Abstract
The infralimbic prefrontal cortex (IL) has been shown to be critical for the regulation of flexible behavior, but its precise function remains unclear. This region has been shown to be critical for the acquisition, consolidation, and expression of extinction learning, leading many to hypothesize that IL suppresses behavior as part of a “stop” network. However, this framework is at odds with IL function in habitual behavior in which the IL has been shown to be required for the expression and acquisition of ongoing habitual behavior. Here, we will review the current state of knowledge of IL anatomy and function in behavioral flexibility and provide a testable framework for a single IL mechanism underlying its function in both extinction and habit learning.
Addiction is characterized both by the inability to terminate or reduce drug taking even in the face of adverse consequences and by chronic relapse to drug taking. Investigation into the neurobiological substrates of the loss of behavioral flexibility in addiction has relied heavily on animal models of addictive behavior, including habit and extinction learning paradigms that investigate distinct components of inflexible behavior.
The prefrontal cortex (PFC) has repeatedly been identified as critical for the cognitive control of flexible actions (Jentsch and Taylor 1999; Everitt et al. 2008; George and Koob 2010; Willcocks and McNally 2012). Subregions of the PFC have distinct connectivity that likely underlies their separable roles in behavior. The infralimbic PFC (IL) has been shown to have a unique role in the development and expression of inflexible reward seeking. Interestingly, IL has been shown to have opposing effects on behavior when compared with the adjacent prelimbic PFC (PL) or, more broadly, the dorsomedial PFC (dmPFC). Perhaps most surprising are the apparently contradictory roles of IL in different measures of behavioral flexibility. In the current review, we discuss models of inflexible drug seeking and the role of IL in the regulation of these behavioral processes. In particular, we focus on the precise function of IL in the acquisition and extinction of drug seeking and the development and expression of habitual drug-seeking behavior. We propose a framework for reconciling the seemingly contradictory role of IL in the regulation of flexible reward seeking through the suppression of the ability of established action–outcome contingencies to guide behavior.
Inflexible behaviors: resistance to extinction and habitual reward seeking
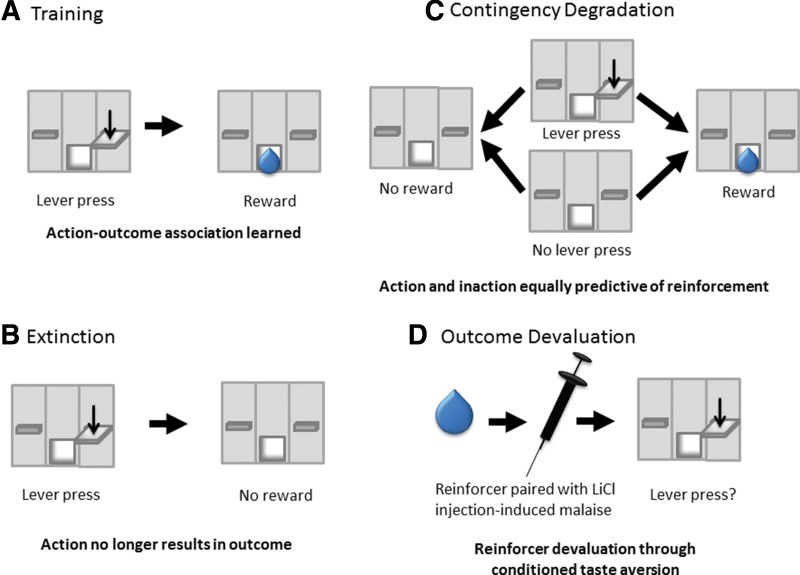
In models of inflexible behavior, animals learn to associate actions, contexts, or stimuli with outcomes, including reward delivery (Fig. 1) or footshock. In extinction, animals must learn that this contingency has been broken: the presentation of outcome-paired stimuli or performing reward-seeking behavior no longer results in outcome presentation. Extinction is a new learning process that does not appear to involve the “forgetting” or “erasure” of the memory of the previously learned contingency (Rescorla 2001; Bouton 2004). Rather, reward-seeking or freezing behavior is inhibited in response to the newly formed memory that the action– or stimulus–outcome association is no longer intact. A number of behavioral models have been suggested to describe the learning and memory processes through which extinction is acquired and expressed, although many believe that extinction occurs as expectations are updated across repeated experience of the loss of action– or stimuli–outcome relationship (Rescorla 2001; Bouton 2004; Delamater and Westbrook 2013). While the fear and reward-seeking literatures diverge in a number of areas, a considerable literature implicates overlapping circuits in the acquisition and expression of extinction in these separate paradigms (Peters et al. 2009).
Figure 1.
Instrumental extinction and habit models. (A) For extinction and habit models, animals first learn an association between their action (e.g., lever press) and an outcome (e.g., reinforcer delivery). (B) In extinction training, the action–outcome relationship is terminated. Lever pressing no longer results in reinforcer delivery. Acquisition of extinction requires learning that the previously reinforced action no longer produces the delivery of a reinforcer, and the expression of extinction learning requires suppression of the previously reinforced action. (C) Habitual behavior is behavior that is no longer mediated by the relationship between actions and their outcome. One method of assessing habits is to disrupt the contingency between action and outcome. In contingency degradation, action and inaction are equivalently likely to be associated with the delivery of the reinforcer. Goal-directed animals are expected to reduce responding for the previously learned contingency when it is degraded through noncontingent reinforcement while habitual animals are insensitive to this change in contingency. (D) Habitual behavior can also be assessed through altering the value of the outcome. Typically, the outcome is devalued through association of the reinforcer with illness (i.e., a conditioned taste aversion). As with contingency degradation, when behavior is sensitive to the action–outcome contingency (i.e., goal-directed), a reduction in behavior is expected. Habitual animals, whose behavior is stimulus-driven, are insensitive to the effects of outcome devaluation on instrumental responding.
After repeated performance, behaviors can become habitual and even compulsive (Adams 1982; Dickinson 1985; Balleine and Dickinson 1998; Dickinson et al. 2002; Miles et al. 2003). Early on, behavior is mediated by its relationship to its outcome (action–outcome contingency) in which drug seeking is performed explicitly because of the drug's reinforcing properties. Over time, these actions transition to stimulus–response habits. The action–outcome relationship no longer drives behavior, but rather, exposure to environmental stimuli (i.e., contexts or cues) that have been repeatedly paired with the drug initiates drug seeking. Because habitual behavior is not dependent on the action–outcome association, animal models use contingency degradation and/or outcome devaluation methods to assess whether reward seeking is habitual or goal-directed. In these tests, animals that are performing goal-directed actions in relation to their consequences discontinue responding when the action–outcome contingency is disrupted, or when the value of the outcome is lost. By definition, however, habitual behaviors are those where responding continues despite the fact that the action no longer produces the desired outcome.
Connectivity of the rodent infralimbic prefrontal cortex
The medial PFC (mPFC) of the rodent consists of at least three distinct subregions—the anterior cingulate (ACC), prelimbic (PL), and infralimbic areas (Van Eden and Uylings 1985). Connectivity studies suggest that IL, the most ventral portions of PL, and medial orbitofrontal cortex that constitute the ventral mPFC (vmPFC), share overlapping targets. In contrast, the more dorsal portions of PL and the ACC, together with the dmPFC, are more similar in their subcortical connectivity (Berendse et al. 1992). The distinction between these subregions in the mPFC has been supported by functional, cytoarchitectonic, anatomical, and neurochemical data, although the overlap between these definitions is not perfect (Heidbreder and Groenewegen 2003).
In general, dmPFC subregions have extensive connectivity with sensory and motor cortices, whereas the vmPFC primarily interconnects with limbic cortices. IL extensively innervates amygdalar nuclei, including dense inputs to the central and basolateral nuclei and GABAergic cells that compose the intercalated nucleus of the amygdala (Cassell and Wright 1986; Mcdonald et al. 1996). The basal nuclei of the amygdala send projections to the ventral portions of the mPFC, including the IL. Of increasing interest to addiction research are the connections between the mPFC and the hypothalamus. While dmPFC projections are primarily concentrated within the posterior hypothalamus, IL projections are widely distributed throughout the hypothalamus, most notably including the dorsomedial and lateral hypothalamus (Hurley et al. 1991). The mPFC has extensive projections to the striatum with a dense projection from IL extensively innervating the medial shell of the nucleus accumbens (NAcS), and some innervation of the medial core. Notably, the more superficial layers of IL project primarily onto matrix compartments of striatal targets, while deep layers of IL have stronger inputs to the patch compartment (Gerfen 1984; Berendse et al. 1992). It appears that this is generally true across the mPFC, although the extent of deep layer innervation of the patch compartment is greater in the IL than more dorsal mPFC subregions. Others have suggested that this results in a robust impact of vmPFC on dopaminergic signaling due to the interactions between patch compartment neurons and midbrain reward structures (Gerfen 1984; Berendse et al. 1992; Heidbreder and Groenewegen 2003).
In addition to indirect connections with the dopamine system, the PFC projects to the ventral tegmental area (VTA) and the substantia nigra pars compacta, and in turn the mPFC is directly targeted by dopaminergic neurons primarily from the VTA (Thierry et al. 1973). The more ventral portions of the mPFC, including the IL, are more extensively connected with these midbrain dopamine areas than dorsal subregions. The vmPFC has been shown to have elevated basal dopamine levels as compared to dmPFC (Hedou et al. 1999a,b, 2001), potentially consistent with elevated dopaminergic innervation. The overlapping projection targets of IL and components of the limbic corticostriatal circuitry enables both integration and parallel processing of reward and cognitive information (Haber 2008; Haber and Knutson 2010), potentially facilitating the transition from a flexible regulation of behavior by the prefrontal cortex to automatic behavior driven by dorsal striatal structures (Everitt and Robbins 2013).
Infralimbic PFC controls extinction of instrumental and Pavlovian associations
While multiple subregions of the mPFC have distinct roles in extinction, the IL, in particular, has been shown to be critical for the acquisition, consolidation, and expression of extinction (Peters et al. 2009; Millan et al. 2011). This evidence comes from studies of both fear and drug conditioning where a significant body of literature shows an overlap in the neural circuits that mediate behavior, but with notable distinctions, particularly in the involvement of subcortical structures (cf, Peters et al. 2009). This suggests that much of the circuitry-mediating extinction is general across types of aversive and appetitive learning, although instances where discrepancies exist will be discussed below.
Fear extinction training has been shown to induce increased excitability of IL neurons (Santini et al. 2008) and activity (Milad and Quirk 2002; Santini et al. 2004; Knapska and Maren 2009). Consistent with this, fear extinction training up-regulates the MAPK signaling pathway, which has been shown to be required for long-term extinction memory (Hugues et al. 2004, 2006). Loss of IL (but not PL) function through pharmacological inactivation or lesion blocks the consolidation of extinction learning (Morgan et al. 1993; Quirk et al. 2000; Lebrón et al. 2004; Sierra-Mercado et al. 2006; Laurent and Westbrook 2009). Consolidation of fear extinction is IL protein synthesis dependent, suggesting that ongoing IL plasticity may be necessary for the consolidation of the memory (Santini et al. 2004). Consistent with a role for IL plasticity in fear extinction, infusions of the proplasticity neurotrophin BDNF into the IL mimic previous extinction training (Peters et al. 2010). Interestingly, the infusion of BDNF does not appear to simply promote the acquisition of extinction, but rather disrupts the expression of the Pavlovian fear memory even without extinction training. This suggests that disrupting IL stability can prevent the expression of previously established behavior.
Investigation into the extinction of drug-seeking behavior has also implicated IL as a critical cortical region (Gass and Chandler 2013). As with fear extinction, general IL activity patterns have been shown to be high during critical learning periods, including early in extinction (Francois et al. 2014). After extinction of cocaine seeking, inactivation of the IL produces spontaneous reinstatement of cocaine-seeking responses (Peters et al. 2008). Interestingly, these authors also observed that activation of IL with a glutamate receptor agonist prevented drug-primed reinstatement. It has also been shown that modulation of glutamate signaling through infusions of D-cycloserine in IL can promote sucrose extinction (Peters and De Vries 2013), although IL D-cycloserine administration does not impact contextual control over Pavlovian extinction (Torregrossa et al. 2010). Similar to findings in fear learning, IL activity appears to be required for the consolidation of extinction: inactivation of IL immediately after extinction training sessions resulted in decreased expression of extinction (LaLumiere et al. 2010), but extinction could be augmented by promoting IL AMPA activity. Consistent with a role for IL glutamate signaling in extinction, the extinction of cocaine seeking but not withdrawal after noncontingent drug delivery resulted in increased expression of excitatory amino acid transporters (Miguéns et al. 2008).
Neuromodulator signaling in IL also plays a critical role in extinction learning. Adrenergic signaling in IL bidirectionally regulates the consolidation of extinction for cocaine (LaLumiere et al. 2010). Infusions of a β2-adrenergic agonist promoted consolidation of extinction, while antagonism reduced consolidation. A role for dopamine signaling in extinction has also been reported. Antagonism of IL dopamine D2 receptors prevents consolidation, but not learning, of fear extinction, suggesting that activity at D2 receptors is required for normal consolidation of this memory (Mueller et al. 2010).
While the bulk of the investigation of IL in drug seeking has focused on extinction of cocaine-seeking behavior, recent studies have suggested that IL is also involved in the extinction of alcohol seeking. As with fear, extinction of alcohol seeking is associated with elevated c-Fos expression in IL (Marchant et al. 2010). IL plasticity may also play a role in the extinction of ethanol seeking, as expression of the proplasticity molecule polysialylated neural cell adhesion molecule (PSA-NCAM) is predictive of innate differences in the ability to extinguish ethanol-seeking behavior (Barker et al. 2012). Further, loss of PSA-NCAM resulted directly in the inability to extinguish over multiple training sessions. Interestingly, D2 agonism has been shown to induce PSA-NCAM expression (Castillo-Gómez et al. 2008), providing a potential mechanism by which IL dopamine D2 activity may drive the plasticity underlying consolidation of extinction learning.
In contrast to fear and cocaine extinction, IL inactivation does not appear to reinstate alcohol seeking (Willcocks and McNally 2012), highlighting the need to consider the neurocircuitry of addictive behavior separately for different drugs of abuse. The role of IL in extinction of heroin appears to stand in contrast to other drugs of abuse, where it may drive reinstatement of heroin seeking (Bossert et al. 2012; Peters et al. 2013). In addition, these data highlight the importance of considering a loss of plasticity separately from a loss of function. In the absence of PSA-NCAM-mediated plasticity, the IL may be maintaining stable activity patterns that were established under reinforced conditions. The loss of plasticity may drive ongoing behavior under extinction conditions because of a failure to update the action–outcome expectancy. However, under lesion or inactivation conditions, the IL provides no input to cortical and subcortical targets.
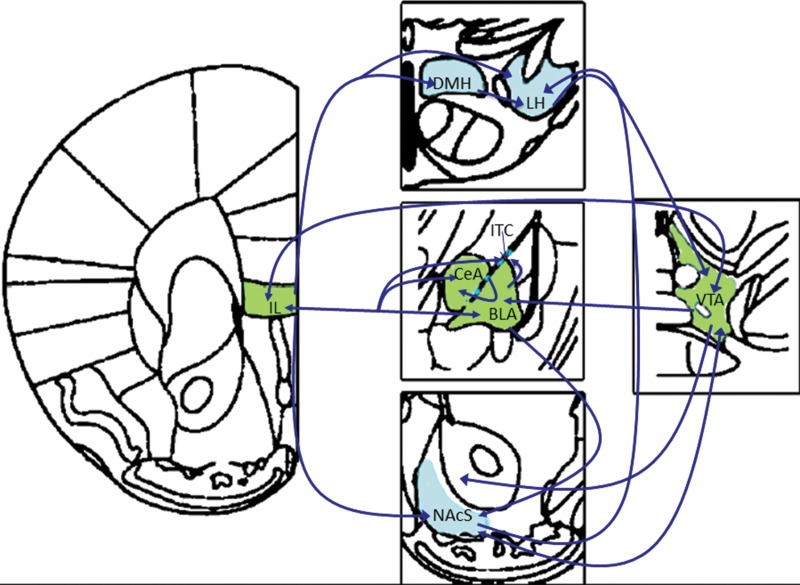
The IL projects to a number of target regions relevant to the expression and extinction of drug-seeking behavior (Fig. 2). Notably, disconnection of the IL from NAcS through contralateral inactivation prevents the expression of extinction, mimicking findings from bilateral IL lesions (Peters et al. 2008). NAcS glutamatergic plasticity has been reported after extinction training (Thompson and Swanson 2010) and, together, these findings suggest that IL glutamatergic projections to NAcS promote the plasticity observed after extinction training. The IL also projects extensively to the dorsomedial hypothalamus (DMH). Interestingly, the expression of extinction of ethanol seeking has been associated with increased c-Fos expression in IL projections to DMH (Marchant et al. 2010). The DMH has previously been associated with motivated behavior, and investigation into control of DMH activity by prefrontal networks is expected to provide considerable information regarding the neurocircuitry of extinction, and potentially how the extinction of alcohol and food seeking differs from cocaine- and fear-motivated learning. One potential mechanism by which the role of IL in extinction differs for various drugs of abuse is through the engagement of subcortical targets. The loss of IL may not affect the extinction of ethanol seeking as a result of increased engagement of DMH or other neuroanatomical substrates, although psychostimulants may not involve these structures to the same degree, resulting in a higher reliance on IL function.
Figure 2.
Subcortical targets of IL involved with extinction and habit learning. The IL projects to a number of subcortical targets, many of which have been shown to be critical substrates of extinction and habit learning. In addition, these subcortical regions have extensive interconnectivity, implicating a large potential network in mediating the acquisition and expression of extinction and stimulus–response habits. Although a number of additional targets exist, the dorsomedial hypothalamus has been identified as a substrate of extinction learning, and may be involved in habitual behavior as well. Additionally, amygdalar nuclei, which are extensively connected with the prefrontal cortex, the ventral striatum, and dopaminergic nuclei, have been implicated in extinction and habit formation, and are likely to be an important focus of investigation into the regulation of flexible reward seeking. The accumbens shell, which receives input from IL as well as a number of other limbic and midbrain structures, has been identified as a key regulator of extinction. Reports have implicated brain regions in green in both extinction and habitual behavior. To date, brain regions highlighted in blue have only been associated with extinction learning and their role in habitual behavior is as yet unclear.
Infralimbic PFC is required for the development of habitual behavior
As a general framework, it is thought that the shift away from goal-directed behavior to habitual reward seeking is paralleled by a decrease in PFC control of behavior, with stimulus–response habits instead mediated by subcortical regions including the striatum (Yin and Knowlton 2006). However, the role for IL in habitual reward seeking appears to be at odds with this general framework for the role of PFC in goal-directed behavior as well as with the ability of IL activity to drive behavioral flexibility in models of extinction. When IL is lesioned before training, animals are able to acquire action–outcome contingencies, and to remain sensitive to outcome devaluation even after extended training when control (sham lesion) animals are habitual (Killcross and Coutureau 2003). The IL is not merely critical in the acquisition of habitual behavior, but rather, inactivation of IL after training to habit restores goal-directed behavior. This suggests that intact IL function is required for the expression of stimulus–response habits even after their acquisition (Coutureau and Killcross 2003).
Using a T-maze task, Smith et al. (2012) investigated the role of IL glutamatergic pyramidal cells in online control of habitual behavior. Complementary to previous findings, this group also observed that IL inhibition during behavior disrupted the expression of habitual reward seeking. They extended this to demonstrate that disruption of the IL must occur during performance of the behavior to alter the expression of habits. In contrast, neither pre-task nor post-task disruption during potential “anticipation” or consolidation intervals prevented the expression of habits. Additional work has demonstrated that IL must be online during the acquisition of habitual performance, not simply the expression of habits (Smith and Graybiel 2013). These data build upon additional research from this group, indicating that across the development of habits, IL develops “task-bracketing” activity (i.e., activity at the beginning and end of trials) in the superficial layers that are primarily corticocortical projections (Smith and Graybiel 2013). In contrast, deep-layer projection neurons exhibit an increase in activity across the development of habits that stands in contrast to a decrease in activity in the more dorsal PL. Together, these data indicate that, as is the case for extinction learning, IL is required for both the acquisition and expression of habitual reward-seeking behavior, and that particular patterns of activity may be required for this to occur.
In a response conflict paradigm in which the performance of an automatic behavior in an inappropriate context was assessed, inactivation of IL promoted the inhibition of inappropriate responding (Haddon and Killcross 2011a). This finding is in direct contrast to data on the roles of more dorsal PL and ACC in which loss of function drives inappropriate responding that is insensitive to context (Marquis et al. 2007). Together, these data suggest that IL lesions enable context and outcome information to promote flexible action despite competing drive of automatic, “overtrained” behavior.
While there is a significant body of literature indicating that dopamine signaling in the striatum is critical for the formation of habitual behavior (Nelson and Killcross 2006), a number of studies have also established a role for IL dopamine signaling in habit formation. Infusions of dopamine in the IL, but not dmPFC, restores goal-directed behavior after animals have become insensitive to outcome devaluation (i.e., habitual) (Hitchcott et al. 2007). More recent data have suggested that the ability of dopamine to promote flexible, goal-directed reward seeking may be mediated through D2-like receptors, as agonism of dopamine D2 receptors in IL restores sensitivity to changes in action–outcome relationship at a time point in which animals look habitual (Barker et al. 2013). Interestingly, antagonism of dopamine D1 receptors in IL similarly promoted flexible goal-directed actions, suggesting that a balance of IL D1:D2 signaling may be critical for the expression of actions versus habits. Systemic data have shown opposing roles for dopamine D1 and D2 receptors on amphetamine-induced acceleration of habit formation. Systemic administration of dopamine D1 antagonists reverse this acceleration, although D2 antagonists facilitate habit formation, potentially consistent with data from the IL (Nelson and Killcross 2013). While PL dopamine signaling has been shown to modulate behavior in a response conflict paradigm (Haddon and Killcross 2011b), to our knowledge, the effect of IL dopamine manipulations on the regulation of context-specific behavior has not yet been investigated. However, IL dopamine signaling has been shown to modulate compulsive-like reward seeking in a paradigm involving competition between reward seeking and avoidance of adverse consequences in which entry to a reward- and punishment-paired context was investigated. As in habitual reward-seeking behavior, D2 agonism or D1 antagonism was found to reduce compulsive-like continued reward seeking (Barker et al. 2013).
The precise mechanism by which both dopamine D2 agonism and IL inactivation or lesioning drive goal-directed behavior is unclear. One possibility is that D2 agonism reduces inflexible behavior through decreasing pyramidal cell excitability, ultimately reducing IL regulation of behavior. Alternatively, a growing literature suggests that, as is well established in the striatum, prefrontal dopamine D1- and D2-containing neurons may be in part separate populations of cells, with distinct subcortical projection targets (Vincent et al. 1995; Gee et al. 2012). Striatal dopamine D1- and D2-containing neurons have distinct projection targets and compose the direct and indirect loops in the basal ganglia, participating in dopamine regulation of cortical input—and likely addictive behavior—through these distinct pathways (Gerfen et al. 1990; Surmeier et al. 1996; Onn et al. 2000; Kravitz and Kreitzer 2012). It is similarly possible that a shift in D1 versus D2 signaling differentially affect behavior through distinct outputs. A considerable amount of research has investigated the role of prefrontal dopamine in working memory, suggesting that PFC dopamine regulates prefrontal behavior in a dose-dependent manner. When dopamine activity is either too high or too low, it has been proposed that animals perform PFC-mediated tasks suboptimally (Arnsten 2007). However, more recent work has suggested that this “inverted-U” relationship between PFC dopamine signaling and PFC-mediated performance may be specific to working memory and attention (Floresco 2013). Investigation of behavioral flexibility in set shifting tasks implicates specific dopamine receptor subtypes in behavioral performance. Dopamine D2 receptor antagonism impaired set-shifting, while inhibition of the D4 receptor (a member of the D2-family of dopamine receptors) had opposing effects on behavior (Floresco et al. 2006). Furthermore, loss of either the D1 or D2 receptor activity in the mPFC can prevent the ability to update behavior in response to changes in reward value, but does not appear to block perception of the value change (Winter et al. 2009). While these data are not specific to IL, they highlight the importance of considering neuromodulator signaling carefully in complex assessments of behavioral flexibility.
Discrepancies in the role of infralimbic PFC in the regulation of reward seeking: frameworks for unifying theories
The absence of IL prevents the acquisition and expression of stimulus–response habits, indicating that IL function is critical for habitual behavior. Similarly, loss of IL activity can also enable selection of appropriate response strategies in situations of conflict. This suggests that the loss of IL promotes goal-directed actions that are sensitive to changes in action–outcome contingency. Somewhat counter intuitively, loss of IL also results in the inability to acquire or express extinction learning when action– or stimulus–outcome contingencies are no longer in place. The role of IL in extinction has led to the suggestion that the IL is part of a “stop” circuitry that mediates both fear and drug-seeking through projections to intercalated nucleus of the amygdala or the accumbens shell, respectively (Peters et al. 2008, 2009; Gass and Chandler 2013). However, it should be pointed out that this proposed model does not appear to account for the role of IL in habitual behaviors where loss of IL can reduce on-going habitual reward seeking.
Since acquisition and expression of extinction learning shares many features in common with habitual behavior, we, as have others, suggest that IL plays a complementary role in both forms of flexible behavior. For many types of extinction, IL function is critical to both the acquisition and the expression of extinction, and extinction can be bidirectionally altered through manipulation of IL plasticity or activity. Given the similarities between goal-directed behaviors and extinction, it is surprising that IL appears to play an opposing role in the acquisition and expression of stimulus–response habits. While loss of IL often prevents extinction, the same manipulations promote the expression of goal-directed behavior and indeed prohibit the development of habitual behavior that renders animals sensitive to changes in action–outcome relationships (i.e., sensitive to contingency changes).
There are at least three potential frameworks suggested or alluded to in a considerable literature through which IL manipulations have distinct consequences on these separate forms of behavioral flexibility [(Fig. 3; Coutureau and Killcross 2003; Killcross and Coutureau 2003; Peters et al. 2009) for additional discussion of these hypotheses]. First, the IL may engage discrete subcortical networks to drive extinction and/or habit acquisition and expression. Second, the IL is driven by distinct inputs mediating each behavior. Third, IL function is similar in both extinction and habit, but it elicits a component of behavior that appears artificially distinct. We propose that the latter framework—that IL functions similarly in both forms of behavioral flexibility—is the most likely mechanism (it should be noted that these processes are not mutually exclusive and could occur in combination). In particular, we suggest that through active inhibition of established action–outcome mediated behavior, the IL suppresses goal-directed behavior and therefore promotes the expression of habitual reward seeking as well as the acquisition and expression of extinction learning. In the absence of IL, the suppression of learned action–outcome relationships required to express and acquire extinction learning may be restricted. This may result in the inability to acquire or express the extinction of reward seeking when IL function is lost after extinction. Similarly, in assessments of habitual behavior, the loss of IL activity renders animals sensitive to changes in the outcome value. Again, we suggest that when IL suppression of the action–outcome contingency is lost, devaluation of the outcome is associated with a decrease in responding consistent with goal-directed behavior.
Figure 3.
Testing the model. Based on the existing literature, we have outlined four separate, but potentially coexisting, hypotheses for the separable role of the IL in the acquisition and expression of extinction and habit learning. Assessing the precise role of IL function in behavioral flexibility will likely require the elegant combination of behavioral, genetic, and molecular techniques. (a,b) IL may promote extinction and habit learning through interactions with distinct input and output sites. To determine the neuroanatomical substrates that participate in IL activity to drive the expression of extinction learning and habitual behaviors, inactivation and lesion studies can be performed. The timing of these procedures can disentangle the role of IL and its projection targets in the acquisition and expression of habitual behavior and extinction. While these traditional methods enable significant anatomical specificity, the use of optogenetic and DREADD strategies can further refine our understanding of the neuroanatomy of these behaviors to include cell-type specificity together with regional selectivity. If separate neural circuits mediate these behaviors, we would expect distinctions in the effect of lesions on these behaviors—e.g., disconnection of IL from projection targets or input structures may impact one, neither, or both of these behaviors enabling a greater understanding of the neurocircuitry of habitual behavior and extinction. (c) These anatomical studies should be incorporated with novel behavioral approaches to assessing the overarching role of IL in behavior. One hypothesis is that IL function facilitates the acquisition of stimulus–response behavior, including habits and extinction. We propose that the most simple way to disentangle these hypotheses is through the critical evaluation of loss of IL function in the acquisition of novel stimulus–response behaviors and in return to previously required action–outcome strategies. Specifically, inactivation or lesion of IL (or IL circuits) could be performed in tandem with assessments of the expression or acquisition of stimulus–response behaviors other than habit. (d) Alternatively, IL may promote habits and extinction learning through the suppression of the ability of action–outcome relationships to guide behavior. Testing this hypothesis would require suppression of previously acquired action–outcome relationships, in tandem with alterations of IL function, to determine whether loss of IL still results in a reversion to goal-directed behavior in the absence of the neurons encoding this relationship.
While we favor the above scenario, an alternative explanation might be that the IL functions similarly in the acquisition and expression of habit and extinction by driving the acquisition of stimulus–response associations—in other words, expediting the development of stimulus–response habits and facilitating the acquisition of novel stimulus- and context-mediated learning that may enable the acquisition and expression of extinction (cf Rescorla 2001; Bouton 2004; Delamater and Westbrook 2013). The IL may be acting in both habit and extinction to encode novel stimulus–response associations that override previously established contingencies. To our knowledge, no definitive data exist to discount this hypothesis. However, there is evidence that may argue against a role for the IL in the facilitation of stimulus–response learning. For example, it has been shown that the loss of IL does not prevent the acquisition of stimulus–response behaviors, specifically Pavlovian autoshaping (Chudasama and Robbins 2003). While these data do not convincingly show that IL does not have a role in facilitating the acquisition of these stimulus–response relationships, it clearly shows that the absence of IL does not prevent the formation of them, as it does stimulus–response habits and extinction learning.
Finally, the above-proposed hypotheses do not discount the additional possibility that IL activity is either driven by or drives separate subcortical networks in the acquisition and expression of extinction versus habitual behavior. Rather, we suggest that these accounts of IL function in flexible behavior are complementary. Evidence indicates that separate, but overlapping, neurocircuitry mediate the extinction of distinct reinforcers and habits, including additional prefrontal subregions and subcortical structures within limbic corticostriatal circuitry. While a more substantial literature has implicated the critical roles of the nucleus accumbens and amygdala in extinction (Peters et al. 2009), a growing literature has identified these same neuroanatomical substrates in the development and expression of habitual reward seeking (Belin and Everitt 2008; Lingawi and Balleine 2012). Indeed, given evidence of layer specificity in IL activity in habitual behavior (Smith and Graybiel 2013), and likely coordination of IL and PL activity in extinction (Peters et al. 2009; Sierra-Mercado et al. 2011), consideration of IL interaction with other cortical subregions should be made. The neuroanatomical substrates that cooperate with IL to drive extinction and habitual behavior are readily testable through the combination of lesion and inactivation studies, and the more temporally refined and cell-type-specific use of optogenetic strategies.
Potential for translation to the human disease state
The rodent IL has been shown to play a critical role in the development and expression of inflexible behavior that characterizes a number of mental illnesses, including addiction and disorders such as post-traumatic stress disorder. While not universally accepted, there is some speculation that the human subgenual vmPFC may be homologous to the rodent IL. Not only is the connectivity overlapping, but also data have linked the human vmPFC—including Brodmann's areas 25, 14, 11, 13 ventral 24 and 32—with extinction and habitual behavior. Increased vmPFC activity has been shown to be associated with the expression of goal-directed behavior in humans (de Wit et al. 2009), and it has been observed that connectivity between the vmPFC and striatal subregions can predict the “goal-directedness” of actions in healthy populations. This is consistent with the idea that innate differences in these connections may drive the propensity to develop habitual response strategies (de Wit et al. 2012). Activity in the subgenual vmPFC has also been shown to be associated with extinction learning in humans and may underlie extinction abnormalities in disease states (Quirk and Beer 2006; Hartley et al. 2011; Milad et al. 2013). Alhough a majority of these reports have focused on the investigation of fear extinction, given the high overlap between the role of IL in distinct forms of extinction learning in the rodent, it is tempting to speculate that considerable similarity between vmPFC function in fear and reward extinction will exist.
Consistent with roles for habitual behavior and extinction learning in disease states, vmPFC abnormalities have been observed in addiction (Bechara 2003; Sinha and Li 2007; George and Koob 2010; Courtney et al. 2012), post-traumatic stress disorder, and depression (Shin et al. 2001; Li and Sinha 2008; Peters et al. 2009). Indeed, decreased vmPFC activity has been observed in alcoholics showing elevated propensity toward habitual behavior (Sjoerds et al. 2013). Others have suggested that the human vmPFC may be involved in the suppression of irrelevant information, potentially by integrating information from separable components of limbic corticostriatal circuitry (Nieuwenhuis and Takashima 2011). If indeed these structures are performing comparable functions, and working toward the suppression of previously established action–outcome associations, an understanding of the causes of dysfunction and mechanisms of restoration of vmPFC function is expected to be critical for the treatment and prevention of mental illnesses associated with inflexible behavior.
Conclusions and Summary
The IL PFC is highly interconnected with neuroanatomical substrates that regulate complex cognitive behavior. Its position within the limbic corticostriatal circuitry that mediates reward-seeking behavior makes the IL a key target for investigation into the loss of behavioral flexibility. The IL function has been shown to be crucial for the acquisition and expression of extinction, as well as for the acquisition and expression of habitual behavior. We suggest that these data argue that the IL is critical for the active suppression of established action–outcome relationships, and that its absence results in the use of established action–outcome contingencies to guide behavior. Future research should focus on identifying the precise mechanism through which the IL regulates the acquisition and expression of behavioral flexibility, as well as the neurotransmitter and neuroanatomical targets that mediate this role, with the aim of developing effective prevention and treatment strategies for psychiatric illnesses characterized by inflexible behavior, including addiction, obsessive compulsive disorder, and post-traumatic stress disorder.
Funding
This research was supported by National Institutes of Health Grants A007474 and AA020135 (to J.M.B.); AA012870, DA 011717, DA015222 (to J.R.T.); and AA010761, AA010983 and AA019967 (to L.J.C.).
Footnotes
Article is online at http://www.learnmem.org/cgi/doi/10.1101/lm.035501.114.
References
- Adams CD 1982. Variations in the sensitivity of instrumental responding to reinforcer devaluation. Q J Exp Psychol B 34: 77–98 [Google Scholar]
- Arnsten AFT 2007. Catecholamine and second messenger influences on prefrontal cortical networks of “representational knowledge”: a rational bridge between genetics and the symptoms of mental illness. Cereb Cortex 17 Suppl 1: i6–i15 [DOI] [PubMed] [Google Scholar]
- Balleine BW, Dickinson A 1998. Goal-directed instrumental action: contingency and incentive learning and their cortical substrates. Neuropharmacology 37: 407–419 [DOI] [PubMed] [Google Scholar]
- Barker JM, Torregrossa MM, Taylor JR 2012. Low prefrontal PSA-NCAM confers risk for alcoholism-related behavior. Nat Neurosci 15: 1356–1358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barker JM, Torregrossa MM, Taylor JR 2013. Bidirectional modulation of infralimbic dopamine D1 and D2 receptor activity regulates flexible reward seeking. Front Neurosci 7: 126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bechara A 2003. Risky business: emotion, decision-making, and addiction. J Gambl Stud 19: 23–51 [DOI] [PubMed] [Google Scholar]
- Belin D, Everitt BJ 2008. Cocaine seeking habits depend upon dopamine-dependent serial connectivity linking the ventral with the dorsal striatum. Neuron 57: 432–441 [DOI] [PubMed] [Google Scholar]
- Berendse HW, Galis-de Graaf Y, Groenewegen HJ 1992. Topographical organization and relationship with ventral striatal compartments of prefrontal corticostriatal projections in the rat. J Comp Neurol 316: 314–347 [DOI] [PubMed] [Google Scholar]
- Bossert JM, Stern AL, Theberge FRM, Marchant NJ, Wang H-L, Morales M, Shaham Y 2012. Role of projections from ventral medial prefrontal cortex to nucleus accumbens shell in context-induced reinstatement of heroin seeking. J Neurosci 32: 4982–4991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouton ME 2004. Context and behavioral processes in extinction. Learn Mem 11: 485–494 [DOI] [PubMed] [Google Scholar]
- Cassell MD, Wright DJ 1986. Topography of projections from the medial prefrontal cortex to the amygdala in the rat. Brain Res Bull 17: 321–333 [DOI] [PubMed] [Google Scholar]
- Castillo-Gómez E, Gómez-Climent MA, Varea E, Guirado R, Blasco-Ibáñez JM, Crespo C, Martínez-Guijarro FJ, Nácher J 2008. Dopamine acting through D2 receptors modulates the expression of PSA-NCAM, a molecule related to neuronal structural plasticity, in the medial prefrontal cortex of adult rats. Exp Neurol 214: 97–111 [DOI] [PubMed] [Google Scholar]
- Chudasama Y, Robbins TW 2003. Dissociable contributions of the orbitofrontal and infralimbic cortex to Pavlovian autoshaping and discrimination reversal learning: further evidence for the functional heterogeneity of the rodent frontal cortex. J Neurosci 23: 8771–8780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney KE, Ghahremani DG, Ray LA 2012. Fronto-striatal functional connectivity during response inhibition in alcohol dependence. Addict Biol 18: 593–604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutureau E, Killcross S 2003. Inactivation of the infralimbic prefrontal cortex reinstates goal-directed responding in overtrained rats. Behav Brain Res 146: 167–174 [DOI] [PubMed] [Google Scholar]
- Delamater AR, Westbrook RF 2013. Psychological and neural mechanisms of experimental extinction: a selective review. Neurobiol Learn Mem. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit S, Corlett PR, Aitken MR, Dickinson A, Fletcher PC 2009. Differential engagement of the ventromedial prefrontal cortex by goal-directed and habitual behavior toward food pictures in humans. J Neurosci 29: 11330–11338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit S, Watson P, Harsay HA, Cohen MX, van de Vijver I, Ridderinkhof KR 2012. Corticostriatal connectivity underlies individual differences in the balance between habitual and goal-directed action control. J Neurosci 32: 12066–12075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson A 1985. Actions and habits: the development of behavioural autonomy. Philos Trans R Soc Lond B Biol Sci 308: 67–78 [Google Scholar]
- Dickinson A, Wood N, Smith JW 2002. Alcohol seeking by rats: action or habit? Q J Exp Psychol B 55: 331–348 [DOI] [PubMed] [Google Scholar]
- Everitt BJ, Robbins TW 2013. From the ventral to the dorsal striatum: devolving views of their roles in drug addiction. Neurosci Biobehav Rev. 10.1016/j.neubiorev.2013.02.010 [DOI] [PubMed] [Google Scholar]
- Everitt BJ, Belin D, Economidou D, Pelloux Y, Dalley JW, Robbins TW 2008. Review. Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction. Philos Trans R Soc Lond B Biol Sci 363: 3125–3135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Floresco SB 2013. Prefrontal dopamine and behavioral flexibility: shifting from an “inverted-U” toward a family of functions. Front Neurosci 7: 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Floresco SB, Magyar O, Ghods-Sharifi S, Vexelman C, Tse MTL 2006. Multiple dopamine receptor subtypes in the medial prefrontal cortex of the rat regulate set-shifting. Neuropsychopharmacology 31: 297–309 [DOI] [PubMed] [Google Scholar]
- Francois J, Huxter J, Conway MW, Lowry JP, Tricklebank MD, Gilmour G 2014. Differential contributions of infralimbic prefrontal cortex and nucleus accumbens during reward-based learning and extinction. J Neurosci 34: 596–607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gass JT, Chandler LJ 2013. The plasticity of extinction: contribution of the prefrontal cortex in treating addiction through inhibitory learning. Front Psychiatry 4: 46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee S, Ellwood I, Patel T, Luongo F, Deisseroth K, Sohal VS 2012. Synaptic activity unmasks dopamine D2 receptor modulation of a specific class of layer V pyramidal neurons in prefrontal cortex. J Neurosci 32: 4959–4971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- George O, Koob GF 2010. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev 35: 232–247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerfen CR 1984. The neostriatal mosaic: compartmentalization of corticostriatal input and striatonigral output systems. Nature 311: 461–464 [DOI] [PubMed] [Google Scholar]
- Gerfen CR, Engber TM, Mahan LC, Susel Z, Chase TN, Monsma FJ, Sibley DR 1990. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 250: 1429–1432 [DOI] [PubMed] [Google Scholar]
- Haber S 2008. Parallel and integrative processing through the Basal Ganglia reward circuit: lessons from addiction. Biol Psychiatry 64: 173–174 [DOI] [PubMed] [Google Scholar]
- Haber SN, Knutson B 2010. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology 35: 4–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddon JE, Killcross S 2011a. Inactivation of the infralimbic prefrontal cortex in rats reduces the influence of inappropriate habitual responding in a response-conflict task. Neuroscience 199: 205–212 [DOI] [PubMed] [Google Scholar]
- Haddon JE, Killcross S 2011b. Rat prefrontal dopamine and cognitive control: impaired and enhanced conflict performance. Behav Neurosci 125: 344–349 [DOI] [PubMed] [Google Scholar]
- Hartley CA, Fischl B, Phelps EA 2011. Brain structure correlates of individual differences in the acquisition and inhibition of conditioned fear. Cereb Cortex 21: 1954–1962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedou G, Feldon J, Heidbreder CA 1999a. Effects of cocaine on dopamine in subregions of the rat prefrontal cortex and their efferents to subterritories of the nucleus accumbens. Eur J Pharmacol 372: 143–155 [DOI] [PubMed] [Google Scholar]
- Hedou G, Homberg J, Feldon J, Heidbreder CA 1999b. Amphetamine microinfusion in the dorso-ventral axis of the prefrontal cortex differentially modulates dopamine neurotransmission in the shell–core subterritories of the nucleus accumbens. Ann N Y Acad Sci 877: 823–827 [DOI] [PubMed] [Google Scholar]
- Hedou G, Homberg J, Feldon J, Heidbreder CA 2001. Expression of sensitization to amphetamine and dynamics of dopamine neurotransmission in different laminae of the rat medial prefrontal cortex. Neuropharmacology 40: 366–382 [DOI] [PubMed] [Google Scholar]
- Heidbreder CA, Groenewegen HJ 2003. The medial prefrontal cortex in the rat: evidence for a dorso-ventral distinction based upon functional and anatomical characteristics. Neurosci Biobehav Rev 27: 555–579 [DOI] [PubMed] [Google Scholar]
- Hitchcott PK, Quinn JJ, Taylor JR 2007. Bidirectional modulation of goal-directed actions by prefrontal cortical dopamine. Cereb Cortex 17: 2820–2827 [DOI] [PubMed] [Google Scholar]
- Hugues S, Deschaux O, Garcia R 2004. Postextinction infusion of a mitogen-activated protein kinase inhibitor into the medial prefrontal cortex impairs memory of the extinction of conditioned fear. Learn Mem 11: 540–543 [DOI] [PubMed] [Google Scholar]
- Hugues S, Chessel A, Lena I, Marsault R, Garcia R 2006. Prefrontal infusion of PD098059 immediately after fear extinction training blocks extinction-associated prefrontal synaptic plasticity and decreases prefrontal ERK2 phosphorylation. Synapse 60: 280–287 [DOI] [PubMed] [Google Scholar]
- Hurley KM, Herbert H, Moga MM, Saper CB 1991. Efferent projections of the infralimbic cortex of the rat. J Comp Neurol 308: 249–276 [DOI] [PubMed] [Google Scholar]
- Jentsch JD, Taylor JR 1999. Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology (Berl) 146: 373–390 [DOI] [PubMed] [Google Scholar]
- Killcross S, Coutureau E 2003. Coordination of actions and habits in the medial prefrontal cortex of rats. Cereb Cortex 13: 400–408 [DOI] [PubMed] [Google Scholar]
- Knapska E, Maren S 2009. Reciprocal patterns of c-Fos expression in the medial prefrontal cortex and amygdala after extinction and renewal of conditioned fear. Learn Mem 16: 486–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kravitz AV, Kreitzer AC 2012. Striatal mechanisms underlying movement, reinforcement, and punishment. Physiology (Bethesda) 27: 167–177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaLumiere RT, Niehoff KE, Kalivas PW 2010. The infralimbic cortex regulates the consolidation of extinction after cocaine self-administration. Learn Mem 17: 168–175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurent V, Westbrook RF 2009. Inactivation of the infralimbic but not the prelimbic cortex impairs consolidation and retrieval of fear extinction. Learn Mem 16: 520–529 [DOI] [PubMed] [Google Scholar]
- Lebrón K, Milad MR, Quirk GJ 2004. Delayed recall of fear extinction in rats with lesions of ventral medial prefrontal cortex. Learn Mem 11: 544–548 [DOI] [PubMed] [Google Scholar]
- Li CR, Sinha R 2008. Inhibitory control and emotional stress regulation: neuroimaging evidence for frontal-limbic dysfunction in psycho-stimulant addiction. Neurosci Biobehav Rev 32: 581–597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lingawi NW, Balleine BW 2012. Amygdala central nucleus interacts with dorsolateral striatum to regulate the acquisition of habits. J Neurosci 32: 1073–1081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchant NJ, Furlong TM, McNally GP 2010. Medial dorsal hypothalamus mediates the inhibition of reward seeking after extinction. J Neurosci 30: 14102–14115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marquis J-P, Killcross S, Haddon JE 2007. Inactivation of the prelimbic, but not infralimbic, prefrontal cortex impairs the contextual control of response conflict in rats. Eur J Neurosci 25: 559–566 [DOI] [PubMed] [Google Scholar]
- Mcdonald AJ, Mascagni F, Guo L 1996. Projections of the medial and lateral prefrontal cortices to the amygdala: a Phaseolus vulgaris leucoagglutinin study in the rat. Neuroscience 71: 55–75 [DOI] [PubMed] [Google Scholar]
- Miguéns M, Crespo JA, Del Olmo N, Higuera-Matas A, Montoya GL, García-Lecumberri C, Ambrosio E 2008. Differential cocaine-induced modulation of glutamate and dopamine transporters after contingent and non-contingent administration. Neuropharmacology 55: 771–779 [DOI] [PubMed] [Google Scholar]
- Milad MR, Quirk GJ 2002. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature 420: 70–74 [DOI] [PubMed] [Google Scholar]
- Milad MR, Furtak SC, Greenberg JL, Keshaviah A, Im JJ, Falkenstein MJ, Jenike M, Rauch SL, Wilhelm S 2013. Deficits in conditioned fear extinction in obsessive-compulsive disorder and neurobiological changes in the fear circuit. JAMA Psychiatry 70: 608–618 [DOI] [PubMed] [Google Scholar]
- Miles FJ, Everitt BJ, Dickinson A 2003. Oral cocaine seeking by rats: action or habit? Behav Neurosci 117: 927–938 [DOI] [PubMed] [Google Scholar]
- Millan EZ, Marchant NJ, McNally GP 2011. Extinction of drug seeking. Behav Brain Res 217: 454–462 [DOI] [PubMed] [Google Scholar]
- Morgan MA, Romanski LM, LeDoux JE 1993. Extinction of emotional learning: contribution of medial prefrontal cortex. Neurosci Lett 163: 109–113 [DOI] [PubMed] [Google Scholar]
- Mueller D, Bravo-Rivera C, Quirk GJ 2010. Infralimbic D2 receptors are necessary for fear extinction and extinction-related tone responses. Biol Psychiatry 68: 1055–1060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson A, Killcross S 2006. Amphetamine exposure enhances habit formation. J Neurosci 26: 3805–3812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson AJD, Killcross S 2013. Accelerated habit formation following amphetamine exposure is reversed by D1, but enhanced by D2, receptor antagonists. Front Neurosci 7: 76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieuwenhuis ILC, Takashima A 2011. The role of the ventromedial prefrontal cortex in memory consolidation. Behav Brain Res 218: 325–334 [DOI] [PubMed] [Google Scholar]
- Onn SP, West AR, Grace AA 2000. Dopamine-mediated regulation of striatal neuronal and network interactions. Trends Neurosci 23: S48–S56 [DOI] [PubMed] [Google Scholar]
- Peters J, De Vries TJ 2013. D-cycloserine administered directly to infralimbic medial prefrontal cortex enhances extinction memory in sucrose-seeking animals. Neuroscience 230: 24–30 [DOI] [PubMed] [Google Scholar]
- Peters J, LaLumiere RT, Kalivas PW 2008. Infralimbic prefrontal cortex is responsible for inhibiting cocaine seeking in extinguished rats. J Neurosci 28: 6046–6053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, Kalivas PW, Quirk GJ 2009. Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem 16: 279–288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, Dieppa-Perea LM, Melendez LM, Quirk GJ 2010. Induction of fear extinction with hippocampal-infralimbic BDNF. Science 328: 1288–1290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, Pattij T, De Vries TJ 2013. Targeting cocaine versus heroin memories: divergent roles within ventromedial prefrontal cortex. Trends Pharmacol Sci. 34: 689–6950 [DOI] [PubMed] [Google Scholar]
- Quirk GJ, Beer JS 2006. Prefrontal involvement in the regulation of emotion: convergence of rat and human studies. Curr Opin Neurobiol 16: 723–727 [DOI] [PubMed] [Google Scholar]
- Quirk GJ, Russo GK, Barron JL, Lebron K 2000. The role of ventromedial prefrontal cortex in the recovery of extinguished fear. J Neurosci 20: 6225–6231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rescorla RA 2001. Experimental extinction. In Handbook of contemporary learning theories, pp. 119–154 Lawrence Erlbaum Associates, Mahwah, NJ [Google Scholar]
- Santini E, Ge H, Ren K, Peña de Ortiz S, Quirk GJ 2004. Consolidation of fear extinction requires protein synthesis in the medial prefrontal cortex. J Neurosci 24: 5704–5710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santini E, Quirk GJ, Porter JT 2008. Fear conditioning and extinction differentially modify the intrinsic excitability of infralimbic neurons. J Neurosci 28: 4028–4036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin LM, Whalen PJ, Pitman RK, Bush G, Macklin ML, Lasko NB, Orr SP, McInerney SC, Rauch SL 2001. An fMRI study of anterior cingulate function in posttraumatic stress disorder. Biol Psychiatry 50: 932–942 [DOI] [PubMed] [Google Scholar]
- Sierra-Mercado D, Corcoran KA, Lebrón-Milad K, Quirk GJ 2006. Inactivation of the ventromedial prefrontal cortex reduces expression of conditioned fear and impairs subsequent recall of extinction. Eur J Neurosci 24: 1751–1758 [DOI] [PubMed] [Google Scholar]
- Sierra-Mercado D, Padilla-Coreano N, Quirk GJ 2011. Dissociable roles of prelimbic and infralimbic cortices, ventral hippocampus, and basolateral amygdala in the expression and extinction of conditioned fear. Neuropsychopharmacology 36: 529–538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha R, Li CSR 2007. Imaging stress- and cue-induced drug and alcohol craving: association with relapse and clinical implications. Drug Alcohol Rev 26: 25–31 [DOI] [PubMed] [Google Scholar]
- Sjoerds Z, de Wit S, van den Brink W, Robbins TW, Beekman ATF, Penninx BWJH, Veltman DJ 2013. Behavioral and neuroimaging evidence for overreliance on habit learning in alcohol-dependent patients. Transl Psychiatry 3: e337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith KS, Graybiel AM 2013. A dual operator view of habitual behavior reflecting cortical and striatal dynamics. Neuron 79: 361–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith KS, Virkud A, Deisseroth K, Graybiel AM 2012. Reversible online control of habitual behavior by optogenetic perturbation of medial prefrontal cortex. Proc Natl Acad Sci 109: 18932–18937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surmeier DJ, Song WJ, Yan Z 1996. Coordinated expression of dopamine receptors in neostriatal medium spiny neurons. J Neurosci 16: 6579–6591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thierry AM, Blanc G, Sobel A, Stinus L, Golwinski J 1973. Dopaminergic terminals in the rat cortex. Science 182: 499–501 [DOI] [PubMed] [Google Scholar]
- Thompson RH, Swanson LW 2010. Hypothesis-driven structural connectivity analysis supports network over hierarchical model of brain architecture. Proc Natl Acad Sci 107: 15235–15239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torregrossa MM, Sanchez H, Taylor JR 2010. D-cycloserine reduces the context specificity of Pavlovian extinction of cocaine cues through actions in the nucleus accumbens. J Neurosci 30: 10526–10533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Eden CG, Uylings HB 1985. Cytoarchitectonic development of the prefrontal cortex in the rat. J Comp Neurol 241: 253–267 [DOI] [PubMed] [Google Scholar]
- Vincent SL, Khan Y, Benes FM 1995. Cellular colocalization of dopamine D1 and D2 receptors in rat medial prefrontal cortex. Synapse 19: 112–120 [DOI] [PubMed] [Google Scholar]
- Willcocks AL, McNally GP 2012. The role of medial prefrontal cortex in extinction and reinstatement of alcohol-seeking in rats. Eur J Neurosci. [DOI] [PubMed] [Google Scholar]
- Winter S, Dieckmann M, Schwabe K 2009. Dopamine in the prefrontal cortex regulates rats behavioral flexibility to changing reward value. Behav Brain Res 198: 206–213 [DOI] [PubMed] [Google Scholar]
- Yin HH, Knowlton BJ 2006. The role of the basal ganglia in habit formation. Neuroscience 7: 464–476 [DOI] [PubMed] [Google Scholar]