Abstract
Purpose. To compare tear film metrics in patients with type 1 diabetes mellitus (DM) and healthy controls and investigate the association between peripheral neuropathy and ocular surface quality. Methods. Dry eye symptoms were quantified in 53 patients with type 1 DM and 40 age-matched controls. Ocular examination included tear film lipid layer thickness grading, tear film stability and quantity measurement, and retinal photography. DM individuals additionally underwent a detailed neuropathy assessment. Results. Neither mean age nor dry eye symptom scores differed significantly between the DM and control groups (P = 0.12 and P = 0.33, resp.). Tear lipid thickness (P = 0.02), stability (P < 0.0001), and quantity (P = 0.01) were significantly lower in the DM group. Corneal sensitivity was also reduced in the DM group (P < 0.001) and tear film stability was inversely associated with total neuropathy score (r = −0.29, P = 0.03). Conclusion. The DM group exhibited significantly reduced tear film stability, secretion, and lipid layer quality relative to the age-matched control group. The negative correlation between tear film parameters and total neuropathy score suggests that ocular surface abnormalities occur in parallel with diabetic peripheral neuropathy.
1. Introduction
While cataract and retinopathy have been extensively researched in patients with diabetes mellitus (DM), only a fraction of the published research has been dedicated to ocular surface complications. However, dry eye symptoms and signs of epithelial fragility, punctate keratopathy, persistent epithelial defects, and decreased corneal sensitivity are not uncommon in DM [1–3]. Compromised innervation of the cornea in patients with DM has also been described [4–6]. Tear film dysfunction, characterised by impairment in tear quantity and quality, can occur in association with abnormal corneal innervation due to the intimate, functional relationship between the cornea and the preocular tear film [7, 8]. The resulting dry eye is a recognised cause of debilitating, chronic ocular irritation symptoms [9]. The restrictions on life imposed by this chronic condition can be significant and, in terms of impact on quality of life, have been equated in more severe cases to those induced by dialysis and severe angina [10].
Tear film irregularity has been reported in DM, especially in association with that of extended disease duration and severity, as defined by stage of retinopathy [2, 11]. It has been postulated that damage to the microvasculature and denervation of the lacrimal gland may contribute to impaired lacrimation in DM [2, 7, 12]. Despite this neural link, few studies have explored whether the ocular surface alters in association with peripheral neuropathy [7, 13, 14], although anomalous innervation of the lacrimal gland has been reported in those with diabetic sensory neuropathy [2, 7].
This prospective study sought to compare the tear film in DM with that of control subjects and examine the relationship of the preocular tear film with markers of peripheral neuropathy.
2. Materials and Methods
The study adhered to the tenets of the Declaration of Helsinki and was conducted with Regional Ethics Committee approval (NTX/09/12/122). Two hundred and seven patients, identified by a specialist endocrinologist (Author, Geoffrey D. Braatvedt) as having a history of type 1 DM, were invited to participate. A history of laser therapy for retinopathy, cumulative contact lens wear of ≥3 months, ocular surgery, ocular trauma, or ocular or systemic disease that may affect the ocular surface, or a diagnosis of peripheral neuropathy unrelated to DM precluded participation in the study. Consequently, 53 individuals with DM were deemed eligible and were willing to participate in the study. Forty healthy, nondiabetic volunteers (HbA1c < 41 mmol/mol) were recruited as controls.
For each participant, a detailed ocular and general medical history, including smoking and alcohol use, was obtained. Dry eye symptoms were recorded and scored with the McMonnies dry eye questionnaire [15] and general ocular surface and eyelid examination was performed by slit-lamp biomicroscopy (Topcon Medical Systems, NJ, USA). Tear film interferometry with the Keeler Tearscope Plus (Keeler Ltd, Berkshire, UK) enabled tear film lipid patterns to be graded according to Guillon's classification [16]. Lipid patterns corresponding to increasing lipid layer thickness were subsequently assigned numerical lipid layer grades 0–5, for the purpose of statistical analysis, as 0 (absent), 1 (open meshwork), 2 (closed meshwork), 3 (wave), 4 (amorphous), or 5 (coloured fringes) (Figure 1). Tear film stability was recorded with the aid of the Tearscope Plus with the fine grid insert, as the noninvasive tear breakup time (NIBUT). An average of 3 readings was calculated for each measurement. The Phenol Red Thread (PRT) test (Zone-Quick, FCI Ophthalmics, Pembroke, MA, USA) provided an index of tear quantity. Central corneal sensitivity was measured using noncontact corneal aesthesiometry with the NCCA (Glasgow Caledonian University, UK) and adopting a double staircase method of threshold determination [17]. All ocular assessments were performed at a single visit, on the right eye only, with the exception of subjects reporting unilateral surgery or trauma to the right eye, in which case the left eye was examined. Tests were performed in the same order for each participant, from least to most invasive, to minimize the effect of reflex tearing.
Figure 1.
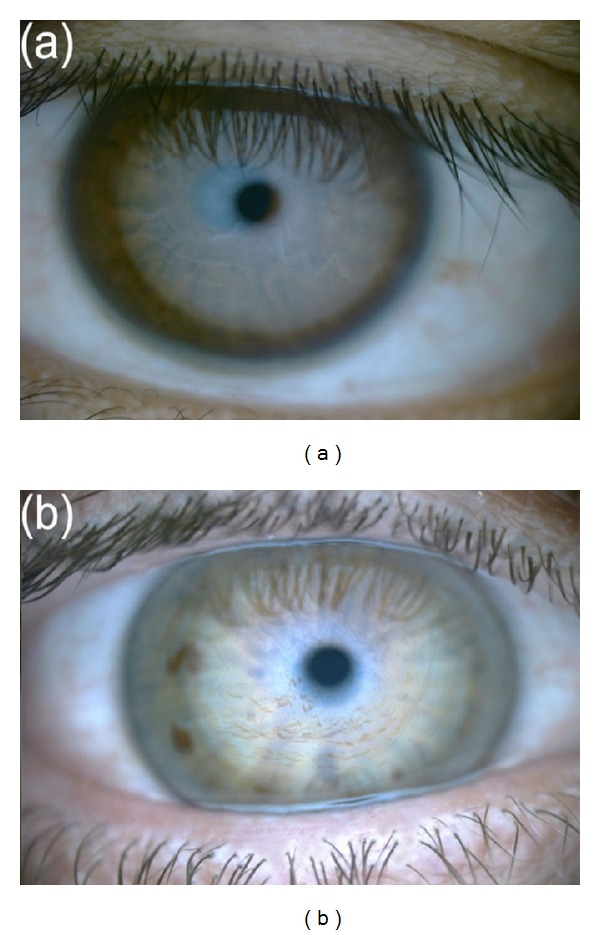
Representative examples of tear film—lipid layer grades: (a) grade 3: wave pattern; (b) grade 5: colour fringe pattern.
Digital images of the central and peripheral retina were captured (Non-Mydriatic Retinal Camera DR-DGi, Canon Inc., USA) and graded according to the Early Treatment Diabetic Retinopathy Study (ETDRS) criteria [18] for diabetic retinopathy by an independent, fellowship-trained, medical retina specialist (Author, Monika Pradhan).
The DM group underwent neuropathic assessment by an experienced neurologist (Author, Dean Kilfoyle). An overall neuropathy score (total neuropathy score or TNS) was obtained from a previously validated and recognised combination of the symptomatic neuropathy score [19], a clinical neuropathic assessment by the neurologist, biothesiometry (quantitative sensory testing) on the medial malleolus and great toe, and nerve conduction study (NCS), with increasing score representing increasing severity of neuropathy [19].
Statistical analysis was undertaken with IBM SPSS v19.0 (Chicago, IL, USA). Regression analysis was performed between the results of the tear film assessment techniques, corneal sensitivity, diabetic retinopathy grading, and total neuropathy score. Positively skewed raw NIBUT data were logarithmically transformed prior to parametric statistical testing. One-factor ANOVA and Friedman tests were performed between variables with normally distributed and non-normally distributed data, respectively, to test differences between controls and DM groups. Pearson and Spearman correlation (2-tailed) analyses were performed for data that did and did not approximate normal distributions, respectively. A P value of less than 0.05 was considered statistically significant.
3. Results and Discussion
3.1. Results
Patient characteristics including gender, age, ethnicity, and duration of diabetes are described in Table 1. All but two participants in the DM group self-identified as being of European descent while the control participants included 47.5% NZ European, 22.5% Indian, and 20% Asian (excluding Indian). The mean age of the DM group (49 ± 12 years) did not differ significantly from that of the control group (44 ± 15 years) (P = 0.12) and no significant differences in tear film characteristics were identified between the ethnic subgroups (P > 0.05).
Table 1.
Participant characteristics for the patients with type 1 diabetes and control group.
| Diabetes | Controls | |
|---|---|---|
| Subjects (n) | 53 | 40 |
| M : F ratio | 26 : 27 | 17 : 23 |
| Age (years) | 48.6 ± 11.8 | 44.3 ± 14.7 |
| HbA1c (mmol/mol) | 61.3 ± 12.0 | 35.0 ± 2.5 |
| Ethnicity | ||
| European | 51 | 19 |
| Indian | 0 | 9 |
| Asian (excluding Indian) | 1 | 8 |
| Maori | 1 | 0 |
| Others | 0 | 4 |
| Mean diabetes Duration (years) |
25.8 ± 11.4 | |
| <10 | 5 | |
| 10–20 | 10 | |
| 21–31 | 22 | |
| >31 | 16 |
Dry eye symptom scores and clinical findings were compared for the diabetes and control groups (Table 2). The mean dry eye symptom score was not statistically significantly different between the diabetes and control groups (P = 0.33). However, clinical tests showed significant differences between the two groups, with lipid layer grading (P = 0.02), NIBUT (P < 0.0001), and PRT test (P = 0.01) results all statistically, and clinically, significantly lower in the DM group. NIBUT was observed to be lower in females than in males in both the control (P = 0.06) and DM (P < 0.01) groups. The patients with DM exhibited reduced corneal sensitivity compared to controls (P < 0.0001) (corneal sensitivity and subbasal nerve density reported in detail elsewhere) [20]. The total neuropathy score (maximum possible score 40) ranged between 0 and 21 (5.3 ± 5.1) in the patients with diabetes.
Table 2.
Comparison of symptoms and tear characteristics for control and patient groups (mean/median), together with the significance of their differences (ANOVA/Friedmann). Noninvasive tear breakup time (NIBUT) values are extrapolated from logarithmically transformed data.
| Diabetes | Healthy controls | ANOVA/Friedmann (P values) |
|
|---|---|---|---|
| McMonnies questionnaire (mean ± SD) |
8.8 ± 6.7 | 7.6 ± 4.6 | 0.33 |
| Lipid layer thickness grade (median) | 2 | 3 | 0.02 |
| NIBUT (s) (mean ± SD) | 6.0 ± 1.9 | 8.2 ± 2.5 | <0.0001 |
| Phenol red thread test (mm) (mean ± SD) | 13.7 ± 4.7 | 16.3 ± 4.9 | 0.01 |
| Corneal sensitivity threshold (mBAR) | 1.3 ± 1.3 | 0.2 ± 1.3 | <0.001 |
Retinopathy was not observed in the majority of patients (60%) while 21% exhibited mild and 19% exhibited moderate DR. Regression analysis between interferometry, tear film stability, tear quantity, diabetic retinopathy grade, and total neuropathy score (Table 3) highlighted a positive relationship between lipid layer grading and both NIBUT (r = 0.56, P < 0.01) and tear quantity (r = 0.38, P < 0.01). Tear film stability was noted to be inversely related to total neuropathy score (r = −0.29, P = 0.03). The association between corneal sensitivity and total neuropathy score failed to reach statistical significance (r = 0.24, P = 0.08).
Table 3.
Correlation analysis between age, diabetes duration, McMonnies questionnaire scores (DEQ), phenol red thread test (PRTT) (mm), tear film interferometry including lipid layer grade, stability (NIBUT), diabetic retinopathy grading, and total neuropathy score (TNS) in those with DM (n = 53).
| Correlation (r values) | Probability (P values) | |
|---|---|---|
| Age versus NIBUT | −0.28∗ | 0.05 |
| Age versus TNS | 0.41∗∗ | 0.00 |
| DEQ versus TNS | −0.05 | 0.73 |
| Lipid layer thickness versus NIBUT | 0.56∗∗ | 0.00 |
| Lipid layer thickness versus PRTT | 0.39∗∗ | 0.00 |
| NIBUT versus diabetes duration | −0.29∗ | 0.03 |
| NIBUT versus TNS | −0.29∗ | 0.03 |
| NIBUT versus retinopathy grade | −0.03 | 0.82 |
| PRTT versus TNS | 0.01 | 0.96 |
| PRTT versus retinopathy grade | −0.16 | 0.26 |
∗∗Correlation is significant at the 0.01 level (2-tailed).
∗Correlation is significant at the 0.05 level (2-tailed).
3.2. Discussion
Comparison of the status of the tear film between patients with type 1 DM and age-matched healthy control subjects demonstrated a significantly poorer tear film quality in diabetes. Lipid layer grade, tear film stability, and tear quantity (basal with minimal reflex tear secretion) [21] were all significantly reduced in patients with type 1 DM, confirming compromised protection of the ocular surface in patients with diabetes.
Dry eye symptoms were observed to increase in severity with advancing age in both groups, in the current study, consistent with reports in the literature [22, 23]. Interestingly, age-matched subjects in both groups reported similar dry eye symptom severity, despite clear differences in tear quality (P < 0.0001). This lack of difference in symptoms is believed to be related to the impaired sensitivity of the ocular surface in diabetes.
Tear film stability and lipid layer thickness have been shown to be influenced by gender [24], with older women tending to exhibit thinner and more contaminated lipid layers [25]. The current study also reported reduced tear film stability in females compared to males, both in the DM group (P < 0.01) and in the control group (P = 0.06). A decrease in circulating androgens in postmenopausal women is believed to play an important role in affecting meibomian gland function, supporting female gender as a risk factor for dry eye disease [26].
In addition to a reduction in tear film stability, several studies have reported a diminished tear secretion in the diabetic eye [7, 11, 27], as observed in the present research. This further degrades the quality of tear film in an already compromised diabetic eye.
Reduced tear film stability has been associated with the presence of superficial punctate keratitis [8]. Functional abnormalities of corneal innervation may contribute to the incidence of superficial punctate keratitis in these patients through its adverse effect on tear film instability [28]. An inverse association between corneal innervation and peripheral neuropathy has been reported previously [6, 29]. The current study supports this relationship, with a modest inverse correlation observed between NIBUT and total neuropathy score (a measure of peripheral neuropathy) (r = −0.29, P = 0.03).
Retinopathy grade was observed to be unrelated to tear film stability in the current study (r = −0.03, P = 0.82), contrary to previous observations [11, 30]. However, it should be noted that, as history of laser treated retinopathy was an exclusion criterion in the current study, patients with more than moderate retinopathy were generally ineligible to participate. This restriction in the range of disease severity in the cohort enrolled in the current study likely contributed to the absence of a relationship in our study.
In this New Zealand-based study, the control group comprised patients of a variety of ethnic backgrounds reflecting the multiethnic population [31]. Type 1 DM in New Zealand is predominantly reported in those with European (Caucasian) heritage [32] and this was observed in the present cohort. Although ethnicity has previously been shown to be a determinant of tear film stability [33], no significant difference was observed between the ethnic subgroups of the control subjects in the present study.
There is potential for lipid, aqueous, and mucins, the major components of the tear film, to be adversely affected in patients with diabetes. The meibomian and lacrimal glands are responsible for the secretion of the lipid and aqueous portions of the preocular tear film, respectively. Meibomian glands are innervated by parasympathetic fibres with a smaller contribution from sympathetic and sensory neurons [34]. Disease or any damage to these neurons leads to dry eye in an animal model [35]. Two human studies have previously reported a compromised tear lipid layer in DM patients, as confirmed in the current study [8, 36]. Clinical observation of noncontiguous or absent lipid layers is associated with significantly increased tear film evaporation [37], one of the key factors in dry eye development [38].
Elevated expression of advanced glycation end products in the lacrimal gland has been postulated as a reason for changes in lacrimal gland function described in diabetes [12]. A reduction in goblet cell numbers, compromising mucin quantity, may also contribute to the tear film instability observed in diabetes [7, 39]. Goblet cell loss in those with diabetic peripheral neuropathy and poor metabolic control has been previously reported [7].
The reduced tear production identified in patients with diabetes (P = 0.01) in the current study lends support to the concept that lacrimal gland function might be adversely affected by a neuropathic mechanism [40], resulting in dysfunction of the ocular surface secretory glands via their innervation. Such dysfunction could arise from a peripheral neuropathy involving the afferent sensory nerves from the ocular surface affecting corneal sensitivity and the autonomic (efferent) nerves responsible for innervating the tear-component secreting glands and the lacrimal and meibomian glands [35, 40, 41]. This is supported by the current results of reduced corneal sensitivity; however the previously reported association between corneal sensitivity and peripheral neuropathy failed to reach statistical significance in the current study (r = 0.24, P = 0.08) [42]. Reduced tear secretion has previously been identified in patients with type 2 diabetes relative to healthy controls [8, 11].
4. Conclusion
In summary, the current study confirms the underlying threat to ocular surface health in patients with type 1 DM compared to control subjects. The reduction in tear production in patients with DM and the association between reduced tear film stability and diabetic peripheral polyneuropathy add credence to the hypothesis that diabetic peripheral neuropathy is associated with, or directly affects, secretory lacrimal gland function [43]. Hence, patients with DM, particularly the older female, are at a greater risk of dry eye and compromised preocular tear film.
Acknowledgments
This work was supported by Save Sight Society NZ Incorporated and New Zealand Optometric and Vision Research Foundation (NZOVRF). The statistical analysis consultation was provided by Dr. Avinesh Pillai from the Department of Statistics, University of Auckland.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
References
- 1.Hyndiuk RA, Kazarian EL, Schultz RO, Seideman S. Neurotrophic corneal ulcers in diabetes mellitus. Archives of Ophthalmology. 1977;95(12):2193–2196. doi: 10.1001/archopht.1977.04450120099012. [DOI] [PubMed] [Google Scholar]
- 2.Kaiserman I, Kaiserman N, Nakar S, Vinker S. Dry eye in diabetic patients. American Journal of Ophthalmology. 2005;139(3):498–503. doi: 10.1016/j.ajo.2004.10.022. [DOI] [PubMed] [Google Scholar]
- 3.Schultz RO, Peters MA, Sobocinski K, Nassif K, Schultz KJ. Diabetic keratopathy as a manifestation of peripheral neuropathy. American Journal of Ophthalmology. 1983;96(3):368–371. doi: 10.1016/s0002-9394(14)77829-8. [DOI] [PubMed] [Google Scholar]
- 4.Kallinikos P, Berhanu M, O’Donnell C, Boulton AJM, Efron N, Malik RA. Corneal nerve tortuosity in diabetic patients with neuropathy. Investigative Ophthalmology and Visual Science. 2004;45(2):418–422. doi: 10.1167/iovs.03-0637. [DOI] [PubMed] [Google Scholar]
- 5.Malik RA, Kallinikos P, Abbott CA, et al. Corneal confocal microscopy: a non-invasive surrogate of nerve fibre damage and repair in diabetic patients. Diabetologia. 2003;46(5):683–688. doi: 10.1007/s00125-003-1086-8. [DOI] [PubMed] [Google Scholar]
- 6.Tavakoli M, Kallinikos P, Iqbal A, et al. Corneal confocal microscopy detects improvement in corneal nerve morphology with an improvement in risk factors for diabetic neuropathy. Diabetic Medicine. 2011;28(10):1261–1267. doi: 10.1111/j.1464-5491.2011.03372.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dogru M, Goebbels MJ. Tear secretion and tear film function in insulin dependent diabetics. British Journal of Ophthalmology. 2000;84, article 1210 doi: 10.1136/bjo.84.10.1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Inoue K, Kato S, Ohara C, Numaga J, Amano S, Oshika T. Ocular and systemic factors relevant to diabetic keratoepitheliopathy. Cornea. 2001;20(8):798–801. doi: 10.1097/00003226-200111000-00004. [DOI] [PubMed] [Google Scholar]
- 9.Herrero-Vanrell R, Peral A. International dry eye workshop (DEWS). Update of the disease. Archivos de la Sociedad Espanola de Oftalmologia. 2007;82(12):733–734. doi: 10.4321/s0365-66912007001200002. [DOI] [PubMed] [Google Scholar]
- 10.Buchholz P, Steeds CS, Stern LS, et al. Utility assessment to measure the impact of dry eye disease. The Ocular Surface. 2006;4(3):155–161. doi: 10.1016/s1542-0124(12)70043-5. [DOI] [PubMed] [Google Scholar]
- 11.Saito J, Enoki M, Hara M, Morishige N, Chikama T, Nishida T. Correlation of corneal sensation, but not of basal or reflex tear secretion, with the stage of diabetic retinopathy. Cornea. 2003;22(1):15–18. doi: 10.1097/00003226-200301000-00004. [DOI] [PubMed] [Google Scholar]
- 12.Alves M, Calegari VC, Cunha DA, Saad MJA, Velloso LA, Rocha EM. Increased expression of advanced glycation end-products and their receptor, and activation of nuclear factor kappa-B in lacrimal glands of diabetic rats. Diabetologia. 2005;48(12):2675–2681. doi: 10.1007/s00125-005-0010-9. [DOI] [PubMed] [Google Scholar]
- 13.Rosenberg ME, Tervo TMT, Immonen IJ, Muller LJ, Gronhagen-Riska C, Vesaluoma MH. Corneal structure and sensitivity in type 1 diabetes mellitus. Investigative Ophthalmology & Visual Science. 2000;41(10):2915–2921. [PubMed] [Google Scholar]
- 14.Hom M, De Land P. Self-reported dry eyes and diabetic history. Optometry. 2006;77(11):554–558. doi: 10.1016/j.optm.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 15.McMonnies CW, Ho A. Responses to a dry eye questionnaire from a normal population. Journal of the American Optometric Association. 1987;58(7):588–591. [PubMed] [Google Scholar]
- 16.Guillon J. Non-invasive tearscope plus routine for contact lens fitting. Contact Lens and Anterior Eye. 1998;21(1):S31–S40. doi: 10.1016/s1367-0484(98)80035-0. [DOI] [PubMed] [Google Scholar]
- 17.Patel DV, Tavakoli M, Craig JP, Efron N, McGhee CNJ. Corneal sensitivity and slit scanning in vivo confocal microscopy of the subbasal nerve plexus of the normal central and peripheral human cornea. Cornea. 2009;28(7):735–740. doi: 10.1097/ICO.0b013e318193e0e3. [DOI] [PubMed] [Google Scholar]
- 18.Grading diabetic retinopathy from stereoscopic color fundus photographs—an extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991;98:786–806. [PubMed] [Google Scholar]
- 19.Cornblath DR, Chaudhry V, Carter K, et al. Total neuropathy score: validation and reliability study. Neurology. 1999;53(8):1660–1664. doi: 10.1212/wnl.53.8.1660. [DOI] [PubMed] [Google Scholar]
- 20.Misra S. Corneal microstructure in diabetes mellitus and its association with peripheral neuropathy and cardiac autonomic neuropathy [Ph.D. thesis] Auckland, New Zealand: University of Auckland; 2013. [Google Scholar]
- 21.Tomlinson A, Blades KJ, Pearce EI. What does the phenol red thread test actually measure? Optometry & Vision Science. 2001;78(3):142–146. doi: 10.1097/00006324-200103000-00005. [DOI] [PubMed] [Google Scholar]
- 22.Schein OD, Tielsch JM, Munoz B, Bandeen-Roche K, West S. Relation between signs and symptoms of dry eye in the elderly: a population-based perspective. Ophthalmology. 1997;104(9):1395–1401. doi: 10.1016/s0161-6420(97)30125-0. [DOI] [PubMed] [Google Scholar]
- 23.Tomlinson A, Giesbrecht C. Effect of age on human tear film evaporation in normals. Advances in Experimental Medicine and Biology. 1994;350:271–274. doi: 10.1007/978-1-4615-2417-5_46. [DOI] [PubMed] [Google Scholar]
- 24.Craig JP, Tomlinson A. Age and gender effects on the normal tear film. Advances in Experimental Medicine and Biology. 1998;438:411–415. doi: 10.1007/978-1-4615-5359-5_57. [DOI] [PubMed] [Google Scholar]
- 25.Maïssa C, Guillon M. Tear film dynamics and lipid layer characteristics—effect of age and gender. Contact Lens and Anterior Eye. 2010;33(4):176–182. doi: 10.1016/j.clae.2010.02.003. [DOI] [PubMed] [Google Scholar]
- 26.Sullivan DA, Sullivan BD, Ullman MD, et al. Androgen influence on the meibomian gland. Investigative Ophthalmology & Visual Science. 2000;41(12):3732–3742. [PubMed] [Google Scholar]
- 27.Ozdemir M, Buyukbese MA, Cetinkaya A, Ozdemir G. Risk factors for ocular surface disorders in patients with diabetes mellitus. Diabetes Research and Clinical Practice. 2003;59(3):195–199. doi: 10.1016/s0168-8227(02)00244-9. [DOI] [PubMed] [Google Scholar]
- 28.Herse PR. A review of manifestations of diabetes mellitus in the anterior eye and cornea. American Journal of Optometry and Physiological Optics. 1988;65(3):224–230. doi: 10.1097/00006324-198803000-00013. [DOI] [PubMed] [Google Scholar]
- 29.Tavakoli M, Quattrini C, Abbott C, et al. Corneal confocal microscopy: A novel noninvasive test to diagnose and stratify the severity of human diabetic neuropathy. Diabetes Care. 2010;33(8):1792–1797. doi: 10.2337/dc10-0253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ozdemir M, Temizdemir H. Age- and gender-related tear function changes in normal population. Eye. 2010;24(1):79–83. doi: 10.1038/eye.2009.21. [DOI] [PubMed] [Google Scholar]
- 31.Zealand SN, editor. 2013 Census QuickStats about Culture and Identity. Statistics New Zealand; 2014. [Google Scholar]
- 32.Willis JA, Scott RS, Darlow BA, et al. Incidence of type 1 diabetes mellitus diagnosed before age 20 years in Canterbury, New Zealand over the last 30 years. Journal of Pediatric Endocrinology and Metabolism. 2002;15(5):637–643. doi: 10.1515/jpem.2002.15.5.637. [DOI] [PubMed] [Google Scholar]
- 33.Patel S, Virhia SK, Farrell P. Stability of the precorneal tear film in Chinese, African, Indian, and Caucasian eyes. Optometry and Vision Science. 1995;72(12):911–915. doi: 10.1097/00006324-199512000-00011. [DOI] [PubMed] [Google Scholar]
- 34.Chung CW, Tigges M, Stone RA. Peptidergic innervation of the primate meibomian gland. Investigative Ophthalmology and Visual Science. 1996;37(1):238–245. [PubMed] [Google Scholar]
- 35.Song XJ, Li D, Farley W, et al. Neurturin-deficient mice develop dry eye and keratoconjunctivitis sicca. Investigative Ophthalmology and Visual Science. 2003;44(10):4223–4229. doi: 10.1167/iovs.02-1319. [DOI] [PubMed] [Google Scholar]
- 36.Inoue K, Okugawa K, Amano S, et al. Blinking and superficial punctate keratopathy in patients with diabetes mellitus. Eye. 2005;19(4):418–421. doi: 10.1038/sj.eye.6701497. [DOI] [PubMed] [Google Scholar]
- 37.Craig JP, Tomlinson A. Importance of the lipid layer in human tear film stability and evaporation. Optometry and Vision Science. 1997;74(1):8–13. doi: 10.1097/00006324-199701000-00014. [DOI] [PubMed] [Google Scholar]
- 38.The definition and classification of dry eye disease: Report of the definition and classification subcommittee of the international Dry Eye WorkShop (2007) The Ocular Surface. 2007;5(2):75–92. doi: 10.1016/s1542-0124(12)70081-2. [DOI] [PubMed] [Google Scholar]
- 39.Tseng SCG, Hirst LW, Maumenee AE. Possible mechanisms for the loss of goblet cells in mucin-deficient disorders. Ophthalmology. 1984;91(6):545–552. doi: 10.1016/s0161-6420(84)34251-8. [DOI] [PubMed] [Google Scholar]
- 40.Cousen P, Cackett P, Bennett H, Swa K, Dhillon B. Tear production and corneal sensitivity in diabetes. Journal of Diabetes and its Complications. 2007;21(6):371–373. doi: 10.1016/j.jdiacomp.2006.05.008. [DOI] [PubMed] [Google Scholar]
- 41.Ishida N, Rao GN, Del Cerro M, Aquavella JV. Corneal nerve alterations in diabetes mellitus. Archives of Ophthalmology. 1984;102(9):1380–1384. doi: 10.1001/archopht.1984.01040031122038. [DOI] [PubMed] [Google Scholar]
- 42.Pritchard N, Edwards K, Vagenas D, et al. Corneal sensitivity as an ophthalmic marker of diabetic neuropathy. Optometry and Vision Science. 2010;87(12):1003–1008. doi: 10.1097/OPX.0b013e3181fd6188. [DOI] [PubMed] [Google Scholar]
- 43.Zagon IS, Klocek MS, Sassani JW, McLaughlin PJ. Dry eye reversal and corneal sensation restoration with topical naltrexone in diabetes mellitus. Archives of Ophthalmology. 2009;127(11):1468–1473. doi: 10.1001/archophthalmol.2009.270. [DOI] [PMC free article] [PubMed] [Google Scholar]


