Abstract
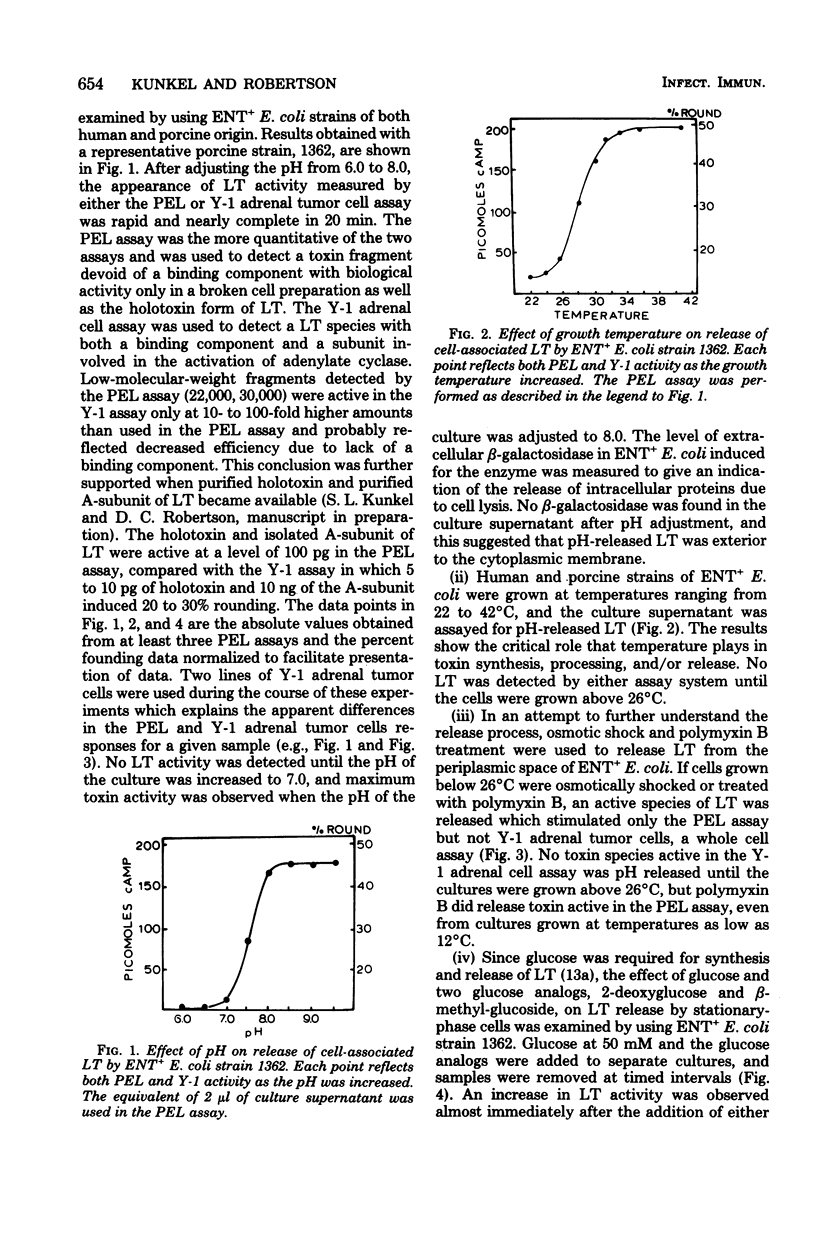
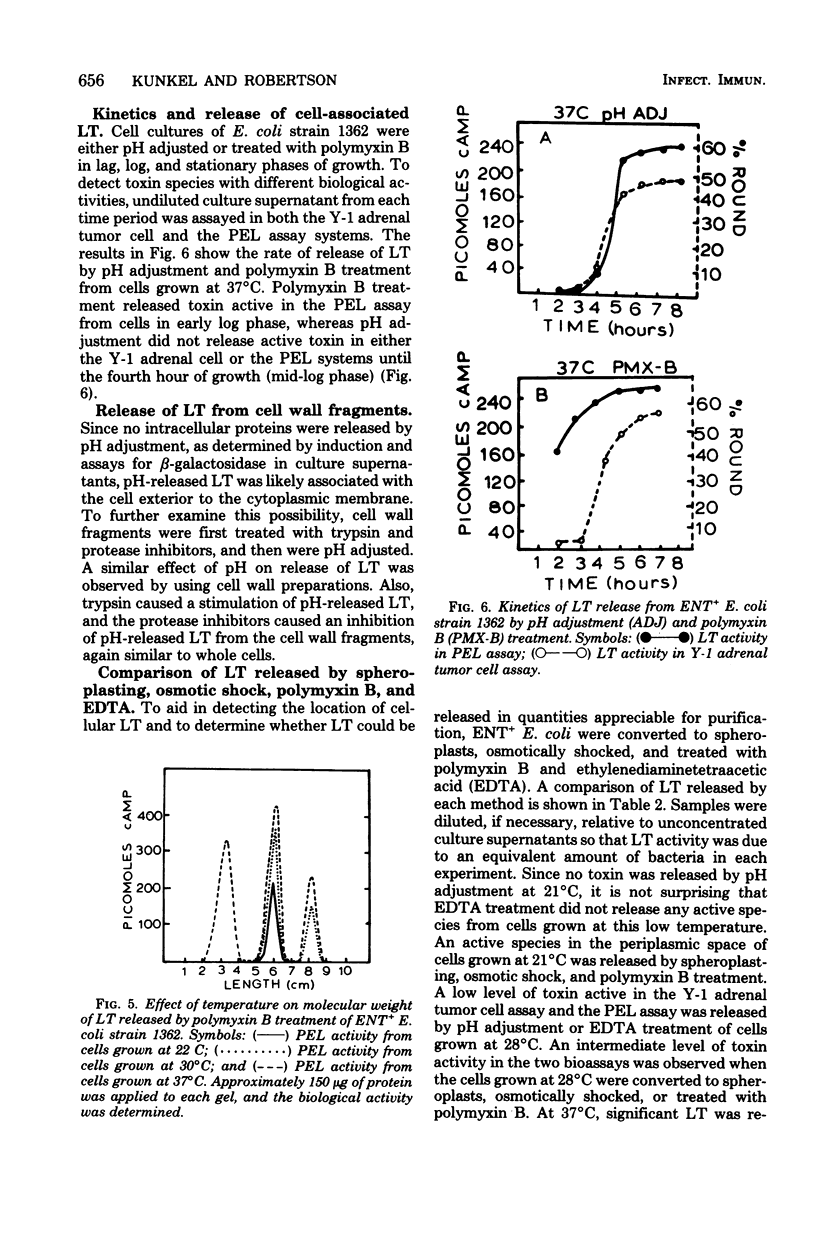
Various conditions affecting the release of heat-labile enterotoxin (LT) by enterotoxigenic Escherichia coli have been examined. The pH of a defined medium containing three amino acids, M-9 salts, and 0.5% glucose decreased to less than 7.0 in early log phase of growth, and no extracellular LT was detected. Adjustment of the pH at 8 h from 6.0 to 8.0 resulted in a concomitant increase in LT activity in culture supernatants. The release of cell-associated LT was significantly reduced by preincubation with protease inhibitors and increased by preincubation with trypsin. Cell-associated LT was not released by pH adjustment of cells grown at 21°C; however, polymyxin B treatment released a toxin species active in only the pigeon erythrocyte lysate (PEL) assay system. As the growth temperature was increased, polymyxin B released toxin species which exhibited both PEL and Y-1 adrenal tumor cell activity. Polymyxin B extracts of enterotoxigenic E. coli in early log phase grown at 37°C possessed only PEL activity, whereas extracts from cells in late-log and stationary phases had biological activity in both assay systems. Also, LT released by pH adjustment from mid-log to stationary phase was active in both PEL and Y-1 adrenal tumor cell assays. Gel electrophoresis of polymyxin B extracts revealed at least three molecular weight species active in either the PEL (22,000 daltons and 30,000 daltons) or both the PEL and the Y-1 adrenal tumor cell assay (72,000 daltons), depending on the growth temperature. These observations may help to explain the chemical and biological heterogeneity of most LT preparations and facilitate purification of LT by increasing the yield of enterotoxin.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alderete J. F., Robertson D. C. Purification and chemical characterization of the heat-stable enterotoxin produced by porcine strains of enterotoxigenic Escherichia coli. Infect Immun. 1978 Mar;19(3):1021–1030. doi: 10.1128/iai.19.3.1021-1030.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ames G. F. Resolution of bacterial proteins by polyacrylamide gel electrophoresis on slabs. Membrane, soluble, and periplasmic fractions. J Biol Chem. 1974 Jan 25;249(2):634–644. [PubMed] [Google Scholar]
- Brockman R. W., Heppel L. A. On the localization of alkaline phosphatase and cyclic phosphodiesterase in Escherichia coli. Biochemistry. 1968 Jul;7(7):2554–2562. doi: 10.1021/bi00847a016. [DOI] [PubMed] [Google Scholar]
- Cerny G., Teuber M. Differential release of periplasmic versus cytoplasmic enzymes from Escherichia coli B by polymixin B. Arch Mikrobiol. 1971;78(2):166–179. doi: 10.1007/BF00424873. [DOI] [PubMed] [Google Scholar]
- Cheng K. J., Ingram J. M., Costerton J. W. Alkaline phosphatase localization and spheroplast formation of Pseudomonas aeruginosa. Can J Microbiol. 1970 Dec;16(12):1319–1324. doi: 10.1139/m70-218. [DOI] [PubMed] [Google Scholar]
- DUFF J. T., WRIGHT G. G., YARINSKY A. Activation of Clostridium botulinum type E toxin by trypsin. J Bacteriol. 1956 Oct;72(4):455–460. doi: 10.1128/jb.72.4.455-460.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dafni Z., Robbins J. B. Purification of heat-labile enterotoxin from Escherichia coli O78:H11 by affinity chromatography with antserum to Vibrio cholerae toxin. J Infect Dis. 1976 Mar;133 (Suppl):138–141. doi: 10.1093/infdis/133.supplement_1.s138. [DOI] [PubMed] [Google Scholar]
- Donta S. T., Moon H. W., Whipp S. C. Detection of heat-labile Escherichia coli enterotoxin with the use of adrenal cells in tissue culture. Science. 1974 Jan 25;183(4122):334–336. doi: 10.1126/science.183.4122.334. [DOI] [PubMed] [Google Scholar]
- Dorner F. Escherichia coli enterotoxin. Purification and partial characterization. J Biol Chem. 1975 Nov 25;250(22):8712–8719. [PubMed] [Google Scholar]
- Evans D. J., Jr, Evans D. G., Richardson S. H., Gorbach S. L. Purification of the polymyxin-released, heat-labile enterotoxin of Escherichia coli. J Infect Dis. 1976 Mar;133 (Suppl):97–102. doi: 10.1093/infdis/133.supplement_1.s97. [DOI] [PubMed] [Google Scholar]
- Gill D. M., King C. A. The mechanism of action of cholera toxin in pigeon erythrocyte lysates. J Biol Chem. 1975 Aug 25;250(16):6424–6432. [PubMed] [Google Scholar]
- Gill D. M. Multiple roles of erythrocyte supernatant in the activation of adenylate cyclase by Vibrio cholerae toxin in vitro. J Infect Dis. 1976 Mar;133 (Suppl):55–63. doi: 10.1093/infdis/133.supplement_1.s55. [DOI] [PubMed] [Google Scholar]
- Gilligan P. H., Robertson D. C. Nutritional requirements for synthesis of heat-labile enterotoxin by enterotoxigenic strains of Escherichia coli. Infect Immun. 1979 Jan;23(1):99–107. doi: 10.1128/iai.23.1.99-107.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lampen J. O. Release of penicillinase by Bacillus licheniformis. J Gen Microbiol. 1967 Aug;48(2):261–268. doi: 10.1099/00221287-48-2-261. [DOI] [PubMed] [Google Scholar]
- Lariviére S., Gyles C. L., Barnum D. A. Preliminary characterization of the heat-labile enterotoxin of Escherichia coli F11(P155). J Infect Dis. 1973 Sep;128(3):312–320. doi: 10.1093/infdis/128.3.312. [DOI] [PubMed] [Google Scholar]
- Leive L. Release of lipopolysaccharide by EDTA treatment of E. coli. Biochem Biophys Res Commun. 1965 Nov 22;21(4):290–296. doi: 10.1016/0006-291x(65)90191-9. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B., Meijers J., Peters R., van der Hoek P., van Alphen L. Electrophoretic resolution of the "major outer membrane protein" of Escherichia coli K12 into four bands. FEBS Lett. 1975 Oct 15;58(1):254–258. doi: 10.1016/0014-5793(75)80272-9. [DOI] [PubMed] [Google Scholar]
- MALAMY M. H., HORECKER B. L. PURIFICATION AND CRYSTALLIZATION OF THE ALKALINE PHOSPHATASE OF ESCHERICHIA COLI. Biochemistry. 1964 Dec;3:1893–1897. doi: 10.1021/bi00900a018. [DOI] [PubMed] [Google Scholar]
- MacaAlister T. J., Irvin R. T., Costerton J. W. Cell surface-localized alkaline phosphatase of Escherichia coli as visualized by reaction product deposition and ferritin-labeled antibodies. J Bacteriol. 1977 Apr;130(1):318–328. doi: 10.1128/jb.130.1.318-328.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nisonson I., Tannenbaum M., Neu H. C. Surface localization of Escherichia coli 5'-nucleotidase by electron microscopy. J Bacteriol. 1969 Nov;100(2):1083–1090. doi: 10.1128/jb.100.2.1083-1090.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nossal N. G., Heppel L. A. The release of enzymes by osmotic shock from Escherichia coli in exponential phase. J Biol Chem. 1966 Jul 10;241(13):3055–3062. [PubMed] [Google Scholar]
- ORSKOV I., ORSKOV F., SOJKA W. J., LEACH J. M. Simultaneous occurrence of E. coli B and Lantigens in strains from diseased swine. Influence of cultivation temperature. Two new E. coli Kantigens: K 87 and K 88. Acta Pathol Microbiol Scand. 1961;53:404–422. [PubMed] [Google Scholar]
- REVEL H. R., LURIA S. E., ROTMAN B. Biosynthesis of B-D-galactosidase controlled by phage-carried genes. I. Induced beta-D-galactosidase biosynthesis after transduction of gene z-plus by phage. Proc Natl Acad Sci U S A. 1961 Dec 15;47:1956–1967. doi: 10.1073/pnas.47.12.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rappaport R. S., Sagin J. F., Pierzchala W. A., Bonde G., Rubin B. A., Tint H. Activation of Heat-labile Escherichia coli enterotoxin by trypsin. J Infect Dis. 1976 Mar;133 (Suppl):41–54. doi: 10.1093/infdis/133.supplement_1.s41. [DOI] [PubMed] [Google Scholar]
- Sack R. B. Human diarrheal disease caused by enterotoxigenic Escherichia coli. Annu Rev Microbiol. 1975;29:333–353. doi: 10.1146/annurev.mi.29.100175.002001. [DOI] [PubMed] [Google Scholar]
- Schenkein I., Green R. F., Santos D. S., Maas W. K. Partial purification and characterization of a heat-labile enterotoxin of Escherichia coli. Infect Immun. 1976 Jun;13(6):1710–1720. doi: 10.1128/iai.13.6.1710-1720.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witholt B., Boekhout M., Brock M., Kingma J., Heerikhuizen H. V., Leij L. D. An efficient and reproducible procedure for the formation of spheroplasts from variously grown Escherichia coli. Anal Biochem. 1976 Jul;74(1):160–170. doi: 10.1016/0003-2697(76)90320-1. [DOI] [PubMed] [Google Scholar]



