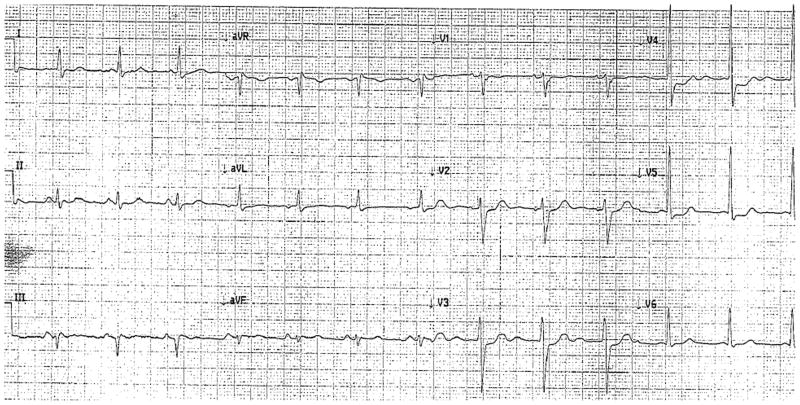
Five days after the onset of substernal chest pain, a 60-year-old man with a history of hypertension, smoking (60 pack-year history), severe bullous emphysema, and epilepsy presented with acutely worsening chest pain. Pharmacologic management for non-ST elevation myocardial infarction was initiated based on 12-lead ECG findings of subtle anterolateral ST segment changes (V2-5) (Figure 1) and a troponin I level of 6.36 ng/mL (normal <0.04 ng/mL).
Figure 1.
The electrocardiogram showed subtle anterolateral ST segment changes in the anterolateral leads (V2–V5).
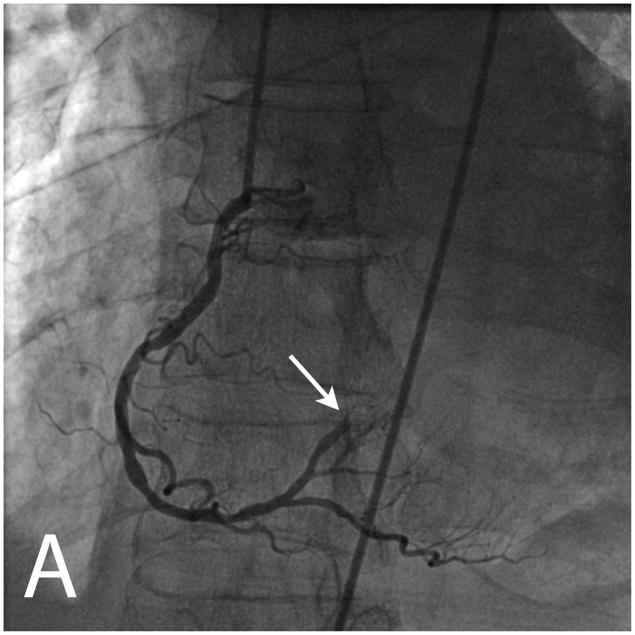
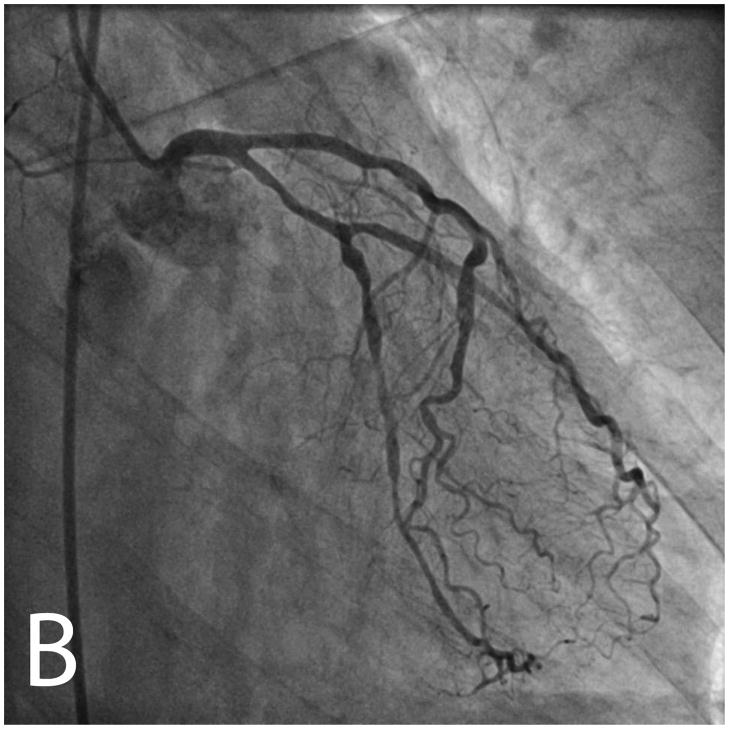
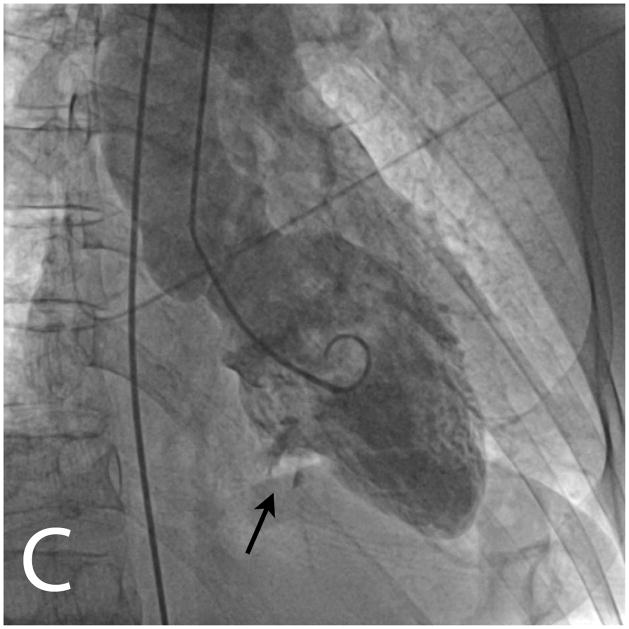
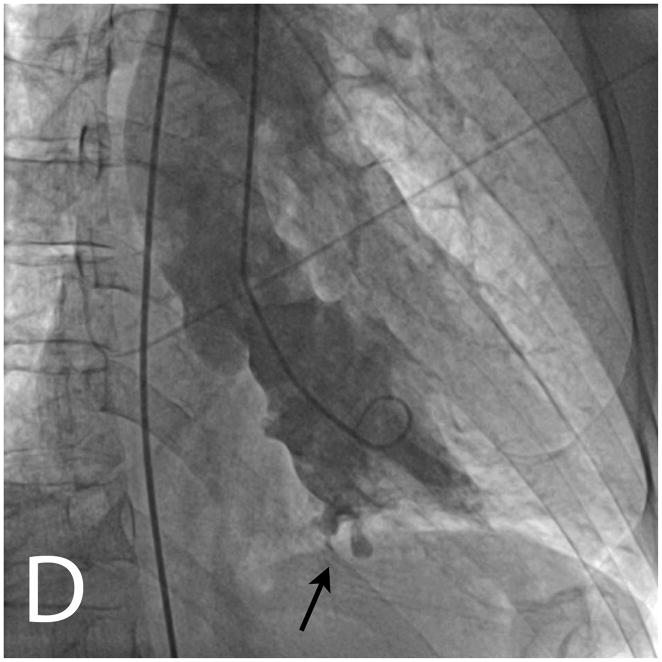
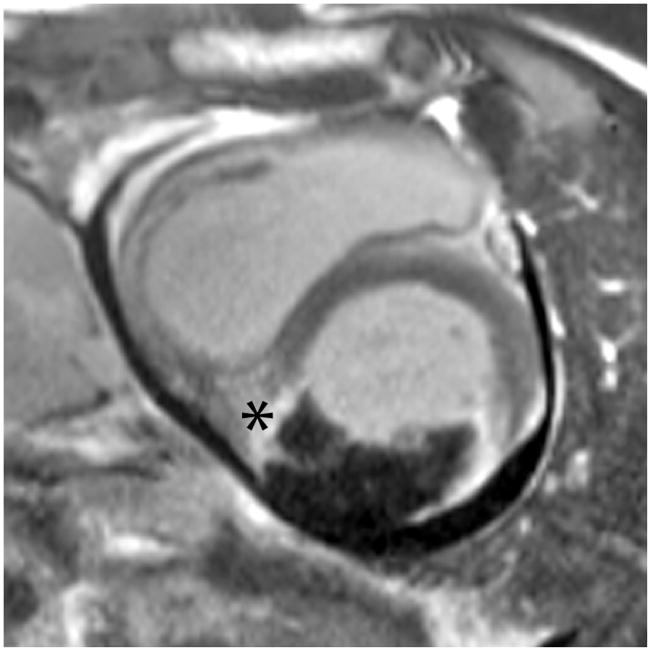
Invasive angiography demonstrated a distal occlusion of a right-sided posterolateral branch and a non-occlusive stenosis in the distal circumflex artery (Figure 2, A and B). Two small craters of contrast protruded outside the contrast-filled left ventricular contour on cineangiography within a dyskinetic basal inferior wall (Figure 2, C and D; Supplemental Material, Movie 1). Because the occlusion was distal, percutaneous coronary intervention was not performed. Transthoracic echocardiography showed heterogeneous echogenicity within a 28 mm-thick, dyskinetic inferior left ventricular wall (Supplemental Material, Movie 2) and a mass protruding into the left atrium (Figure 3). This unusual mass was suspicious for an intramyocardial hematoma based on the clinical setting and its location within myocardium subtended by the occluded artery. Left ventricular ejection fraction was visually estimated to be 45%. There were wall motion abnormalities in the basal and mid anterolateral, inferolateral and inferior walls extending into the apical inferior wall. In retrospect, the anterolateral ST changes likely represented a combination of inferior and inferolateral infarct and ischemia. All antiplatelet/antithrombotic agents except for aspirin were discontinued and the patient was urgently transferred for surgical evaluation and cardiovascular MRI (CMR).
Figure 2.
Invasive angiography showed an abrupt occlusion of a right sided posterolateral branch (white arrow, panel A) and diffuse non-occlusive stenoses in all three epicardial coronary arteries (panels A and B). Ventriculography in an RAO projection showed a dyskinetic basal inferior wall with two craters of contrast extending outside the contour of the contrast-filled left ventricular cavity (black arrows, panels C and D).
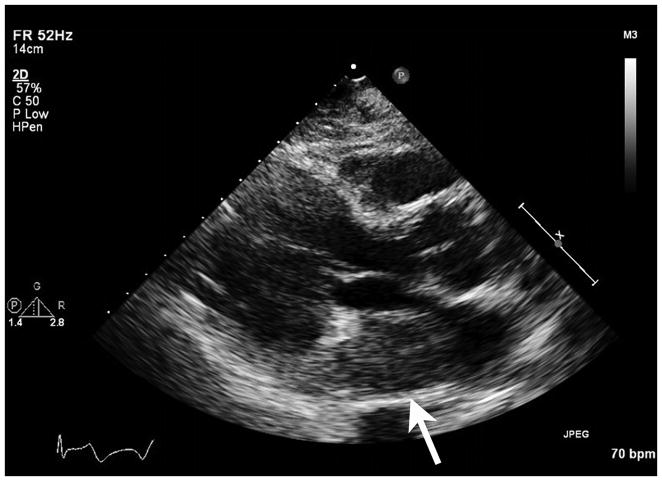
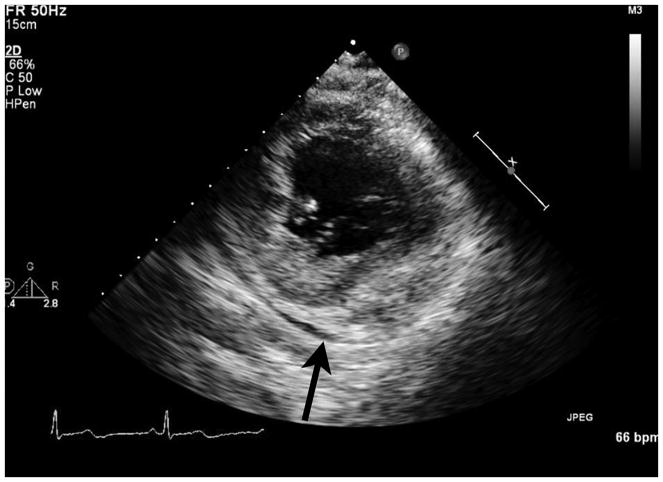
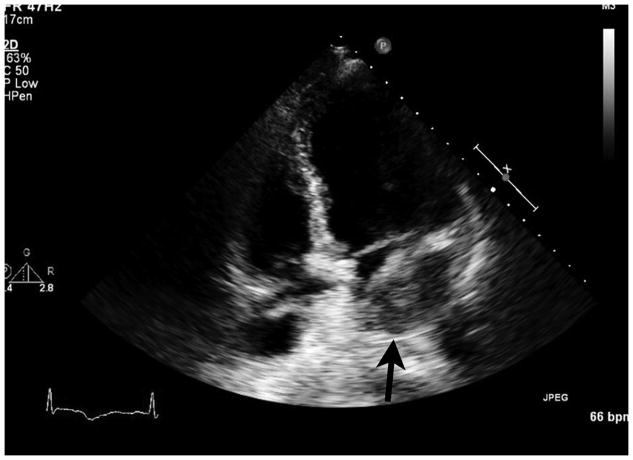
Figure 3.
Transthoracic echocardiography showed a mass protruding into the left atrium on the parasternal long axis view (arrow, left panel). On the parasternal short axis view, the basal inferior wall was 28 mm thick, heterogeneously echogenic and dyskinetic (arrow, middle panel). An off-axis apical four chamber view showed the mass in the posterior left atrium (arrow, right panel).
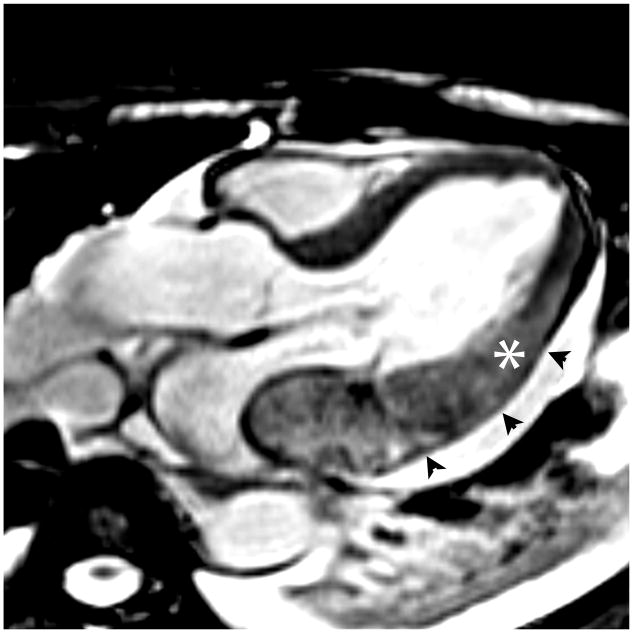
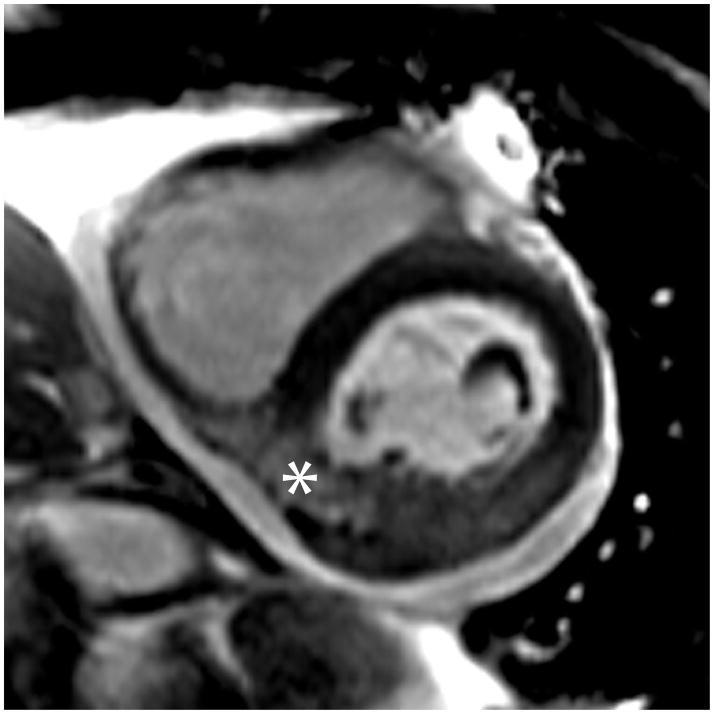
By CMR, the regional wall motion abnormalities and visually estimated ejection fraction on real time cine images were similar to the previous echo. Perfusion imaging during administration of gadolinium contrast agent (gadopentetate dimeglumine 0.15 mmol/kg) and early gadolinium enhancement images (3 minutes following contrast administration) demonstrated a hypoperfused intramyocardial hematoma in the basal and mid inferoseptum, inferior and inferolateral walls of the left ventricle and also in the posterior and lateral left atrium (Figure 4; Supplemental Material, Movie 3). Combining information from short axis late gadolinium enhancement (LGE) and bright-blood T2-weighted images demonstrated that the intramyocardial hematoma was constrained by visceral pericardium in the inferior wall (Figure 5). Thus, this portion of the intramyocardial hematoma met criteria for a left ventricular pseudoaneurysm. The intramyocardial hematoma dissected from the basal inferior/inferolateral wall through the plane of the mitral valve. Posteriorly, the wall of the left atrium was identified on long axis bright-blood T2-weighted images, constraining the intramyocardial hematoma from rupture into the left atrium (Figure 5). The left atrial wall was distorted, partially compressing the left atrial cavity (Supplemental Material, Movie 4). The portions of the intramyocardial hematoma constrained by the left atrial wall and within the left ventricular myocardium had similar characteristics on first pass perfusion and early gadolinium enhancement images (Figure4; Supplemental Material, Movie 3). Pericardial enhancement and a small pericardial effusion were seen. The intramyocardial hematoma was surrounded by transmural myocardial enhancement (infarction) on late gadolinium enhancement images. Pre-contrast bright-blood T2-weighted images demonstrated increased myocardial signal intensity in the infarcted region, characteristic of edema associated with acute myocardial injury.
Figure 4.
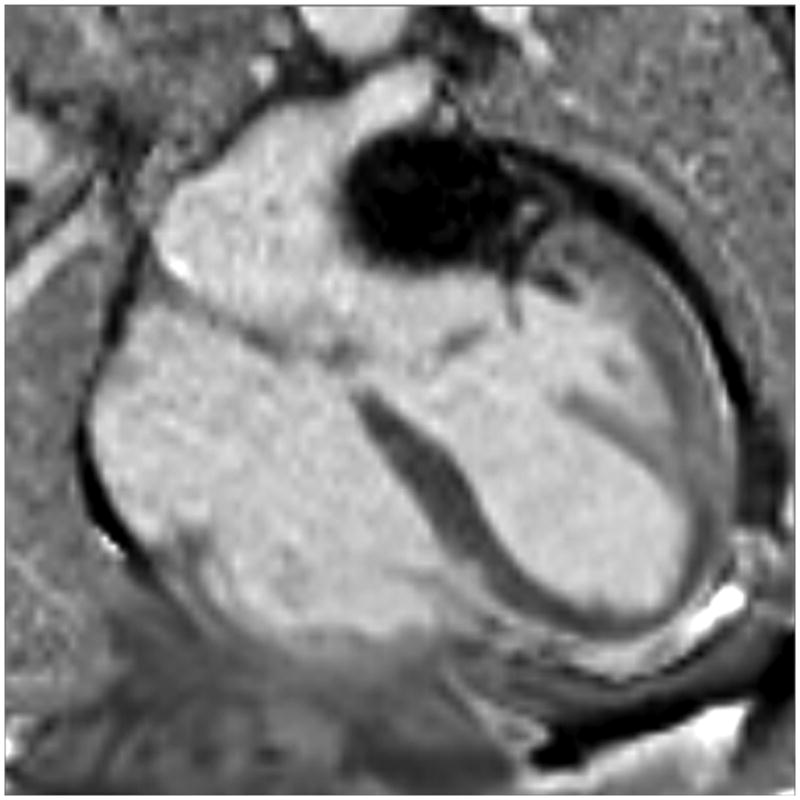
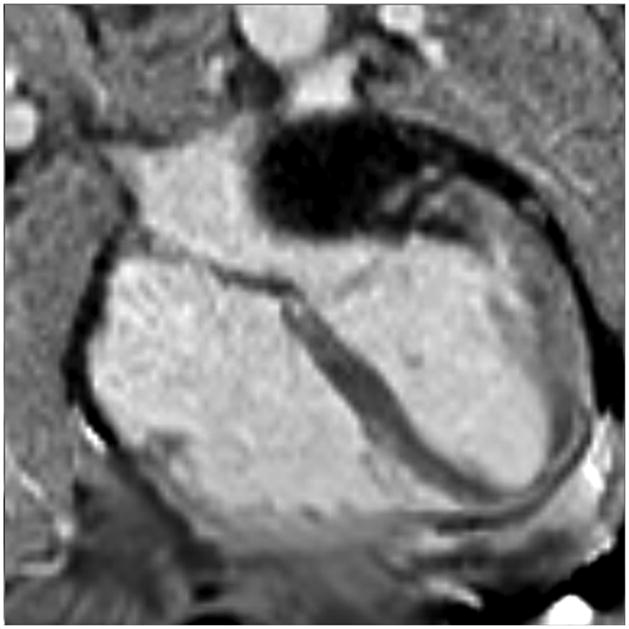
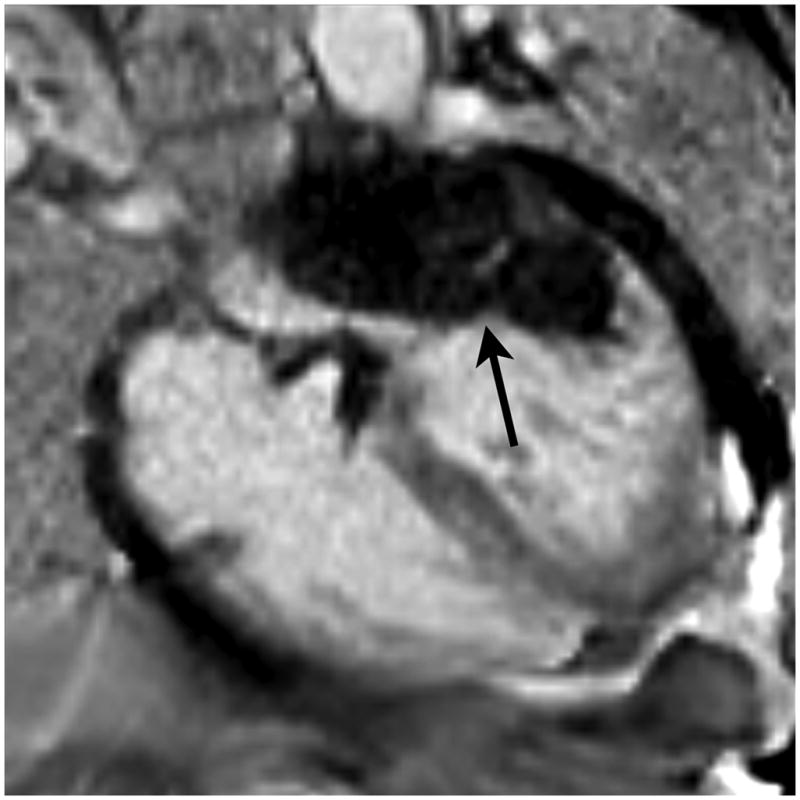
The intramyocardial hematoma extended from the inferolateral wall through the mitral annulus into the left atrial wall. Single-shot, inversion recovery, steady-state free precession images were obtained in a parallel 4-chamber orientation three minutes after administration of gadolinium contrast. The image to the left was at a level slightly below the aortic root; subsequent images moved toward the diaphragm. They demonstrated an intramyocardial hematoma extending from the basal and mid inferolateral walls into the posterolateral left atrial wall beneath the mitral valve annulus (arrow).
Figure 5.
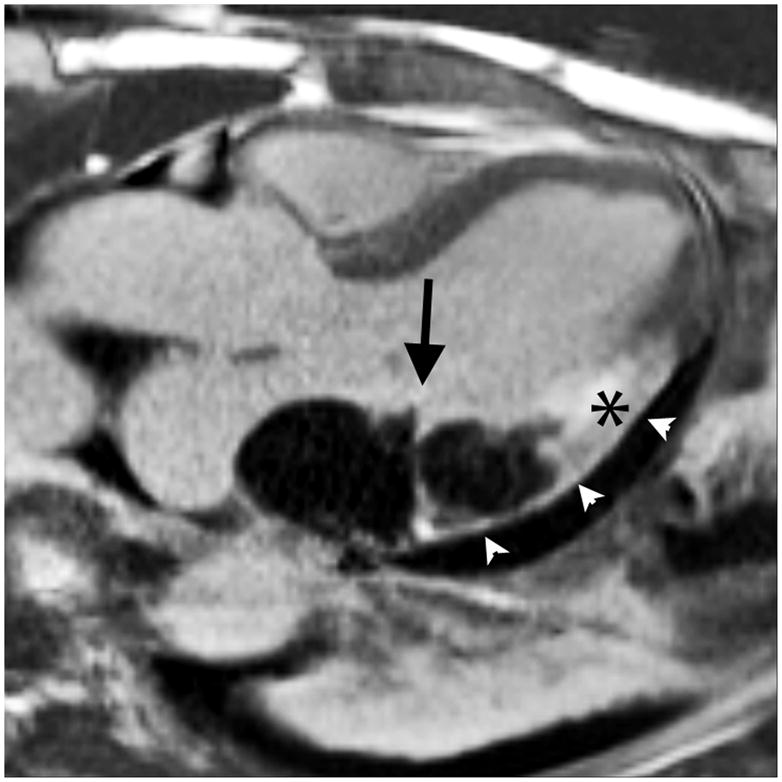
The intramyocardial hematoma was constrained inferiorly by visceral pericardium (pseudoaneurysm) and posteriorly by the left atrial wall. Bright-blood T2-weighted images and late gadolinium enhancement (LGE) images were obtained in the 3 chamber and short axis orientations, corresponding to the parasternal views in Figure 2. The left atrial wall was seen as a dark line separating the hematoma from the left atrial blood pool on the 3 chamber T2-weighted image (arrowheads). The short axis images demonstrated that the intramyocardial hematoma had not ruptured into the pericardial space but appeared to be directly adjacent to it, contained by the visceral pericardium. The 3 chamber LGE image showed the intramyocardial hematoma on both sides of the atrioventricular groove (arrow). Myocardium surrounding the intramyocardial hematoma was transmurally infarcted on LGE images, with edema demonstrated on T2-weighted images (asterices).
Surgical consultation was obtained but conservative management was recommended due to the extensive nature of the defect, the likely friable tissue, and the lack of reliable borders for surgical repair. Further imaging with contrast CT and serial echocardiograms documented a slight increase in the size of the pericardial effusion over the subsequent two days. The patient died suddenly in the hospital six days after his initial presentation, presumably due to myocardial rupture.
Both dissecting intramyocardial hematoma and left atrial intramural hematoma are rare, but the combination of the two has only been reported four times to our knowledge 1–4. All previously reported cases were also in the setting of acute inferior myocardial infarction. Three of the four previously described patients died within days of symptom onset, while one survived past one year. The survivor was one of two patients managed surgically4. Previous descriptions of this entity as a “pseudoaneurysm with subepicardial dissection4,” an “incomplete left atrial rupture,2” and a “complex cardiac rupture,3” have implied a pathophysiology between intramyocardial hemorrhage and rupture. Mechanistically, intramyocardial blood under left ventricular pressure presumably dissects within tissue planes of the left ventricle, then beneath the coronary sinus and within the wall of the left atrium 2, 4.
Dissecting intramyocardial hematoma associated with an intramural left atrial hematoma is a rare mechanical complication of myocardial infarction. The prognosis of patients presenting with dissecting intramyocardial hematoma with extension into the left atrial wall is poor, both with surgical and conservative management. In this case, echocardiography and cardiovascular MRI aided in understanding how the impending rupture was constrained and guided surgical risk assessment.
Supplementary Material
Invasive left ventricular cineangiography in an RAO projection showed a dyskinetic basal inferior wall with two craters of contrast extending outside the contour of the contrast-filled left ventricular cavity. Best viewed with Windows Media Player.
Transthoracic echocardiography in parasternal short axis showed a dyskinetic, 28 mm-thick inferior left ventricular wall with heterogeneous echogenicity. Best viewed with Windows Media Player.
The intramyocardial hematoma was hypoperfused during first-pass gadolinium perfusion imaging. It extended from the inferolateral wall through the mitral annulus into the posterior and lateral left atrium in this parallel 4 chamber orientation. Best viewed with Windows Media Player.
The intramyocardial hematoma was seen in the posterior and lateral left atrial wall, partially compressing the left atrial cavity, on this segmented breath-held 4 chamber cine. The basal anterolateral wall was akinetic. Best viewed with Windows Media Player.
References
- 1.Kovacic JC, Horton MD, Campbell TJ, Wilson SH. Left atrial hematoma complicating inferior myocardial infarction. J Am Soc Echocardiogr. 2004;17:1201–1203. doi: 10.1016/j.echo.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 2.Chida K, Ohkawa S, Nagashima K, Imai T, Kuboki K, Maeda S, Watanabe C. An autopsy case of incomplete left atrial rupture following left atrial infarction associated with left ventricular myocardial infarction. Jpn Circ J. 1995;59:299–302. doi: 10.1253/jcj.59.299. [DOI] [PubMed] [Google Scholar]
- 3.Gallego P, Oliver JM, Gonzalez A, Dominguez FJ, Sanchez-Recalde A, Mesa JM. Left atrial dissection: Pathogenesis, clinical course, and transesophageal echocardiographic recognition. J Am Soc Echocardiogr. 2001;14:813–820. doi: 10.1067/mje.2001.113366. [DOI] [PubMed] [Google Scholar]
- 4.Nathaniel C, Lane S, Palma R, Scheinerman SJ, Missri JC. Pseudoaneurysm causing partial obliteration of the left atrium: Case report and review. Cathet Cardiovasc Diagn. 1996;38:83–86. doi: 10.1002/(SICI)1097-0304(199605)38:1<83::AID-CCD19>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Invasive left ventricular cineangiography in an RAO projection showed a dyskinetic basal inferior wall with two craters of contrast extending outside the contour of the contrast-filled left ventricular cavity. Best viewed with Windows Media Player.
Transthoracic echocardiography in parasternal short axis showed a dyskinetic, 28 mm-thick inferior left ventricular wall with heterogeneous echogenicity. Best viewed with Windows Media Player.
The intramyocardial hematoma was hypoperfused during first-pass gadolinium perfusion imaging. It extended from the inferolateral wall through the mitral annulus into the posterior and lateral left atrium in this parallel 4 chamber orientation. Best viewed with Windows Media Player.
The intramyocardial hematoma was seen in the posterior and lateral left atrial wall, partially compressing the left atrial cavity, on this segmented breath-held 4 chamber cine. The basal anterolateral wall was akinetic. Best viewed with Windows Media Player.