Significance
The possibility that terminally differentiated hypertrophic chondrocytes could survive and become osteoblasts in vivo has been debated for more than a century. We show that hypertrophic chondrocytes can survive the cartilage-to-bone transition and become osteoblasts and osteocytes during endochondral bone formation and in bone repair. Our discovery provides the basis for a conceptual change of a chondrocyte-to-osteoblast lineage continuum, with new insights into the process of endochondral bone formation, the ontogeny of bone cells, and bone homeostasis. Furthermore, our findings have implications for current concepts on mechanisms of skeletal disorders and bone repair and regeneration.
Keywords: osteoblast ontogeny, chondrocyte lineage, bone repair
Abstract
According to current dogma, chondrocytes and osteoblasts are considered independent lineages derived from a common osteochondroprogenitor. In endochondral bone formation, chondrocytes undergo a series of differentiation steps to form the growth plate, and it generally is accepted that death is the ultimate fate of terminally differentiated hypertrophic chondrocytes (HCs). Osteoblasts, accompanying vascular invasion, lay down endochondral bone to replace cartilage. However, whether an HC can become an osteoblast and contribute to the full osteogenic lineage has been the subject of a century-long debate. Here we use a cell-specific tamoxifen-inducible genetic recombination approach to track the fate of murine HCs and show that they can survive the cartilage-to-bone transition and become osteogenic cells in fetal and postnatal endochondral bones and persist into adulthood. This discovery of a chondrocyte-to-osteoblast lineage continuum revises concepts of the ontogeny of osteoblasts, with implications for the control of bone homeostasis and the interpretation of the underlying pathological bases of bone disorders.
In vertebrates, the endochondral bones of the axial and appendicular skeleton (1) develop from mesenchymal progenitors that form condensations in the approximate shape of the future skeletal elements. These progenitors differentiate into chondrocytes, which proliferate, mature, and undergo hypertrophy, forming an avascular cartilaginous template surrounded by a perichondrium. The first osteoblasts differentiate from mesenchymal precursors in the perichondrium and produce a bone collar, which will become the future cortical bone (1). Blood vessels then invade through the bone collar into the hypertrophic cartilage, bringing in osteoblast progenitors from the perichondrium (2), which lay down bone matrix to form the primary ossification center (POC); the cartilage matrix is degraded; and the proximal and distal growth plates, comprising layers of differentiating chondrocytes and spongy/trabecular bone (the primary spongiosa), form (2). Thereafter, linear bone growth continues by endochondral ossification mediated by the growth plate, whereas osteoblasts in the perichondrium form cortical bone on the outer circumference.
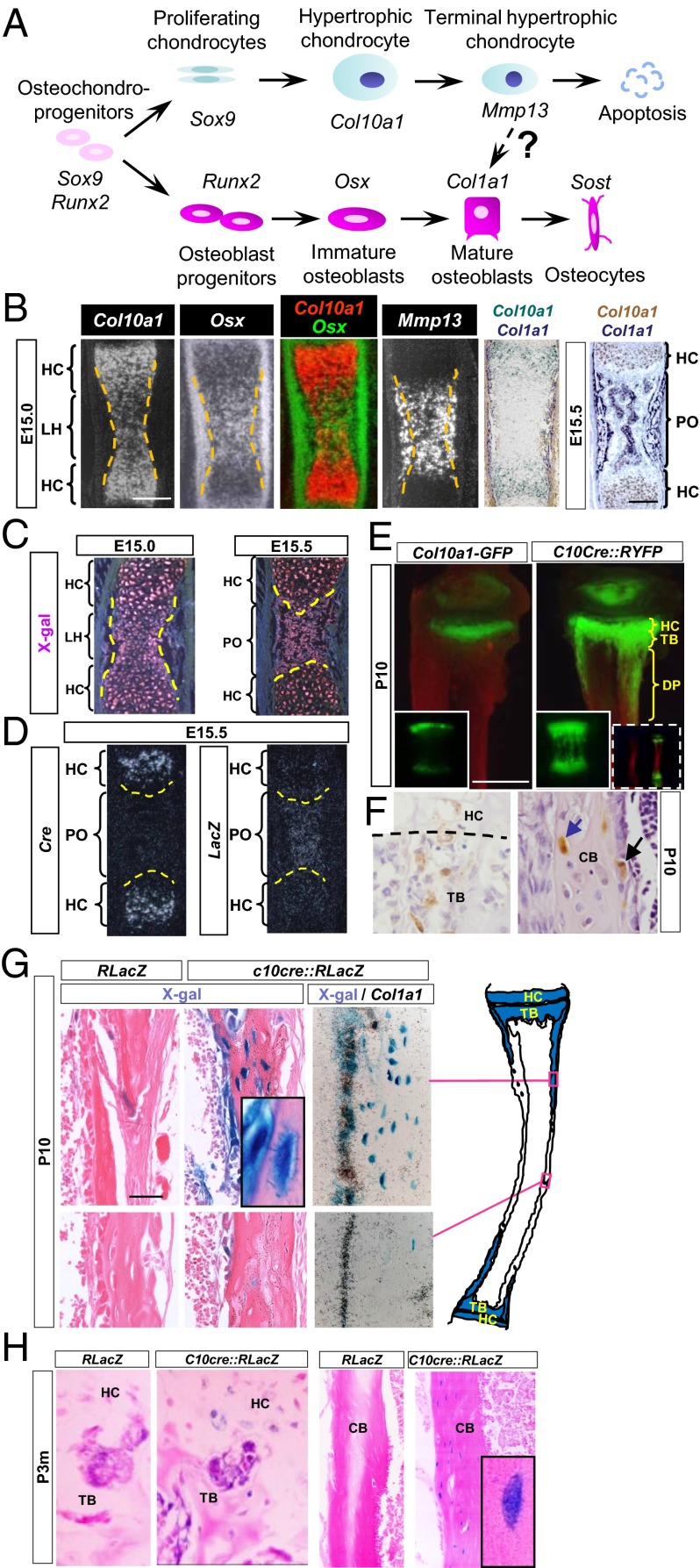
Chondrocytes and osteoblasts are regarded as separate lineages in development, being derived from common mesenchymal progenitors that express the transcription factors sex determining region Y (SRY)-box 9 (Sox9) and runt related transcription factor 2 (Runx2) (1). Lineage determination toward the chondrocyte or osteoblast fate is controlled by the relative expression of Sox9 and Runx2 (3–5) (Fig. 1A). Sox9 controls chondrocyte proliferation and their progression into hypertrophy (6). Collagen X is the most specific marker of hypertrophic chondrocytes (HCs), the Col10a1 gene being expressed only in prehypertrophic and hypertrophic chondrocytes in the growth plate (7). By contrast, Runx2 is essential for specifying the osteoblast lineage and directly regulates another transcription factor, osterix (Osx) (8, 9), but is also expressed by HCs. It regulates Col10a1 and matrix metalloproteinase-13 (Mmp13) expression in HCs (10, 11). Osx is expressed in prehypertrophic chondrocytes and osteoblasts and is essential for preosteoblast differentiation (9): it directly transactivates Col1a1, which encodes collagen I, a marker of differentiated osteoblasts.
Fig. 1.
HCs contribute to osteoblastic lineage in mouse endochondral bone. (A) Current view of chondrocyte and osteoblast lineages. (B) In situ hybridization showing mRNA expression of indicated genes during POC formation in E15.0 and E15.5 tibia. Dotted lines indicate the cartilage and perichondrium border. (Scale bar, 200 μm.) (C) LacZ expression (by X-Gal staining, which is pink under dark field) in HCs at E15.0 and HC-derived cells at E15.5 in C10cre::RLacZ tibia. Dotted lines indicate the chondro-osseous junction. (D) Cre and LacZ mRNA at E15.5 in C10cre::RLacZ tibia. (E) Fluorescent signals in P10 tibia of Col10a1-GFP and C10cre::RYFP mice. (Insets) Vertebra. Whole tibiae (wild type and C10cre::RYFP) are shown in dashed Insets. (Scale bar, 1 mm.) (F) P10 tibia of C10cre::Z/EG mouse stained by GFP antibody (brown). Dotted line represents the chondro-osseous junction. GFP-expressing osteoblasts and osteocytes are denoted by black and blue arrows, respectively. (G) X-Gal staining (blue) of P10 C10cre::RLacZ and control tibiae. (Inset) LacZ+ osteocyte and bone surface osteoblast LacZ+ cell. Col1a1 in situ hybridization reveals X-Gal, Col1a1 double-positive cells in the endosteum. The locations of the sections are denoted in the cartoon on the right. (Scale bar, 100 µm.) (H) X-Gal staining of 3-mo-old tibia from C10cre::RLacZ and control. CB, cortical bone; DP, diaphysis; LH, zone of late HCs; PO, primary ossification center; PS, primary spongiosa; TB, trabecular region.
Because maintenance of bone throughout life requires continuous renewal of osteoblasts, their lineage has been the subject of intense interest. The possibility that HCs are an alternative source of osteoblasts is controversial (12). Support comes from imaging, morphological, and ultrastructural studies in vivo, in which HCs were observed at the chondro-osseous junction and osteoblasts in chondrocyte lacuna (13–18). Recent lineage studies failed to resolve this issue because non–HC-specific reagents were used to track the fate of HCs (2, 19, 20) or because the half-life of fluorescent protein tracers in HCs may not be sufficient to span a possible HC-to-osteoblast transition (21). Studies showing the presence of apoptotic nuclei in HCs and abundant apoptogens in the microenvironment of HCs at the chondro-osseous junction provided the current concept that death, by apoptosis or extended autophagy, is the fate of HCs in endochondral ossification (12, 22). However, these studies cannot preclude that some HCs survive.
To determine whether HCs contribute to the osteoblast lineage in vivo, we used the Cre/loxP genetic recombination approach to tag specifically HCs and follow their fate. We show that the descendants of HCs may become Col1a1-expressing osteoblasts and sclerostin (SOST)-expressing osteocytes in prenatal and postnatal bones and in bone injury repair. We therefore provide evidence that the HC is part of a continuum that directly contributes to the osteoblast lineage.
Results and Discussion
Col10a1-Cre Activity Specifically Labels HCs for Lineage Analyses.
At embryonic day 15 (E15.0) in the tibia, just before the formation of the POC that separates the hypertrophic zone (HZ) into the proximal and distal parts of the developing skeletal element, osteoblast differentiation, characterized by Col1a1 expression, begins in the bone collar immediately adjacent to HCs in the HZ. By contrast, Col10a1 expression is restricted specifically to HCs and is not expressed in the bone collar. At this stage in the middle of the HZ, down-regulation of Col10a1 expression and up-regulation of Mmp13 and Osx were observed (Fig. 1B) (23, 24). We denote HCs in this region as “late HCs” (LHs). Col10a1-expressing HCs and LHs, however, do not express the differentiated osteoblast marker, Col1a1 (Fig. 1B).
The expression of preosteoblastic markers in LHs before the formation of the POC raises the possibility that these cells may transition to an osteoblastic fate (Fig. 1B). To tag and trace the fate of HCs, we used their specific expression of Col10a1. We generated, by gene targeting in embryonic stem cells, a Col10a1-Cre (abbreviated C10cre) mouse that expresses Cre recombinase under the control of the endogenous Col10a1 promoter (25) (Fig. S1A) and crossed it with mice harboring Cre-reporter in the Rosa26R locus that encodes either β-galactosidase (RLacZ) or yellow fluorescent protein (RYFP), or LacZ/enhanced green fluorescent protein (Z/EG) under the control of the β-actin promoter. Cre activity irreversibly marks, by expression of reporter gene, HCs in which recombination has occurred. This reporter continues to express in their progeny/descendants, even when Cre is not active. In C10cre::RLacZ mice before the POC is formed at around E15.5 in tibia, only HCs express Cre (E14.5; Fig. S1A) and LacZ activity as reflected by X-Gal staining (E15.0; Fig. 1C). No expression of Cre or LacZ was detected in the bone collar/periosteum.
HC-Derived Cells Are Present in Fetal, Neonatal, and Adult Bone.
At E15.5 and later, while Cre transcripts were restricted to HCs, LacZ+ cells also were present in the newly formed POC (Fig. 1 C and D and Fig. S1B). The presence of Cre−;LacZ+ cells in the POC reflects previous Cre-mediated activation of RLacZ transcription in HCs, which continues in the HC descendent cells, indicating that HC-derived cells survive in the POC. To assess whether HC-derived cells also were present in postnatal bone, we compared Col10a1-GFP mice (Fig. 1E), in which GFP expression is regulated by the endogenous Col10a1 gene, with C10cre::RYFP mice. At postnatal day 10 (P10) in Col10a1-GFP mice, GFP expression clearly was restricted to the HCs in the HZ, whereas in C10cre::RYFP mice, YFP expression extended beyond the HZ, indicating the presence of descendant cells in all the endochondral bones studied (Fig. 1E and Fig. S2). Importantly, no LacZ+ or YFP+ cells were found in the perichondrium/periosteum. There were no YFP-expressing cells in the calvaria at P10 (Fig. S2), which develops by direct differentiation of osteoblasts from mesenchymal cells (intramembranous ossification), confirming the specificity of Cre recombinase activity in HCs.
The HC-derived YFP+ cells, morphologically resembling osteoblasts, were found close to the chondro-osseous junction, on the surface of trabeculae, and in the endosteum (Fig. 1F). YFP+ and LacZ+ also were present within the cortical bone matrix as osteocyte-like cells with extended cellular processes typical of osteocytes (Fig. 1 F and G). In C10cre::RLacZ mice, LacZ+ HC-derived cells found in the endosteum did not express Col10a1 but expressed Col1a1, suggesting lineage progression of HCs to osteoblasts and osteocytes in vivo (Fig. 1G). The presence of LacZ+ cells at the chondro-osseous junction and in cortical bone of 3-mo-old (P3m) C10cre::RLacZ mice (Fig. 1H) suggests that HCs continue to commit to the osteogenic fate in adulthood and may thrive as osteocytes. To exclude the possibility that Col10a1 heterozygosity caused by targeted Cre insertion and consequential reduced expression of collagen X may induce HCs to become osteoblasts, we generated transgenic mice expressing a BAC-C10cre transgene under the control of a Col10a1 promoter and flanking regulatory elements. BAC-C10cre::RLacZ transgenic mice also showed specific expression of Cre in HCs and the presence of HC-derived cells in the trabecular bone (Fig. S3), similar to that of C10cre::RLacZ mice. These data suggest that heterozygosity for Col10a1 does not cause the lineage transition and are consistent with previous reports showing that bone development is not affected in Col10a1 null mice (26, 27).
Tamoxifen-Inducible C10CreERt Activity Facilitates Lineage Tracing of Tagged Populations of HCs.
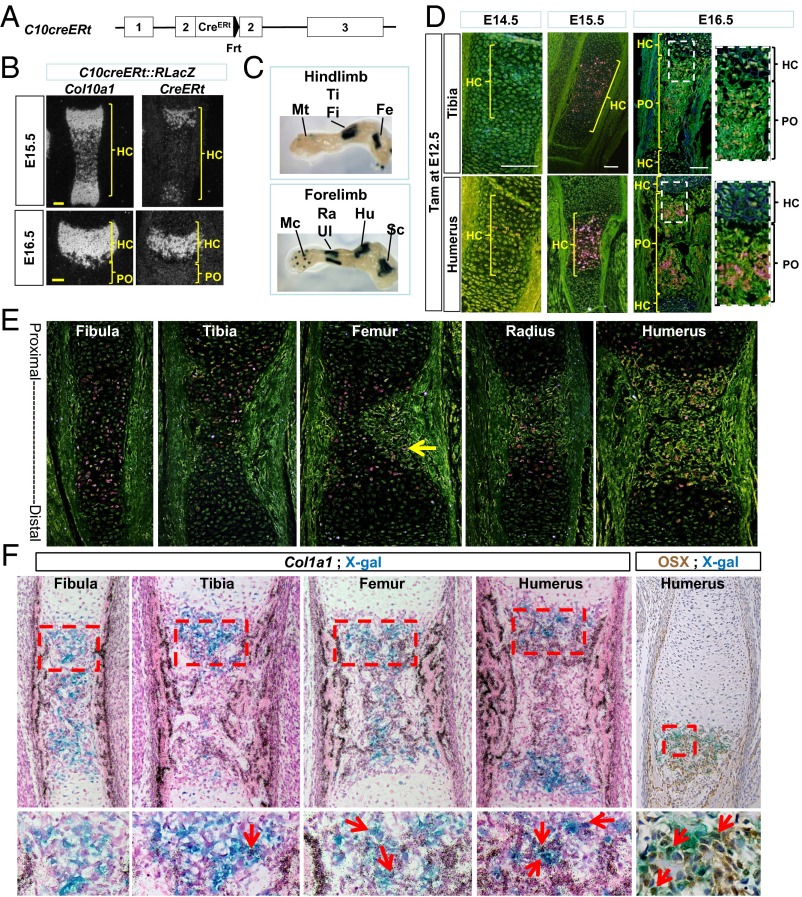
In the C10cre::RLacZ and C10cre::RYFP systems, HCs are continuously marked by the reporters for as long as Col10a1 is expressed. To follow a specific population of HCs, we adopted a genetically controlled pulse-chase approach. We generated mice expressing CreERt controlled by the endogenous Col10a1 gene by gene targeting (Fig. 2A and Fig. S4A). CreERt is a Cre recombinase fused with a modified estrogen receptor ligand-binding domain (ERt) that becomes active through nuclear localization in the presence of tamoxifen (tam) (28). In Col10a1-CreERt (abbreviated C10creERt) mice, the temporal and spatial pattern of CreERt mRNA expression is indistinguishable from that of Col10a1 and C10cre (Fig. 2B and Fig. S4B). A pulse injection of tam in C10creERt::RlacZ mice marks HCs generated during a defined time window in development.
Fig. 2.
Generation and characterization of the C10creERt mouse line. (A) Targeted Col10a1::CreERt allele (detailed in Fig. S4). (B) In situ hybridization showing Col10a1 and CreERt mRNA in C10creERt::RLacZ at E14.5 and E16.5. (C) Proximal-to-distal temporal progression of hypertrophy and transition shown in X-Gal–stained E15.5 C10cre::RLacZ limb skeletal elements. (D) X-Gal–stained tibia and humerus of E14.5–E16.5 C10creERt::RLacZ mice after tam injection at E12.5. (E) X-Gal–stained bone sections harvested 24 h post tam injection at E14.5 from the same C10creERt::RLacZ fetus. Arrow indicates LacZ+ (pink) cell in the forming POC of femur. (F) Col1a1 in situ hybridization on X-Gal–stained sections of different E16.0 bones from the same C10creERt::RLacZ fetus, harvested 36 h post tam injection at E14.5. Arrows in the enlarged areas of the POC indicate LacZ+ and Col1a1 double-positive cells. (Scale bar, 100 μm.) Fe, femur; Fi, fibula; Hu, humerus; Mc, metacarpal; Mt, metatarsal; PO, primary ossification center; Ra, radius; Sc, scapula; Ti, tibia; Ul, ulna. B, D, and E are dark-field images.
To test the specificity of the tam-inducible Cre activity, we administered tam to C10creERt::RlacZ mice at E12.5, when chondrogenesis begins, and compared the different bones according to their developmental sequence, in which the distal bones lag behind the proximal elements (29)(Fig. 2C). At E14.5, LacZ+ cells were not detected in the humerus or tibia (Fig. 2D). HCs normally differentiate around E13.5 and E14.5, but none of them was LacZ+, most likely because of the time lag required for CreERt activation and recombinase action. There was no leakage of Cre activity in the absence of tam (Fig. S5A); nonspecific X-Gal staining was negligible in fetal and early postnatal stages (Fig. S5 B and C).
At E15.5, many LacZ+ HCs were observed in the HZ of the humerus and a smaller population in the tibia (Fig. 2D), consistent with the general earlier development of the forelimbs (29) and the time required for Cre activation of β-galactosidase activity following hypertrophy. At E16.5, there were fewer LacZ+ HCs in the HZ, but many were present in the developing POC of the humerus (Fig. 2D), consistent with a transition of HCs into bone.
HC Derivatives Transit to the Primary Spongiosa and Become Col1a1-Expressing Cells.
To visualize HCs in their transit from the HZ to their descendants in the POC, we exploited the different temporal progression of chondrocyte differentiation and ossification from proximal to distal skeletal elements (29). We administered tam at E14.5, just before POC formation in the long bones. By 24 h, in the same fetus, the distal elements of the hindlimb, fibula, and tibia had just initiated the formation of the POC, and all LacZ+ cells were HCs (Fig. 2E). In the femur, POC formation was more advanced; other than LacZ+ HCs, a few LacZ+ cells could be found in the forming POC (arrow in Fig. 2E). POC formation had just been completed in the radius of the forelimb, and more LacZ+ cells were present there (Fig. 2E). The POC of the humerus was most mature, and many LacZ+ cells were present (Fig. 2E). These LacZ+ cells were not expressing Col1a1 (Fig. S6); however, at 36 h after tam injection, some LacZ+ cells expressed Col1a1 and OSX, predominantly in the proximal elements, especially the humerus (arrows in Fig. 2F). Importantly, at no stage were LacZ+ cells found in the bone collar, confirming the HC origin of these cells. These data reveal that HCs give rise to immature OSX-expressing osteoblasts (preosteoblasts) and the more mature Col1a1-expressing differentiated osteoblasts to the POC.
Our data are in contrast to a cell fate mapping study of chondrocytes using a Col2a1CreERt mouse (30), in which after administration of tam at E14.5 to mark Col2a1-expressing chondrocytes, LacZ+ cells were not detected in the primary spongiosa of the humerus at E17.5 (2) (Fig. S7). Interestingly, using the same Col2a1CreERt strain, when we injected tam a day earlier at E13.5, we detected LacZ+ cells in the primary spongiosa of the femur at E17.5 (Fig. S7). The data suggest that the 3-d time frame of tam activity between E14.5 and E17.5 may be insufficient to mark and track proliferating chondrocytes all the way to HC-derived osteoblasts in the primary spongiosa and that tam injection at E14.5 also labeled the perichondrial cells. The observed labeling of perichondrial cells most likely is a result of Col2a1CreERt expression in osteoblast progenitors in the perichondrium, reflecting the perichondrial origin of osteoblasts.
Osteoblasts and Osteocytes Derived from Fetal HCs Are Present in Neonatal and Adult Bone.
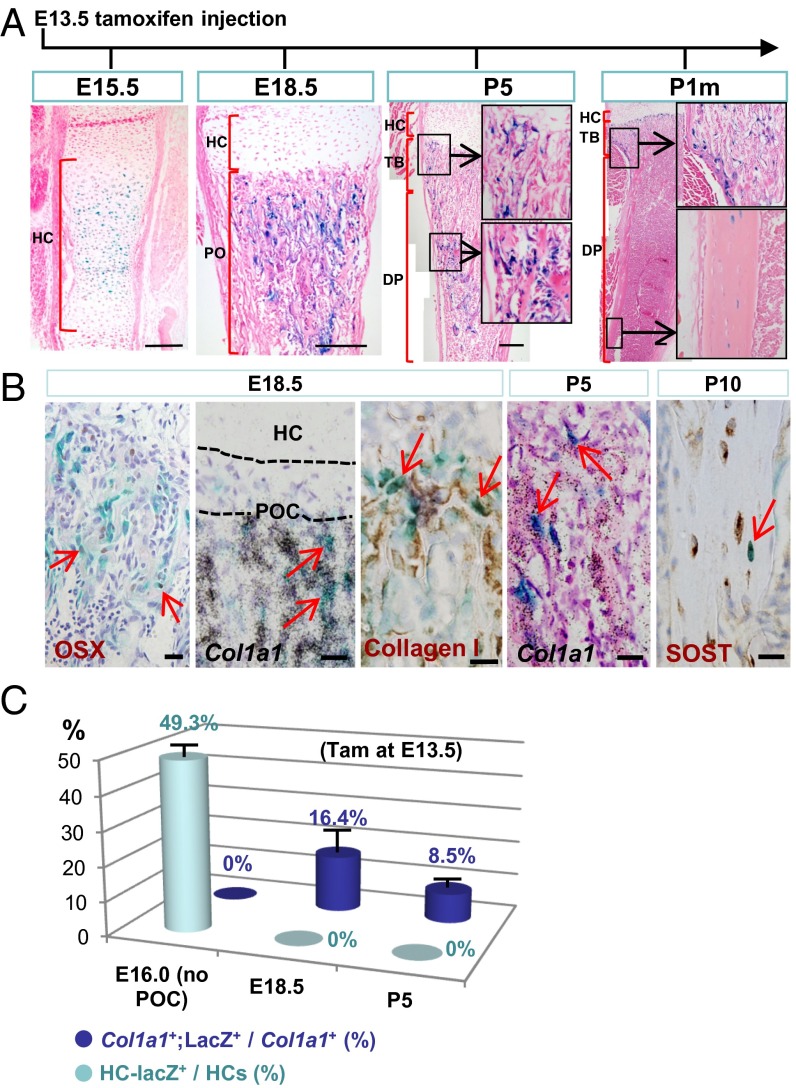
To assess whether fetal HC-derived cells persist in postnatal bone long term, we tracked the fate of HCs in the tibia, which grows at a relatively constant rate (29). We injected tam at E13.5 and monitored the fate of HC descendants daily until E18.5, and then at P5 and P1m. LacZ+ cells were detected at every stage, with HC-derived cells present in the trabecular and cortical bones, including the endosteum region (Fig. 3A and Fig. S8A). Again, HC-derived cells were not detected in the bone collar and periosteum, where osteoblasts are derived directly from mesenchymal progenitors (8). A parallel set of experiments was performed using the RYFP reporter, with similar results (Fig. S8B), independently confirming the HC-to-osteoblast transition.
Fig. 3.
HCs contribute to the full osteoblast lineage revealed by tam-inducible lineage tracing. (A and B) X-Gal staining of C10creERt::RLacZ tibiae sections at E15.5 and E18.5 and postnatal stages (P5, P1m) after tam injection at E13.5. In B, LacZ+ cells were found to express OSX, Col1a1, and SOST. (Scale bar, 200 μm.) (C) The proportion of tagged HCs [HC-LacZ+/HCs (%)] in the HZ and tagged Col1a1-expressing cells [Col1a1+;LacZ+/Col1a1+ (%)] in fetal and postnatal ossification centers. Tam was injected at E13.5, and tibiae were X-Gal stained at E16.0, E18.5, and P5 (n = 5 for each stage). Col1a1-expressing cells (by in situ hybridization) in the trabecular bone and endosteum were counted. DP, diaphysis; PO, primary ossification center; TB, trabecular region.
Molecular characterization of the HC-derived cells showed that the LacZ+ cells in fetal and postnatal bone expressed OSX and Col1a1, consistent with preosteoblast and osteoblast identities, respectively (Fig. 3B). At P10, LacZ+ cells expressing sclerostin also were found in cortical bone, with morphological features of osteocytes (Fig. 3B), suggesting that once committed, differentiation of the labeled HCs to osteoblasts and osteocytes follows that of the canonical osteoblast-to-osteocyte lineage, and these cells persist into adulthood.
Most HC-Derived Cells Become Osteoblasts.
To gain insight into the dynamics of chondrocytic-to-osteoblastic transition, we administered tam at E13.5 and estimated the proportions of LacZ+ cells in the HC population and in the Col1a1+ osteoblastic population in the tibia at E16.0, E18.5, and P5. At E16.0, in the absence of POC, about 49.3% of the HCs were labeled (Fig. 3C). At E18.5 and P5, none of the HCs in the HZ was LacZ+ and about 16.4% (at E18.5) and 8.5% (at P5) of the Col1a1-expressing osteoblastic cell population were LacZ+ in the trabecular and endosteal compartments (Fig. 3C). Given that approximately half the HCs were labeled at E16.5 and that the continuous contribution of osteoblasts from the perichondrium Col1a1+;LacZ− cells likely would dilute the overall proportion of Col1a1+;LacZ+ cells, the data probably are an underestimation of the relative proportion of HC-derived osteoblasts to the total. However, the data do indicate a significant contribution of osteogenic activity from HC-derived cells. The exact proportion of total osteoblasts derived from HCs could not be determined, because it was not possible to simultaneously lineage-trace osteoblasts derived from the perichondrium/periosteum.
Approximately 80% of the LacZ+ HC-derived cells expressed Col1a1 at both E18.5 and P5, suggesting that most HC-derived cells become osteoblasts and that some HCs probably undergo apoptosis. However, because of the technical challenges of tagging and tracking populations of HCs in vivo, we cannot determine the fraction of HCs that undergo apoptosis.
Postnatal HCs Contribute to Bone Formation and Repair.
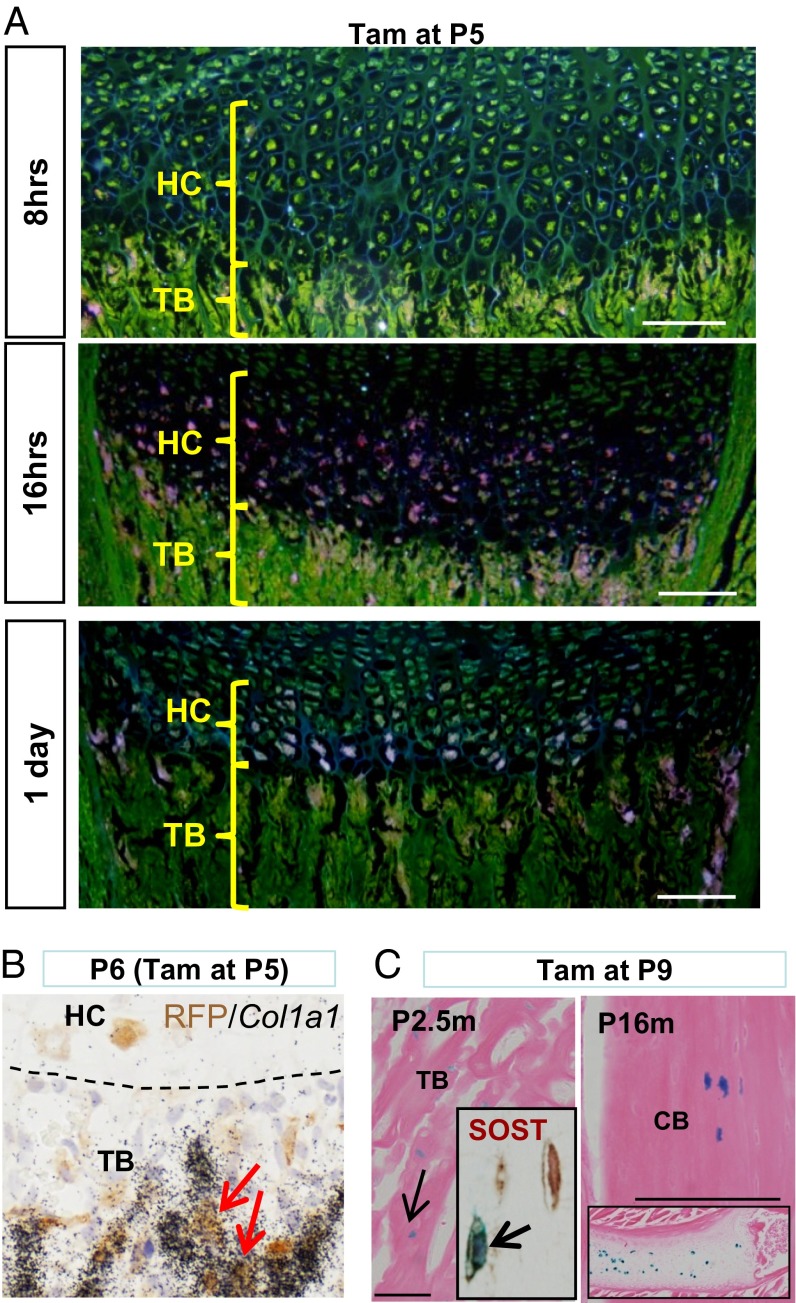
We further investigated whether this HC-to-osteoblast transition occurs during postnatal growth of endochondral bone by injecting tam at P5. At 8 h post injection, there were no LacZ+ HCs, reflecting the time lag required for activation of Cre and its RLacZ target (Fig. 4A). However, at 16 and 24 h post tam injection, we observed LacZ+ cells in the HZ and chondro-osseus junction (Fig. 4A), consistent with previous reports suggesting that HCs transit through the HZ within 48 h (25, 31). Some labeled HC descendants were detected expressing Col1a1 24 h post injection (Fig. 4B), suggesting that the time required to differentiate into osteoblast is faster postnatally. We also injected tam at P9 and found LacZ+ cells in the trabecular and cortical bones of the tibia at 2.5 and 16 mo post injection (Fig. 4C). Our data show that the HC-to-bone transition occurs during postnatal bone growth and that HC-derived cells may be long-lived within the mature bone.
Fig. 4.
Postnatal HCs may become osteoblasts and osteocytes. (A) LacZ activity (dark fields of X-Gal staining) in C10creERt::RLacZ tibia 8, 16, and 24 h following tam injection at P5. (Scale bar, 20 μm.) (B) Col1a1 mRNA expression in red fluorescent protein (RFP)-labeled cells in trabeculae of C10creERt::RtdTomato tibia 24 h after tam injection at P5. (C) LacZ+ osteocytes were found in 2.5- and 16-mo-old tibia after tam injection at P9. Insets show SOST-expressing LacZ+ osteocyte in P2.5m tibia, and persisting LacZ+ HCs in P16m tibia (top) and rib (bottom). (Scale bar, 100 μm.) CB, cortical bone; PO, primary ossification center; TB, trabecular region.
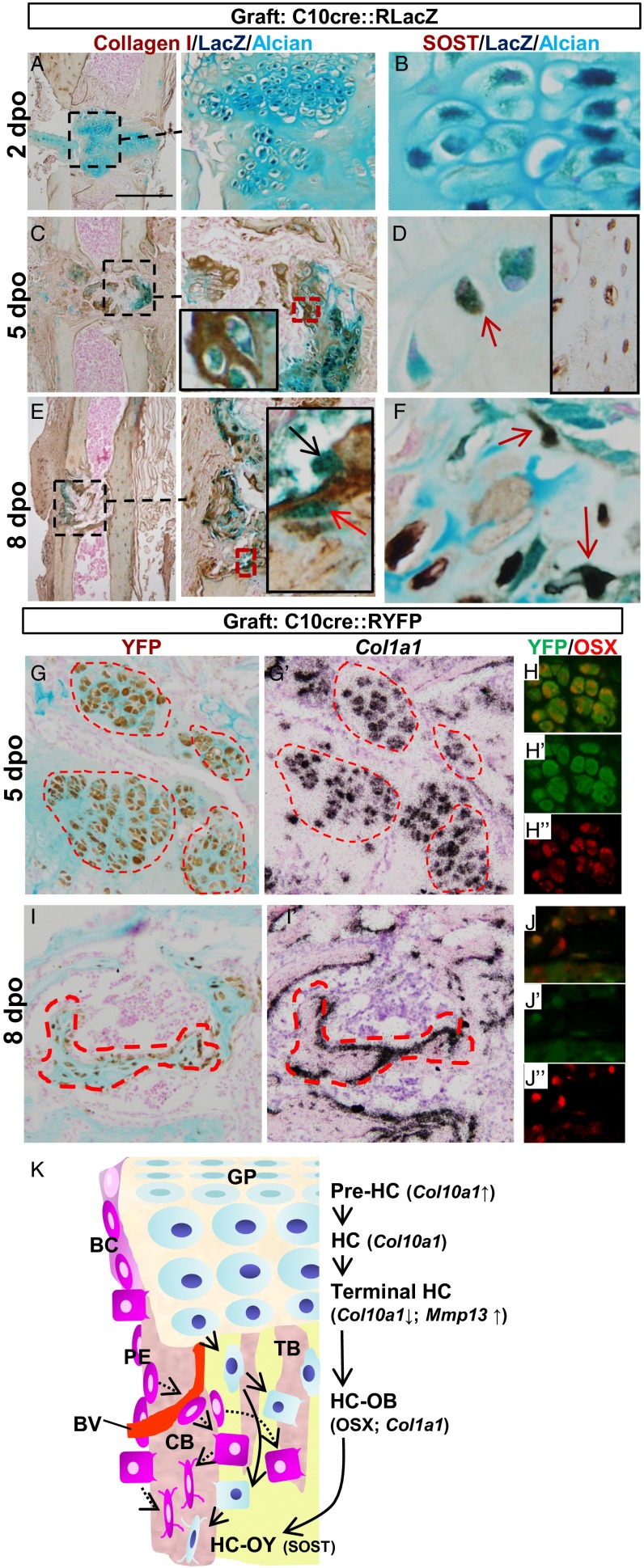
We further asked whether HCs may contribute to bone repair. We grafted pieces of hypertrophic cartilage isolated from P10 C10Cre::RLacZ/YFP pups into bone injury sites generated by drilling into the tibia of 3-mo-old adult female mice. Two days post operation (2 dpo), Alcian blue staining showed that the graft remained cartilaginous with LacZ+ HCs. Notably, collagen I and SOST were not expressed in the graft at this stage (Fig. 5 A and B). However, at 5 dpo, Alcian blue staining was reduced in the graft but collagen I and SOST were expressed (Fig. 5 C and D), indicating that remodeling of the cartilage matrix and bone formation had started. By 8 dpo, little cartilage was left at the injury site. However, LacZ+ osteoblast- and osteocyte-like cells could be found (Fig. 5D), with the latter expressing SOST (Fig. 5F). Because the HC-derived cells in the graft also expressed Col1a1 and OSX (Fig. 5 G–J), we conclude that postnatal HCs also can commit to osteogenic fate during bone repair.
Fig. 5.
HC-derived osteoblasts and osteocytes contribute to bone repair and revised concept of osteoblast ontogeny in endochondral bone. (A–F) Fate of LacZ-tagged HCs in grafts of P10 C10cre::RLacZ hypertrophic cartilage inserted into the injury sites in tibia of P3m adult females. Tibiae were analyzed for indicated markers 2, 5, and 8 dpo. Alcian blue and collagen I immunostaining marks cartilage and bone matrix, respectively. LacZ+ (blue) osteoblasts (black arrow) and osteocytes (red arrows) of graft origin were identified in the bone repair site. (G–J) Similar to A–F, graft hypertrophic cartilages from C10cre::RYFP mice were inserted into the bone injury site. YFP+ cells expressing Col1a1 and OSX were detected at 5 and 8 dpo. (K) A model for the ontogeny of osteoblasts in endochondral bone. Sources of osteoblasts are direct differentiation from periosteal mesenchymal cells to form cortical bone (CB), perichondrium-derived osteoblast progenitors accompanying vascular invasion of the POC, and HC transition to osteoblast lineages. BC, bone collar; BV, blood vessel; GP, growth plate; OB, osteoblast; OY, osteocyte; PE, periosteum; TB, trabecular bone.
An HC-to-Osteoblast Lineage Continuum in Endochondral Bone Formation.
The hypothesis that chondrocytes may become osteoblasts was proposed more than a century ago (32, 33); however despite data consistent with such a concept, this has eluded verification in vivo. The plasticity of chondrocytes in culture and their ability to change their phenotype to fibroblastic or osteoblastic states are well documented (34, 35). The reversion of HCs to a prehypertrophic-like state in response to endoplasmic reticulum (ER) stress suggests that hypertrophy is not an irreversible state in vivo (25). Here, we show that in normal endochondral bone formation, HCs can survive and become osteoblasts and osteocytes, contributing to trabecular bone, the endosteum, and mature bone. In addition, we show that postnatal HCs may contribute directly to bone repair by becoming osteoblasts and osteocytes. Our data are consistent with a recent report showing that grafted cartilage supports the regeneration of bone in vivo (36). We have resolved the longstanding question of whether an HC can become an osteoblast and contribute to the full osteogenic lineage. This discovery provides a conceptual change with regard to the origin of osteoblasts of endochondral bone and has important implications for bone biology.
We propose an osteoblast lineage model whereby HCs and perichondrial/periosteal osteoblast progenitors contribute to the osteogenic pool during endochondral bone formation and growth (Fig. 5K). The differential origin of osteoblasts may assure a steady supply of osteoblasts for building bone and provide diverse sources of progenitors for fracture repair. Whether the osteoblasts from differing origins have different, similar, or equal roles and contribution in maintaining bone homeostasis during growth, during aging, or in fracture repair are important issues to be addressed in the future. Knowing that HCs are a source of osteoblasts in bone, we also should re-examine the causes of bone phenotypes arising from mutations in genes that are expressed in cartilage.
Materials and Methods
Genetically Modified Mouse Strains.
Generation of the Col10a1-Cre mouse line was described previously (25). Generation of the Col10a1-CreERt mouse is described in Fig. S4. The strategy of generating the Col10a1-GFP mouse strain essentially was the same as that of generating the Col10a1-CreERt mouse: GFP fragment was isolated from the pEGFP-N1 plasmid (Clontech), fused with the neo cassette, and targeted into exon 2 of Col10a1. Reporter mouse strains used are listed in SI Materials and Methods.
X-Gal Staining, Immunohistochemistry, and in Situ Hybridization.
Tam (T5648; Sigma) was dissolved stepwise in ethanol and corn oil to a final concentration of 10 mg/mL. To induce Cre recombinase activity, tam was injected at a dosage of 0.1 mg/g body weight. Whole mount X-Gal staining was performed for LacZ reporter mice; in situ hybridization and antibody staining were performed on paraffin sections as previously described (23).
Bone Repair.
Hypertrophic cartilages were dissected from the distal tibiae of P10 donor mice. The bone collar was removed and the cartilage graft kept in HBSS. At the same time, the medial side of the right proximal tibia of an anesthetized 3-mo-old wild-type female was exposed and a 0.4-mm hole was drilled through the bone at the midpoint between the growth plate and the middle of the tibia. The grafts were inserted into the bone injury site, and the skin was sutured. The mice resumed normal activity after the operation, and the right tibiae were harvested at different time points for analyses.
Additional details are provided in SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Patrick Tam and C. C. Hui for helpful advice. We thank Nelson Dung for making constructs, Christine Ng and Sarah Fu for generating ES cell clones, Chi Leung So for blastocyst injection, and Anna Niewiadomsky for histological support. This work was funded by grants from the University Grants Committee of Hong Kong (AoE/M-04/04) and Hong Kong Research Grants Council (GRF7628-13M).
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1302703111/-/DCSupplemental.
References
- 1.Karsenty G, Kronenberg HM, Settembre C. Genetic control of bone formation. Annu Rev Cell Dev Biol. 2009;25:629–648. doi: 10.1146/annurev.cellbio.042308.113308. [DOI] [PubMed] [Google Scholar]
- 2.Maes C, et al. Osteoblast precursors, but not mature osteoblasts, move into developing and fractured bones along with invading blood vessels. Dev Cell. 2010;19(2):329–344. doi: 10.1016/j.devcel.2010.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Day TF, Guo X, Garrett-Beal L, Yang Y. Wnt/beta-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev Cell. 2005;8(5):739–750. doi: 10.1016/j.devcel.2005.03.016. [DOI] [PubMed] [Google Scholar]
- 4.Akiyama H, et al. Osteo-chondroprogenitor cells are derived from Sox9 expressing precursors. Proc Natl Acad Sci USA. 2005;102(41):14665–14670. doi: 10.1073/pnas.0504750102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhou G, et al. Dominance of SOX9 function over RUNX2 during skeletogenesis. Proc Natl Acad Sci USA. 2006;103(50):19004–19009. doi: 10.1073/pnas.0605170103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akiyama H, Chaboissier MC, Martin JF, Schedl A, de Crombrugghe B. The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev. 2002;16(21):2813–2828. doi: 10.1101/gad.1017802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kong RY, et al. Intron-exon structure, alternative use of promoter and expression of the mouse collagen X gene, Col10a-1. Eur J Biochem. 1993;213(1):99–111. doi: 10.1111/j.1432-1033.1993.tb17739.x. [DOI] [PubMed] [Google Scholar]
- 8.Otto F, et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell. 1997;89(5):765–771. doi: 10.1016/s0092-8674(00)80259-7. [DOI] [PubMed] [Google Scholar]
- 9.Nakashima K, et al. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002;108(1):17–29. doi: 10.1016/s0092-8674(01)00622-5. [DOI] [PubMed] [Google Scholar]
- 10.Li F, et al. Runx2 contributes to murine Col10a1 gene regulation through direct interaction with its cis-enhancer. J Bone Miner Res. 2011;26(12):2899–2910. doi: 10.1002/jbmr.504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hirata M, et al. C/EBPβ and RUNX2 cooperate to degrade cartilage with MMP-13 as the target and HIF-2α as the inducer in chondrocytes. Hum Mol Genet. 2012;21(5):1111–1123. doi: 10.1093/hmg/ddr540. [DOI] [PubMed] [Google Scholar]
- 12.Shapiro IM, Adams CS, Freeman T, Srinivas V. Fate of the hypertrophic chondrocyte: Microenvironmental perspectives on apoptosis and survival in the epiphyseal growth plate. Birth Defects Res C Embryo Today. 2005;75(4):330–339. doi: 10.1002/bdrc.20057. [DOI] [PubMed] [Google Scholar]
- 13.Boyde A, Shapiro IM. Morphological observations concerning the pattern of mineralization of the normal and the rachitic chick growth cartilage. Anat Embryol (Berl) 1987;175(4):457–466. doi: 10.1007/BF00309681. [DOI] [PubMed] [Google Scholar]
- 14.Yoshioka C, Yagi T. Electron microscopic observations on the fate of hypertrophic chondrocytes in condylar cartilage of rat mandible. J Craniofac Genet Dev Biol. 1988;8(3):253–264. [PubMed] [Google Scholar]
- 15.Riminucci M, et al. Vis-à-vis cells and the priming of bone formation. J Bone Miner Res. 1998;13(12):1852–1861. doi: 10.1359/jbmr.1998.13.12.1852. [DOI] [PubMed] [Google Scholar]
- 16.Farnum CE, Turgai J, Wilsman NJ. Visualization of living terminal hypertrophic chondrocytes of growth plate cartilage in situ by differential interference contrast microscopy and time-lapse cinematography. J Orthop Res. 1990;8(5):750–763. doi: 10.1002/jor.1100080517. [DOI] [PubMed] [Google Scholar]
- 17.Crelin ES, Koch WE. An autoradiographic study of chondrocyte transformation into chondroclasts and osteocytes during bone formation in vitro. Anat Rec. 1967;158(4):473–483. doi: 10.1002/ar.1091580410. [DOI] [PubMed] [Google Scholar]
- 18.Roach HI, Erenpreisa J, Aigner T. Osteogenic differentiation of hypertrophic chondrocytes involves asymmetric cell divisions and apoptosis. J Cell Biol. 1995;131(2):483–494. doi: 10.1083/jcb.131.2.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Metzger D, Clifford J, Chiba H, Chambon P. Conditional site-specific recombination in mammalian cells using a ligand-dependent chimeric Cre recombinase. Proc Natl Acad Sci USA. 1995;92(15):6991–6995. doi: 10.1073/pnas.92.15.6991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hilton MJ, Tu X, Long F. Tamoxifen-inducible gene deletion reveals a distinct cell type associated with trabecular bone, and direct regulation of PTHrP expression and chondrocyte morphology by Ihh in growth region cartilage. Dev Biol. 2007;308(1):93–105. doi: 10.1016/j.ydbio.2007.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Maye P, et al. Generation and characterization of Col10a1-mcherry reporter mice. Genesis. 2011;49(5):410–418. doi: 10.1002/dvg.20733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gibson G. Active role of chondrocyte apoptosis in endochondral ossification. Microsc Res Tech. 1998;43(2):191–204. doi: 10.1002/(SICI)1097-0029(19981015)43:2<191::AID-JEMT10>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 23.Stickens D, et al. Altered endochondral bone development in matrix metalloproteinase 13-deficient mice. Development. 2004;131(23):5883–5895. doi: 10.1242/dev.01461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Inada M, et al. Critical roles for collagenase-3 (Mmp13) in development of growth plate cartilage and in endochondral ossification. Proc Natl Acad Sci USA. 2004;101(49):17192–17197. doi: 10.1073/pnas.0407788101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tsang KY, et al. Surviving endoplasmic reticulum stress is coupled to altered chondrocyte differentiation and function. PLoS Biol. 2007;5(3):e44. doi: 10.1371/journal.pbio.0050044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rosati R, et al. Normal long bone growth and development in type X collagen-null mice. Nat Genet. 1994;8(2):129–135. doi: 10.1038/ng1094-129. [DOI] [PubMed] [Google Scholar]
- 27.Kwan KM, et al. Abnormal compartmentalization of cartilage matrix components in mice lacking collagen X: Implications for function. J Cell Biol. 1997;136(2):459–471. doi: 10.1083/jcb.136.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Feil R, et al. Ligand-activated site-specific recombination in mice. Proc Natl Acad Sci USA. 1996;93(20):10887–10890. doi: 10.1073/pnas.93.20.10887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Patton JT, Kaufman MH. The timing of ossification of the limb bones, and growth rates of various long bones of the fore and hind limbs of the prenatal and early postnatal laboratory mouse. J Anat. 1995;186(Pt 1):175–185. [PMC free article] [PubMed] [Google Scholar]
- 30.Nakamura E, Nguyen M-T, Mackem S. Kinetics of tamoxifen-regulated Cre activity in mice using a cartilage-specific CreER(T) to assay temporal activity windows along the proximodistal limb skeleton. Dev Dyn. 2006;235(9):2603–2612. doi: 10.1002/dvdy.20892. [DOI] [PubMed] [Google Scholar]
- 31.Farnum CE, Wilsman NJ. Determination of proliferative characteristics of growth plate chondrocytes by labeling with bromodeoxyuridine. Calcif Tissue Int. 1993;52(2):110–119. doi: 10.1007/BF00308319. [DOI] [PubMed] [Google Scholar]
- 32.Brachet A. Etude sur la resorption du cartilage et le developpement des os longs chez les oiseaux. J Anat Physiol. 1893;10:391–417. [Google Scholar]
- 33.van der Stricht O. Recherches sur le cartilage articulaire des oiseaux. Arch Biol (Liege) 1890;10:1–41. [Google Scholar]
- 34.Ishizeki K, Takigawa M, Nawa T, Suzuki F. Mouse Meckel’s cartilage chondrocytes evoke bone-like matrix and further transform into osteocyte-like cells in culture. Anat Rec. 1996;245(1):25–35. doi: 10.1002/(SICI)1097-0185(199605)245:1<25::AID-AR5>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 35.Zerega B, Cermelli S, Bianco P, Cancedda R, Cancedda FD. Parathyroid hormone [PTH(1-34)] and parathyroid hormone-related protein [PTHrP(1-34)] promote reversion of hypertrophic chondrocytes to a prehypertrophic proliferating phenotype and prevent terminal differentiation of osteoblast-like cells. J Bone Miner Res. 1999;14(8):1281–1289. doi: 10.1359/jbmr.1999.14.8.1281. [DOI] [PubMed] [Google Scholar]
- 36.Bahney CS, et al. Stem cell-derived endochondral cartilage stimulates bone healing by tissue transformation. J Bone Miner Res. 2014;29(5):1269–1282. doi: 10.1002/jbmr.2148. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.