Abstract
Objective(s):
The relationship between tramadol, as an antinociceptive drug, and locus coeruleus (LC), the main noradrenergic nucleus of the brain that affects regulation and modulation of pain through descending noradrenergic pathways was investigated.
Materials and Methods:
Male Sprague-Dawley rats were divided into four groups of 10 rats. The rats were fixed in stereotaxic instrument and then a probe was inserted into LC. Pain was induced by subcutaneous injection of 50 μl of 2.5% formalin 40 minutes after initiation of microdialysis in right hind paw, and nociceptive pain scores were calculated every 5 minutes. Subsequently noradrenaline (NA) and its metabolite, 3-methoxy-4-hydroxyphenylglycol (MHPG), were collected and measured by microdialysis of locus coeruleus in freely moving rats every 15 minutes during formalin injection.
Results:
Nociceptive pain scores observed in formalin test had the highest nociceptive sensation 5 minutes after injection. Significant rises in concentrations of NA and MHPG, in samples taken between 30 and 45 min after initiation of the locus coeruleus microdialysis, coincided with the peak of the pain after injection of formalin.
Conclusion:
According to concurrency of the highest nociceptive sensation and peak of NE and MHPG concentrations, tramadol can indirectly affect the LC by blocking the pain signals from different parts of the brain such as periaqueductal gray mater, central nucleus of amygdale or the spinal cord.
Keywords: Antinociceptive, Locus coeruleus, Microdialysis, Stereotaxic instrument, Tramadol
Introduction
Locus coeruleus (LC) or A6 region, in rat brain, is a cluster of noradrenalin-containing neurons located adjacent to the fourth ventricle in the pontine brainstem. LC has the ability of synthesizing neuropeptides, and as an important noradrenergic nucleus it plays an important role in the regulation of pain through descending noradrenergic pain regulatory pathways (1).
LC is densely innervated by serotonergic projections mainly from dorsal raphe (DR) and pericoerulear region (2). Serotonin (5-HT) exerts its effects through receptors like 5-HT1A and 5-HT2, while 5-HT and noradrenaline (NA) have partly opposite effects (3). The firing rate of noradrenergic neurons of LC is regulated by 5-HT. DR nucleus can affect LC by activation of 5-HT1A and 5-HT2A receptors (3). LC is the main source of noradrenergic neurons in mammalian brain and sends many projections into different parts of the brain including hypothalamus, thalamic relay nuclei, amygdala, basal telencephalon, the cortex, forebrain, brainstem, cerebellum and the spinal cord (4). There are sufficient evidence that the LC, with the help of excitatory and inhibitory pathways originating from nucleus paragigantocellularis and nucleus prepositus hypoglossi, have major role in modulation of the pain (5). Fluctuations of some neurotransmitters like noradrenaline and its metabolite have major role in pain regulatory function of LC (5). In order for estimating the effects of different drugs on pain sensation, formalin test can be employed as a standard method for inducing acute and chronic pain. The two main advantages of this test in comparison to other methods include the continuity of the test and the possibility to analyze animal behavior while it is freely moving (6).
Tramadol, with the chemical name of (±) cis-2-[(dimethylamino)methyl]-1-(3-methoxyphenyl) cyclohexanol hydrochloride, is a centrally acting synthetic analgesic being widely used in moderate to severe pain and is usually marketed as tramadol hydrochloride (7). Tramadol hydrochloride is a white, crystalline and odorless powder which is readily soluble in water and ethanol and has a pKa of 9.41 (7). This drug has many applications including treatment of rheumatoid arthritis, diabetic neuropathy, restless legs syndrome, post-herpetic neuralgia, acute opioid withdrawal and fibromyalgia (8). Tramadol is not in the category of non steroidal anti-inflammatory drugs (NSAIDs), thus it does not have the threat of stomach ulcers and gastrointernal bleeding that may happen with NSAIDs (8). The exact antinociceptive mechanisms of tramadol and its effects on NA and 5-HT concentrations in the spinal cord are not yet completely understood, but some suggested mechanisms are:
-
1)
Binding and stimulating the receptors; tramadol is a very weak agonist of μ-opioid receptors
-
2)
Serotonin Release
-
3)
Inhibition of NA and 5-HT reuptake
-
4)
Antagonizing the NMDA receptor
-
5)
Antagonizing the (α7)5 nicotinic acetylcholine receptor
-
6)
M1 and M3 muscarinic acetylcholine receptor antagonist (9).
Tramadol is metabolized to O-desmethyltramadol, with a similar but more potent effect to tramadol (10). The effects of tramadol, however, have not been understood.
Microdialysis is a technique for analysis of low molecular weight substances that can be found in extracellular fluids. It is a good way of studying the pharmacokinetics of drugs and molecules with less tissue disturbance (11). This technique involves the stereotactic insertion of a microdialysis probe into a selected area of the brain with a physiological solution being pumped through the probe. This sampling method has a historical background in the field of physiology and neuroscience since it is commonly used for collection of neurotransmitters (11).
The objective of this study was to find the relationship between tramadol, an antinociceptive drug, and LC, the main noradrenergic nucleus of the brain that affects regulation and modulation of pain through descending noradrenergic pathways (12).
Materials and Methods
Ethics
The protocol of this study was approved by Shiraz University of Medical Sciences Ethics Committee.
Animals
Forty 90-day-old male Sprague-Dawley rats, weighing 280–320 g, were divided into four groups of 10 rats. The rats were housed in standard Plexiglas® boxes with free access to food and water under light/dark of 12/12 hr (7 AM to 7 PM) cycles at a constant temperature of 21°C (13).
Study groups
The animals were randomly divided into four groups of 10 rats: group one (main control or control formalin and tramadol test)—the animals were injected with normal saline (IP and SC in the right hind paw); group two (control tramadol test)—the rats were injected with normal saline (IP) and formalin (SC in the right hind paw); group three (control formalin test)—the animals were injected with tramadol (IP) and normal saline (SC in the right hind paw); and group four (main test group)—the animals were injected with tramadol (IP) and formalin (SC in the hind paw).
Probe implantation
To implant the microdialysis probe, rats were anesthetized with sodium pentobarbital (50 mg/kg IP) and placed in stereotaxic apparatus (Stolting, USA). A hole was made in the skull and a probe was implanted into the left LC (A/P: -9.8 mm from the bregma, L/M: 1.2 mm D/V: -7.1 mm, vertically) (14).
After the operation, rats were placed in a single Plexiglas® cage (area of 50 cm2) for gaining their consciousness. The rats had free access to food and water (15).
Tramadol preparation and injection
Pure powder of tablet grade tramadol hydroch-loride was purchased from Sun Pharmaceutical Industries, India and 2 mg of powder was dissolved in 2 ml normal saline.
Microdialysis
Microdialysis was started 24 hours after insertion of the microdialysis probe. Concentric probes of microdialysis (4.2 mm active membrane; OD 0.52 mm) (CMA/Microdialysis, Inc., Acton) with polycarbonate membrane with a molecular weight cut-off value of 20 kd and an exposed membrane surface of 0.5×2.0 mm were used. Following 24 hours of the stereotaxic operation, ACSF (Artificial cerebrospinal fluid) was perfused through a microdialysis probe with a flow rate of 2 μl/min. Samples were collected in 15 min intervals and 30 μl of the perfused fluid were collected in every tube located in a cool micro-fraction collector. Microdialysate samples were collected continuously and immediately afterwards refrigerated at -80°C for analysis (16, 17).
ACSF preparation
The composition of ACSF was 125 mM NaCl, 3 mM KCl, 0.75 mM MgSO4 and 1.2 mM CaCl2, pH 7.4 (17).
Formalin test
For induction of pain, 50 μl of 2.5% formalin was applied (SC) with a 27 G needle to the right hind paw of rats. Afterward, each animal’s responses were monitored with the help of objective behavioral criteria, such as early favoring, biting, flinching and licking of the hind paw and scored as follows:
0- Animal’s injected paw is completely on the floor and can endure the animal’s weight.
1- Animal’s injected paw is partially situated on the floor and is partially elevated.
2- Animal’s injected paw is elevated without any contact with the surface.
3- Animal shakes, licks or bites the injected paw (19).
Verifying the correct probe placement and entrance to the LC
After the end of microdialysis, each rat was sacrificed by ether and its brain was fixed for histological verification (20).
Sample collection
Microdialysis samples were collected every 15 min in 1.5 ml Eppendorf tubes. ACSF was pushed by a microdialysis pump through tubes at a constant rate of 2 μl/min. After 15 min, 30 μl of the perfused ACSF was collected in each tube. These specimens were frozen on dry-ice and stored in an ultra-low temperature freezer at -80°C until HPLC analysis.
HPLC analysis
Microdialysis samples were measured by high performance liquid chromatography (HPLC) system. NA was determined in samples using electrochemical HPLC detector (BAS mod. LC-250) and 5-HIAA was separated by means of a reverse-phase column (C18, BAS Septstik). The instrument was operated with the help of aqueous phosphate buffer solution (0.1 M, pH 3.2) containing 0.2 mM sodium octyl-sulphate, 0.1 mM of EDTA and 18% v/v methanol as mobile phase. The flow rate was set on 0.2 ml/min. Plotting graphs and data analyses were done by Autochro data module software. Concentrations of 5-HIAA were calculated by interpolating the areas obtained after fraction injection with those obtained from 4 known standard concentrations being injected regularly to the HPLC to monitor the linearity of the system. Integration of chromatographic data was performed by Perkin-Elmer 1020 software program (21).
Data analysis
Data were examined by SPSS® ver 18.0 for Windows®. The Student’s t test was used to compare means of two groups. One-way analysis of variance (ANOVA) was used to compare means of three or more groups, and Dunkan multiple range test was used as the post hoc test. A P-value <0.05 was considered statistically significant (22).
Results
Formalin test
After the injection of formalin in group 2 (control of tramadol), the mean±SEM of nociceptive score in the first 5 min was 2.51±0.13. In the second 5 min, it reduced to 1.97±0.17 (Figure 1). The value then increased to 2.12±0.09 in the second phase of nociception (Figure 1). In group 4, the mean±SEM of pain score in the first 5 min was 2.08±0.16, and for the second 5 min, it reduced to 1.32±0.2 (Figure 1). For the third to sixth 5 min, the mean±SEM of nociceptive score increased to 1.83±0.15 (Figure 1). Therefore, there was a significant differences (P=0.027) in the second 5 min between control and group 4 of rats (Figure 1). On the other hand, SC injection of normal saline in the right hind paw of rats in groups 1 and 3 did not show any changes in animals behaviors (score=0).
Figure 1.
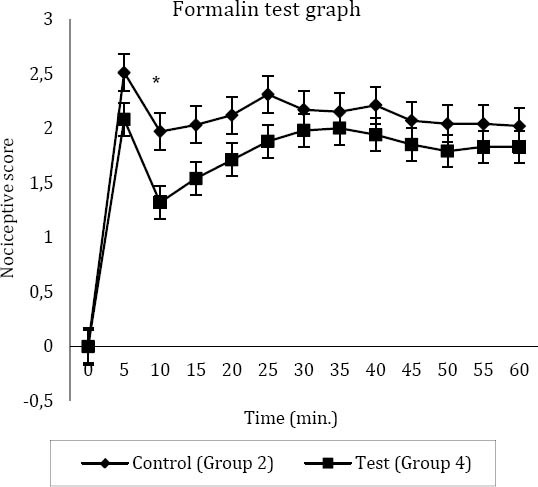
The nociceptive score in main test group (group 4) and control of tramadol (group 2)
Higher nociceptive scores show more pain and lower nociceptive scores represents less pain
* Significant difference between the test and the control (n=8) (P=0.027)
0: Animal’s injected paw is completely on the floor and can endure the animal’s weight
1: Animal’s injected paw situates partially on the floor and is partially elevated
2: Animal’s injected paw is elevated without any contact with the surface
3: Animal shakes, licks or bites the injected paw (19)
Microdialysis
Noradrenaline
In rats of group two (control of tramadol), the baseline mean±SEM for NA before formalin injection was 135.36±5.11 picograms per milliliter (pg/ml). Fifteen min after injection of formalin—phase one or acute phase of formalin test—it increased to 581.44±47.98 (pg/ml), and then it reduced to127.26±9.29 (pg/ml) (Figure 2). The increase in NA concentration in the acute phase of formalin test was significant (P<0.05) (Figure 2). In group 4, the mean±SEM for concentrations of NA before formalin injection was 122.07±5.21 (pg/ml). Fifteen min after injection of formalin, it reached to 223.02±15.92 (pg/ml), and then it decreased to 164.75±15.56 (pg/ml) (Figure 2). In group 3 (control of formalin), there was no significant difference between the samples; the mean±SEM for concentrations of NA was 126.06± 8.98 (pg/ml). In group 1 (main control or control of formalin and tramadol), the mean±SEM for baseline NA was 139.47±8.85 (pg/ml) (Figure 2).
Figure 2.
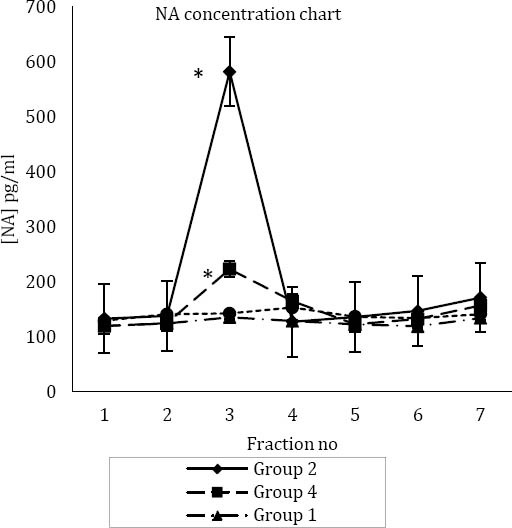
(A) Mean±SEM concentration of noradrenaline (NA) in four study groups
*Significant difference with group 1, 3 and 4. (SC) in hind paw;
* *: Significant difference with group 1, 2 and 3
Fraction numbers are according to collection of microdialysis samples every 15 min
Group one (main control or control of formalin and tramadol test): injection of normal saline (IP) and injection of normal saline (SC) in right hind paw
Group two (control of tramadol test): injection of normal saline (IP) and injection of formalin (SC) in right hind paw
Group three (control of formalin test): injection of tramadol (IP) and injection of normal saline (SC) in right hind paw
Group four (main test group test): injection of tramadol (IP) and injection of formalin
MHPG
In groups 1 and 3, no significant differences were observed between MHPG concentrations in microdialysis samples (Figure 3). The mean±SEM for MHPG concentrations in samples of group 3 was 363.41±22.42 (pg/ml), while in rats of group 1, it was 377.56±34.79 (pg/ml) (Figure 3). In group 2, the mean±SEM for baseline MHPG level before formalin injection was 413.65±33.29 (pg/ml). In the third sample (third 15 min), there was a sharp increase in the concentration of MHPG to 1465.9±137.47 (pg/ml), and then it decreased to 428.78±17.59 (pg/ml) (Figure 3).
Figure 3.
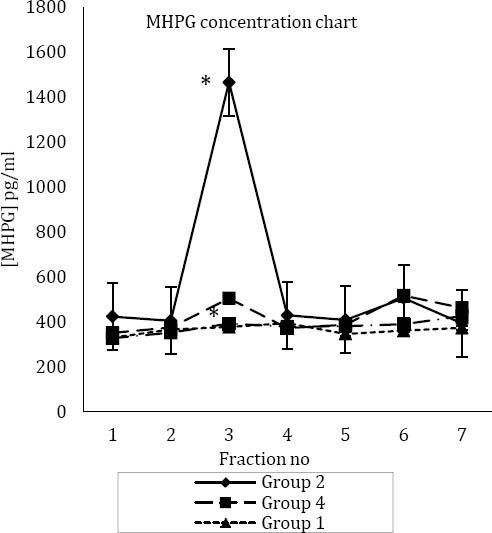
Mean±SEM concentration of 3-methoxy-4-hydroxyphenylglycol (MHPG) in four study groups
*: Significant difference with group 1, 3 and 4
* *: Significant difference with group 1, 2 and 3
Fraction numbers are according to collection of microdialysis samples every 15 min
Group one (main control or control of formalin and tramadol test): injection of normal saline (IP) and injection of normal saline (SC) in right hind paw
Group two (control of tramadol test): injection of normal saline
(IP) and injection of formalin (SC) in right hind paw
Group three (control of formalin test): injection of tramadol (IP)
and injection of normal saline (SC) in right hind paw
Group four (main test group test): injection of tramadol (IP) and
injection of formalin (SC) in hind paw
Discussion
In this study, the significant increase in the concentration of NA and MHPG in the third 15-min (third sample of microdialysis) was associated with the highest nociceptive sensation in rats, in the formalin test. In group 2, the increase was about 4 folds higher in comparison to the baseline; therefore, it had a significant difference with other groups. Although there was a significant increase in group 4, it was not as much as group 2. There was no significant difference between the samples in the absence of the pain sensation—in groups 1 and 3. The pontine noradrenergic areas including A5, A6 (LC) and A7, are believed to act as an antinociceptive inducing component (23). There are some projections from these neurons to the dorsal horn of the spinal cord and release NA to suppress passage the pain messages (24). These areas have an important role in regulating, modulating and suppressing the pain and antinociceptive effects observed after being directly stimulated. Among these nuclei, LC, the largest nucleus of noradrenergic neurons in the central nervous system, has an important role (25). Local infusion of prazosin as a typical α1-adrenoceptor antagonist into LC significantly decreased concentrations of extracellular NA in the nucleus. On the other hand, α2-adrenoceptors in LC play a major role in modulating the feedback of LC neurons to excitation (26).
A wide range of brain functions such as memory, stress adaptation emotion and vigilance is related to LC nucleus (27). Different parts of the brain send projections into LC, thereby they can affect the nucleus. On the other hand, LC has extensive noradrenergic fiber connections to forebrain, brainstem, cerebellum, hippocampus, thalamus, cerebral cortex, periaqueductal gray (PAG) matter, nucleus of the solitary tract, nucleus paragigantocellularis, nucleus prepositus hypoglossi and Barrington’s nucleus in the pons (28). From limbic areas including the central nucleus of amygdala (CeA), bed nucleus of the stria terminalis and the spinal cord, adrenergic projections that innervate the LC, are included in this response (29). After sensory stimulations, the adrenergic projections inhibit LC nucleus. Inhibition of α-adrenoceptors has increased the activity of LC to a physiological excitatory stimulus compression of the hind paw at amounts pretty well less than those necessary to increase their spontaneous activity. Therefore, it can exert modulatory effects through functionally various brain regions (30). Any increase in the discharge of LC neurons is associated with a rise in the amount of extracellular NA in the thalamus and cortex (31). For instance, the stimulation of the primary motor cortex is associated with the increase in the discharge rate of LC and the descending antinociceptive effects (30). Furthermore, after blocking LC or the descending noradrenergic pathways by lidocaine or intrathecal administration of an α2-adrenoceptor antagonist, the spinal antinociceptive effect that was induced by electric M1 stimulation (with an electrode placed in M1 at frequency of 300 Hz) in the groups of neuropathic models of rats was reduced (32, 33, 34). LC has an important role in regulation and suppression of pain so that after inactivation of LC by lidocaine injection, the descending adrenergic antinociceptive pathways were significantly blocked (31). The exact antinociceptive mechanism of tramadol, a centrally acting synthetic analgesic, is not completely known yet, (33) however, some of the suggested mechanisms are binding and stimulating the receptors, accumulation of monoamines like NA and 5-HT, especially in nerve endings and inhibition of their reuptake, and finally its action through 5-HT descending pathways (19). Dubuisson and Dennis showed that the presence or absence of PAG stimulation can affect the pain intensity (6) According to Sanchez-Blazquez, et al., there are opioid receptors, such as mu and delta receptors, in periaqueductal gray matter. Therefore, stimulation of receptors by tramadol in this area could suppress the pain signals from reaching the LC (31). The maximum pain sensation was 5 minutes after formalin injection (Figure 1) which is in line with the increase in concentration of NA and MHPG in the third sample. Formalin was injected 15 min after beginning of microdialysis and took 15 extra min for microdialysis liquid to pass through pipe tubes into the Eppendorf tubes. The elevation of NA and its metabolite, MHPG, in the third sample was associated with the formalin injection time. Therefore, it indicates that after the pain sensation and stimulation of LC, release of NA with stimulation of adrenergic and serotonergic descending pathways has an important role in suppressing the pain (35). The significant difference in pain scores between group 2 and 4, and its reduction in group 4 shows the effect of the drug. There is a decrease in NA and its metabolite concentration in LC after administration of tramadol. Formalin test has some advantages among other pain test models. In this method, pain sensation is continuous and includes acute pain (first 5 min) and chronic pain (15-20 min) after formalin injection (5). Because the main antinociception effect of tramadol through LC is observed in the first phase of the formalin test (Figure 1), it is concluded that tramadol has significant effect on acute pain. These results are in line with other literature data showing that tramadol can indirectly affect the LC by blocking the pain signals from different parts of the brain such as PAG, CAN and the spinal cord (36). By blocking the reuptake of NA and MHPG in nerve terminals, the pain signals cannot transfer completely into the CNS. On the other hand tramadol as an opioid drug, by stimulating receptors in these parts of the brain, partially blocks transmission of the pain signals to LC.
Conclusion
The results of this study demonstrated that tramadol may block the pain signals from different parts of the brain like PAG, CAN and the spinal cord by blocking NA and MHPG reuptake in nerve terminals; therefore, the pain signals cannot transfer completely into the CNS. Moreover, tramadol as an opioid drug, by stimulating receptors in various parts of the brain in relationship with LC, inhibits the pain signals from reaching LC completely. Therefore, antinociceptive effects of tramadol can appear by its indirect effect on LC.
Acknowledgment
The results described in this paper were part of student thesis which was supported by Shiraz University of Medical Sciences and Pharmaceutical Sciences Research Center, Shiraz, Iran.
References
- 1.Clavier RM. Afferent projections to the self- stimulation regions of the dorsal pons, including the locus coeruleus, in the rat as demonstrated by the horseradish peroxidase technique. Brain Res Bull. 1979;4:497–504. doi: 10.1016/0361-9230(79)90034-0. [DOI] [PubMed] [Google Scholar]
- 2.Morgane PJ, Jacobs MS. Raphe projections to the locus coeruleus in the rat. Brain Res Bull. 1979;4:519–534. doi: 10.1016/0361-9230(79)90037-6. [DOI] [PubMed] [Google Scholar]
- 3.Yevtushenko OO, Oros MM, Reynolds GP. Early response to selective serotonin reuptake inhibitors in panic disorder is associated with a functional HT1A receptor gene polymorphism. J Affect Disord. 2010;123:308–311. doi: 10.1016/j.jad.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 4.Stone EA, Lin Y, Sarfraz Y, Quartermain D. The role of the central noradrenergic system in behavioral inhibition. Brain Res Rev. 20l1;67:193–208. doi: 10.1016/j.brainresrev.2011.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sajedianfard J, Khatami S, Semnanian S, Naghdi N, Jorjani M. In vivo measurement of noradrenaline in the locus coeruleus of rats during the formalin test: A microdialysis study. Eur J Pharmacol. 2005;512:153–156. doi: 10.1016/j.ejphar.2005.02.032. [DOI] [PubMed] [Google Scholar]
- 6.Dubuisson D, Dennis SG. The formalin test: a quantitative study of the analgesic effects of morphine, meperidine, and brain stem stimulation in rats and cats. Pain. 1978;4:161–174. doi: 10.1016/0304-3959(77)90130-0. [DOI] [PubMed] [Google Scholar]
- 7.Budd K. The role of tramadol in acute pain management. Acute Pain. 1999;2:189–196. [Google Scholar]
- 8.Sindrup SrH, Andersen G, Madsen C, Smith T, Brosen K, Jensen TS. Tramadol relieves pain and allodynia in polyneuropathy: a randomised, double- blind, controlled trial. Pain. 1999;83:85–90. doi: 10.1016/s0304-3959(99)00079-2. [DOI] [PubMed] [Google Scholar]
- 9.Lehtonen Pi, Sten T, Aitio O, Kurkela M, Vuorensola K, Finel M, et al. Glucuronidation of racemic O-desmethyltramadol, the active metabolite of tramadol. Eur J Pharm Sci. 2010;41:523–530. doi: 10.1016/j.ejps.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 10.Vlase L, Leucuta SE, Imre S. Determination of tramadol and O-desmethyltramadol in human plasma by high-performance liquid chromatography with mass spectrometry detection. Talanta. 2008;75:1104–1109. doi: 10.1016/j.talanta.2008.01.006. [DOI] [PubMed] [Google Scholar]
- 11.Plock N, Kloft C. Microdialysi-theoretical background and recent implementation in applied life-sciences. Eur J Pharm Sci. 2005;25:1–24. doi: 10.1016/j.ejps.2005.01.017. [DOI] [PubMed] [Google Scholar]
- 12.Maeda M, Tsuruoka M, Hayashi B, Nagasawa I, Inoue T. Descending pathways from activated locus coeruleus/subcoeruleus following unilateral hindpaw inflammation in the rat. Brain Res Bull. 2009;78:170–174. doi: 10.1016/j.brainresbull.2008.09.005. [DOI] [PubMed] [Google Scholar]
- 13.Viard E, Sapru HN. Carotid baroreflex in the rat: role of glutamate receptors in the medial subnucleus of the solitary tract. Neuroscience. 2004;126:785–794. doi: 10.1016/j.neuroscience.2004.04.016. [DOI] [PubMed] [Google Scholar]
- 14.Kawahara H, Kawahara Y, Westerink BHC. The role of afferents to the locus coeruleus in the handling stress-induced increase in the release of noradrenaline in the medial prefrontal cortex: a dual- probe microdialysis study in the rat brain. Eur J Pharmacol. 2000;387:279–286. doi: 10.1016/s0014-2999(99)00793-1. [DOI] [PubMed] [Google Scholar]
- 15.Renno WM, Mullet MA, Williams FG, Beitz AJ. Construction of 1 mm microdialysis probe for amino acids dialysis in rats. J Neurosci Methods. 1998;79:217–228. doi: 10.1016/s0165-0270(97)00192-1. [DOI] [PubMed] [Google Scholar]
- 16.DiFrancesco R, DiCenzo R, Vicente G, Donnelly J, Martin TM, Colon LA, et al. Determination of lopinavir cerebral spinal fluid and plasma ultrafiltrate concentrations by liquid chromatography coupled to tandem mass spectrometry. J Pharm Biomed Anal. 2007;44:1139–1146. doi: 10.1016/j.jpba.2007.05.020. [DOI] [PubMed] [Google Scholar]
- 17.Alimoradian A, Sajedianfard J, Baha-aldini Beigy F, Panjehshahin MR, Owji AA. Relationship of dopamine of the nucleus accumbens with Intra- infralimbic apomorphine microinjection. Iran J Basic Med Sci. 2013;16:743–750. [PMC free article] [PubMed] [Google Scholar]
- 18.Sakiyama Y, Sujaku T, Furuta A. A novel automated method for measuring the effect of analgesics on formalin-evoked licking behavior in rats. J Neurosci Methods. 2008;167:167–175. doi: 10.1016/j.jneumeth.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 19.Lee B-C, Eom K-H, Soh K-S. Primo-vessels and primo-nodes in rat brain, spine and sciatic nerve. J Acupunct Meridian Stud. 2010;3:111–115. doi: 10.1016/S2005-2901(10)60020-9. [DOI] [PubMed] [Google Scholar]
- 20.Morales-Villagran A, Lopez-Perez S, Medina-Ceja L, Tapia R. Cortical catecholamine changes and seizures induced by 4-aminopyridine in awake rats, studied with a dual microdialysis-electrical recording technique. Neurosci Lett. 1999;275:133–136. doi: 10.1016/s0304-3940(99)00759-4. [DOI] [PubMed] [Google Scholar]
- 21.Sauernheimer C, Williams KM, Brune K, Geisslinger G. Application of microdialysis to the pharmacokinetics of analgesics: Problems with reduction of dialysis efficiency in vivo. J Pharmacol Toxicol Methods. 1994;32:149–154. doi: 10.1016/1056-8719(94)90068-x. [DOI] [PubMed] [Google Scholar]
- 22.Cho HG, Lee HS, Bae M, Joo K. Chronic arthritis increases tyrosine hydroxylase mRNA levels in the pontine noradrenergic cell groups. Brain Res. 1995;695:96–99. doi: 10.1016/0006-8993(95)00937-l. [DOI] [PubMed] [Google Scholar]
- 23.Gutierrez-Guzman BE, Hernandez-Perez JJ, Lopez-Vazquez MA, Fregozo CS, Guevara MA, Olvera-Cortes ME. Serotonin depletion of supramammillary/posterior hypothalamus nuclei produces place learning deficiencies and alters the concomitant hippocampal theta activity in rats. Eur J Pharmacol. 2012;682:99–109. doi: 10.1016/j.ejphar.2012.02.024. [DOI] [PubMed] [Google Scholar]
- 24.Milner D, Lara JP, Lopez de Miguel MP, Lopez-Gonzalez MV, Spyer KM, Gonzalez-Baron S. A5 region modulation of the cardiorespiratory responses evoked from parabrachial cell bodies in the anaesthetised rat. Brain Res. 2003;982:108–118. doi: 10.1016/s0006-8993(03)03005-1. [DOI] [PubMed] [Google Scholar]
- 25.Pudovkina OL, Westerink BHC. Functional role of alpha1-adrenoceptors in the locus coeruleus: a microdialysis study. Brain Res. 2005;1061:50–56. doi: 10.1016/j.brainres.2005.08.049. [DOI] [PubMed] [Google Scholar]
- 26.Nitz D, Siegel JM. GABA release in the locus coeruleus as a function of sleep/wake state. Neuroscience. 1997;78:795–801. doi: 10.1016/s0306-4522(96)00549-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jodoj E, Chiang C, Aston-Jones G. Potent excitatory influence of prefrontal cortex activity on noradrenergic locus coeruleus neurons. Neuroscience. 1998;83:63–79. doi: 10.1016/s0306-4522(97)00372-2. [DOI] [PubMed] [Google Scholar]
- 28.Zhong XL, Lv KR, Zhang Q, Yu JT, Xing YY, Wang ND, et al. Low-frequency stimulation of bilateral anterior nucleus of thalamus inhibits amygdale- kindled seizures in rats. Brain Res Bull. 2011;86:422427. doi: 10.1016/j.brainresbull.2011.08.014. [DOI] [PubMed] [Google Scholar]
- 29.Liu N, Ho IK, Rockhold RW. Contribution of glutamatergic systems in locus coeruleus to nucleus paragigantocellularis stimulation-evoked behavior. Pharmacol Biochem Behav. 1999;63:555–567. doi: 10.1016/s0091-3057(99)00005-2. [DOI] [PubMed] [Google Scholar]
- 30.Safari MS, Haghparast A, Semnanian S. Effect of lidocaine administration at the nucleus locus coeruleus level on lateral hypothalamus-induced antinociception in the rat. Pharmacol Biochem Behav. 2009;92:629–634. doi: 10.1016/j.pbb.2009.03.002. [DOI] [PubMed] [Google Scholar]
- 31.Sanchez-Blazquez P, Gomez-Serranillos P, Garzon J. Agonists determine the pattern of G-protein activation in mu-opioid receptor-mediated supraspinal analgesia. Brain Res Bull. 2001;54:229–235. doi: 10.1016/s0361-9230(00)00448-2. [DOI] [PubMed] [Google Scholar]
- 32.Simson PE, Weiss JM. Blockade of a2-adrenergic receptors, but not blockade of y-aminobutyric acid, serotonin, or opiate receptors, augments responsiveness of locus coeruleus neurons to excitatory stimulation. Neuropharmacology. 1989;28:651–660. doi: 10.1016/0028-3908(89)90147-0. [DOI] [PubMed] [Google Scholar]
- 33.Sakiyama Y, Sujaku T, Furuta A. A novel automated method for measuring the effect of analgesics on formalin-evoked licking behavior in rats. J Neurosci Methods. 2008;167:167–175. doi: 10.1016/j.jneumeth.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 34.Jodoj E, Chiang C, Aston-Jones G. Potent excitatory influence of prefrontal cortex activity on noradrenergic locus coeruleus neurons. Neuroscience. 1998;83:63–79. doi: 10.1016/s0306-4522(97)00372-2. [DOI] [PubMed] [Google Scholar]
- 35.Movassaghi S, Sharifi ZN, Soleimani M, Joghataii MT, Hashemi M, Shafaroodi H, et al. Effect of pentoxifylline on ischemia- induced brain damage and spatial memory impairment in rat. Iran J Basic Med Sci. 2012;15:1083–1090. [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang CE, Yang S-W, Guo Y-G, Qiao J-T, Dafny N. Locus coeruleus stimulation modulates the nociceptive response in parafascicular neurons: an analysis of descending and ascending pathways. Brain Res Bull. 1997;42:273–278. doi: 10.1016/s0361-9230(96)00262-6. [DOI] [PubMed] [Google Scholar]


