Abstract
AIM: To study whether H pylori are associated with chronic cholecystitis.
METHODS: The subjects were divided into three groups: H pylori-infected cholecystitis group, H pylori-negative cholecystitis group and control group. Pathologic changes of the gallbladder were observed by optic and electronic microscopes and the levels of interleukin-1, 6 and 8 (IL-1, 6 and 8) were detected by radioimmunoassay.
RESULTS: Histological evidence of chronic cholecystitis including degeneration, necrosis, inflammatory cell infiltration, were found in the region where H pylori colonized. Levels of IL-1, 6 and 8 in gallbladder mucosa homogenates were significantly higher in H pylori-infected cholecystitis group than those in H pylori-negative cholecystitis group and control group.
CONCLUSION: H pylori infection may be related to cholecystitis.
Keywords: H pylori, Chronic cholecystitis, Interleukin, Colonization, Gallbladder mucosa
INTRODUCTION
H pylori have definite pathogenic action and related to gastritis, peptic ulcer and gastric carcinoma[1-3]. Previous studies have demonstrated that H pylori correlate with diseases of the extra-gastrointestine, the liver and the cholecyst[4-8]. We have isolated H pylori from the gallbladder and cultured H pylori, and preliminarily proved that there exist live H pylori in the gallbladder. In the present study, we carried out electron microscopic observation and immunohistochemistry to the relationship between H pylori and chronic cholecystitis.
MATERIALS AND METHODS
Subjects
A total of 81 cases with chronic cholecystitis were divided into H pylori-negative group (n = 59) and H pylori-infected group (n = 22), based on previous studies, PCR amplification and culture results for H pylori. Besides, a control group was used including another 20 cases who were proved to have no obvious inflammation in the gallbladder mucosa except polyps or H pylori infection after a cholecystectomy due to a gallbladder polyp.
Investigation of gastric mucosa metaplasia of epithelial cells of gallbladder mucosa
By histochemical staining of AB/PAS mucus, and based on histology of mucosal epithelial cells and characteristics of cells secreting mucus, an observation was made on gastric mucosa metaplasia of epithelial cells of gallbladder mucosa and colonization of H pylori in the epithelial cells of gallbladder mucosa. The relationship between H pylori and the epithelial cells of the gallbladder was observed by optic microscopy, W-S silver stain and immunohistochemical stain using anti-H pylori antibodies. The resected gallbladder specimens from cases with chronic cholecystitis were immobilized with 3% glutaral, embedded and sliced for transmission electron microscopic investigation.
Relationship between H pylori and cholecystitis
Inflammatory changes of epithelial cells of the gallbladder mucosa in regions where H pylori colonized were observed by optic microscopy and ultrastructural changes of epithelial cells of the gallbladder were observed by electron microscopy.
Assay of interleukins in homogenates of gallbladder mucosa
The radio-immune analytical reagent kits including IL-1, IL-6 and IL-8 were purchased from Dongya Research Institute of Biotechnology, Beijing. About 1 g of resected gallbladder mucosa was added into ultrapure water (Center of Molecular Biology, Research Institute of Surgery, Daping Hospital, Third Military Medical University, Chongqing) and homogenized in an IS-l homogenizer (Medical Machine Factory, Zhejiang) and the homogenates were centrifuged at 4000 r/min for l5 min, after which the supernatants were collected and frozen at -70°C. The EC-l200 radio-immune auto-γ counting device (Zhongjia Corporation of ChinaAcademy of Sciences) was employed for radio-immune assay and a fully automatic biochemical assay device of Beckman Synchron CX (USA) was used for quantification of proteins of gallbladder mucosa homogenates.
Statistical analysis
Data were expressed as mean ± SD and processed with Chi-square test and Student’s t-test. P < 0.05 was considered significant.
RESULTS
Metaplasia of gastric mucosa in gallbladder
In cases with a gallbladder polyp, no mucus stained positive for PAS was found in gallbladder mucosa in the region beyond the polyp, nor was positive substance of PAS, i.e., metaplasia of gastric mucosa, found in epithelial cells of mucosa. In cases with chronic lithic cholecystitis, the epithelium of gallbladder mucosa was column-like. AB/PAS stain showed that neutral mucus was positive, with metaplasia of epithelial cells of gastric mucosa (Figure 1).
Figure 1.
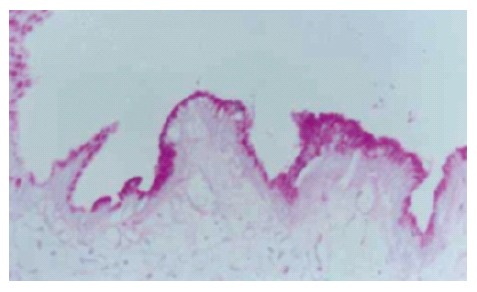
Specimens of chronic cholecystitis. Many positive PAS materials appear in epithelial cells of gallbladder mucosa (PAS × 200).
Gastric metaplasia of gallbladder mucosa appeared in 25 cases, accounting for 30.86% (25/81) of all cases with chronic cholecystitis, however, it was not found in the control group (Table 1). This suggested that gallbladder mucosa was apt to gastric metaplasia in cases with chronic lithiasis cholecystitis, especially in those with H pylori infection in the gallbladder, of gastric metaplasia of gallbladder mucosa (18/22, 81.82%) was significantly higher than that in cases with negative H pylori (7/59, 11.86%, P < 0.01). It indicated that gastric metaplasia of gallbladder mucosa might relate to H pylori infection in the gallbladder.
Table 1.
Gastric metaplasia of gallbladder mucosa in cases with chronic cholecystitis
| Group | n | Gastric metaplasia positive (n) | Gastric metaplasia negative (n) |
| Control | 20 | 0 | 20 |
| Chronic cholecystitis | |||
| Positive H pylori | 22 | 18 | 4 |
| Negative H pylori | 59 | 7 | 52 |
Colonization of H pylori in gallbladder and its relation to cholecystitis
The optic microscopy showed that H pylori were scattered or aggregated on, or located within certain distance from the epithelial cells of gallbladder mucosa and that individual H pylori distributed inside epithelial cells or existed in intercellular space. At the regions with H pylori, column-like cells secreted neutral mucus by AB/PAS stain, indicating that the gallbladder mucosa had gastric metaplasia and that gastric mucosa were absent in some regions where H pylori located. Electron microscopy showed that H pylori were located on, stuck to, or entered the epithelial cells of gallbladder mucosa, where, however, no tight junction or adhesiveness could be seen. Moreover, optic microscopy revealed degeneration of the epithelial cells of gallbladder mucosa, infiltration and exudation of inflammatory cells, exfoliation of the epithelial cells, or chronic inflammation such as mucous layer shrinkage, decrease or even disappearance of epithelial cells and glands at sites where H pylori were present. Sometimes, there could be seen that inflammatory cells aggregated around H pylori and the latter were swallowed (Figure 2). On the other hand, only a few inflammatory cells infiltrated the mucosa, with intact epithelial cells, in most cases with chronic cholecystitis without H pylori infection. Exceptionally, even in these cases, there emerged acute inflammatory manifestations including infiltration of large numbers of inflammatory cells, degeneration, apoptosis and exudation of epithelial cells or chronic inflammatory manifestations including atrophy of glands. In the gallbladder epithelial cells that were proved to have H pylori infection by electron microscopy, changes such as destructed epithelial cell membranes, loose cellular connection, dilatation of mitochondria, decrease or disappearance of crest, and dilatation of endoplasmic reticulum could be seen, which were severer compared with those without H pylori infection (Figure 3). The levels of IL-l, IL-6 and IL-8 of gallbladder mucosa homogenates were expressed as ng/g protein, and are shown in Table 2.
Figure 2.
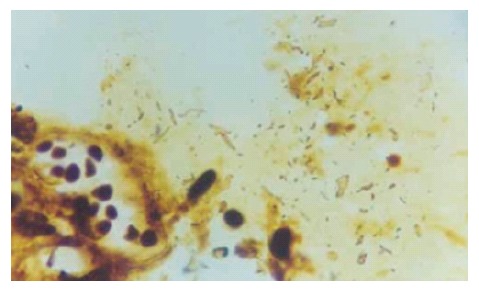
Helicobacter-like bacteria and inflammatory cells in mucus on gallbladder mucosa (WS × 200).
Figure 3.
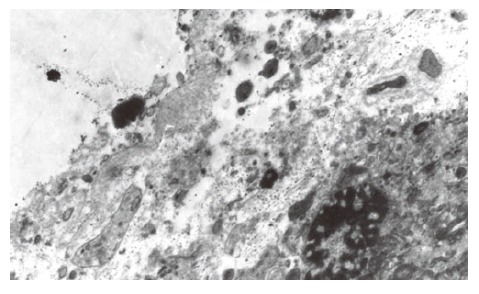
Electron microscopic images of H pylori on the epithelial cells of gallbladder (× 6000).
Table 2.
Levels of IL-l, IL-6 and IL-8 of gallbladder mucosa homogenates (ng/g protein)
| Groups | n | IL-1 | IL-6 | IL-8 |
| Control | 20 | 21.65 ± 4.28 | 77.10 ± 10.56 | 101.35 ± 19.39 |
| Chronic cholecystitis | ||||
| Negative H pylori | 59 | 68.76 ± 15.08b | 159.54 ± 37.65b | 152.10 ± 46.57 |
| Positive H pylori | 22 | 142.68 ± 25.41bd | 241.50 ± 80.60bd | 593.18 ± 93.59bd |
P < 0.01 vs control group;
P < 0.01 vs negative H pylori group.
In cases with chronic cholecystitis, the levels of IL-l, IL-6 and IL-8 in gallbladders in both negative H pylori group and positive H pylori group were significantly higher than those in control group (P < 0.01). Moreover, there was a significant difference between positive H pylori group and negative H pylori group in levels of IL-1, IL-6 and IL-8 in the gallbladder (P < 0.01).
DISCUSSION
The gallbladder and stomach are originated from endoblasts and have similar tissue structures, with the mucosa covered with a slime layer [9-11]. AB/PAS stain showed that the epithelial cells of gallbladder mucosa secreted neutral mucus, with gastric metaplasia, in about 31% (25/81) of cases with cholecystolithiasis. Caselli et al[12-14] also demonstrated that the epithelial cells of gallbladder mucosa had gastric metaplasia in cases with cholecystolithiasis. Roa et al[15,16] found that pepsinogenI, II were expressed in the epithelial cells of the gallbladder. The significance of gastric metaplasia of gallbladder mucosa lies in that the gastric metaplasia provides conditions for H pylori colonization in the gallbladder. The results of our study showed that compared with cases without H pylori infection in the gallbladder, there was a significantly higher incidence rate of gastric metaplasia in epithelial cells of gallbladder mucosa in cases with H pylori infection. It also proved that the gastric metaplasia of gallbladder mucosa closely correlated with H pylori infection in the gallbladder. Nevertheless, there were cases with H pylori infection but without gastric metaplasia in the gallbladder; meanwhile, there were cases with gastric metaplasia but without H pylori infection. These findings indicate that there is no absolute causality between gastric metaplasia and H pylori infection in the gallbladder. We hypothesize that many epithelial cells of the gastrointestinal tract have receptors for H pylori colonization factors. Therefore, H pylori can colonize on the epithelial cells of the gallbladder mucosa with no gastric metaplasia[17].
We also found that H pylori were separated from or adhered to the epithelial cells of gallbladder mucosa and that some H pylori penetrated through epithelial cells of the gallbladder. It that H pylori had a weak ability to pass through cells. At sites where H pylori aggregated, the epithelial cells of gallbladder mucosa were degenerated, erosive and even apoptotic. In some parts, inflammatory cells infiltrated, which became more obvious with increases in the number of H pylori. Electron microscopy revealed that at sites infected with H pylori, the integrity of the cell membrane of epithelial cells was destructed, with swelling of mitochondria and dilatation of endoplasmic reticulum. It showed that colonization of H pylori in the gallbladder cause inflammation of the gallbladder, mainly chronic nonsuppurative inflammation, which in turn provides an important condition for H pylori as one of the etiological factors leading to cholecystitis. Damage of the epithelial cells of gallbladder mucosa caused by H pylori may relate to specific virulence factors of H pylori such as cytotoxin-associated protein (CagA) and vacuoles toxin (VacA) , as well as urease, lipopolysaccharides and mucus enzyme of H pylori[18].
H pylori can also damage the epithelial cells of gallbladder mucosa through mediating inflammation and immunoreaction. The levels of IL-l, IL-6 and IL-8 in gallbladder mucosa homogenates in both H pylori negative and positive groups were significantly higher than those in control group (P < 0.01). It indicates that chronic lithic cholecystitis is associated with these three cytokines. We also found that in cholecystitis specimens with or without H pylori infection, levels of IL-1, IL-6 and IL-8 were significantly higher than those in control group, indicating that these interleukins may participate in pathogenesis of chronic cholecystitis. This may accord with the function of IL in H pylori-related gastritis and gastric ulcer[19,20]. After infection with H pylori, the urease, lipase and heat shock proteins secreted by H pylori can activate regional epithelial cells of mucosa and vascular endothelial cells expressing IL-1, IL-6 and other cytokines such as ICAM-I, hence stimulating and chemotaxy intravascular lymphocytes and monocytes to shift to H pylori-infected sites. IL-6 can activate and induce differentiation of T cells through other cytokines and enhance the function of monocytes and NK cells, resulting in inflammation and injury at sites infected with H pylori. Our study verified that CagA of H pylori exerted strong action in stimulating epithelial cells and other cells expressing IL-8, which can activate and chemotactic neutrophils and lymphocytes[21]. In cases with chronic cholecystitis infected with H pylori, levels of IL-1, IL-6 and IL-8 of gallbladder mucosa were significantly higher than those in cases without H pylori infection. It suggests that H pylori participate in and aggravate cholecystitis, destruction of epithelial cells of the gallbladder and atrophy of the gallbladder[22]. Taken together, our study indicates that H pylori infection in the gallbladder may be one of the etiological factors leading to cholecystitis. The precise mechanism requires further verifications.
Footnotes
Supported by the National Natural Science Foundation of China, No. 39970039
S- Editor Liu Y L- Editor Zhu LH E- Editor Ma WH
References
- 1.Tsuji S, Kawano S. Peptic ulcer recurrence and Helicobacter pylori: evidence from Japan. J Gastroenterol. 2003;38:410–411. doi: 10.1007/s005350300074. [DOI] [PubMed] [Google Scholar]
- 2.Apostolov E, Al-Soud WA, Nilsson I, Kornilovska I, Usenko V, Lyzogubov V, Gaydar Y, Wadström T, Ljungh A. Helicobacter pylori and other Helicobacter species in gallbladder and liver of patients with chronic cholecystitis detected by immunological and molecular methods. Scand J Gastroenterol. 2005;40:96–102. doi: 10.1080/00365520410009546. [DOI] [PubMed] [Google Scholar]
- 3.Guo XL, Wang LE, Du SY, Fan CL, Li L, Wang P, Yuan Y. Association of cyclooxygenase-2 expression with Hp-cagA infection in gastric cancer. World J Gastroenterol. 2003;9:246–249. doi: 10.3748/wjg.v9.i2.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ohara T, Kanoh Y, Higuchi K, Arakawa T, Morisita T. Eradication therapy of Helicobacter pylori directly induces apoptosis in inflammation-related immunocytes in the gastric mucosa--possible mechanism for cure of peptic ulcer disease and MALT lymphoma with a low-grade malignancy. Hepatogastroenterology. 2003;50:607–609. [PubMed] [Google Scholar]
- 5.Gibbons AH. Helicobacter pylori: a clinician's view. Hosp Med. 2003;64:535–538. doi: 10.12968/hosp.2003.64.9.2246. [DOI] [PubMed] [Google Scholar]
- 6.D'Elios MM, Amedei A, Del Prete G. Helicobacter pylori antigen-specific T-cell responses at gastric level in chronic gastritis, peptic ulcer, gastric cancer and low-grade mucosa-associated lymphoid tissue (MALT) lymphoma. Microbes Infect. 2003;5:723–730. doi: 10.1016/s1286-4579(03)00114-x. [DOI] [PubMed] [Google Scholar]
- 7.Konturek PC, Brzozowski T, Konturek SJ, Kwiecień S, Pajdo R, Drozdowicz D, Stachura J, Karczewska E, Hahn EG. Functional and morphological aspects of Helicobacter pylori-induced gastric cancer in Mongolian gerbils. Eur J Gastroenterol Hepatol. 2003;15:745–754. doi: 10.1097/01.meg.0000059155.68845.9d. [DOI] [PubMed] [Google Scholar]
- 8.Randi G, Franceschi S, La Vecchia C. Gallbladder cancer worldwide: geographical distribution and risk factors. Int J Cancer. 2006;118:1591–1602. doi: 10.1002/ijc.21683. [DOI] [PubMed] [Google Scholar]
- 9.Tsukanov VV, Grishchenko NN. Association of Helicobacter pylori with chronic cholecystitis. Eksp Klin Gastroenterol. 2003;(6):80–82. [PubMed] [Google Scholar]
- 10.Osadchuk MA, Geras'kina TB. Chronic cholecystitis--some lithogenic aspects. Ter Arkh. 1997;69:27–30. [PubMed] [Google Scholar]
- 11.Pradhan SB, Dali S. Relation between gallbladder neoplasm and Helicobacter hepaticus infection. Kathmandu Univ Med J (KUMJ) 2004;2:331–335. [PubMed] [Google Scholar]
- 12.Peng XN, Fan XG, Huang Y, Wang ZM, Cheng YP. The study on relationship between helicobacter infection and primary liver carcinoma. Shijie Huaren Xiaohua Zazhi. 2002;10:902–906. [Google Scholar]
- 13.Jiao JZ, Nie QH, Zhao CL, Wu YS, Wen SX, Wu Q. Clinical relationship between Helicobacter pylori infection and chronic hepatopathy. Shijie Huaren Xiaohua Zazhi. 2003;11:851–853. [Google Scholar]
- 14.Huang C, Wu ZM, Zheng ZX. Clinical significance of 13C breath test in preoperative examination for patients with cholecystolithiasis cholecystitis. Shijie Huaren Xiaohua Zazhi. 2002;10:1336–1338. [Google Scholar]
- 15.Chen W, Li D, Cannan RJ, Stubbs RS. Common presence of Helicobacter DNA in the gallbladder of patients with gallstone diseases and controls. Dig Liver Dis. 2003;35:237–243. doi: 10.1016/s1590-8658(03)00060-4. [DOI] [PubMed] [Google Scholar]
- 16.Leong RW, Sung JJ. Review article: Helicobacter species and hepatobiliary diseases. Aliment Pharmacol Ther. 2002;16:1037–1045. doi: 10.1046/j.1365-2036.2002.01282.x. [DOI] [PubMed] [Google Scholar]
- 17.Monstein HJ, Jonsson Y, Zdolsek J, Svanvik J. Identification of Helicobacter pylori DNA in human cholesterol gallstones. Scand J Gastroenterol. 2002;37:112–119. doi: 10.1080/003655202753387455. [DOI] [PubMed] [Google Scholar]
- 18.Roa I, Araya JC, Shiraishi T, Yatani R, Wistuba I, Villaseca M, de Aretxabala X. Immunohistochemical demonstration of pepsinogens I and II in the gallbladder. Rev Med Chil. 1992;120:1351–1358. [PubMed] [Google Scholar]
- 19.Straubinger RK, Greiter A, McDonough SP, Gerold A, Scanziani E, Soldati S, Dailidiene D, Dailide G, Berg DE, Simpson KW. Quantitative evaluation of inflammatory and immune responses in the early stages of chronic Helicobacter pylori infection. Infect Immun. 2003;71:2693–2703. doi: 10.1128/IAI.71.5.2693-2703.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kamangar F, Limburg P, Taylor P, Dawsey S. Re: Helicobacter pylori and interleukin 1 genotyping: an opportunity to identify high-risk individuals for gastric carcinoma. J Natl Cancer Inst. 2003;95:760; author reply 760–761. doi: 10.1093/jnci/95.10.760. [DOI] [PubMed] [Google Scholar]
- 21.Morland CM, Fear J, Joplin R, Adams DH. Inflammatory cytokines stimulate human biliary epithelial cells to express interleukin-8 and monocyte chemotactic protein-1. Biochem Soc Trans. 1997;25:232S. doi: 10.1042/bst025232s. [DOI] [PubMed] [Google Scholar]
- 22.Amieva MR, Vogelmann R, Covacci A, Tompkins LS, Nelson WJ, Falkow S. Disruption of the epithelial apical-junctional complex by Helicobacter pylori CagA. Science. 2003;300:1430–1434. doi: 10.1126/science.1081919. [DOI] [PMC free article] [PubMed] [Google Scholar]


