Abstract
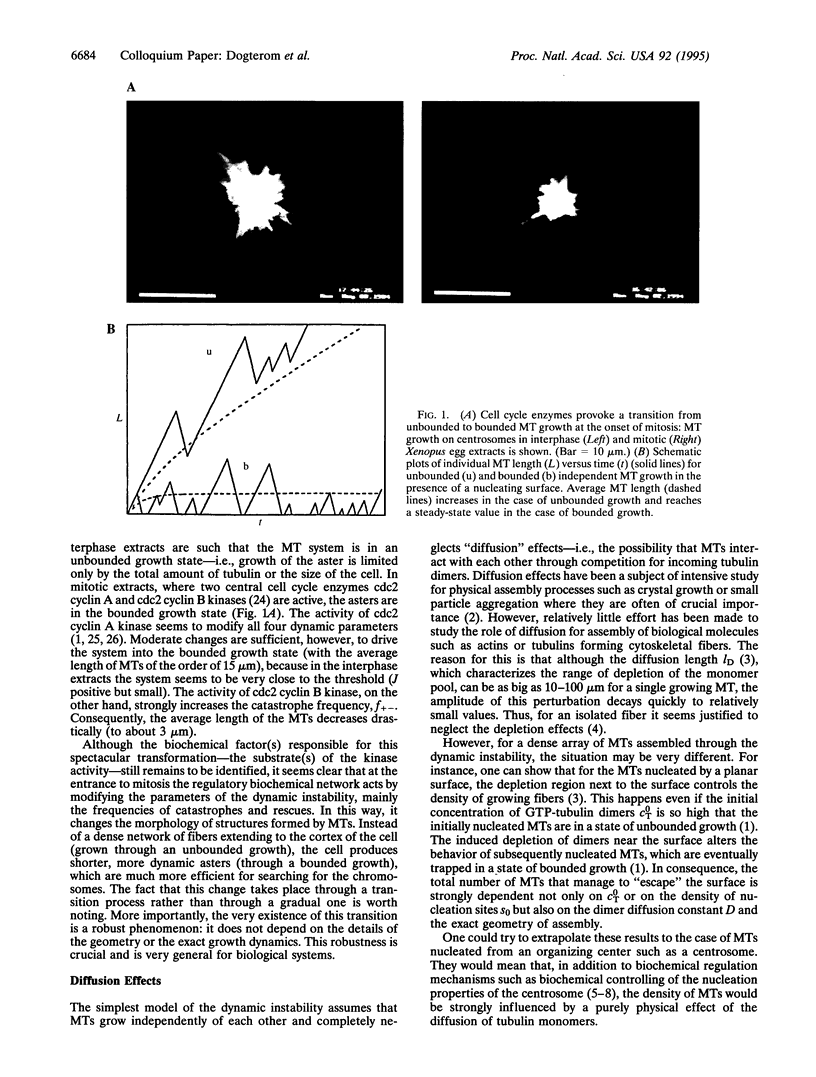
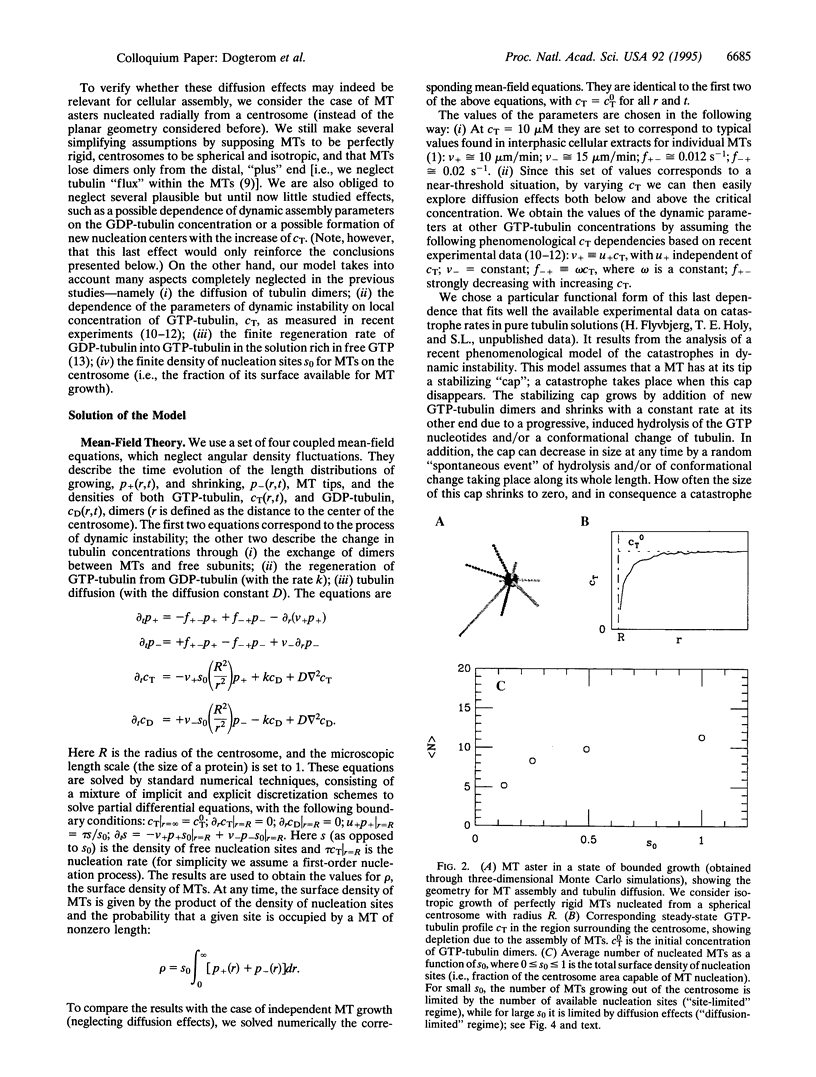
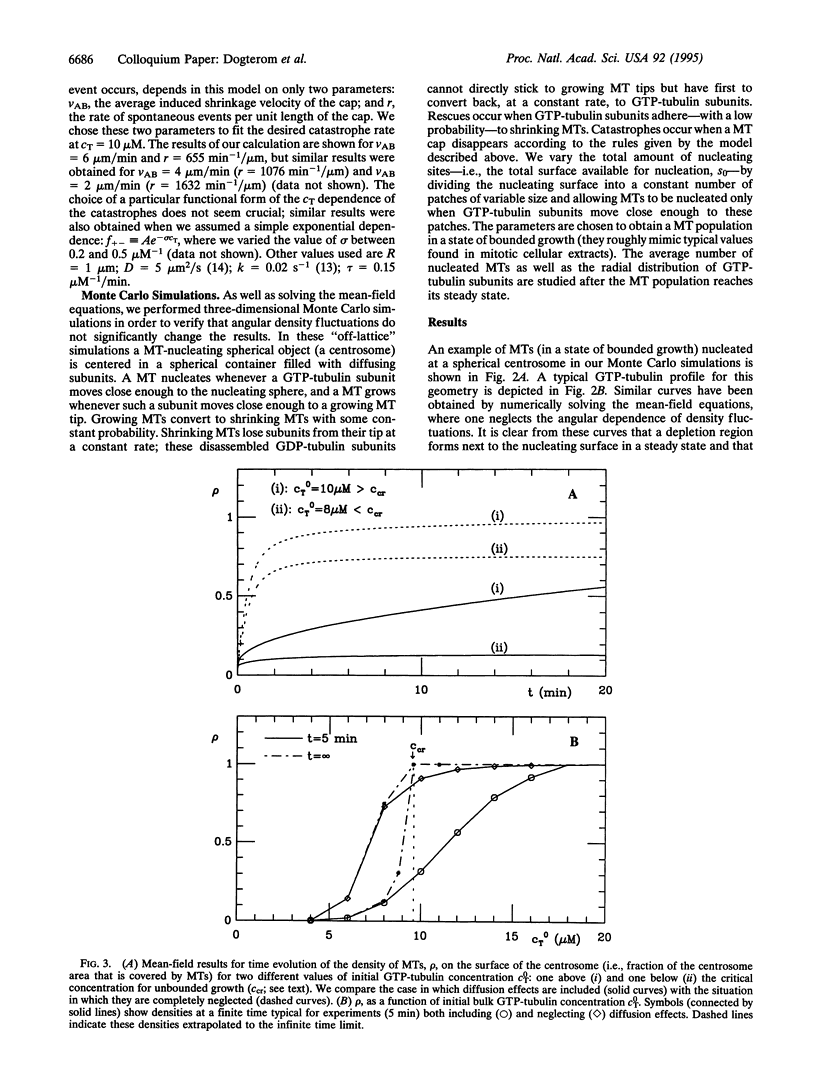
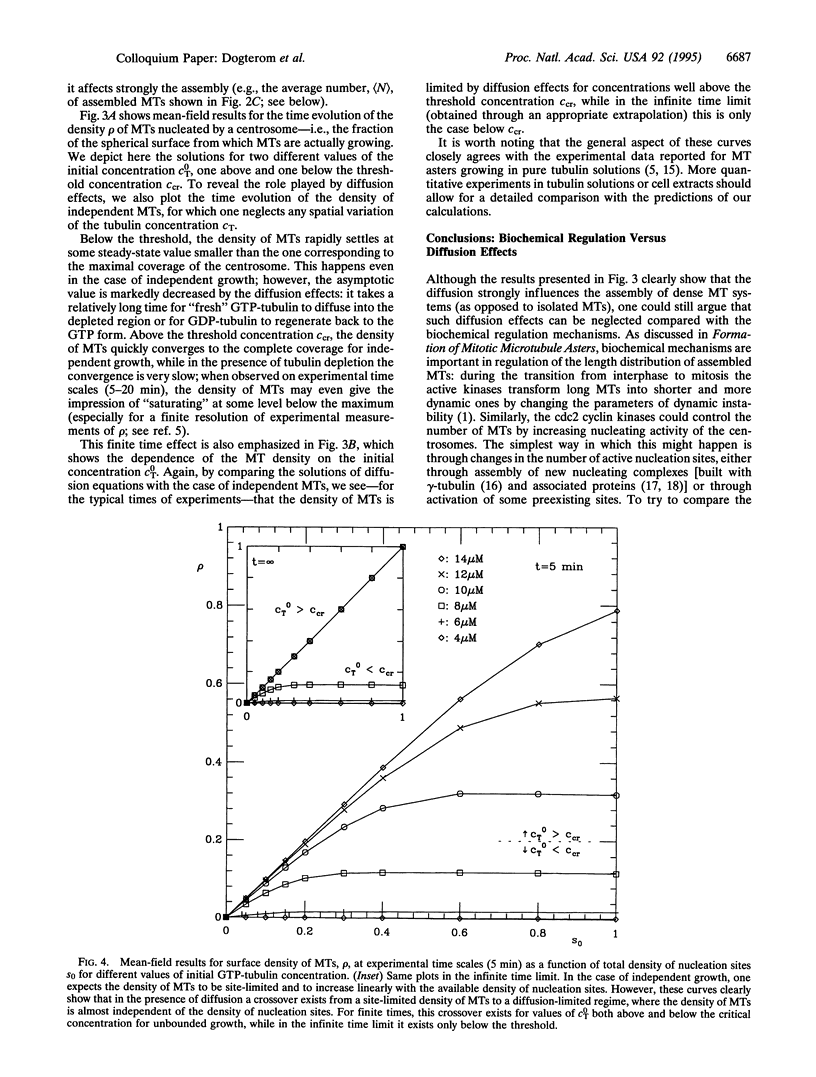
Microtubule asters forming the mitotic spindle are assembled around two centrosomes through the process of dynamic instability in which microtubules alternate between growing and shrinking states. By modifying the dynamics of this assembly process, cell cycle enzymes, such as cdc2 cyclin kinases, regulate length distributions in the asters. It is believed that the same enzymes control the number of assembled microtubules by changing the "nucleating activity" of the centrosomes. Here we show that assembly of microtubule asters may be strongly altered by effects connected with diffusion of tubulin monomers. Theoretical analysis of a simple model describing assembly of microtubule asters clearly shows the existence of a region surrounding the centrosome depleted in GTP tubulin. The number of assembled microtubules may in some cases be limited by this depletion effect rather than by the number of available nucleation sites on the centrosome.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bayley P. Why microtubules grow and shrink. Nature. 1993 May 27;363(6427):309–309. doi: 10.1038/363309a0. [DOI] [PubMed] [Google Scholar]
- Belmont L. D., Hyman A. A., Sawin K. E., Mitchison T. J. Real-time visualization of cell cycle-dependent changes in microtubule dynamics in cytoplasmic extracts. Cell. 1990 Aug 10;62(3):579–589. doi: 10.1016/0092-8674(90)90022-7. [DOI] [PubMed] [Google Scholar]
- Bré M. H., Karsenti E. Effects of brain microtubule-associated proteins on microtubule dynamics and the nucleating activity of centrosomes. Cell Motil Cytoskeleton. 1990;15(2):88–98. doi: 10.1002/cm.970150205. [DOI] [PubMed] [Google Scholar]
- Buendia B., Draetta G., Karsenti E. Regulation of the microtubule nucleating activity of centrosomes in Xenopus egg extracts: role of cyclin A-associated protein kinase. J Cell Biol. 1992 Mar;116(6):1431–1442. doi: 10.1083/jcb.116.6.1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dogterom M, Leibler S. Physical aspects of the growth and regulation of microtubule structures. Phys Rev Lett. 1993 Mar 1;70(9):1347–1350. doi: 10.1103/PhysRevLett.70.1347. [DOI] [PubMed] [Google Scholar]
- Drechsel D. N., Hyman A. A., Cobb M. H., Kirschner M. W. Modulation of the dynamic instability of tubulin assembly by the microtubule-associated protein tau. Mol Biol Cell. 1992 Oct;3(10):1141–1154. doi: 10.1091/mbc.3.10.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi H. C., Palacios M. J., McNamara L., Cleveland D. W. Gamma-tubulin is a centrosomal protein required for cell cycle-dependent microtubule nucleation. Nature. 1992 Mar 5;356(6364):80–83. doi: 10.1038/356080a0. [DOI] [PubMed] [Google Scholar]
- Kalt A., Schliwa M. Molecular components of the centrosome. Trends Cell Biol. 1993 Apr;3(4):118–128. doi: 10.1016/0962-8924(93)90174-y. [DOI] [PubMed] [Google Scholar]
- Kellogg D. R., Sullivan W., Theurkauf W., Oegema K., Raff J. W., Alberts B. M. Studies on the centrosome and cytoplasmic organization in the early Drosophila embryo. Cold Spring Harb Symp Quant Biol. 1991;56:649–662. doi: 10.1101/sqb.1991.056.01.074. [DOI] [PubMed] [Google Scholar]
- Masuda H., Sevik M., Cande W. Z. In vitro microtubule-nucleating activity of spindle pole bodies in fission yeast Schizosaccharomyces pombe: cell cycle-dependent activation in xenopus cell-free extracts. J Cell Biol. 1992 Jun;117(5):1055–1066. doi: 10.1083/jcb.117.5.1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melki R., Carlier M. F., Pantaloni D. Oscillations in microtubule polymerization: the rate of GTP regeneration on tubulin controls the period. EMBO J. 1988 Sep;7(9):2653–2659. doi: 10.1002/j.1460-2075.1988.tb03118.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchison T. J., Sawin K. E. Tubulin flux in the mitotic spindle: where does it come from, where is it going? Cell Motil Cytoskeleton. 1990;16(2):93–98. doi: 10.1002/cm.970160202. [DOI] [PubMed] [Google Scholar]
- Mitchison T., Kirschner M. Dynamic instability of microtubule growth. Nature. 1984 Nov 15;312(5991):237–242. doi: 10.1038/312237a0. [DOI] [PubMed] [Google Scholar]
- Mitchison T., Kirschner M. Microtubule assembly nucleated by isolated centrosomes. Nature. 1984 Nov 15;312(5991):232–237. doi: 10.1038/312232a0. [DOI] [PubMed] [Google Scholar]
- Moudjou M., Paintrand M., Vigues B., Bornens M. A human centrosomal protein is immunologically related to basal body-associated proteins from lower eucaryotes and is involved in the nucleation of microtubules. J Cell Biol. 1991 Oct;115(1):129–140. doi: 10.1083/jcb.115.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nigg E. A. Cellular substrates of p34(cdc2) and its companion cyclin-dependent kinases. Trends Cell Biol. 1993 Sep;3(9):296–301. doi: 10.1016/0962-8924(93)90011-o. [DOI] [PubMed] [Google Scholar]
- Oakley B. R. Gamma-tubulin: the microtubule organizer? Trends Cell Biol. 1992 Jan;2(1):1–5. doi: 10.1016/0962-8924(92)90125-7. [DOI] [PubMed] [Google Scholar]
- Ohta K., Shiina N., Okumura E., Hisanaga S., Kishimoto T., Endo S., Gotoh Y., Nishida E., Sakai H. Microtubule nucleating activity of centrosomes in cell-free extracts from Xenopus eggs: involvement of phosphorylation and accumulation of pericentriolar material. J Cell Sci. 1993 Jan;104(Pt 1):125–137. doi: 10.1242/jcs.104.1.125. [DOI] [PubMed] [Google Scholar]
- Raff J. W., Kellogg D. R., Alberts B. M. Drosophila gamma-tubulin is part of a complex containing two previously identified centrosomal MAPs. J Cell Biol. 1993 May;121(4):823–835. doi: 10.1083/jcb.121.4.823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmon E. D., Saxton W. M., Leslie R. J., Karow M. L., McIntosh J. R. Diffusion coefficient of fluorescein-labeled tubulin in the cytoplasm of embryonic cells of a sea urchin: video image analysis of fluorescence redistribution after photobleaching. J Cell Biol. 1984 Dec;99(6):2157–2164. doi: 10.1083/jcb.99.6.2157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verde F., Dogterom M., Stelzer E., Karsenti E., Leibler S. Control of microtubule dynamics and length by cyclin A- and cyclin B-dependent kinases in Xenopus egg extracts. J Cell Biol. 1992 Sep;118(5):1097–1108. doi: 10.1083/jcb.118.5.1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verde F., Labbé J. C., Dorée M., Karsenti E. Regulation of microtubule dynamics by cdc2 protein kinase in cell-free extracts of Xenopus eggs. Nature. 1990 Jan 18;343(6255):233–238. doi: 10.1038/343233a0. [DOI] [PubMed] [Google Scholar]
- Walker R. A., O'Brien E. T., Pryer N. K., Soboeiro M. F., Voter W. A., Erickson H. P., Salmon E. D. Dynamic instability of individual microtubules analyzed by video light microscopy: rate constants and transition frequencies. J Cell Biol. 1988 Oct;107(4):1437–1448. doi: 10.1083/jcb.107.4.1437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker R. A., Pryer N. K., Salmon E. D. Dilution of individual microtubules observed in real time in vitro: evidence that cap size is small and independent of elongation rate. J Cell Biol. 1991 Jul;114(1):73–81. doi: 10.1083/jcb.114.1.73. [DOI] [PMC free article] [PubMed] [Google Scholar]