Abstract
Homeoprotein (HP) transcription factors were originally identified for their embryonic cell-autonomous developmental functions. In this review, we discuss their postnatal and adult physiological functions based on the study of Otx2, Engrailed-1 and Engrailed-2 (collectively Engrailed). For Engrailed, we discuss its function in the cell-autonomous regulation of ventral midbrain dopaminergic neuron survival and physiology and in the non-cell-autonomous maintenance of axons. For Otx2, we describe how the protein is expressed in the choroid plexus and transported into cortical parvalbumin cells where it regulates plasticity in the visual cortex. These two examples illustrate how the understanding of HP postnatal and adult functions, including signalling functions, may lead to the identification of disease-associated genetic pathways and to the development of original therapeutic strategies.
Keywords: homeoprotein, signalling, Parkinson's, plasticity, synapse
1. Introduction
Homeoprotein (HP) transcription factors are the products of a family of developmental genes with multiple functions, including the definition of body axis, lineage decision and cell/growth cone migration and guidance [1–3]. They are characterized by their highly conserved 60 amino acid-long DNA-binding domain, the homeodomain (HD). In addition to mediating DNA binding, the HD encompasses two short sequences allowing HP secretion and internalization to impart non-cell-autonomous functions [4]. Such functions have been studied in vivo for Engrailed-1 (En1) and Engrailed-2 (En2), collectively Engrailed or En1/2, for Otx2 and for Pax6 [5]. However, based on sequence alignments, it is likely that non-cell-autonomous functions are a property of many HPs, and a dozen HPs have been verified in vitro.
It is important to consider that HP function can extend beyond transcription regulation. For example, HPs can interact with mRNA and regulate translation [6]. HPs typically have numerous binding domains and regulatory motifs outside the HD that allow interaction with not only other transcription factors but also translation factors, such as eIF4E [7], and many other molecules that are part of signalling pathways, such as kinases [8] or even complex sugars [9]. A recent review on Hox proteins attests to the reach of HP regulation, as they are implicated in DNA transcription, repair, initiation and replication, mRNA translation and signal transduction [10]. As HPs transfer into cells through non-conventional mechanisms thereby gaining direct access to the cytoplasm and nucleus [4], the non-cell-autonomous activity of HPs are likely to impact directly many aspects of cell function, just as cell-autonomous expression does. In the context of development, HPs are found throughout plant and animal kingdoms and we have proposed that HPs are evolutionarily early morphogens that by definition can signal non-cell-autonomously and provide positional information [11]. Given that HPs can have such diverse targets, both cell autonomous and non-cell autonomous, the study of HP activity must clearly encompass many aspects of cell biology.
Before describing the present knowledge on the postnatal and adult functions of HPs, it is useful to recall what is known concerning their non-cell-autonomous embryonic functions (leaving aside the huge body of literature on their developmental cell-autonomous activity). A very early function is the definition of the extent of the eye anlagen by Pax6 intercellular transfer. Blocking the passage of Pax6 in vivo by local expression of a single-chain antibody (aPax6-scfv) or by injection of a monoclonal anti-Pax6 antibody at the midblastula stage prevents the extension of the eye anlagen in the zebrafish and leads to defects in eye development [12]. Another recently discovered non-cell-autonomous embryonic function of Pax6 is to enhance oligodendrocyte precursor cell migration in the chick neural tube [13].
An embryonic non-cell-autonomous function of En1/2 is to guide retinal ganglion cell (RGC) axons along the anterior–posterior (A–P) axis of the optic tectum in the chick and frog. En1/2 shows graded expression along the A–P axis and the in vivo expression of a single-chain antibody (aEn1/2-scfv) results in the invasion of the posterior tectum by temporal growth cones [14]. The effect of En1/2 on axon guidance in vitro is to attract and repel nasal and temporal growth cones, respectively [15]. This activity is entirely due to the local regulation of mRNA translation through an interaction with the mTOR pathway [15]. Interestingly, this guidance effect of En1/2 also requires Ephrin/Eph and Adenosine signalling [16]. This cooperation between Engrailed signalling and classical signalling pathways was also demonstrated in the fly wing disc where Engrailed interacts with Decapentaplegic (DPP) to induce the formation of the anterior cross vein [17]. An important aspect of En1/2 guidance activity is that many of the translated mRNAs encode mitochondrial proteins, including Ndufs1 and Ndufs3, two key subunits of Complex I of the electron chain transfer [18]. Consequently, En1/2 internalization results in rapid bursts of ATP synthesis and secretion [16].
The third HP for which non-cell-autonomous functions have been identified is Otx2 [5]. The role of Otx2 transfer in the regulation of cerebral cortex plasticity will be developed in the main core of this review which from now on will be focused on postnatal and adult cell-autonomous and non-cell-autonomous activities of En1/2 and Otx2.
2. Engrailed1/2 influences mitochondrial function
As already mentioned, HPs are not only expressed during development but also in the adult. En1/2 is expressed in the adult tectum/superior colliculus and in the dopaminergic nuclei of the ventral midbrain [19,20], while En2 is additionally expressed by granule cells in the cerebellum [2,21]. Functions have been studied for En1 in the visual system (frog tectum) and for En1/2 in the ventral midbrain. Holt and co-workers [22], in a search for mRNAs present in the retinal growth cones and translated locally in response to guidance cues, observed that En1 is the most active morphogen when it comes to regulating translation and subsequently used En1 to identify newly synthesized proteins. In a series of elegant experiments, they showed that laminB2, normally a nuclear type V intermediate filament, is locally translated following En1 internalization in retinal axons. Very interestingly, they showed that laminB2 is necessary to maintain normal mitochondrial morphology and membrane potential in the mature free-swimming tadpole. Moreover, knock-out experiments led to axon destabilization in the tadpole, a phenotype that was rescued in laminB2 gain-of-function experiments. The activity of laminB2 is local and not nuclear as it persisted when the nuclear localization sequence was deleted from laminB2. Although the latter data do not formally show it, it is tempting to speculate that the retrograde transsynaptic transfer of En1 regulates synapse viability.
The ability to regulate mitochondrial activity is very important as many neurological and psychiatric diseases have a strong metabolic facet. In this context, adult En1/2 expression in the ventral midbrain dopaminergic (mDA) nuclei, the Substantia Nigra pars compacta (SNpc) and ventral tegmental area (VTA), which also expresses Otx2, is of great interest. In 2007, it was shown that the loss of one En1 allele (En1−/+ mouse) leads to the progressive death of mDA neurons [11]. The neurons start dying at six weeks after birth and death continues progressively until, after 1 year, 40% and 20% mDA neurons are lost in the SNpc and VTA, respectively. Recombinant En1 and En2 are internalized by live cells allowing functional access to cytoplasm and nucleus. When infused in the midbrain of En1−/+ mice, they provoke a complete arrest of cell death [11]. Thus, En1/2 can be considered a therapeutic protein and this experiment importantly suggests that non-cell-autonomous and cell-autonomous properties of HPs can be shared as the exogenously added protein can substitute for the endogenous one.
En1 is expressed by mDA neurons and its activity is thus considered primarily cell autonomous. However, it was important to test the protective capacity of exogenous En1/2 in bona fide mouse models of Parkinson's disease (PD). In three distinct models (6-OHDA, MPTP and A30P α-synuclein mutant), infused En1 protects mDA neurons from cell death [18]. En1 protein infusion also increases the amount of dopamine (DA) per neuron (DA was measured in the striatum) while all effects, including amphetamine-induced rotational behaviour of unilaterally infused mice, were abolished by the specific inhibition of Ndufs1 mRNA translation (using an appropriate siRNA), thus implicating mitochondrial metabolic pathways [18]. Very curiously, the effect of En1 lasted over three months (J Fuchs 2010, unpublished results) suggesting an epigenetic effect of the protein. Taken together, the rescue, the DA content upregulation and the long-lasting effect suggest that En1/2 might be worth considering as a disease-modifying agent in PD. A scheme illustrating the effects of En1/2 on neuron physiology is proposed in figure 1.
Figure 1.
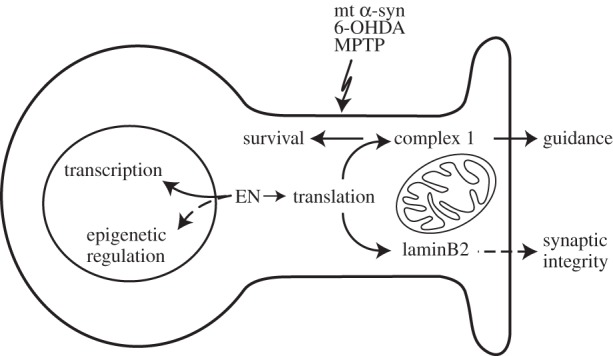
Effect of En1/2 on neuron physiology and mDA neuron pathophysiology. In the nucleus, En1/2 acts as transcription factor due to its DNA-binding HD. Preliminary experiments suggest that En1/2 might also induce epigenetic modifications (as indicated by the observation of long-lasting behavioural effects after a short-time infusion of the protein). In the cytoplasm, En1/2 stimulates the translation of nuclear-encoded subunits of complex I of the mitochondrial respiratory chain leading to two effects: (i) the guidance of RGC axons in the optic tectum during development and (ii) the increased survival of adult mDA neurons after a challenge with PD modelling agents (6-OHDA, MPTP and α-synuclein-A30P). During development and in the adult, En1/2 also induces the translation of laminB2 in RGC axons, which is essential for normal mitochondrial physiology, axon maintenance and potentially synapse integrity.
3. More on gene dosage and therapeutic proteins
The latter studies on the En1 heterozygote phenotype and rescue raise the issue of the HP dosage in the adult and of their use as therapeutic proteins. These aspects were investigated with Otx2 protein. By using a mouse line expressing only one allele of Otx2 (Otx2−/+) and another mouse line expressing a hypomorph of Otx2 (knock-in mutation of R36A/K37A, termed AA), Bernard et al. [23] studied the adult eye phenotypes of wild-type, Otx2AA/+, Otx2−/+ and Otx2AA/AA mice (null mutants do not develop the head and Otx2AA/– mice do not survive to adulthood). In the adult, all of these mutants present morphological and functional abnormalities in the eye, including impaired vision. However, the more severe the mutation (i.e. lower total Otx2 activity), the sooner the defect appeared in the adult. This study demonstrated the importance of gene dosage in the development of age-dependent pathologies.
Still in the context of Otx2 and eye pathology, a mouse model of glaucoma was developed based on the injection of N-methyl-d-aspartate in the eyecup leading to RGC death. The injection of low amounts of Otx2 (between 3 and 30 ng of the protein) is sufficient to prevent RGC death and to maintain normal visual acuity in the optomoter test [24]. A single injection induces long-lasting Otx2 effects and parallels the long-lasting behavioural effect of En1/2 after internalization into mDA neurons suggesting that these two HPs can be considered as therapeutic proteins, at least in mouse models of two human diseases.
4. Non-cell-autonomous Otx2 regulates visual cortex plasticity
In the course of postnatal development, the cortex goes through critical periods (CPs) of plasticity that allow environmental influence during a vulnerable time window. There are several CPs for different senses that differ in time and duration (opening and closure times). Since the pioneering studies by Wiesel & Hubel [25], the window of plasticity for the binocular V1 cortical area has provided a paradigm that still dominates the scientific literature. Before CP onset at postnatal day 20 (P20) or after its closure at P40 (in the mouse), blinding an eye temporarily does not decrease its visual acuity, thus binocular vision is maintained. In contrast, the same short and reversible blinding during the CP has irreversible consequences leading to a unilateral loss of visual acuity and to amblyopia [26].
CP onset corresponds to the maturation of parvalbumin (PV)-expressing fast spiking GABAergic interneurons (PV cells) present in layers 3 and 4 of the visual cortex. Among several markers of PV cell maturation is the assembly of the sugar-rich perineuronal nets (PNNs) that surround mature PV cells and can be specifically decorated by certain lectins [27,28]. In a collaborative study with the group of Takao Hensch, it was observed that Otx2 content increases in PV cells in parallel with their maturation [29]. Interestingly, this content does not result from local Otx2 expression but from its import from external sources, such as the choroid plexus or, possibly, the retina [29,30].
Eye opening is rapidly followed by an initial assembly of the PNNs on PV cells through a mechanism not yet elucidated but certainly involving neural activity and possibly a passage of Otx2 from eye to brain [29]. The glycosaminoglycans (GAGs) present in the PNNs serve as binding sites for Otx2 as demonstrated by genetic studies [31] and by the identification of a short Otx2 sequence (RKQRRERTTFTRAQL) that specifically recognizes the GAGs expressed by PV cells [9]. Otx2 accumulation in PV cells permits the opening (first concentration threshold) and closing (second threshold) of a CP for binocular vision. This scheme (a temporal version of the French Flag Problem [32]) has the consequence that Otx2 import is necessary throughout adulthood to maintain the CP for binocular vision in a closed state (figure 2a). In a search for sources of adult Otx2, the choroid plexus, a site of high Otx2 expression at all developmental stages and throughout adulthood, was identified and found to release Otx2 into the ventricles of the adult brain [30]. Indeed, recombining Otx2 in the adult choroid plexus reduces Otx2 content in PV cells [30].
Figure 2.
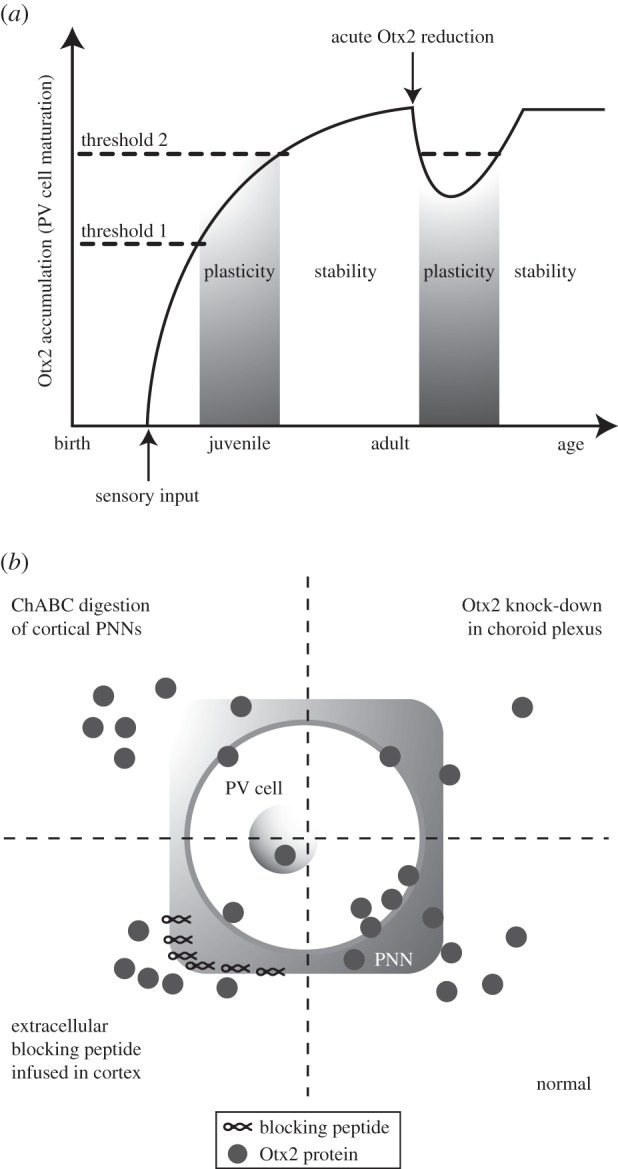
Otx2 HP regulates visual cortex plasticity. (a) A temporal French Flag models Otx2 function in PV cells of the visual cortex. Otx2 accumulates in PV cells after eye opening in the mouse and opens a CP of heightened plasticity once a first concentration threshold is reached. Continued accumulation leads to a second threshold after which the CP closes. Otx2 import throughout adulthood maintains the cortical network in a stabilized non-plastic state. (b) Adult therapies requiring cortical plasticity may be achieved by reducing Otx2 accumulation in PV cells to levels below the second threshold by using one of several methods. Normally, extracellular Otx2 is specifically captured by the GAG-rich PNN that surrounds PV cells and is then internalized. Specific accumulation of Otx2 can be reduced through enzymatic digestion of PNNs by using chondroitinaseABC (ChABC) or through cortical infusion of a blocking peptide that saturates PNN binding sites. Finally, cortical Otx2 can be reduced by knocking down its expression in the choroid plexus, thus lowering its content in the cerebrospinal fluid that bathes the cortex.
Recombining Otx2 in the adult choroid plexus or competing with its import by PV cells through the infusion of the RK-peptide (RKQRRERTTFTRAQL) could reopen plasticity in the adult (figure 2). Indeed, closing an eye following either protocol can install amblyopia in the adult mouse and, more interestingly, the same protocols restore visual acuity and binocular vision in amblyopic mice [9,30]. Interestingly, reactivation of plasticity was also obtained by hydrolysing the complex sugars present in PNNs [33]. This result, added to the finding that the ratio between 4-sulfation/6-sulfation in PNN proteoglycans modifies Otx2 accumulation by PV cells [31], confirms that an important function of PNNs is to regulate Otx2 internalization and to control cerebral cortex plasticity during development and in the adult (figure 2). Because Otx2 is imported in supragranular layer PV cells throughout the cerebral cortex, it is possible that its role in regulating plasticity extends beyond the visual cortex [30].
5. Perspectives
The interest in adult HP functions, cell autonomous or non-cell autonomous, is indeed very recent and much research remains to be accomplished. Concerning downstream HP targets, it is clear that adult targets can differ, in part, from embryonic ones and that cell-autonomous and non-cell-autonomous targets are not necessary identical. In addition, further studies will be necessary to distinguish between different types of targets: transcriptional (direct and indirect), translational (as in the case of Ndufs1/3 and laminB2) and epigenetic (suggested by long-lasting effects of a time-restricted application of En1/2 or Otx2). It is of high interest that identified translational targets have a connection with mitochondrial physiopathology and the regulation of neuronal metabolism with consequences on neuronal physiology and survival. It is of note that an eIF4E-binding domain is necessary for En1/2 to regulate translation and that such domains have been conserved in more than 200 HPs [7]. What was found in the case of mouse models of PD (a regulation of mitochondrial function through En1/2-dependent translation of complex I subunits) might thus be a more general property of HPs and not only of Engrailed and the midbrain.
Identifying HP genetic targets has been on the agenda of many laboratories and we shall not discuss this issue here, only to recall again that targets may change through development and may depend on whether or not the protein function is cell autonomous. This is quite obvious as the cellular context provides cofactors that differ with age and cellular identity and as the epigenetic state of the cell can modify HP access to the chromatin. In addition, transferring proteins might be modified or carry some cofactors, including lipids or non-coding RNAs, that will influence gene expression. A more general possibility is that HPs may modify the epigenetic status of cells as suggested by their long-lasting effects, at least in the cases that were discussed in this brief review. If so, it can be envisaged that the progressive death of DA cells observed in the SN of En1+/− mice involves, in addition to mitochondrial failure, some changes in chromatin structure. The fact that En1/2 regulates the translation of laminB2, a nuclear scaffolding protein, suggests that a regulation by Engrailed at the level of the global structure of the nucleus can be foreseen even though laminB2 activity in the stability of tadpole RGC axons is reported at the mitochondrial level [22].
If we now concentrate on purely non-cell-autonomous activities, several questions—some general and some more specific to Otx2 in the cortex—can be raised. A very general issue is the identification of the transfer mechanisms. Sequences have been identified in the HD that are necessary for internalization and secretion. In fact the first identified internalization sequence corresponds to the third helix of the HD of Antennapedia (a fly HP) and is highly conserved among all HPs. It is sufficient for internalization and is now used as a vector peptide [4]. The mechanism for its internalization is not fully understood but involves a disturbance of the membrane that follows the insertion of a tryptophan (in position 48 of the HD) into the lipid bilayer [4]. A secretion sequence is also present in the HD though the precise mechanism of secretion is still under study by several groups [34]. Again this sequence is highly conserved suggesting that most HPs can transfer between cells; presently, the ability of 12 HPs to transfer has been confirmed either in vivo and/or in vitro. It is of note that linking the internalization and secretion sequences allows the resulting chimeric peptide to enter the basolateral membrane of a tight-junction epithelium and to gain access to the apical extracellular side, demonstrating that the two sequences are necessary and sufficient for internalization and secretion and suggesting a polarized traffic mechanism [35].
A second general issue concerns co-signalling and synergy between HPs and classical factors. It was shown that En1/2 cooperates with EphrinA5 in the optic tectum and with DPP in the fly wing disc [14,16,17]. A similar synergy was found between Pax6 and Netrin signalling in the ventral neural tube [13]. The essence of this synergy, illustrated in figure 3, is based on the hypothesis that HPs sensitize the classical receptors to otherwise inefficient concentrations of their agonists. This suggests that HP signalling only works at physiological concentrations of the other signalling factors. For example, an excess of EphrinA5 induces temporal growth cone collapse in the absence of Engrailed, but at sub-threshold (probably physiological) EphrinA5 concentrations, the addition of nanomolar amounts of En1 is necessary to observe collapse [14]. From an evolutionary point of view, as HP signalling is shared between plants and animals [4], it is possible that HP signalling represents a very primitive mechanism already active in the first multicellular organisms and that the other pathways were recruited during evolution and have added robustness to HP signalling.
Figure 3.
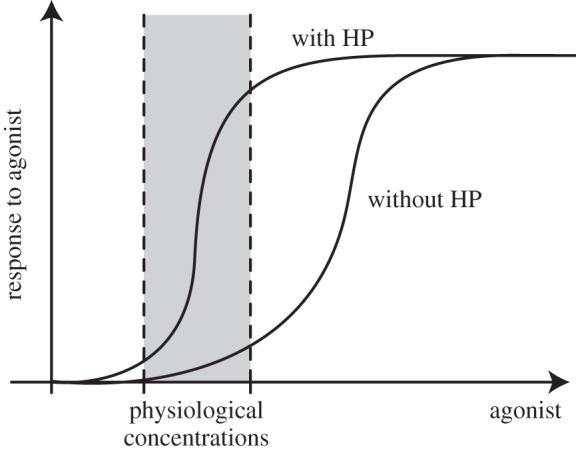
A model for the co-signalling and synergy between HPs and classical signalling factors. In artificial situations, response to a signalling molecule (agonist) is possible at high agonist concentrations with or without HP. However, at physiological concentrations, proper response to the agonist is made possible by the potentiating effect of HP signalling.
A final issue is the specific recognition of PV cells by Otx2 and more generally of HPs by target cells. As explained above, Otx2 infused in the visual cortex or secreted by the choroid plexus finds its way to PV cells. This recognition is mediated by a unique sequence motif in Otx2 for specific binding to GAGs present in the extracellular matrix [9]. Similarly to a variant of Otx2 mutated in the recognition domain, Engrailed infused in the same conditions did not specifically accumulate in PV cells [9]. These findings raise the hypothesis of a sugar code for HP recognition by various cell types. Going forward, it will be of interest to purify and sequence endogenous GAG chains that bind distinct HPs. Because all PV cells are enwrapped by PNNs, it is conceivable that Otx2 secreted by choroid plexus is captured by PV cells throughout the cerebral cortex. This places this factor in a good position to coordinate the activity of different cortical areas and potentially play a global role in cognition.
All in all, the study of HP activity in the adult is coming of age with the unexpected twist of adult non-cell-autonomous functions. This novel physiological pathway is under study by several groups and both non-cell-autonomous and cell-autonomous functions will certainly further contribute to the understanding of brain development, physiology and pathologies. It has not escaped our attention that HPs are also expressed outside of the brain. Given general HP adult expression and the conservation of the transfer sequences, we are convinced that the work summarized here represents only the tip of the iceberg.
Acknowledgements
We thank France Maloumian for her help with figure design.
Funding statement
This work was supported by Collège de France, Centre National de la Recherche Scientifique (CNRS) and Institut National de la Santé et de la Recherche Médicale (INSERM). Support from Agence Nationale pour la Recherche (ANR-10-BLAN-141-01 & ANR-11-BLAN-069467) is also acknowledged.
References
- 1.Gehring WJ. 1987. Homeo boxes in the study of development. Science 236, 1245–1252. ( 10.1126/science.2884726) [DOI] [PubMed] [Google Scholar]
- 2.Davis CA, Joyner AL. 1988. Expression patterns of the homeo box-containing genes En-1 and En-2 and the proto-oncogene int-1 diverge during mouse development. Genes Dev. 2, 1736–1744. ( 10.1101/gad.2.12b.1736) [DOI] [PubMed] [Google Scholar]
- 3.Duboule D, Morata G. 1994. Colinearity and functional hierarchy among genes of the homeotic complexes. Trends Genet. 10, 358–364. ( 10.1016/0168-9525(94)90132-5) [DOI] [PubMed] [Google Scholar]
- 4.Joliot A, Prochiantz A. 2004. Transduction peptides: from technology to physiology. Nat. Cell Biol. 6, 189–196. ( 10.1038/ncb0304-189) [DOI] [PubMed] [Google Scholar]
- 5.Spatazza J, Di Lullo E, Joliot A, Dupont E, Moya KL, Prochiantz A. 2013. Homeoprotein signaling in development, health, and disease: a shaking of dogmas offers challenges and promises from bench to bed. Pharmacol. Rev. 65, 90–104. ( 10.1124/pr.112.006577) [DOI] [PubMed] [Google Scholar]
- 6.Dubnau J, Struhl G. 1996. RNA recognition and translational regulation by a homeodomain protein. Nature 379, 694–699. ( 10.1038/379694a0) [DOI] [PubMed] [Google Scholar]
- 7.Topisirovic I, Borden KL. 2005. Homeodomain proteins and eukaryotic translation initiation factor 4E (eIF4E): an unexpected relationship. Histol. Histopathol. 20, 1275–1284. [DOI] [PubMed] [Google Scholar]
- 8.Kim YH, Choi CY, Lee SJ, Conti MA, Kim Y. 1998. Homeodomain-interacting protein kinases, a novel family of co-repressors for homeodomain transcription factors. J. Biol. Chem. 273, 25 875–25 879. ( 10.1074/jbc.273.40.25875) [DOI] [PubMed] [Google Scholar]
- 9.Beurdeley M, Spatazza J, Lee H, Sugiyama S, Bernard C, Di Nardo AA, Hensch TK, Prochiantz A. 2012. Otx2 binding to perineuronal nets persistently regulates plasticity in the mature visual cortex. J. Neurosci. 32, 9429–9437. ( 10.1523/JNEUROSCI.0394-12.2012) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rezsohazy R. 2014. Non-transcriptional interactions of Hox proteins: inventory, facts, and future directions. Dev. Dyn. 243, 117–131. ( 10.1002/dvdy.24060) [DOI] [PubMed] [Google Scholar]
- 11.Sonnier L, Le Pen G, Hartmann A, Bizot JC, Trovero F, Krebs MO, Prochiantz A. 2007. Progressive loss of dopaminergic neurons in the ventral midbrain of adult mice heterozygote for Engrailed1. J. Neurosci. 27, 1063–1071. ( 10.1523/JNEUROSCI.4583-06.2007) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lesaffre B, Joliot A, Prochiantz A, Volovitch M. 2007. Direct non-cell autonomous Pax6 activity regulates eye development in the zebrafish. Neural Dev. 2, 2 ( 10.1186/1749-8104-2-2) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Di Lullo E, Haton C, Le Poupon C, Volovitch M, Joliot A, Thomas JL, Prochiantz A. 2011. Paracrine Pax6 activity regulates oligodendrocyte precursor cell migration in the chick embryonic neural tube. Development 138, 4991–5001. ( 10.1242/dev.066282) [DOI] [PubMed] [Google Scholar]
- 14.Wizenmann A, et al. 2009. Extracellular engrailed participates in the topographic guidance of retinal axons in vivo. Neuron 64, 355–366. ( 10.1016/j.neuron.2009.09.018) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Brunet I, Weinl C, Piper M, Trembleau A, Volovitch M, Harris W, Prochiantz A, Holt C. 2005. The transcription factor Engrailed-2 guides retinal axons. Nature 438, 94–98. ( 10.1038/nature04110) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stettler O, Joshi RL, Wizenmann A, Reingruber J, Holcman D, Bouillot C, Castagner F, Prochiantz A, Moya KL. 2012. Engrailed homeoprotein recruits the adenosine A1 receptor to potentiate ephrin A5 function in retinal growth cones. Development 139, 215–224. ( 10.1242/dev.063875) [DOI] [PubMed] [Google Scholar]
- 17.Layalle S, Volovitch M, Mugat B, Bonneaud N, Parmentier ML, Prochiantz A, Joliot A, Maschat F. 2011. Engrailed homeoprotein acts as a signaling molecule in the developing fly. Development 138, 2315–2323. ( 10.1242/dev.057059) [DOI] [PubMed] [Google Scholar]
- 18.Alvarez-Fischer D, et al. 2011. Engrailed protects mouse midbrain dopaminergic neurons against mitochondrial complex I insults. Nat. Neurosci. 14, 1260–1266. ( 10.1038/nn.2916) [DOI] [PubMed] [Google Scholar]
- 19.Alberi L, Sgado P, Simon HH. 2004. Engrailed genes are cell-autonomously required to prevent apoptosis in mesencephalic dopaminergic neurons. Development 131, 3229–3236. ( 10.1242/dev.01128) [DOI] [PubMed] [Google Scholar]
- 20.Di Nardo AA, Nedelec S, Trembleau A, Volovitch M, Prochiantz A, Montesinos ML. 2007. Dendritic localization and activity-dependent translation of Engrailed1 transcription factor. Mol. Cell Neurosci. 35, 230–236. ( 10.1016/j.mcn.2007.02.015) [DOI] [PubMed] [Google Scholar]
- 21.Joyner AL, Hanks M. 1991. The engrailed genes: evolution and function. Semin. Dev. Biol. 2, 736–741. [Google Scholar]
- 22.Yoon BC, Jung H, Dwivedy A, O'Hare CM, Zivraj KH, Holt CE. 2012. Local translation of extranuclear lamin B promotes axon maintenance. Cell 148, 752–764. ( 10.1016/j.cell.2011.11.064) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bernard C, et al. 2014. Graded Otx2 activities demonstrate dose-sensitive eye and retina phenotypes. Hum. Mol. Genet. 23, 1742–1753. ( 10.1093/hmg/ddt562) [DOI] [PubMed] [Google Scholar]
- 24.Torero Ibad R, Rheey J, Mrejen S, Forster V, Picaud S, Prochiantz A, Moya KL. 2011. Otx2 promotes the survival of damaged adult retinal ganglion cells and protects against excitotoxic loss of visual acuity in vivo. J. Neurosci. 31, 5495–5503. ( 10.1523/JNEUROSCI.0187-11.2011) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wiesel TN, Hubel DH. 1965. Comparison of the effects of unilateral and bilateral eye closure on cortical unit responses in kittens. J. Neurophysiol. 28, 1029–1040. [DOI] [PubMed] [Google Scholar]
- 26.Hensch T. 2005. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888. ( 10.1038/nrn1787) [DOI] [PubMed] [Google Scholar]
- 27.Balmer TS, Carels VM, Frisch JL, Nick TA. 2009. Modulation of perineuronal nets and parvalbumin with developmental song learning. J. Neurosci. 29, 12 878–12 885. ( 10.1523/JNEUROSCI.2974-09.2009) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fawcett J. 2009. Molecular control of brain plasticity and repair. Prog. Brain Res. 175, 501–509. ( 10.1016/S0079-6123(09)17534-9) [DOI] [PubMed] [Google Scholar]
- 29.Sugiyama S, Di Nardo AA, Aizawa S, Matsuo I, Volovitch M, Prochiantz A, Hensch TK. 2008. Experience-dependent transfer of Otx2 homeoprotein into the visual cortex activates postnatal plasticity. Cell 134, 508–520. ( 10.1016/j.cell.2008.05.054) [DOI] [PubMed] [Google Scholar]
- 30.Spatazza J, Lee HH, Di Nardo AA, Tibaldi L, Joliot A, Hensch TK, Prochiantz A. 2013. Choroid-plexus-derived Otx2 homeoprotein constrains adult cortical plasticity. Cell Rep. 3, 1815–1823. ( 10.1016/j.celrep.2013.05.014) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miyata S, Komatsu Y, Yoshimura Y, Taya C, Kitagawa H. 2012. Persistent cortical plasticity by upregulation of chondroitin 6-sulfation. Nat. Neurosci. 15, 414–422. ( 10.1038/nn.3023) [DOI] [PubMed] [Google Scholar]
- 32.Wolpert L. 1969. Positional information and the spatial pattern of cellular differentiation. J. Theor. Biol. 25, 1–47. ( 10.1016/S0022-5193(69)80016-0) [DOI] [PubMed] [Google Scholar]
- 33.Pizzorusso T, Medini P, Berardi N, Chierzi S, Fawcett JW, Maffei L. 2002. Reactivation of ocular dominance plasticity in the adult visual cortex. Science 298, 1248–1251. ( 10.1126/science.1072699) [DOI] [PubMed] [Google Scholar]
- 34.Maizel A, Bensaude O, Prochiantz A, Joliot A. 1999. A short region of its homeodomain is necessary for engrailed nuclear export and secretion. Development 126, 3183–3190. [DOI] [PubMed] [Google Scholar]
- 35.Dupont E, Prochiantz A, Joliot A. 2007. Identification of a signal peptide for unconventional secretion. J. Biol. Chem. 282, 8994–9000. ( 10.1074/jbc.M609246200) [DOI] [PubMed] [Google Scholar]


