Abstract
Inflammatory Bowel Diseases (IBD), including Crohn's Disease and Ulcerative Colitis, have long been associated with a genetic basis, and more recently host immune responses to microbial and environmental agents. Dinitrobenzene sulfonic acid (DNBS)-induced colitis allows one to study the pathogenesis of IBD associated environmental triggers such as stress and diet, the effects of potential therapies, and the mechanisms underlying intestinal inflammation and mucosal injury. In this paper, we investigated the effects of dietary n-3 and n-6 fatty acids on the colonic mucosal inflammatory response to DNBS-induced colitis in rats. All rats were fed identical diets with the exception of different types of fatty acids [safflower oil (SO), canola oil (CO), or fish oil (FO)] for three weeks prior to exposure to intrarectal DNBS. Control rats given intrarectal ethanol continued gaining weight over the 5 day study, whereas, DNBS-treated rats fed lipid diets all lost weight with FO and CO fed rats demonstrating significant weight loss by 48 hr and rats fed SO by 72 hr. Weight gain resumed after 72 hr post DNBS, and by 5 days post DNBS, the FO group had a higher body weight than SO or CO groups. Colonic sections collected 5 days post DNBS-treatment showed focal ulceration, crypt destruction, goblet cell depletion, and mucosal infiltration of both acute and chronic inflammatory cells that differed in severity among diet groups. The SO fed group showed the most severe damage followed by the CO, and FO fed groups that showed the mildest degree of tissue injury. Similarly, colonic myeloperoxidase (MPO) activity, a marker of neutrophil activity was significantly higher in SO followed by CO fed rats, with FO fed rats having significantly lower MPO activity. These results demonstrate the use of DNBS-induced colitis, as outlined in this protocol, to determine the impact of diet in the pathogenesis of IBD.
Keywords: Medicine, Issue 84, Chemical colitis, Inflammatory Bowel Disease, intra rectal administration, intestinal inflammation, transmural inflammation, myeloperoxidase activity
Introduction
Inflammatory bowel disease (IBD) is characterized by chronic relapsing inflammation in the gastro-intestinal (GI) tract, resulting in symptoms of diarrhea, weight loss, and abdominal pain. Ulcerative colitis (UC) and Crohn's disease (CD) are the two major forms of IBD, and can be distinguished by the location of the inflammation within the GI tract. In UC patients, the inflammation typically involves the rectum and extends contiguously up the colon for a variable extent affecting only the superficial mucosa. In contrast, CD can affect any part of the GI tract, although it predominantly affects the ileum and cecum. CD frequently manifests as transmural inflammation often associated with granulomas and leading to stricturing (fibrostenotic) and/or penetrating (fistulizing) disease. Although the etiology of IBD remains elusive, it is well accepted that IBD is multifactorial, involving interactions among the hosts immune system, genetic susceptibility and responses to environmental and microbial factors.
To date, different models of IBD have been proposed that display various clinical, histological and immune responses characteristic of UC and CD. The most commonly used models include genetically modified mice (IL-2, IL-10, SAMP/Yit), infection induced models (Citrobacter rodentium, Salmonella typhimurium), adoptive transfer models (CD45+ RB High, CD62L+ cell transfer into SCID mice) and chemically induced colitis models (Dextran Sodium Sulfate (DSS), Trinitrobenzene sulfonic acid (TNBS), and Dinitrobenzene sulfonic acid (DNBS)). Owing to their low cost and rapid onset of disease, chemical models are considered invaluable to the study of various aspects of IBD. Each of the chemical colitis models listed has advantages as well as limitations in some aspects of their clinical, immunological and histopathological relevance to IBD. DSS is one of the most common chemical methods employed to induce colitis in rodents1. Administration of 3-10% DSS (MW: 42 kDa) for 7-10 days in the drinking water of mice can induce symptoms and signs of colitis including weight loss, diarrhea with blood, colonic shortening, mucosal ulceration and neutrophil infiltration. This model is particularly useful for drug screening studies, as well as exploring the mechanisms of epithelial regeneration, the impact of innate immunity on mucosal homeostasis, and the role of inflammation in promoting intestinal dysplasia and adenocarcinoma development. There are however some drawbacks to the DSS model, including variation in the concentration of DSS needed to induce colitis in different animal facilities, as well as inconsistent water uptake by mice and thus inconsistent exposure to DSS, resulting in variation in the degree, extent and distribution of mucosal injury and ulceration in the colon. All these features lead to heterogeneity of results and limit the ability to compare results across studies from different research groups.
An alternative to the DSS model is the hapten-induced DNBS or TNBS models of colitis. This model employs rectal instillation of the mucosal sensitizing agents DNBS or TNBS, diluted in varying concentrations of ethanol. The administration of ethanol is a prerequisite to break the colonic mucosal barrier to allow penetration of DNBS or TNBS into the lamina propria. DNBS/TNBS will then haptenize the localized colonic and gut microbial proteins to become immunogenic, thereby triggering the host innate and adaptive immune responses. In general, this model is associated with severe and sometimes bloody diarrhea, weight loss and intestinal wall thickening however symptoms vary depending upon the type of rodent used, as well as the timing, dose and degree of exposure to the DNBS or TNBS used in the study. Important distinctions between rats and mice should be noted, with the benefits of lower cost for purchase and board, as well as lower body mass for decreasing per-animal costs of treatment in vivo. This should be set against the more rapid and severe course of colitis in mice, where the more fragile and responsive animals may quickly reach a humane endpoint.
The intestinal inflammation initially results from ethanol induced damage to intestinal epithelial cells, leading to increased epithelial permeability, microbial penetration into the mucosa, haptenization of host proteins, all of this resulting in infiltration of neutrophils, macrophages and Th1 T lymphocytes into the damaged mucosa. In comparison to DNBS, TNBS is considered as a hazardous chemical due to its highly oxidative properties that can pose a risk of explosion upon contact with bases such as sodium and potassium hydroxide. Therefore DNBS is currently regarded as a preferred choice of chemical over TNBS to induce colitis. In rodents, DNBS colitis is considered as one of the most convenient methods to study the following IBD associated modifiers of disease:
Depression and reactivation of colitis: It has been shown that stress, anxiety and depression in IBD patients are frequently associated with disease relapse. DNBS colitis is a suitable model to study the role of depression and its consequences on reactivation of colitis in mice. The method typically involves initial induction of colitis by DNBS, followed by the resolution of the colitis by leaving the mice for 6-8 weeks. The mice are then administered with depression causing agents such as reserpine or by olfactory bulbectomy to induce depression followed by testing them for reactivation of colitis through challenge with a subcolitic dose of DNBS2.
Stress and reactivation of colitis: Stress is another common environmental factor that has been linked to IBD pathogenesis. There is a growing body of evidence that suggests a strong association between chronic stress and the onset of symptoms of UC and CD in rodents as well as in humans and nonhuman primates3. DNBS colitis is a good model to study stress-associated reactivation of colitis in both mice and rats. The method is usually employed in a similar fashion as mentioned above for depression except in place of depression, the animals are exposed to stressors such as sonic and restraint stress4.
Neurogenic inflammation: A number of studies have described either transient or permanent alterations in the enteric nervous system (ENS) structure and function, as seen in tissue samples from animal models and from patients with IBD. DNBS is a good model to explore the effects of inflammation on the ENS5 and to study both noradrenergic and cholinergic neural pathways6.
Injury-repair mechanisms: During IBD pathogenesis, host-derived injury mediators such as reactive oxygen species (ROS), nitric oxide (NO), intercellular adhesion molecule 3 (ICAM-3), and P-selectin have all been shown to play a role in intestinal epithelial disruption. DNBS is an excellent model to study injuries caused by the up-regulation of these mediators as well as repair mechanisms that are regulated by using selective drugs or inhibitors7,8.
Transmural inflammation: In addition to the above mentioned applications of DNBS, the model can also be applied to study transmural inflammation of the gut, a classical feature found in patients with CD. Both DNBS and TNBS-induced colitis are associated with significant infiltration of lymphocytes, into the colonic mucosa making these models particularly useful to study T-cell dependent immune mechanisms.
In comparison with the DSS model, the advantages of DNBS and TNBS-induced colitis include low cost, rapid development of colitis (usually requires 1-3 days to show reproducible ulceration and inflammation) and consistent localized damage to the distal colon. However the drawbacks are a requirement for a greater level of technical expertise, optimization of DNBS/ TNBS dose, and the need for anesthesia for rectal administration.
In this methodological paper we studied the effects of varying concentrations of n-6 and n-3 polyunsaturated fatty acids on altering the colonic mucosal response to DNBS-induced colitis in rats using the vegetable oils safflower oil (SO) and canola oil (CO), and fish oil (FO). It has been shown that n-6 and n-3 fatty acids are important mediators of intestinal inflammatory disease through their role as acyl moieties of cell membrane phosopholipids9. In contrast to the proinflammatory potential of n-6 fatty acids, n-3 fatty acids at sufficiently high intake are potentially potent anti-inflammatory agents. The anti-inflammatory actions of n-3 fatty acids are mediated directly through replacement of arachidonic acid as an eicosanoid substrate, inhibiting arachidonic acid metabolism and indirectly through alteration of proinflammatory gene expression and cell signaling. The n-3 fatty acids also give rise to resolvins, a family of anti-inflammatory mediators. High intake of n-3 fatty acids is associated with a decrease in production of proinflammatory eicosanoids, cytokines, chemokines, reactive oxygen species and expression of adhesion molecules. In this study, rats were fed ad libitum diets identical in all nutrients except fatty acids, with as a percent energy from fat, 20% SO, 20% CO, or 18% fish oil plus 2% safflower oil (FO)10-11. As a percentage of the daily energy, the SO diet provided 15% linoleic acid (LA), with <0.06% α-linolenic acid (ALA) and no eicosapentaenoic acid (EPA) or docosahexaenoic acid (DHA), the CO diet had 4.2% LA and 1.9% ALA with no EPA or DHA, and the FO diet provided 1.4% EPA, 4.9% DHA, 0.32% LA, and 0.12% ALA. Three weeks after initiation of the lipid diets, the mice were administered intrarectal DNBS or 50% ethanol and sacrificed 5 days later. The inflammatory response was evaluated by assessment of weight loss, histological damage scores and tissue myeloperoxidase activity.
Protocol
Procedures involving animal subjects follow animal care guidelines outlined by the Institutional Animal Care and Use Committee (IACUC) at the University of British Columbia.
Procedures mentioned in this protocol will be described for both rats and mice, while the representative results will be presented from Sprague-Dawley rats.
1. Preparation of DNBS and Intrarectal Administration
For rectal instillation of DNBS in rats, freshly prepare 15-30 mg of DNBS/250 µl of 50% ethanol. For mice, prepare 2-6 mg of DNBS/100 µl of 50% ethanol. Note: Optimize the exact DNBS concentration to be administered at each facility. Male Sprague-Dawley rats, and C57BL/6 mice are preferred for induction of DNBS colitis, however other mouse and rat strains can be used. Mice and rats treated only with 50% ethanol can be used as control groups however given the effects of ethanol are mild or absent, one can omit the inclusion of this group for a study12.
Attach a 1 ml syringe to a 19 G needle that has a polyethylene catheter (PE-90 - rats or PE-50 - mice) fastened to its end.
Lightly anesthetize the rat or mouse resting on a heating pad (e.g. by administering 1.5% isoflurane via nose cone, animals should still have blink and swallowing reflexes and display regular respiration). Apply tear gel to its eyes to avoid the eyes drying out.
Gently apply pressure with fingertips to the posterior end of the animal to remove any stool that may be present in the distal colon.
Gently insert the catheter intrarectally, with the catheter reaching approximately 8 cm proximal to the anus for rats, or 3-4 cm for mice. Inject a small amount of the solution while inserting the catheter to lubricate, allowing easier insertion. Note: If any resistance is felt while inserting the catheter, do not continue to insert as this may lead to perforation of the intestinal wall. Remove the catheter and attempt to gently reinsert, again lubricating the colon as proceeding.
Slowly inject 250 (rats) or 100 (mice) µl of DNBS, or an equal volume or 50% ethanol for controls. After injecting the DNBS, position the animal head-down for 90 sec to avoid loss of the DNBS.
After DNBS administration, feed the animals 8% sucrose water in 0.2% saline to prevent dehydration during the first week.
Carefully observe the animals, with daily monitoring for weight loss and signs of dehydration or distress for the duration of the experiment. Note: Weight loss may rapidly approach the CCAC recommendations of maximum permissible weight loss. Since this mostly derived from dehydration, weight loss should be used as an indicator for administering lactated Ringer's solution to limit dehydration. Please be sure to consult with the facility veterinarian on health issues as appropriate.
2. Collecting Tissues and Assessing Histological Damage in DNBS Challenged Mice
At the desired time point after challenge with DNBS, euthanize animals by overdosing with isoflurane in an anesthetic chamber supplemented with sufficient oxygen. Observe the animal until no rising or falling of the chest is seen and there is no palpable heartbeat. Confirm the animal has been correctly euthanized by absence of a response to toe pinch or touching of the cornea, followed by cervical dislocation.
Open the abdominal cavity and remove the entire large intestine. Open the intestine longitudinally; macroscopic mucosal ulcers should be observed approximately 5-6 cm proximal from the distal end of the colon in rats (2-3 cm in mice). Note: After removing the large intestine, assess macroscopic damage scores if desired. Collect segments of colon from corresponding anatomical location from control animals. Note: The individuals scoring the tissue for macroscopic damage scores should be blinded to the identity of the treatment groups. Score tissue sections using the following or similar criteria: severity and extent of ulceration (0-10), summed with scores for absence or presence of diarrhea (0 or 1), adhesions (0, 1, or 2) and maximal colonic wall thickness in millimeters11.
Collect 0.5 cm sections proximal to the site of maximal macroscopic damage. Snap freeze one 0.5 cm section in liquid nitrogen to later assess myeloperoxidase (MPO) activity (a measure of neutrophil infiltration), and place the other section in 10% neutral buffered formalin for histological analysis. Note: Snap frozen tissues for myeloperoxidase assay can be stored at -80 °C for up to 1 month.
Allow the tissues collected for histological analysis in step 2.3 to fix in formalin overnight, and then wash with 70% ethanol. Embed tissues in paraffin and cut to desired thickness (3-5 µm), stain with hemotoxylin and eosin (HE) for histological scoring.
Have two observers score at least three tissue sections from each animal under a light microscope to determine the histological damage scores for each group. Note: The individuals scoring the tissue for histological damage scores should be blinded to the identity of the treatment groups. Score tissue sections using the following or similar criteria: extent of inflammatory cell infiltrate (0 = absent, 3 = transmural), loss of mucosal architecture (0 = absent, 3 = severe), presence or absence of crypt abscesses (0-1), and goblet cell depletion (0-3). Determine histological damage score as the sum of these scores11.
3. Performing Myeloperoxidase (MPO) Assay on DNBS Challenged Intestinal Tissue
Assay MPO activity in the 0.5 cm colonic segments obtained in step 2.3. Homogenize tissues in MPO homogenization buffer (0.5% hexadecyltrimethyl-ammonium bromide in 50 mM potassium phosphate buffer (pH 6.0)) to give a 50 mg colon segment/ml of homogenization buffer suspension.
Aliquot the homogenized solution into 1 ml portions and centrifuge at 4 °C for 2 min at 10,000 x g.
Mix 100 µl of each aliquot with 2.9 ml of 50 mM phosphate buffer (pH 6.0) containing 0.167 mg/ml of o-dianisidinedihydrochloride and 0.0005% hydrogen peroxide in a cuvette.
Measure the rate of absorbance using a spectrophotometer at 460 nm. Note: MPO activity is defined as the quantity of enzyme degrading 1 µmol/min of H2O2 to H2O at room temperature and is expressed in units per mg of tissue.
Representative Results
Challenge of rats (Sprague-Dawley) or mice (C57BL/6) with DNBS typically results in transient weight loss and diarrhea, sometimes with blood present in the stool. However, factors such as genetics, diet and the intestinal microbiota can modify susceptibility to DNBS induced colitis. Therefore, animals included in the experiments should be from the same institution, either derived locally or ordered in from one animal supplier. All animals should be carefully monitored for the degree of weight loss and symptoms of distress suffered during colitis.
The results presented in Figures 1-4 represent an experiment in which the effects of three dietary oils: safflower oil, canola oil, and fish oil on the resulting severity of DNBS colitis were assessed. Statistical differences among groups were determined using ANOVA followed by post hoc Tukey's test, using a data analysis software program. A P value of <0.05 was considered significant.
Following intrarectal challenge with DNBS (20 mg DNBS/250 μl of 50% ethanol), significant weight loss was observed by 48-hr in rats supplemented with FO and CO and by 72 hr in rats supplemented with SO (P<0.05). Maximum weight loss is seen at 48 hr in rats supplemented with FO and by 72 hr in rats supplemented with CO and SO. Weight gain resumed after 72 hr, and by 5 days post DNBS, FO and SO groups show body weights similar to baseline values. Control rats supplemented with the diets and challenged with intrarectal ethanol continue to gain weight over the 5 day study period.
The intestinal damage caused by DNBS can be widespread, however the area of maximal damage is generally localized to the distal 6 cm of the colon in rats, and the distal 3 cm in mice. Macroscopically this can be observed as colonic wall thickening, mucosal ulceration, and occasional adhesion of the distal colon to adjacent organs, such as the bladder. On closer histological assessment, extensive epithelial injury, in some areas leading to the formation of ulcers, immune cell infiltration (predominantly neutrophils and lymphocytes), cryptitis, mucus depletion, and edema is observed. Although these histological signs of inflammation and tissue damage are evident in all three dietary oil supplemented groups upon challenge with DNBS, the most severe damage is seen in the safflower oil group, followed by the canola oil group, whereas rats in the fish oil group demonstrate minimal epithelial injury and a limited infiltration of inflammatory cells into the mucosa (Figure 2).
Differences in histological damage can be quantified to allow for a better comparison between groups. Criteria used to assess histological damage included the extent of inflammatory cell infiltrate, loss of mucosal architecture, goblet cell depletion, and the presence of crypt abscesses. Shown in Figure 3 are the histological damage scores for the three dietary oil groups during DNBS colitis. As is seen in the H&E stained sections in Figure 2, the safflower oil supplemented group has the greatest extent of damage, followed by the canola oil group, while the fish oil group show the least amount of damage.
The extent of inflammatory cell infiltrate, particularly of neutrophils, can be further examined by performing a myeloperoxidase (MPO) activity assay. MPO is considered a marker of inflammation, as it is an enzyme found in the granules of neutrophil granulocytes and other cells of myeloid lineage. Assessment of MPO activity in the rat colons at day 5 post-DNBS exposure reveals an increase in activity in all three groups (Figure 4). The greatest MPO activity is observed in the safflower oil group, followed by the canola oil group, while the lowest MPO activity is seen in the fish oil group, corresponding to the histological damage scores assessed in Figure 3.
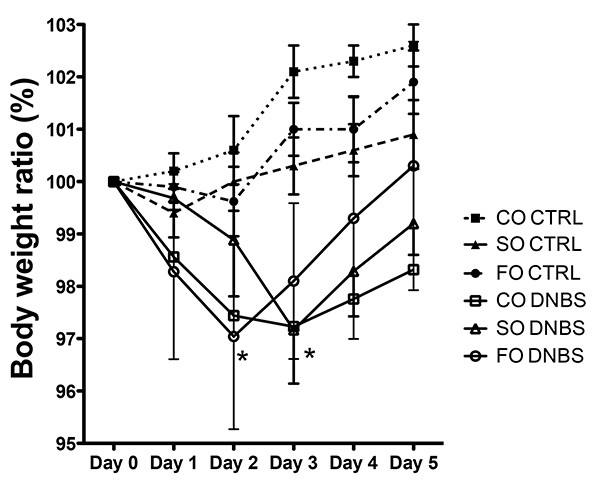 Figure 1. Changes in body weight (% of baseline) following intrarectal administration of 50% ethanol (control) or dinitrobenzene sulfonic acid (DNBS) in adult female Sprague-Dawley rats (250-500 g) fed SO, CO, or FO. The rats are then monitored for loss of body weight from day 1 until day 5 post treatment. The percentage loss of body weight is plotted against days post DNBS treatment. Values are means ± SEM, n=5/ group. When compared to non-DNBS treated counterparts, rats fed with CO and FO had a significant reduction in their initial body weights as early as day 1 post DNBS treatment whereas on day 3 all the three groups of rats had significant weight loss relative to baseline weight (P <0.01). All colitis groups demonstrate weight gain after 72 hr, with weight returning to baseline in the FO and SO fed groups. Rats in the ethanol control groups demonstrate weight gain over the 5 day study period. Click here to view larger image.
Figure 1. Changes in body weight (% of baseline) following intrarectal administration of 50% ethanol (control) or dinitrobenzene sulfonic acid (DNBS) in adult female Sprague-Dawley rats (250-500 g) fed SO, CO, or FO. The rats are then monitored for loss of body weight from day 1 until day 5 post treatment. The percentage loss of body weight is plotted against days post DNBS treatment. Values are means ± SEM, n=5/ group. When compared to non-DNBS treated counterparts, rats fed with CO and FO had a significant reduction in their initial body weights as early as day 1 post DNBS treatment whereas on day 3 all the three groups of rats had significant weight loss relative to baseline weight (P <0.01). All colitis groups demonstrate weight gain after 72 hr, with weight returning to baseline in the FO and SO fed groups. Rats in the ethanol control groups demonstrate weight gain over the 5 day study period. Click here to view larger image.
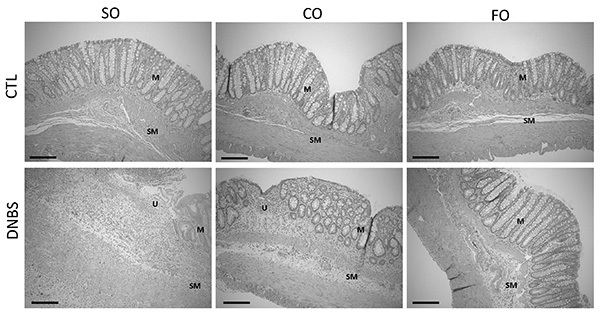 Figure 2. Histological appearance of representative H&E stained colon tissue sections 5 days after intrarectal administration of 50% ethanol (CTL) or DNBS to adult female rats fed SO, CO, or FO. The tissue is collected in 10% formalin and allowed to fix for a minimum of 24 hr followed by tissue sectioning and HE staining. The tissue sections from rats fed SO and CO demonstrate significant disruption of epithelial integrity with varying degree of epithelial injury, crypt drop out, ulcer formation and transmural immune cell infiltration, with more marked changes seen with SO. In CO fed mice, evidence of epithelial regeneration and restitution is seen. In contrast, rats fed FO maintained epithelial integrity and had limited infiltration of inflammatory cells confined to the lamina propria. (M-mucosa, SM-sub-mucosa and U-ulceration; original magnification 100X; scale bar: 100 µm). Click here to view larger image.
Figure 2. Histological appearance of representative H&E stained colon tissue sections 5 days after intrarectal administration of 50% ethanol (CTL) or DNBS to adult female rats fed SO, CO, or FO. The tissue is collected in 10% formalin and allowed to fix for a minimum of 24 hr followed by tissue sectioning and HE staining. The tissue sections from rats fed SO and CO demonstrate significant disruption of epithelial integrity with varying degree of epithelial injury, crypt drop out, ulcer formation and transmural immune cell infiltration, with more marked changes seen with SO. In CO fed mice, evidence of epithelial regeneration and restitution is seen. In contrast, rats fed FO maintained epithelial integrity and had limited infiltration of inflammatory cells confined to the lamina propria. (M-mucosa, SM-sub-mucosa and U-ulceration; original magnification 100X; scale bar: 100 µm). Click here to view larger image.
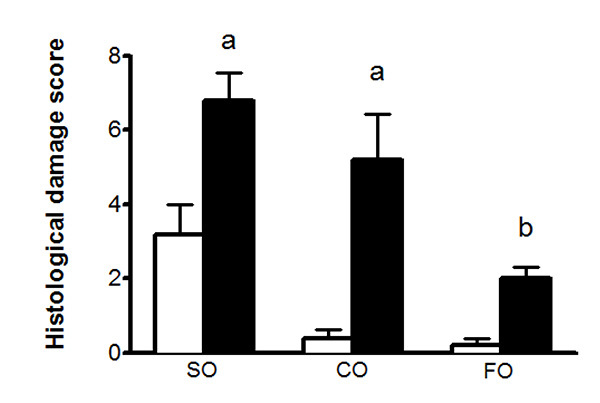 Figure 3. Histological damage score of colonic tissues 5 days after intrarectal administration of 50% ethanol (clear bar) or DNBS (solid bar) to adult female rats fed SO, CO, or FO. Tissue sections are scored by an experienced investigator, blinded to the treatments, using a set criteria based on histological damage. The criteria used for this study includes: inflammatory cell infiltration (0=absent, 3=transmural), loss of mucosal integrity (0=absent, 3= severe), crypt abscess formation (0=absent, 1=present) goblet cell depletion (0=absent, 3=high). The histological damage score is determined as a sum of scores obtained by scoring a minimum of three tissue sections from each animal. The results clearly demonstrate a reduced histological damage in FO fed rats when compared to SO and CO fed rats. Values are means ± SEM, n=5/ group. Compared with respective ethanol controls, DNBS administration is associated with a significant increase in damage score (P<0.05); colitis groups with different symbols are significantly different (P<0.01). Click here to view larger image.
Figure 3. Histological damage score of colonic tissues 5 days after intrarectal administration of 50% ethanol (clear bar) or DNBS (solid bar) to adult female rats fed SO, CO, or FO. Tissue sections are scored by an experienced investigator, blinded to the treatments, using a set criteria based on histological damage. The criteria used for this study includes: inflammatory cell infiltration (0=absent, 3=transmural), loss of mucosal integrity (0=absent, 3= severe), crypt abscess formation (0=absent, 1=present) goblet cell depletion (0=absent, 3=high). The histological damage score is determined as a sum of scores obtained by scoring a minimum of three tissue sections from each animal. The results clearly demonstrate a reduced histological damage in FO fed rats when compared to SO and CO fed rats. Values are means ± SEM, n=5/ group. Compared with respective ethanol controls, DNBS administration is associated with a significant increase in damage score (P<0.05); colitis groups with different symbols are significantly different (P<0.01). Click here to view larger image.
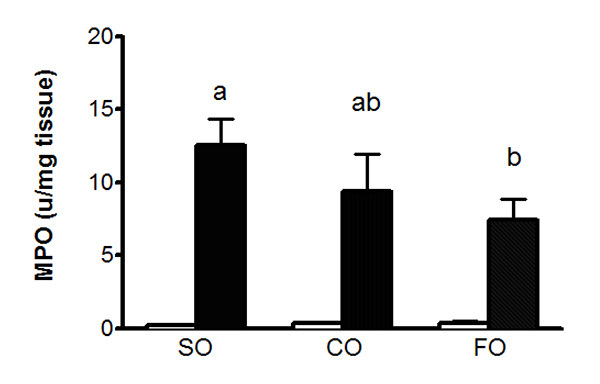 Figure 4. MPO activity in colonic tissues 5 days after intrarectal administration of 50% ethanol (clear bar) or DNBS (solid bar) to adult female rats fed SO, CO, or FO. Inflamed colonic tissues are snap frozen in liquid nitrogen and assayed for myeloperoxidase activity, as an index of granulocyte infiltration. Colitic rats fed FO show reduced MPO activity compared to SO and CO. Values are means ± SEM, n= 5/ group. Compared with respective ethanol controls, DNBS administration is associated with a significant increase in MPO activity (P<0.001); colitis groups with different symbols are significantly different (P<0.05). Click here to view larger image.
Figure 4. MPO activity in colonic tissues 5 days after intrarectal administration of 50% ethanol (clear bar) or DNBS (solid bar) to adult female rats fed SO, CO, or FO. Inflamed colonic tissues are snap frozen in liquid nitrogen and assayed for myeloperoxidase activity, as an index of granulocyte infiltration. Colitic rats fed FO show reduced MPO activity compared to SO and CO. Values are means ± SEM, n= 5/ group. Compared with respective ethanol controls, DNBS administration is associated with a significant increase in MPO activity (P<0.001); colitis groups with different symbols are significantly different (P<0.05). Click here to view larger image.
Discussion
The DNBS model described in this protocol is a valuable, inexpensive, and reproducible colitis model that can be utilized to study various aspects of IBD. When administered intrarectally to rats or mice, DNBS induces a substantial degree of inflammation and tissue injury in the colon, resembling human Crohn's disease in terms of its various histological features including transmural infiltration of polymorphonuclear cells and predominant NF-κB-dependent Th1 activation.
This model has been used to study various factors thought to impact on IBD such as stress, depression, and diet, as well as enteric nervous system involvement, injury-repair mechanisms, transmural inflammation, and preclinical therapies. The DNBS model is also a suitable alternative to TNBS-induced inflammation in mice and rats and offers a less expensive alternative to other commonly employed chemical models of colitis such as DSS. The steps outlined in this protocol will allow one to successfully execute this model.
Consistent with other experimental models of colitis13-16, the current study reinforces the variable effects of dietary fat on intestinal mucosal immune responses where high intake of EPA and DHA consumed prior to and following DNBS exposure attenuates, while high intake of ARA with minimal n-3 fatty acids exacerbates the colitis. The beneficial effects of dietary n-3 fatty acids observed in this study are due in part to n-3 fatty acids effectively lower tissue phospholipid ARA both by inhibiting synthesis of ARA from LA and by replacing ARA with EPA and DHA. This change shifts the fatty acid substrate available for release and subsequent eicosanoid synthesis toward a less proinflammatory environment. In contrast, the high intake of n-6 fatty acids is associated with a high colonic ARA with subsequent synthesis of proinflammatory mediators that exacerbate mucosal immune response. In keeping with our recent study in Citrobacter rodentium induced colitis, rats fed canola oil with less ARA than safflower oil, but with n-3 ALA and no EPA or DHA had an intermediate inflammatory response16.
There are several critical steps that can impact on the successful implementation of the DNBS model. Different studies have used varying concentrations of DNBS ranging from 3-6 mg/mouse or 15-30 mg/rat to establish colitis. Therefore, it is important for individual investigators to optimize the dosage of DNBS for their studies, since optimal doses may vary from one animal facility to the other. It is also important to note that the concentration of DNBS will differ according to the type and species of animal that an investigator is using (Ex: certain strains may be more susceptible and others more resistant to DNBS). Care should be taken to ensure that the concentration of DNBS is not too high as this can cause the rapid death of animals resulting from bowel perforation and sepsis. Another critical step to using this model is developing the appropriate method for intrarectal catheter insertion into the distal colon of the animal. The presence of stool in the colon may prevent adequate insertion of catheter during instillation. To avoid this, gently apply pressure at the posterior end of the anus of lightly anaesthetized mice to remove any stool in the distal colon/ rectum. It is recommended to insert the catheter approximately 3-4 cm from anus, although it may be extended to a maximum of 6-8 cm in rats until the splenic flexure (a sharp bend between transverse and descending colon)7. It is important to note that during the instillation procedure, lubricating the catheter by injecting DNBS/ethanol in small volumes will allow easier insertion of the catheter. In order to avoid the retrograde reflux of DNBS out of the anus, it is critical to hold the animals in a head-down position for 90 sec or alternatively, the mice can be kept for 15 min in the Trendelenburg position (a supine position where the feet are placed higher than the head by 15-30°).
Tissues can be collected at selected time points depending on the interests of the investigator and whether the study is focusing on acute or chronic inflammation. In addition, the tissues can also be used to study the intestinal ENS and innate and adaptive immune system. However, it is essential to ensure that tissues are properly sampled. It is recommended to collect the tissues in an ample volume of 10% formalin in a 5 ml vials as opposed to smaller volume vials to ensure proper fixation. Appropriate fixation of samples is a key to study both the histopathological damage and visualization of target molecules in tissues by H&E and immunostaining, respectively. Histological scoring of tissues should be performed by experienced individuals who are blinded to the experimental conditions, using fixed criteria set by the individual investigator11,17. Myeloperoxidase (MPO) serves as a good marker of inflammation, tissue injury and neutrophil infiltration into gastrointestinal tissues11. To perform the MPO assays, it is suggested to snap freeze the tissues in liquid nitrogen and on the day of analysis, it is recommended to work with freshly prepared buffers and solutions to obtain the best and most accurate results. This model can also be used to study inflammatory cells that infiltrate the intestine, the mechanisms involved in chemotaxis and transmigration of inflammatory cells and the various pathways involved in the activation and release of other proinflammatory mediators involved in tissue injury as well as mediators involved in modulating the inflammatory response and in tissue repair.
The clinical disadvantages currently seen with using immunosuppressive drugs or targeting one proinflammatory cytokine such as TNF-α should encourage investigators to develop better therapeutic strategies to manage patients with IBD. Models, such as the DNBS model, allows investigators to increase our understanding of the pathogenesis of IBD, potentially identifying new therapeutic targets and developing/improving strategies to control inflammation through the use of novel inflammatory pathways and novel therapies.
Disclosures
The authors declare that they have no competing financial interests.
Acknowledgments
This work was supported by operating grants to KJ from the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Crohn's and Colitis Foundation of Canada (CCFC). VM is supported by a CFRI Fellowship and GB a graduate studentship from the Canadian Institutes for Health Research (CIHR). BAV is a Children with Intestinal and Liver Disorders Foundation (CHILD) Chair of Pediatric IBD Research and the Canada Research Chair in Pediatric Gastroenterology and KJ is a Senior Clinician Scientist supported by CHILD and the Child and Family Research Institute (CFRI) Clinician Scientists Award Program, University of British Columbia.
References
- Perše M, Cerar A. Biotechnol J. Biomed., editor. Dextran sodium sulphate colitis mouse model: traps and tricks. 2012. [DOI] [PMC free article] [PubMed]
- Ghia JE, Blennerhassett P, Deng Y, Verdu E, Khan W, Collins S. Reactivation of inflammatory bowel disease in a mouse model of depression. Gastroenterology. 2009;136(7):2280–2288. doi: 10.1053/j.gastro.2009.02.069. [DOI] [PubMed] [Google Scholar]
- Reber S. Stress and animal models of inflammatory bowel disease-an update on the role of the hypothalamo-pituitary-adrenal axis. Psychoneuroendocrinology. 2012;37(1):1–19. doi: 10.1016/j.psyneuen.2011.05.014. [DOI] [PubMed] [Google Scholar]
- Qiu B, Vallance B, Blennerhassett P, Collins S. The role of CD4+ lymphocytes in the susceptibility of mice to stress-induced reactivation of experimental colitis. Nat. Med. 1999;5(10):1178–1182. doi: 10.1038/13503. [DOI] [PubMed] [Google Scholar]
- Boyer L, et al. Myenteric plexus injury and apoptosis in experimental colitis. Auto. Neurosci. Basic Clin. 2005;117(1):41–53. doi: 10.1016/j.autneu.2004.10.006. [DOI] [PubMed] [Google Scholar]
- Saunders P, et al. Noradrenergic and cholinergic neural pathways mediate stress-induced reactivation of colitis in the rat. Auto. Neurosci. Basic Clin. 2006;124(1-2):56–68. doi: 10.1016/j.autneu.2005.12.002. [DOI] [PubMed] [Google Scholar]
- Cuzzocrea S, et al. Calpain inhibitor I reduces colon injury caused by dinitrobenzene sulphonic acid in the rat. Gut. 2001;48(4):478–488. doi: 10.1136/gut.48.4.478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuzzocrea S, et al. Melatonin reduces dinitrobenzene sulfonic acid-induced colitis. J. Pineal Res. 2001;30(1):1–12. doi: 10.1034/j.1600-079x.2001.300101.x. [DOI] [PubMed] [Google Scholar]
- Innis S, Jacobson K. Dietary lipids in early development and intestinal inflammatory disease. Nutr. Rev. 2007;65:S188–S193. doi: 10.1111/j.1753-4887.2007.tb00361.x. [DOI] [PubMed] [Google Scholar]
- Innis SM, de La Presa Owens S. Dietary fatty acid composition in pregnancy alters neurite membrane fatty acids and dopamine in newborn rat brain. J. Nutr. 2001;131(1):118–122. doi: 10.1093/jn/131.1.118. [DOI] [PubMed] [Google Scholar]
- Jacobson K. Mundra H.,Innis S.M. Intestinal responsiveness to experimental colitis in young rats is altered by maternal diet. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;289:13–20. doi: 10.1152/ajpgi.00459.2004. [DOI] [PubMed] [Google Scholar]
- Sanovic S, Lamb DP, Blennerhassett MG. Damage to the enteric nervous system in experimentalcolitis. Am. J. Pathol. 1999;155(4):1051–107. doi: 10.1016/S0002-9440(10)65207-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vilaseca J, Salas A, Guarner F, Rodriguez R, Martinez M, Malagelada JR. Dietary fish oil reduces progression of chronic inflammatory lesions in a rat model of granulomatous colitis. Gut. 1990;31:539–544. doi: 10.1136/gut.31.5.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andoh A, Tsujikawa T, Ishizuka I, Araki Y, Sasaki M, Koyama S, Fujiyama Y, editors. N-3 fatty acid-rich diet prevents early response of interleukin-6 elevation in trinitrobenzene sulfonic acid-induced enteritis. Int. J. Mol. Med. 2003;12:721–725. [PubMed] [Google Scholar]
- Barros KV, Xavier RA, Abreu GG, Martinez CA, Ribeiro ML, Gambero A, Carvalho PO, Nascimento CM, Silveira VL. Soybean and fish oil mixture increases IL-10, protects against DNA damage and decreases colonic inflammation in rats with dextran sulfate sodium (DSS) colitis. Lipids Health Dis. 2010;9 doi: 10.1186/1476-511X-9-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hekmatdoost A, Wu X, Morampudi V, Innis SM, Jacobson K. Dietary oils modify the host immune response and colonic tissue damage following Citrobacter rodentium infection in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2013;304(10):917–928. doi: 10.1152/ajpgi.00292.2012. [DOI] [PubMed] [Google Scholar]
- Innis SM, Dai C, Wu X, Buchan AM, Jacobson K. Perinatal lipid nutrition alters early intestinal development and programs the response to experimental colitis in young adult rats. Am. J. Physiol. Gastrointest. Liver Physiol. 1152;299(6):1376–1385. doi: 10.1152/ajpgi.00258.2010. [DOI] [PubMed] [Google Scholar]


