysostaphin or gentamicin (the latter served as controls). In every round, 100-μl aliquots from cultures from those wells with growth (½× MIC) were inoculated into fresh TSB and grown to logarithmic phase. These cultures were used for the next round of MIC exposure. Bacteria surviving after 10 rounds were grown for an additional 5 rounds (overnight) in TSB without any selection pressure to allow phenotype reversion, and then MIC determinations were performed to measure the sensitivity to Ply187 after nonselective growth.
Biofilm assay.
Biofilm disruption by the chimeric Ply187 was assayed as described in our recent study (29) using a Live/Dead BacLight staining kit (Invitrogen, Carlsbad, CA). Briefly, biofilms grown on glass coverslips were treated for 30 min with chimeric Ply187 (as per the MICs in Table 1) diluted in EB, EB alone, or gentamicin. The biofilms were then stained using the Live/Dead BacLight bacterial viability kit reagents and examined under an Eclipse 90i fluorescence microscope (Nikon, Melville, NY).
Ply187 treatment and assessment of retinal function.
Mice were treated in compliance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research, and all procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Wayne State University. Endophthalmitis was induced by intravitreal injection of S. aureus (strain RN6390) into C57BL/6 mouse eyes as described previously (30). The chimeric Ply187 (1 μg/μl) diluted in EB was administered intravitreally at 6 h (group I) or 12 h (group II) postinfection. Control groups included (i) eyes infected with S. aureus and treated with EB (EB C) and (ii) eyes infected with S. aureus without any further treatment. Clinical examinations were performed using slit lamp microscopy (Carl Zeiss 30SL-M; Carl Zeiss, Germany). The ocular disease was graded, and clinical scores from 0 to 4.0 were assigned by using a previously described scale (31). A clinical score of 4.0 indicated 100% damage, and based on the clinical scores the percent damage and percent retention of eyes were calculated in both Ply187-treated and EB-treated S. aureus-infected eyes. The effect of Ply187 treatment on retinal function was tested with a scotopic electroretinogram (ERG) as described in our recent studies (30, 31).
Bacterial burden estimations.
The bacterial burdens in both chimeric Ply187- and vehicle-treated eyes were estimated by using a bacterial plate count method. At the desired time point, the eyes were enucleated and homogenized in sterile PBS by using stainless steel beads and a tissue lyser (Qiagen, Valencia, CA). The homogenate was serially diluted in sterile PBS and plated on TSA plates. The results are expressed as the mean number of CFU/eye ± the standard error of the mean (SEM).
Bacterial growth in vitreous humor.
To determine the effect of vitreous humor (VH) on antimicrobial activity of chimeric Ply187, bacteria (105 CFU) were incubated with chimeric Ply187 in the presence or absence of calf VH. Following incubation at 37°C for the indicated times, bacterial growth in VH was determined by serial dilution plating.
In vitro inoculum effect.
In order to study the effect of Ply187 on increasing the CFU of bacteria, S. aureus strain RN6390 was grown to logarithmic phase (OD600, 0.4 to 0.6) and 10-fold serial dilutions were made in PBS, starting from 1:10 to 1:10,000. These bacterial dilutions were treated with Ply187 (as per the MICs in Table 1) for 1 h at room temperature, and CFU counts were determined by dilution plating on TSA plates.
Histopathology and TUNEL assays.
Embedding, sectioning, and hematoxylin and eosin stain (H&E) staining were performed by Excalibur Pathology Inc. (Oklahoma City, OK). For immunostaining, the eyes were fixed in Tissue-Tek OCT (Sakura, Torrance, CA), and 5-μm-thick sagittal sections were collected from each eye and mounted onto microscope slides. Terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling (TUNEL) staining was performed on retinal cryo-sections by using an ApopTag fluorescein in situ apoptosis detection kit (Millipore, Billerica, MA) as per the manufacturer's instructions.
Assessment of inflammation and neutrophil infiltration.
The effect of Ply187 treatment on intraocular inflammation was assessed by measuring cytokine/chemokine levels in retinal tissue lysates via an enzyme-linked immunosorbent assay (ELISA) for mouse interleukin-6 (IL-6), IL-1β, and tumor necrosis factor alpha (TNF-α) (BD Biosciences, San Diego, CA) and macrophage inflammatory protein 2 (MIP-2) and KC (R&D Systems, Minneapolis, MN). Flow cytometry with an Accuri C6 apparatus (BD Biosciences, Ann Arbor, MI) was used to determine polymorphonuclear leukocyte (PMN) infiltration as described previously (31, 32).
Statistical analyses.
The statistical analyses were performed using Prism version 6.02 (GraphPad, San Diego, CA). Student's t test was used to determine statistical significance for comparisons of two groups.
RESULTS
Chimeric Ply187 exhibits staphylolytic activity, disrupts biofilms, and reduces resistance development.
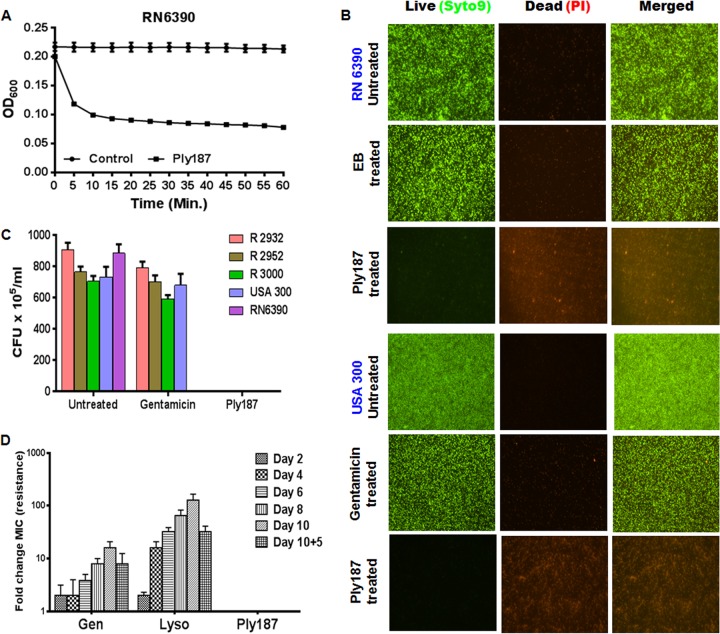
We initiated the study by performing a plate lysis assay to test the lytic activity of Ply187, and we found that the application of either 1 or 10 μg of chimeric Ply187 on a freshly plated S. aureus strain RN6390 lawn resulted in clear zones of inhibition, while 0.1 μg was unable to prevent growth of the cells (data not shown). We also determined the MIC of Ply187 against strains RN6390 and USA300 and three clinical MRSA isolates (Table 1). The lytic activity of Ply187 was assessed by using the turbidity reduction method, and this showed that incubation of RN6390 (Fig. 1A) or the clinical strains/isolates (images not shown) with chimeric Ply187 resulted in a time-dependent reduction in the turbidity of suspension cultures, indicating strong staphylolytic activity.
FIG 1.
Antibacterial activity, biofilm disruption, and resistance development of the chimeric Ply187 toward S. aureus. (A) A turbidity reduction assay was performed by incubating the S. aureus RN6390 strain with chimeric Ply187 (Table 1) diluted in assay buffer and monitoring the reduction in turbidity every 5 min for up to 1 h. OD600, the optical density at 600 nm. (B) Chimeric Ply187, EB, or gentamicin (for antibiotic-resistant strains) was applied to coverslips containing S. aureus RN6390 or CA-MRSA USA300 biofilms for 30 min. Bacterial killing was assessed via Live/Dead staining and subsequent fluorescence microscopy imaging. (C) The viability of biofilm-associated bacteria was enumerated by CFU counts. (D) A resistance development assay was performed against strain RN6390 by repeated exposure of bacteria (105 CFU/well) to 2-fold serial dilutions of Ply187, gentamicin (Gen), and lysostaphin (Lyso). Cells surviving at half of the MIC were used as an inoculum for each subsequent round of exposure. The change in susceptibility was recorded and is presented as the fold change in the MIC (resistance) from day 1 to 10 of exposure and also after 5 days of growth (without any selection pressure) following the 10th day, represented as 10 + 5. The data represent means ± SEM of two independent experiments performed in triplicate.
The capacity of chimeric endolysin to disrupt biofilms was determined by fluorescent imaging of Ply187-treated and untreated biofilms of S. aureus by using the Live/Dead BacLight kit. The green fluorescence (due to SYTO 9) in EB/gentamicin-treated and untreated control biofilms (S. aureus) contrasted with the chimeric Ply187-treated biofilms, which had negligible green fluorescence and only red fluorescence (due to propidium iodide invasion of lysed/disrupted cells), indicating that chimeric Ply187 successfully disrupted biofilms produced by RN6390 and CA-MRSA strain USA300 (Fig. 1B) as well as by other clinical MRSA strains/isolates listed in Table 1 (images not shown). Upon microscopic examination, the residual staining in the Ply187-treated biofilm was determined to be cellular debris, with little to no intact staphylococci. To provide further evidence of biofilm disruption and for quantification of cell death, we enumerated the CFU from both treated and untreated biofilms. Our results indicated that the untreated biofilms showed high CFU counts for all S. aureus strains tested, in contrast to the Ply187-treated biofilms, where the CFU counts were almost 0 (Fig. 1C). As shown in Fig. 1C, the biofilm treated with gentamicin showed complete inhibition of bacterial growth in the MSSA RN6390 strain, while gentamicin had no effect on the biofilms produced by MRSA strains/isolates. Therefore, we concluded that the disruption of biofilms by endolysin was due to its antimicrobial activity (lysis).
To assess the development of resistance against Ply187, we initially attempted to select mutants by serial subculture of S. aureus RN6390 in the presence of sub-MICs of Ply187. Gentamicin and lysostaphin were used as controls. Our data showed that strain RN6390 failed to develop any resistance against Ply187 after up to 10 rounds of exposure. In contrast, this strain exhibited 128- and 16-fold changes in the MICs of lysostaphin and gentamicin, respectively (Fig. 1D). However, this acquired resistance was decreased up to 32-fold in the case of lysostaphin and up to 8-fold in the case of gentamicin when the bacteria were grown in the absence of the lysostaphin or gentamicin for phenotype reversion.
Chimeric Ply187 treatment reduces bacterial burden and attenuates the development of endophthalmitis in mice.
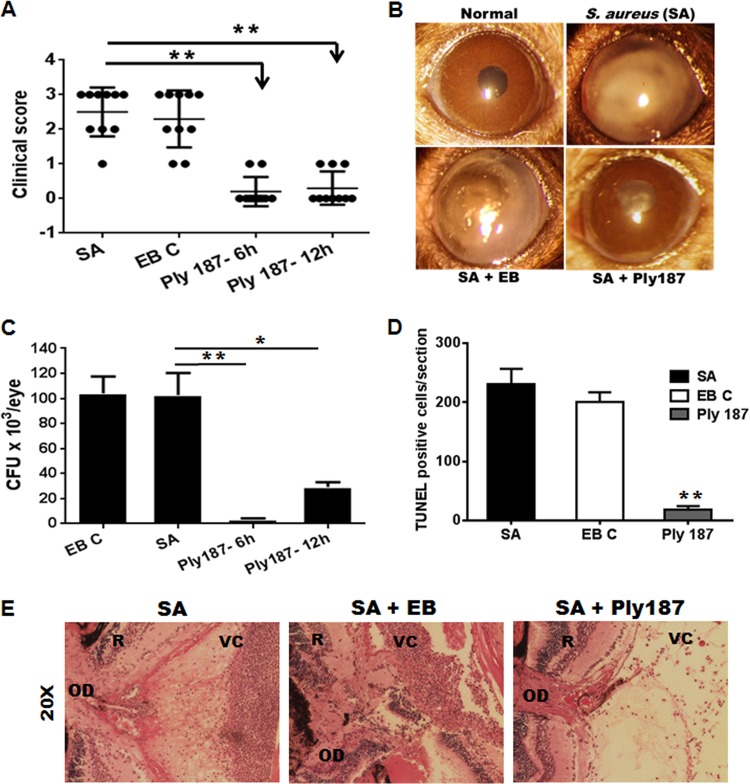
We hypothesized that, similar to antibiotics, chimeric Ply187 would have beneficial effects in protecting eyes from endophthalmitis. To test this hypothesis, we used a C57BL/6 mouse model of S. aureus endophthalmitis. Chimeric Ply187 was administered intravitreally (1 μg/eye in a 1-μl volume of EB) 6 h and 12 h after bacterial infection. Eyes without any treatment or eyes injected with EB served as controls. After 24 h, each infected eye was assigned a clinical score, and the mean clinical scores of chimeric Ply187-treated eyes (Fig. 2A) were significantly (P < 0.005) lower in both the 6-h (mean clinical score, 0.2) and 12-h (mean, 0.3) treatment groups compared to the control EB-treated eyes (mean score, 2.3) and infected control eyes (mean, 2.5). The conversion of clinical scores into the percentage of the eye destroyed or retained (33) revealed 90 to 95% retention of eyes in the treatment group. Figure 2B shows slit lamp images of chimeric Ply187-treated eyes, with clear/transparent corneas and/or anterior chambers, whereas the S. aureus-infected and EB-treated control eyes showed dense corneal opacity, hyopyon, and a severe inflammatory response. We next examined the effect of chimeric Ply187 treatment on bacterial clearance in mouse eyes. The intravitreal injection of chimeric Ply187 at 6 h and 12 h postinfection drastically reduced the bacterial burden in the eyes (Fig. 2C). We observed that, on average, 1.04 × 105 CFU/eye were recovered from infected and EB-treated eyes, compared to 3.6 × 103 and 2.9 × 104 CFU/eye in the 6-h and 12-h treatment groups, respectively.
FIG 2.
Effects of chimeric Ply187on the severity of S. aureus endophthalmitis in mice. (A) The eyes (n = 10) of C57BL/6 mice were infected with S. aureus strain RN6390 and, either 6 h or 12 h postinfection, Ply187 or EB (EB C) was administered by intravitreal injection. Individual eyes were assigned clinical scores (range, 0 to 4; black dots represent an eye) and are presented as mean clinical scores (with SEM [bars]). (B) Slight lamp microscopy examination was performed, and micrographs were taken for representative eyes. (C) Twenty-four hours after Ply187 administration, the eyes were enucleated and homogenized, and the bacterial burden was estimated via serial dilution plating. (D) To measure the retinal cell death, the eyes were embedded in OCT and the cryosections were subjected to TUNEL staining. The total number of dead cells was counted in retinal sections and is presented as the mean (± standard deviation) number of TUNEL-positive cells. (E) For histological analysis following infection, untreated or Ply187 (6 h)-treated eyes were enucleated and H&E stained. Student's t test (**, P < 0.05) was used for the statistical analysis, and comparisons were made between S. aureus (SA) alone, EB C, and Ply187-treated groups. OD, optic disc; R, retina; VC, vitreous chamber; L, lens.
Having shown that chimeric Ply187 treatment attenuates the clinical symptoms of endophthalmitis and reduces the bacterial burden, we determined its protective effects on the retina at the tissue level. First, TUNEL staining was performed to assess retinal cell death, and this showed significantly reduced cell death (average, 17.66 TUNEL+ cells/section) with chimeric Ply187 treatment versus the untreated controls (average, 230 TUNEL+ cells/section) and the EB-treated controls (average, 200.33 TUNEL+ cells/section) (Fig. 2D). Second, H&E staining was performed to evaluate the retinal structural integrity, and our data revealed that the infected eyes of untreated or EB-treated mice showed a heavy cellular infiltrate in the posterior segment, as well as retinal folding (Fig. 2E). In contrast, treatment with chimeric Ply187 preserved the integrity of the retina, and little infiltration was detected in the posterior segment. Taken together, these results indicate that chimeric Ply187 treatment significantly improves the disease outcome.
Chimeric Ply187-treated eyes exhibited reduced inflammatory responses.
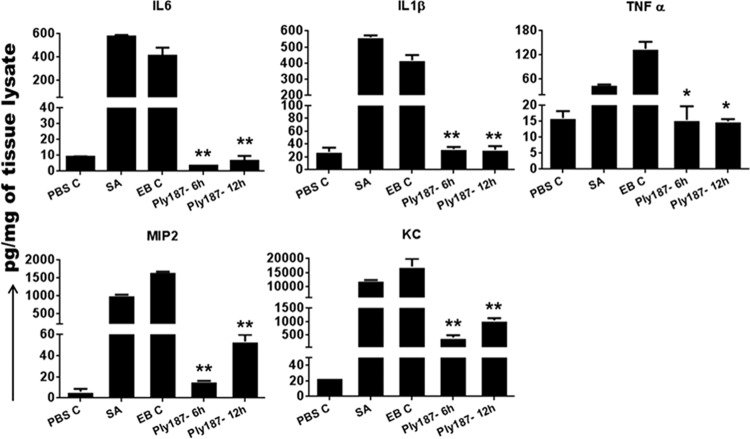
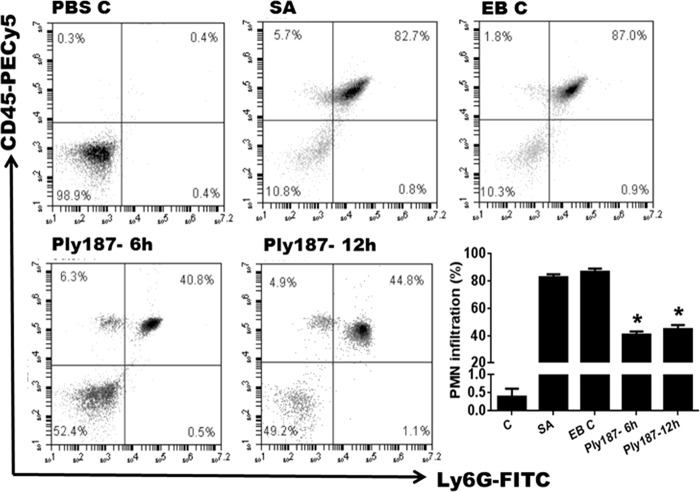
One of the hallmarks of staphylococcal endophthalmitis is the increased levels of inflammatory cytokines and chemokines (34). We observed that chimeric Ply187 treatment significantly suppressed the inflammatory response, as evidenced by dramatically reduced levels of IL-6, IL-1β, TNF-α, MIP-2 (CXCL2), and KC (CXCL1) in the Ply187-treated eyes compared to vehicle-treated and S. aureus control eyes (Fig. 3). The 6-h and 12-h treatment groups had a similar reduction in the levels of the assessed cytokines/chemokines, with the exception of MIP-2, which showed a greater reduction after the 6-h than after the 12-h treatment. Similar to the reductions in the levels of inflammatory mediators, our flow cytometry data revealed an ∼50% reduction in PMN infiltration in eyes treated with chimeric Ply187 compared to vehicle-treated eyes (Fig. 4).
FIG 3.
Effect of chimeric Ply187 on inflammatory cytokines in vivo. Following treatment with chimeric Ply187 or EB (EB C), the eyes (n = 4/group) were enucleated and 10 μg protein lysate was used for the detection of the indicated inflammatory cytokines/chemokines in an ELISA. *, P < 0.05; **, P < 0.005 (Student's t test, SA or EB C versus Ply187 treated).
FIG 4.
Infiltration of PMN in the Ply187-treated mouse retina/vitreous. Infected eyes were given intravitreal injections of either EB or chimeric Ply187 either 6 or 12 h postinfection. Twenty-four hours post-Ply187 treatment, the eyes (n = 6) were enucleated and the retina/vitreous from two eyes were pooled to make single-cell suspensions. The cells were stained with anti-CD45 and anti-Ly6G monoclonal antibodies. Postacquisition, the cells were size gated to differentiate them from debris. The percentage of dually positive PMN was determined in a CD45-verus-Ly6G dot plot (upper right quadrant). The graph shows cumulative data from two independent experiments. *, P < 0.05 (Student's t test, comparison of control versus treated groups).
Chimeric Ply187 treatment allows the maintenance of normal retinal function.
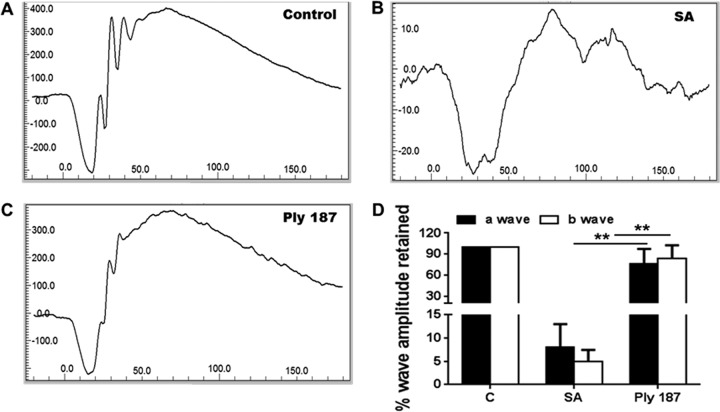
To determine whether chimeric Ply187 treatment also preserves retinal function in infected eyes, we determined scotopic ERG responses. The ERG response showed both normal a-waves (the response generated from photoreceptors) and b-waves (the response generated from the inner retina, mostly the bipolar cells) in the control (uninfected) and chimeric Ply187-treated eyes. Mice that received intravitreal injections of S. aureus demonstrated loss of retinal function with a significant decrease in both a-wave (92%) and b-wave (95%) amplitudes (Fig. 5). Remarkably, mice treated with chimeric Ply187 maintained their normal ERG with no significant decline in a- or b-wave amplitude.
FIG 5.
Effect of Ply187 treatment on retinal function. ERG responses to a 6-dB flash were recorded from (A) uninfected control, (B) S. aureus (SA)-infected, and (C) S. aureus-infected and Ply187-treated eyes (n = 4 to 6). (D) The percentage amplitudes of the a- and b-waves retained in treated and untreated mice were compared to those for normal uninfected mice and are presented as means ± standard deviations. **, P < 0.005 (Student's t test, comparison of control versus treated groups).
DISCUSSION
In the present study, we demonstrated that a single intravitreal injection of chimeric endolysin Ply187AN-KSH3b protected mice from the development of endophthalmitis, as evidenced by reduced bacterial load, intraocular inflammation, and tissue damage and preserved retinal function. Most importantly, repeated exposure of Ply187 did not lead to development of resistance in S. aureus. In view of the limited supply of new antimicrobial agents and the increasing antibiotic resistance among ocular pathogens, our study demonstrates, for the first time, the potential use of phage endolysin therapy in the treatment of staphylococcal endophthalmitis.
In the last 2 decades, phage endolysins have emerged as unique antimicrobial agents that possess exceptionally high specificity and an ability to act against multidrug-resistant microbes (19, 35–37).We recently generated a potent chimeric endolysin, Ply187AN-KSH3b, by fusing the cysteine, histidine-dependent amidohydrolase/endopeptidase domain of endolysin Ply187 from staphylococcal phage 187 with the SH3b cell wall binding domain of LysK from staphylococcal phage K. This chimeric Ply187AN-KSH3b was found to be a more effective antimicrobial agent than the full-length Ply187 or the truncated Ply187 (Ply187AN), and it also outperformed the known high-activity lysin, LysK (38). In this study, we showed that chimeric Ply187 exerts strong lytic activity against the RN6390 strain of S. aureus, which causes endophthalmitis in mice. In addition, Ply187 also showed a strong staphylolytic activity against strain USA300 and three other clinical MRSA isolates. Similar to previous studies (28), our resistance development data showed that S. aureus was unable to develop resistance against Ply187 following repeated exposure. Furthermore, chimeric Ply187 disrupted the biofilms formed by antibiotic-resistant S. aureus strains. As biofilm formation plays an important role in the pathogenesis of ocular infections, including endophthalmitis (39–41), the dispersion of biofilms by chimeric Ply187 further indicated its therapeutic potential in the treatment of endophthalmitis.
Since bacterial toxins and degradative enzymes can cause irreversible damage to the retina (42), it is important to choose antimicrobials with rapid bactericidal activity when treating ocular infection. In cases of suspected bacterial endophthalmitis, intravitreal injection of both vancomycin and an aminoglycoside or an expanded-spectrum cephalosporin is recommended, while vitrectomy may be needed for severe cases (6). However, MDR ocular S. aureus strains are becoming increasingly more prevalent (44), and the chimeric Ply187 could be effective against MDR strains of S. aureus. We showed that intravitreal injection of chimeric Ply187 significantly reduced the bacterial burden in C57BL/6 mouse eyes at either 6 or 12 h postinfection. We noticed that the bacterial burden in the 12-h treatment group was slightly higher than in the 6-h treatment group. To test whether this phenomenon was due to reduced activity of Ply187 in an environment where bacteria are proliferating, we performed an inoculum-effect study. To this end, our data showed that the activity of Ply187 is independent on bacterial load (data not shown). One possible explanation could be that by 12 h after infection, some bacteria might not have access to endolysin, as they have been internalized/phagocytized by infiltrating PMN and monocytes (43–47). As incubation of chimeric Ply187 with vitreous humor did not affect its bactericidal activity (data not shown), the rapid efficacy of chimeric Ply187 in killing bacteria inside the eye suggests that there is no specific cellular factor (e.g., proteases) that can prevent the chimeric endolysin activity. Hence, we propose that endolysins, when delivered intravitreally, can be efficacious in limiting intraocular bacterial growth.
S. aureus produces a variety of virulence factors that are either cell wall-associated molecules or secreted proteins (often toxins). The coordinated actions of these virulence factors lead to tissue destruction and the clinical manifestations of endophthalmitis (48). Our histological analysis revealed that infected eyes treated with the chimeric Ply187 had reduced retinal damage compared to untreated eyes. The rapid decline in ERG response in the control group suggested the dysfunction of retinal cells. This in part could have been due to the death of retinal cells, as reported in previous studies (31, 33). Our TUNEL data also supported these findings and suggested that in bacterial endophthalmitis the cells undergoing apoptosis are mainly retinal cells. Moreover, as the chimeric Ply187-treated eyes retained significant retinal function, this could also have been due to the reduced retinal cell death in the treatment group. Similar to reduced tissue damage, the levels of inflammatory cytokines/chemokines (IL-6, IL-1β, TNF-α, MIP-2, and KC) were also attenuated by chimeric Ply187 treatment. This is advantageous, because an excessive inflammatory response can be harmful to retinal neurons. Neutrophils play an important role in bacterial clearance but, paradoxically, they are also involved in the pathology of endophthalmitis (3, 6). Our data showed that chimeric Ply187 treatment reduced PMN infiltration by 40 to 50% compared to control mice. The reduced PMN infiltration response could have been due to the reduced bacterial burden.
If one were to develop phage endolysins as possible therapeutic agents for humans, safety would always be of great concern. Studies have shown that a rapid release of bacterial ghosts and the sudden release of intracellular bacterial components may amplify the inflammatory response, causing septic shock and multiple organ failure (49). However, none of the endolysin studies performed in vivo so far have reported such side effects (50, 51). Our cytotoxicity analysis showed that chimeric Ply187 does not cause retinal cell death either in vitro (cultured microglia, Müller glia, and RPE cells) or in vivo (data not shown). Similarly, the intravitreal injection of chimeric Ply187 alone does not evoke an inflammatory response in the eyes, suggesting there are no adverse effects from endolysins in the eye.
In conclusion, we demonstrated that a single intravitreal injection of phage endolysin was efficient in protecting mouse eyes from staphylococcal endophthalmitis. Thus, based on this first proof-of-principle study, we suggest that phage lytic enzyme-based therapy be explored for the treatment of endophthalmitis in patients with antibiotic-resistant bacterial infections.
ACKNOWLEDGMENTS
This research was supported by NIH grant R01 EY19888, Research to Prevent Blindness (RPB), and a William and Mary Greve Special Scholar award.
We sincerely thank Michael J. Rybak (Department of Pharmaceutical Sciences, Wayne State University, Detroit, MI) for providing clinical isolates, Deepa Talreja for her technical assistance in some experiments, and Bruce Rottmann for editing the manuscript.
Footnotes
Published ahead of print 2 June 2014
REFERENCES
- 1.Chiquet C, Cornut PL, Benito Y, Thuret G, Maurin M, Lafontaine PO, Pechinot A, Palombi K, Lina G, Bron A, Denis P, Carricajo A, Creuzot C, Romanet JP, Vandenesch F. 2008. Eubacterial PCR for bacterial detection and identification in 100 acute postcataract surgery endophthalmitis. Invest. Ophthalmol. Vis. Sci. 49:1971–1978. 10.1167/iovs.07-1377 [DOI] [PubMed] [Google Scholar]
- 2.Campbell RJ, Bronskill SE, Bell CM, Paterson JM, Whitehead M, Gill SS. 2010. Rapid expansion of intravitreal drug injection procedures, 2000 to 2008: a population-based analysis. Arch. Ophthalmol. 128:359–362. 10.1001/archophthalmol.2010.19 [DOI] [PubMed] [Google Scholar]
- 3.Sadaka A, Durand ML, Gilmore MS. 2012. Bacterial endophthalmitis in the age of outpatient intravitreal therapies and cataract surgeries: host-microbe interactions in intraocular infection. Prog. Retin. Eye Res. 31:316–331. 10.1016/j.preteyeres.2012.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Scott IU, Flynn HW., Jr 2007. Reducing the risk of endophthalmitis following intravitreal injections. Retina 27:10–12. 10.1097/IAE.0b013e3180307271 [DOI] [PubMed] [Google Scholar]
- 5.Speaker MG, Milch FA, Shah MK, Eisner W, Kreiswirth BN. 1991. Role of external bacterial flora in the pathogenesis of acute postoperative endophthalmitis. Ophthalmology 98:639–649. 10.1016/S0161-6420(91)32239-5 [DOI] [PubMed] [Google Scholar]
- 6.Callegan MC, Gilmore MS, Gregory M, Ramadan RT, Wiskur BJ, Moyer AL, Hunt JJ, Novosad BD. 2007. Bacterial endophthalmitis: therapeutic challenges and host-pathogen interactions. Prog. Retin. Eye Res. 26:189–203. 10.1016/j.preteyeres.2006.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lloyd JC, Braga-Mele R. 2009. Incidence of postoperative endophthalmitis in a high-volume cataract surgicentre in Canada. Can. J. Ophthalmol. 44:288–292. 10.3129/i09-052 [DOI] [PubMed] [Google Scholar]
- 8.Yin VT, Weisbrod DJ, Eng KT, Schwartz C, Kohly R, Mandelcorn E, Lam WC, Daneman N, Simor A, Kertes PJ. 2013. Antibiotic resistance of ocular surface flora with repeated use of a topical antibiotic after intravitreal injection. JAMA Ophthalmol. 131:456–461. 10.1001/jamaophthalmol.2013.2379 [DOI] [PubMed] [Google Scholar]
- 9.Storey P, Dollin M, Pitcher J, Reddy S, Vojtko J, Vander J, Hsu J, Garg SJ. 2014. The role of topical antibiotic prophylaxis to prevent endophthalmitis after intravitreal injection. Ophthalmology 121:283–289. 10.1016/j.ophtha.2013.08.037 [DOI] [PubMed] [Google Scholar]
- 10.Alabiad CR, Miller D, Schiffman JC, Davis JL. 2011. Antimicrobial resistance profiles of ocular and nasal flora in patients undergoing intravitreal injections. Am. J. Ophthalmol. 152:999–1004.e1002. 10.1016/j.ajo.2011.05.026 [DOI] [PubMed] [Google Scholar]
- 11.Kowalski RP, Romanowski EG, Mah FS, Sasaki H, Fukuda M, Gordon YJ. 2008. A comparison of moxifloxacin and levofloxacin topical prophylaxis in a fluoroquinolone-resistant Staphylococcus aureus rabbit model. Jpn. J. Ophthalmol. 52:211–216. 10.1007/s10384-008-0530-1 [DOI] [PubMed] [Google Scholar]
- 12.Deramo VA, Lai JC, Winokur J, Luchs J, Udell IJ. 2008. Visual outcome and bacterial sensitivity after methicillin-resistant Staphylococcus aureus-associated acute endophthalmitis. Am. J. Ophthalmol. 145:413–417. 10.1016/j.ajo.2007.10.020 [DOI] [PubMed] [Google Scholar]
- 13.DeLeo FR, Chambers HF. 2009. Reemergence of antibiotic-resistant Staphylococcus aureus in the genomics era. J. Clin. Invest. 119:2464–2474. 10.1172/JCI38226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Alabiad CR, Miller D, Schiffman JC, Davis JL. 2011. Antimicrobial resistance profiles of ocular and nasal flora in patients undergoing intravitreal injections. Am. J. Ophthalmol. 152:999–1004.e1002. 10.1016/j.ajo.2011.05.026 [DOI] [PubMed] [Google Scholar]
- 15.Moss JM, Sanislo SR, Ta CN. 2009. A prospective randomized evaluation of topical gatifloxacin on conjunctival flora in patients undergoing intravitreal injections. Ophthalmology 116:1498–1501. 10.1016/j.ophtha.2009.02.024 [DOI] [PubMed] [Google Scholar]
- 16.Yin VT, Weisbrod DJ, Eng Schwartz KTC, Kohly R, Mandelcorn E, Lam WC, Daneman N, Simor A, Kertes PJ. 2013. Antibiotic resistance of ocular surface flora with repeated use of a topical antibiotic after intravitreal injection. JAMA Ophthalmol. 131:456–461. 10.1001/jamaophthalmol.2013.2379 [DOI] [PubMed] [Google Scholar]
- 17.Donovan DM. 2007. Bacteriophage and peptidoglycan degrading enzymes with antimicrobial applications. Recent Pat. Biotechnol. 1:113–122. 10.2174/187220807780809463 [DOI] [PubMed] [Google Scholar]
- 18.Schmelcher M, Donovan DM, Loessner MJ. 2012. Bacteriophage endolysins as novel antimicrobials. Future Microbiol. 7:1147–1171. 10.2217/fmb.12.97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Doehn JM, Fischer K, Reppe K, Gutbier B, Tschernig T, Hocke AC, Fischetti VA, Loffler J, Suttorp N, Hippenstiel S, Witzenrath M. 2013. Delivery of the endolysin Cpl-1 by inhalation rescues mice with fatal pneumococcal pneumonia. J. Antimicrob. Chemother. 68:2111–2117. 10.1093/jac/dkt131 [DOI] [PubMed] [Google Scholar]
- 20.Nelson D, Loomis L, Fischetti VA. 2001. Prevention and elimination of upper respiratory colonization of mice by group A streptococci by using a bacteriophage lytic enzyme. Proc. Natl. Acad. Sci. U. S. A. 98:4107–4112. 10.1073/pnas.061038398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oechslin F, Daraspe J, Giddey M, Moreillon P, Resch G. 2013. In vitro characterization of PlySK1249, a novel phage lysin, and assessment of its antibacterial activity in a mouse model of Streptococcus agalactiae bacteremia. Antimicrob. Agents Chemother. 57:6276–6283. 10.1128/AAC.01701-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gervasi T, Horn N, Wegmann U, Dugo G, Narbad A, Mayer MJ. 2014. Expression and delivery of an endolysin to combat Clostridium perfringens. Appl. Microbiol. Biotechnol. 98:2495–2505. 10.1007/s00253-013-5128-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gorski A, Targonska M, Borysowski J, Weber-Dabrowska B. 2009. The potential of phage therapy in bacterial infections of the eye. Ophthalmologica 223:162–165. 10.1159/000193293 [DOI] [PubMed] [Google Scholar]
- 24.Proskurov VA. 1970. Treatment of staphylococcal diseases of the eye. Vestn. Oftalmol. 6:82–83 (In Russian.) [PubMed] [Google Scholar]
- 25.Mao J, Schmelcher M, Harty WJ, Foster-Frey J, Donovan DM. 2013. Chimeric Ply187 endolysin kills Staphylococcus aureus more effectively than the parental enzyme. FEMS Microbiol. Lett. 342:30–36. 10.1111/1574-6968.12104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Becker SC, Foster-Frey J, Stodola AJ, Anacker D, Donovan DM. 2009. Differentially conserved staphylococcal SH3b_5 cell wall binding domains confer increased staphylolytic and streptolytic activity to a streptococcal prophage endolysin domain. Gene 443:32–41. 10.1016/j.gene.2009.04.023 [DOI] [PubMed] [Google Scholar]
- 27.Becker SC, Dong S, Baker JR, Foster-Frey J, Pritchard DG, Donovan DM. 2009. LysK CHAP endopeptidase domain is required for lysis of live staphylococcal cells. FEMS Microbiol. Lett. 294:52–60. 10.1111/j.1574-6968.2009.01541.x [DOI] [PubMed] [Google Scholar]
- 28.Rodriguez-Rubio L, Martinez B, Rodriguez A, Donovan DM, Gotz F, Garcia P. 2013. The phage lytic proteins from the Staphylococcus aureus bacteriophage vB_SauS-phiIPLA88 display multiple active catalytic domains and do not trigger staphylococcal resistance. PLoS One 8:e64671. 10.1371/journal.pone.0064671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Talreja D, Muraleedharan C, Gunathilaka G, Zhang Y, Kaye KS, Walia SK, Kumar A. 6 February 2014. Virulence properties of multidrug resistant ocular isolates of Acinetobacter baumannii. Curr. Eye Res. 10.3109/02713683.2013.873055 [DOI] [PubMed] [Google Scholar]
- 30.Kumar A, Singh CN, Glybina IV, Mahmoud TH, Yu FS. 2010. Toll-like receptor 2 ligand-induced protection against bacterial endophthalmitis. J. Infect. Dis. 201:255–263. 10.1086/649589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Talreja D, Kaye KS, Yu F, Walia SK, Kumar A. 2014. Pathogenicity of ocular isolates of Acinetobacter baumannii in a mouse model of bacterial endophthalmitis. Invest. Ophthalmol. Vis. Sci. 55:2392–2402. 10.1167/iovs.13-13401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kochan T, Singla A, Tosi J, Kumar A. 2012. Toll-like receptor 2 ligand pretreatment attenuates retinal microglial inflammatory response but enhances phagocytic activity toward Staphylococcus aureus. Infect. Immun. 80:2076–2088. 10.1128/IAI.00149-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Whiston EA, Sugi N, Kamradt MC, Sack C, Heimer SR, Engelbert M, Wawrousek EF, Gilmore MS, Ksander BR, Gregory MS. 2008. αB-crystallin protects retinal tissue during Staphylococcus aureus-induced endophthalmitis. Infect. Immun. 76:1781–1790. 10.1128/IAI.01285-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Callegan MC, Booth MC, Jett BD, Gilmore MS. 1999. Pathogenesis of gram-positive bacterial endophthalmitis. Infect. Immun. 67:3348–3356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fischetti VA, Nelson D, Schuch R. 2006. Reinventing phage therapy: are the parts greater than the sum? Nat. Biotechnol. 24:1508–1511. 10.1038/nbt1206-1508 [DOI] [PubMed] [Google Scholar]
- 36.Schmelcher M, Donovan DM, Loessner MJ. 2012. Bacteriophage endolysins as novel antimicrobials. Future Microbiol. 7:1147–1171. 10.2217/fmb.12.97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pastagia M, Euler C, Chahales P, Fuentes-Duculan J, Krueger JG, Fischetti VA. 2011. A novel chimeric lysin shows superiority to mupirocin for skin decolonization of methicillin-resistant and -sensitive Staphylococcus aureus strains. Antimicrob. Agents Chemother. 55:738–744. 10.1128/AAC.00890-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mao J, Schmelcher M, Harty WJ, Foster-Frey J, Donovan DM. 2013. Chimeric Ply187 endolysin kills Staphylococcus aureus more effectively than the parental enzyme. FEMS Microbiol. Lett. 342:30–36. 10.1111/1574-6968.12104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Leid JG, Costerton JW, Shirtliff ME, Gilmore MS, Engelbert M. 2002. Immunology of Staphylococcal biofilm infections in the eye: new tools to study biofilm endophthalmitis. DNA Cell Biol. 21:405–413. 10.1089/10445490260099692 [DOI] [PubMed] [Google Scholar]
- 40.Behlau I, Gilmore MS. 2008. Microbial biofilms in ophthalmology and infectious disease. Arch. Ophthalmol. 126:1572–1581. 10.1001/archopht.126.11.1572 [DOI] [PubMed] [Google Scholar]
- 41.Suzuki T, Uno T, Kawamura Y, Joko T, Ohashi Y. 2005. Postoperative low-grade endophthalmitis caused by biofilm-producing coccus bacteria attached to posterior surface of intraocular lens. J. Cataract Refract. Surg. 31:2019–2020. 10.1016/j.jcrs.2005.02.032 [DOI] [PubMed] [Google Scholar]
- 42.Bertino JS., Jr 2009. Impact of antibiotic resistance in the management of ocular infections: the role of current and future antibiotics. Clin. Ophthalmol. 3:507–521. 10.2147/OPTH.S5778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.McDonald M, Blondeau JM. 2010. Emerging antibiotic resistance in ocular infections and the role of fluoroquinolones. J. Cataract Refract. Surg. 36:1588–1598. 10.1016/j.jcrs.2010.06.028 [DOI] [PubMed] [Google Scholar]
- 44.Rogers DE, Tompsett R. 1952. The survival of staphylococci within human leukocytes. J. Exp. Med. 95:209–230. 10.1084/jem.95.2.209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Melly MA, Thomison JB, Rogers DE. 1960. Fate of staphylococci within human leukocytes. J. Exp. Med. 112:1121–1130. 10.1084/jem.112.6.1121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kapral FA, Shayegani MG. 1959. Intracellular survival of staphylococci. J. Exp. Med. 110:123–138. 10.1084/jem.110.1.123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gresham HD, Lowrance JH, Caver TE, Wilson BS, Cheung AL, Lindberg FP. 2000. Survival of Staphylococcus aureus inside neutrophils contributes to infection. J. Immunol. 164:3713–3722. 10.4049/jimmunol.164.7.3713 [DOI] [PubMed] [Google Scholar]
- 48.Shamsuddin N, Kumar A. 2011. TLR2 mediates the innate response of retinal Muller glia to Staphylococcus aureus. J. Immunol. 186:7089–7097. 10.4049/jimmunol.1100565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nau R, Eiffert H. 2002. Modulation of release of proinflammatory bacterial compounds by antibacterials: potential impact on course of inflammation and outcome in sepsis and meningitis. Clin. Microbiol. Rev. 15:95–110. 10.1128/CMR.15.1.95-110.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Entenza JM, Loeffler JM, Grandgirard D, Fischetti VA, Moreillon P. 2005. Therapeutic effects of bacteriophage Cpl-1 lysin against Streptococcus pneumoniae endocarditis in rats. Antimicrob. Agents Chemother. 49:4789–4792. 10.1128/AAC.49.11.4789-4792.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Witzenrath M, Schmeck B, Doehn JM, Tschernig T, Zahlten J, Loeffler JM, Zemlin M, Muller H, Gutbier B, Schutte H, Hippenstiel S, Fischetti VA, Suttorp N, Rosseau S. 2009. Systemic use of the endolysin Cpl-1 rescues mice with fatal pneumococcal pneumonia. Crit. Care Med. 37:642–649. 10.1097/CCM.0b013e31819586a6 [DOI] [PubMed] [Google Scholar]