Abstract
Since extrahepatic bile duct cancer is difficult to diagnose and to cure, a safe and radical surgical strategy is needed. In this review, the modes of infiltration and spread of extrahepatic bile duct cancer and surgical strategy are discussed. Extended hemihepatectomy, with or without pancreatoduodenectomy (PD), plus extrahepatic bile duct resection and regional lymphadenectomy has recently been recognized as the standard curative treatment for hilar bile duct cancer. On the other hand, PD is the choice of treatment for middle and distal bile duct cancer. Major hepatectomy concomitant with PD (hepatopancreatoduodenectomy) has been applied to selected patients with widespread tumors. Preoperative biliary drainage (BD) followed by portal vein embolization (PVE) enables major hepatectomy in patients with hilar bile duct cancer without mortality. BD should be performed considering the surgical procedure, especially, in patients with separated intrahepatic bile ducts caused by hilar bile duct cancer. Right or left trisectoriectomy are indicated according to the tumor spread and biliary anatomy. As a result, extended radical resection offers a chance for cure of hilar bile duct cancer with improved resectability, curability, and a 5-year survival rate of 40%. A 5-year survival rate has ranged from 24% to 39% after PD for middle and distal bile duct cancer.
Keywords: Extrahepatic bile duct cancer, Cholangiocarci-noma, Biliary drainage, Portal vein embolization, Extended hemihepatectomy, Pancreatoduodenectomy, Hepatopancreatoduodenectomy, Right trisectionectomy, Left trisectoriectomy
INTRODUCTION
Extrahepatic bile duct cancer is difficult to diagnose in the early stage. Most patients with bile duct cancer have developed jaundice at presentation, and the tumor stage is already advanced. The anatomical relationships between hepatic hilar structures and modes of tumor extension are complicated, and this has resulted in low resectability and curability, high morbidity and mortality, and poor long-term survival rate[1-9]. Because of the advances of clinical imaging, such as magnetic resonance cholangiography (MRC) and helical computed tomography (CT)[10,11], the results of bile duct cancer diagnosis have improved, and the number of candidates for surgical resection has increased.
Radical resection is still the only a chance for cure, and various types of surgical procedure have been applied[1-3,6,12-14]. Extended hemihepatectomy has recently been recognized as the standard curative treatment for hilar bile duct cancer and has acceptable mortality[15-19]. Pancreatoduodenectomy (PD), on the other hand, is the choice of treatment for middle and distal bile duct cancer[20-24], and major hepatectomy concomitant with PD (HPD) has been applied to selected patients with widespread tumors[17,18,25-27]. However, these extensive radical procedures are not always safe, because there are risks of postoperative liver failure and pancreatic leakage. Radical resection with a safe and beneficial strategy is needed in the treatment of bile duct cancer. Proper choice of surgical procedure according to the modes and patterns of tumor infiltration is important for curative resection. Preoperative treatments, including biliary drainage (BD) and portal vein embolization (PVE), are necessary before major hepatectomy. In this article we review the current treatment of extrahepatic bile duct cancer and elucidate safe and beneficial surgical treatments and the surgeon’s role in treatment.
CLASSIFICATION OF BILE DUCT CANCER
Cholangiocarcinoma is defined as any tumor arising from the ductal epithelium of the biliary tree and is classified as intrahepatic or extrahepatic according to its location[28,29]. Intrahepatic cholangiocarcinoma is usually treated as a hepatic tumor, because it requires hepatic resection alone. In this article we have focused on the surgical treatment of extrahepatic bile duct cancer, which is classified according to the primary site into hilar, middle, and distal bile duct cancer[30] (Figure 1). The longitudinal extent of bile duct cancers that involve the bifurcation of the hepatic duct is classified according to a modified Bismuth-Corlette classification[2,9] (Figure 2).
Figure 1.
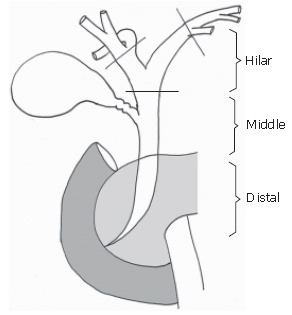
Schema of extrahepatic bile duct.
Figure 2.
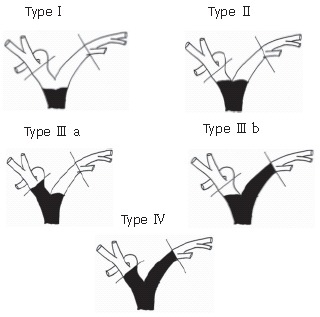
Bismuth-Corlette classification of hilar bile duct cancer[9].
MODE OF INFILTRATION AND SPREAD OF BILE DUCT CANCER
Longitudinal tumor spread along the biliary tree
Infiltration by bile duct cancer includes both longitudinal extension and vertical invasion. Microscopic extension of bile duct cancer beyond the border visualized by cholangiography or the margin observed macroscopically is often detected, and is diagnosed as a microscopically positive margin (R1). Longitudinal extension consists of superficial and submucosal infiltration and includes sometimes direct, lymphatic, or perineural invasion[31-36]. Sakamoto et al[35] pointed out a correlation between the gross type of the tumor and the pattern of infiltration beyond the macroscopic margin at the proximal border. The submucosal spread was predominant in infiltrating type, and the mean length was 6 mm. The superficial extension was predominant in papillary and nodular types, and the mean length was 20 mm. Ebata et al[36] investigated 80 resected specimens with microscopically positive margin and observed invasive (submucosal) extent within 10 mm in all the cases, and non-invasive (superficial) spread within 20 mm in 90% of the cases. Considering these reports, a macroscopic surgical margin over 10 mm is preferable in the invasive type and over 20 mm in the papillary and nodular types. Intraoperative pathologic examination of frozen sections is also advocated to confirm the margin of the cut end of the bile duct. When the margin is positive for cancer, additional resection is necessary to obtain R0 resection whenever possible.
Vertical invasion of the bile duct
Hilar bile duct cancer vertically infiltrates into the hepatoduodenal ligament, in which the hepatic artery and the portal vein are located adjacent to the bile duct. Skeltonization of vessels in the hepatoduodenal ligament and clearance of all perivascular connective tissue is standard surgical procedure[15,37,38]. Hilar bile duct cancer easily invades the right hepatic artery, which usually lies behind the common bile duct, and involves the portal vein. If the tumor shows the signs of invasion of the perivascular connective tissue, en bloc resection of the right hepatic artery is advocated to obtain a negative surgical margin; therefore, right hemihepatectomy is recommended. If tumor invasion of the portal vein is suspected based on the preoperative imaging or the intraoperative findings, combined resection of the portal vein and reconstruction are the treatment of choice to obtain a negative radical margin[39-41].
Need for hepatectomy and caudate lobectomy
When the hilar bile duct cancer has infiltrated the hepatic duct confluence, 3-dimensional knowledge of the hepatic hilum is required to determine the extent of the tumor because of the complicated anatomical relationships. Hilar bile duct cancer spreads not only to the right and left bile ducts but in a craniad and dorsal direction along the thin bile ducts. There are two key points to achieving radical resection of hilar bile duct carcinoma. The first is removal of the liver parenchyma adjacent to the hepatic hilum together with the hilar plate, and extended right or left hepatectomy is preferable for this purpose to remove the infiltration of cancer to the right and left bile ducts. The second key point is caudate lobectomy and resection of the inferior area of segment IV to extirpate cancer infiltration to the thinner bile ducts near the hilum.
The necessity of resecting the caudate lobe has been described by Japanese authors[12,13,42-44]. Nimura et al[43] firstly described caudate lobectomy for hilar bile duct cancer. The clinical efficacy of caudate lobe resection was first described by Sugiura et al[13]. The 5-year survival rate in their retrospective study was 46% with caudate lobectomy and 12% without caudate lobectomy.
Cholangiography of a resected specimen obtained by extended left hemihepatectomy with caudate resection showed that the caudate branches of the bile originated from the confluence of the hepatic ducts and that hilar bile duct cancer invaded these branches (Figure 3).
Figure 3.
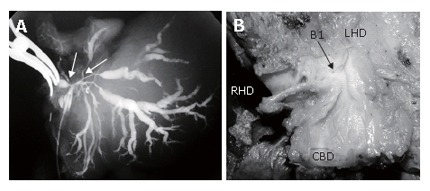
Tumor infiltration of the roots of the caudate branches; A: Cholangiography of a resected specimen. Hilar bile duct cancer has infiltrated the root of the caudate branches (B1) arising from the confluence of the hepatic duct (arrows); B: Gross appearance of the resected tumor. The caudate branch opens into the hilar neoplasm.
DIAGNOSIS
Resectability was assessed and the type of surgical treatment was selected according to the location and extent of the tumor as determined by ultrasonography, helical-CT, direct cholangiography, and MRCP[10]. Patients with para-aortic lymph node metastasis or other distant metastases were not included as candidates for resectional surgery. Direct cholangiography via the percutaneous or endoscopic route provides useful findings, but cholangitis may occur after repeated cholangiography in patients whose right and left hepatic ducts are separated by hilar bile duct cancer because the bile is contaminated with bacteria three days after biliary drainage and undrained bile ducts are infected as a result of reflux of the contaminated bile. Direct cholangiography should be restricted to the time of the first puncture and the evening of the day before surgery if the bile ducts are separated[18]. MRCP provides information regarding the entire biliary tree and eliminates the need for direct cholangiography of the whole biliary tree.
Helical contrast CT angiography as well as conventional angiography reveals not only tumor invasion of the vessels in the hepatoduodenal ligament but also vascular anomalies, especially of the hepatic arteries, which are sometimes a determinant of the operative procedure. Aberrant right hepatic arteries originating from the root of the superior mesenteric artery and aberrant left hepatic arteries originating from the left gastric artery are invaded late by bile duct cancer. Preserving the aberrant arteries increases the probability of curative resection by extended right or left hemihepatectomy.
It is sometimes difficult to make a definite diagnosis of bile duct cancer. Bile cytology or brushing biopsy is the choice of diagnosis, but benign lesions such as primary sclerosing cholangitis, cannot be denied in the treatment of bile duct cancer. The incidence of benign lesion in patients with hilar obstruction ranges from 8% to 15%[45,46]. Surgeons should consider the possibility of benign lesions when treating bile duct cancer. Radical surgery of a lesion suspected of being a malignant neoplasm is justified under the condition of low mortality rate even at the risk of benign diseases.
PREOPERATIVE TREATMENT
Biliary drainage
The aim of preoperative BD is to improve liver function and reduce morbidity and mortality after radical surgery with major hepatectomy, i.e. resection of more than three segment of the liver[47-51]. However, instead of elucidating the benefits of preoperative BD previous reports have emphasized the adverse effects of BD, i.e. infectious complications due to bile contamination and tract seeding[47-58]. Sewnath et al[56] reviewed and summarized randomized controlled and comparative cohort studies, comparing surgery plus preoperative BD with surgery without preoperative BD, and they performed a meta-analysis of the efficacy of preoperative BD. The results however, failed to show an effect of preoperative BD on the surgery. However, the following problems determine the pros and cons of preoperative BD based on the previous reports[18,59]. First, the rate of radical resection or major hepatectomy was too low to examine the effect of BD on surgery in those studies. The rate of resection in randomized controlled studies is only 15% on average and the other procedures were palliative treatments. The average resectability rate was 90% in the comparative cohort studies, but pancreatoduodenectomy composed nearly 90% of resected cases and hemihepatectomy only 2%. To perform major hepatectomy in patients with jaundice for hilar bile duct cancer is most likely to develop postoperative liver failure. Second, in the previous studies, recovery of hepatic function was insufficient, since BD was only performed for 10-14 d and the surgical interventions were carried out at a total bilirubin value of about 10 mg/dL[48,50]. By contrast, the studies from Japan insisted that radical surgery be performed after complete recovery from jaundice, i.e., a total bilirubin decrease to under 2.0 mg/dL[15,16,18]. It takes 4-6 wk for liver function to recover after BD for jaundice[47,60,61]. Finally, most tube-related trouble is due to poor technical skill, such as drainage tube failure and cholangitis caused by unnecessary cholangiography. Slipping of the drainage tube out of the bile duct can be prevented by careful management of the BD tube and by using balloon catheters or pig-tail catheters.
Since suppurative cholangitis is not a rare complication after BD, many reports have pointed out that bile contamination increases postoperative infectious complications and mortality, and that preoperative BD should be avoided. Cholangitis is caused by pushing the contaminated bile into the undrained area during cholangiography or by occlusion of the drainage tube. The most important means of preventing cholangitis is not to perform cholangiography after PTBD in patients whose right and left bile ducts are separated by tumor infiltration. Cholangiography should be limited to the time of PTBD and the afternoon of the day before the operation, because bile juice becomes contaminated by bacteria within 3 d after PTBD.
Once cholangitis has developed regardless of the cause, the treatment of choice is BD by either the percutaneous transhepatic or endoscopic retrograde route. Criticism of preoperative BD due to the risk of cholangitis is nonsense, because doctors claim such opinion indicate that they cannot treat the patients who have cholangitis not by BD. Bile from the BD tube should be cultured, and the sensitivity of isolates to antibiotics should be checked routinely. During the operation, a solution of antibiotic to which the culture are sensitive should be scattered into the abdominal cavity before closing the wound.
The route of BD is either percutaneous transhepatic or endoscopic retrograde. The only drawback of the percutaneous transhepatic route is catheter tract seeding (described below in detail). The endoscopic route, on the other hand, is closely associated with retrograde infection, and thus it is not advocated for hilar obstruction. The biliary stent tube should be replaced every two weeks to avoid tube occlusion by debris in patients in whom an endoscopic retrograde BD (ERBD) tube has been inserted[62].
Points of preoperative BD procedure are listed below. The following should be taken into consideration: (1) Externally drained bile juice should be taken orally[63], (2) Radical operation should be performed after sufficient recovery of liver function[15,16,18], (3) BD should be performed if hepatectomy is scheduled[64], (4) ERBD stent tube should be replaced every two weeks, (5) Bile culture should be performed routinely, and isolates should be tested for the antibiotic sensitivity[58].
For hilar obstruction with interruption of communication between the right and the left hepatic duct, the following should be taken into consideration (1) PTBD of the future remnant liver alone is the first choice[17,18,65,66], (2) The endoscopic route is not advocated[53,57,67,68], (3) To avoid cholangitis, preoperative cholangiography should be performed only when the tube is inserted and on the evening before the operation[18].
Catheter tract seeding is a problem related to PTBD, and its incidence has been reported to be 5%-10%[18,69]. Multiple BD and PTCS may increase the risk of seeding, but the risk can be decreased by the draining only the future remnant liver and using the minimum number of BD tubes. Tract seeding did not affect outcome because the primary tumors were already far-advanced and most patients had multiple recurrences. Sakata et al[69] reported that resection of an isolated metastasis along the catheter tract may prolong survival. Catheter tract seeding is not a rare complication in the treatment of the malignant biliary obstruction, but its incidence is acceptable.
Portal vein embolization
In 1982, Makuuchi et al[70,71] were the first to apply PVE to prevent liver failure after extended hemihepatectomy in patients with hilar cholangiocarcinoma. Major hepatectomy results in massive reduction of the liver volume and a increase in portal pressure. If PVE is performed preoperatively, the portal hypertension can be tolerated, and the rate of liver volume loss decreases due to the atrophy-hypertrophy complex theory. Kubota et al[72] showed indication criteria of PVE according to the volume of liver to be resected, which was calculated from CT, and ICG R15 value. Several recent reports have confirmed the safety of major hepatectomy following PVE for patients with hilar bile duct cancer and obstructive jaundice[16-18,38,73]. The rate of liver volume to be preserved increased by 8%-12% two weeks after PVE[18,74,75]. In Western countries, evaluations of liver volume after PVE have been made after 4-6 wk. The wait between PVE and operation is shorter in the reports from Japan, but was adequate to perform hepatic resection safely.
Safe strategy for major hepatectomy in patients with hilar bile duct cancer
Seyama et al[18] described the safe strategy for hilar bile duct cancer that includes BD and PVE. A flow chart for preoperative treatment is shown in Figure 4. If a patient showed evidence of jaundice or there were dilated bile ducts in the future remnant liver, BD was performed, in principle only in the future remnant liver. Whether PVE was indicated depended on liver function and the volume of the future remnant liver, which was calculated by CT volumetry. In patients with normal liver function, i.e. an ICG R15 value under 10%, PVE was indicated when the remnant hemiliver volume was less than 40%. In patients with jaundice or with an ICG R15 value over 10%, PVE was indicated if the remnant hemiliver volume was less than 50%. Since the standard operative procedure for hilar bile duct cancer is an extended hemihepatectomy, including the whole S1, the remaining hemiliver volume should have a margin from the safety zone. After re-evaluation of the liver volume to be resected, hepatectomy was performed if the patient fulfilled the criteria. Figure 5 showed the intraoperative findings after BD of the future remnant liver followed by PVE of the right portal vein. The right liver was markedly atrophic, and the BD tube was inserted into the bile duct in segment 3, which drained only the future remnant left liver. In this patient, extended right hemihepatectomy was carried out.
Figure 4.
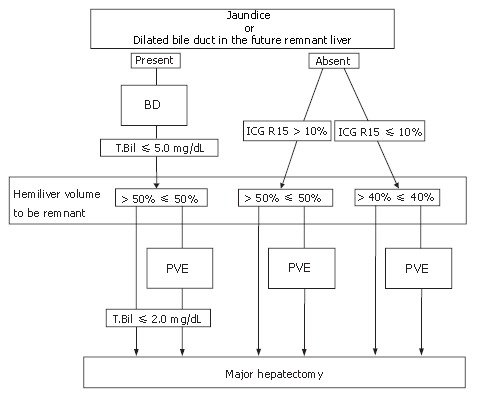
Flowchart of preoperative treatment. If a patient had jaundice or there were dilated bile ducts in the future remnant liver, biliary drainage (BD) was performed. Surgical interventions were scheduled after sufficient recovery of hepatic function. Portal vein embolization (PVE) was carried out to avoid postoperative liver failure, depending on the liver function and the liver volume to be resected.
Figure 5.

Intraoperative view at laparotomy after biliary drainage and portal vein embolization. A percutaneous transhepatic biliary drainage tube (arrow) has been inserted into the bile duct of segment 3. The right liver is markedly atrophic, and there is a clear line of demarcation between the right and left liver.
SURGICAL TREATMENT FOR BILE DUCT CANCER
In view of their modes and patterns of infiltration, the best treatment for hilar bile duct cancer is extrahepatic bile duct resection plus hepatectomy and regional lymphadenectomy, and the best treatment for middle and distal bile duct cancer is pancreatoduodenectomy. Lymph node dissection includes the nodes within the hepatoduodenal ligament, behind the pancreatic head, and along the common hepatic artery. Some authors have reported extended lymphadenectomy including para-aortic lymph nodes, but the incidence of para-aortic lymph node metastasis by bile duct cancer is lower than by pancreatic cancer. Since there is no evidence of a survival benefit[76,77], extended lymph node dissection is not justified.
Extended hemihepatectomy for hilar bile duct carcinoma
Because of its radicality and simplicity, extended hemihepatectomy is recognized as the standard surgical procedure for hilar bile duct cancer[15,17,18,27,38,73]. Extended right hemihepatectomy consists of resection of the right liver, the inferior part of Couinaud’s segment IV, and the entire caudate lobe. Extended left hemihepatectomy consists of resection of the left liver, the hilar part of the anterior segment, and most of the caudate lobe. Even in patients with Bismuth typeIor type II, extended hemihepatectomy is needed to achieve curative resection according to the mode of tumor extension[17,18,73]. If the tumor is predominantly on the right side or centrally located, extended right hepatectomy is selected. Kawasaki et al[17] explained why right hepatectomy is more likely to be associated with a negative margin than left hepatectomy based on the anatomic considerations, i.e. length of the extrahepatic part of each hepatic duct, location of the common hepatic duct in the hepatoduodenal ligament, facility for systematic caudate lobectomy, and ease of portal vein reconstruction. Extended left hemihepatectomy is indicated for left-side-dominant tumor. If the tumor has spread diffusely into the intrapancreatic bile duct, pancreatoduodenectomy is performed simultaneously[27] (HPD, described later).
Right or left trisectoriectomy for hilar bile duct carcinoma
Right or left trisectoriectomy is one of the most extensive resections because of the massive loss of volume of the hepatic parenchyma[78-80]. Hilar bile duct cancer widely invades the hepatic hilum (Bismuth type IV), resulting in jaundice, and sometimes requires trisectoriectomy for curative resection. Right or left trisectoriectomy is advantageous in terms of obtaining a cancer-free margin in the hepatic ducts. A negative hepatic margin was obtained in 75% of cases treated by left trisectoriectomy and 87.5% of cases treated by right trisectoriectomy, both rates are higher than obtained by extended hemihepatectomy[79,80]. Variation in the anatomy of the right lateral branch should be taken into consideration to obtain adequate surgical margin (Figure 6). Type A is an ordinary bifurcation, and extended right hepatectomy is the choice of treatment. If the right lateral branch originates directly from the common hepatic duct (Figure 6 Type B1-2), left trisectoriectomy, which preserves the right lateral branch, is a useful alternative. When the right lateral branch originates from the left hepatic duct, left trisectoriectomy may provide a longer bile duct margin than other hepatic resection (Figure 6 Type C). Since the aberrant right hepatic artery originates from the SMA, which runs behind the portal vein and the right-dorsal aspect of the hepatoduodenal ligament, is hardly invaded by bile duct cancer; left trisectoriectomy, which preserves the right lateral branch of the right hepatic artery, is a useful alternative for advanced hilar cholangiocarcinoma. However, the operative risk of trisectoriectomy is greater than that of extended hemihepatectomy, and preoperative BD of the future remnant liver and PVE are indispensable. Nagino et al[80] reported a mortality rate of 7.1% after right trisectoriectomy despite preoperative PVE. Shimada et al[79] reported 12 consecutive cases of left trisectoriectomy for hilar cholangiocarcinoma with no mortality after BD followed by PVE. Proper patient selection and adequate preoperative treatment are required when radical high-risk operations, such as trisectoriectomy, are preformed for hilar bile duct cancer.
Figure 6.
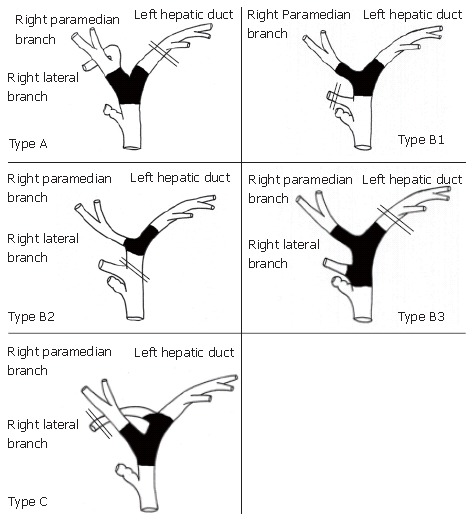
Indications for left trisectoriectomy according to tumor location and the bile duct variations in the anatomy of the right lateral branch of the bile duct. Double lines indicate the scheduled cut end of the bile duct. Type A, ordinary bifurcation; extended right hepatectomy is the choice of treatment. Type B, the right lateral branch originates from the common hepatic duct; left trisectoriectomy, preserving the right lateral branch, is a useful alternative in Types B1 and B2. Type C, the right lateral branch originates from the left hepatic duct; left trisectoriectomy may provide a longer bile duct margin.
Limited resections for hilar bile duct cancer
Parenchyma preserving hepatectomy is a less invasive procedure for hilar bile duct cancer than major hepatectomy. The drawback of the procedure is high incidence of positive margins because of tumor spread along the bile duct, although various type of hepatic resection have been used; segmental, S4 + S1, S4a + S5 + S1, S1, and extended hilar resection[14,81,82]. Other problems include the complexity of the surgical procedure, which takes a great deal of time, intraoperative bleeding, and the difficulty of biliary reconstruction. Consequently, parenchyma preserving hepatectomy for hilar bile duct cancer is the useful alternative in patients in poor general condition or with high-risk factors.
Bile duct resection without hepatectomy is the most minimally invasive, least radical surgical treatment. It is indicated only when the primary tumor is located in the middle of the common hepatic duct and there is no invasion or spread. It is also an option for high-risk patients.
Surgery for middle and distal bile duct cancer
Pancreatoduodenectomy is the treatment of choice for middle and lower bile duct cancer. The pylorus preserving Whipple procedure (PpPD) is commonly performed, and the short- and long-term results of PpPD are comparable to those of standard PD[83-85]. When a middle bile duct cancer arises midway along the extrahepatic duct, the decision has to be made as to whether PD or extended hemihepatectomy is more appropriate according to the tumor location and extension.
Indications for hepatopancreatoduodenectomy
Hepatopancreatoduodenectomy (HPD) has been used to treat biliary malignancies and is a useful alternative for the treatment of bile duct cancer. The clinical indications are longitudinal tumor spread from the hepatic duct to the intrapancreatic bile duct or massive lymph node metastases along the bile duct and behind the pancreatic head. Massive lymph node metastases are not a good indication because the presence of lymph node metastases is itself a negative prognostic factor. However, postoperative hospital mortality has been very high. Nimura et al[26]firstly presented a considerable number of patients who had undergone HPD, hospital mortality was as high as 35% and 5-year survival was only 6%. The causes of postoperative mortality after HPD are postoperative liver failure and leakage of pancreatojejunostomy resulting in bleeding from the major arteries. BD followed by PVE is indispensable to prevent postoperative liver failure[17,18,27]. A two-stage operation, in which reconstruction of the pancreatic duct is the second step, has been reported to reduce the risk of leakage of the pancreatic juice[86-88]. Miyagawa et al[27] firstly reported twelve consecutive cases of HPD without mortality, applying BD followed by PVE and staged reconstruction of the pancreatic duct.
Figure 7 is a cholangiography of a resected specimen obtained by HPD. The common hepatic duct was obstructed (Bismuth type 2), and the tumor had superficially spread to the right and left hepatic duct and the intrapancreatic part of the common bile duct. This case was considered to be a good candidate for HPD, and all the margins were negative in spite of the wide tumor spread. Pathological examination revealed that the depth of invasion was to the subserosa and that there were five lymph node metastases, all close to the bile duct. The postoperative course after HPD was uneventful, and there was no liver failure. The patient is alive without tumor recurrence as of 10 mo after surgery.
Figure 7.
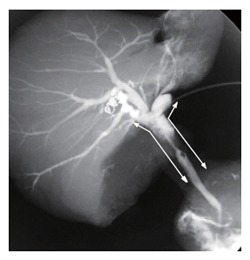
Cholangiography of the resected specimen by HPD. The tumor is located in the common hepatic duct (Bismuth-Corllete type 2), and microscopic examination showed superficial spread to the right and left hepatic duct and the intrapancreatic part of the common bile duct (arrows).
Combined vascular resection and reconstruction
The value of portal vein resection and reconstruction in the treatment of bile duct cancer is still controversial. Previously, invasion of a major vessel meant an unresectable tumor. Recent reports have shown improved results when portal vein resection and reconstruction are performed together with major hepatectomy[39-41,89]. Neuhaus et al[66] reported a 5-year survival rate of 65% after curative resection of hilar bile duct cancer with portal vein resection and reconstruction. Although their case series excluded the 60-d deaths (mortality rate of 17%) and non-curative resections, the results indicated that portal vein reconstruction concomitant with hepatectomy improves the probability of long-term survival. Interestingly, macroscopic portal vein invasion was a significant prognostic factor, but microscopic invasion was not[40,66]. When there is severe adhesion between the tumor and portal vein, combined resection and reconstruction is needed to obtain a negative surgical margin. Kondo et al[89] advocated portal vein resection and reconstruction prior to hepatic dissection during extended right hepatectomy, and the procedure enables no-touch resection. On the other hand, portal vein reconstruction increases the risk of major hepatectomy. The reported mortality of portal vein reconstruction for hilar bile duct cancer is 10%-20%, but recent studies have shown that PVE improves mortality[17,18,73]. PVE is recommended to increase the safety of major hepatectomy with portal vein reconstruction.
The clinical significance of combined resection and reconstruction of the hepatic artery of the remnant liver has not been resolved[17,18,39-41,90,91]. Reconstruction of the hepatic artery has been performed less frequently than reconstruction of the portal vein, because the conditions that require combined resection of the hepatic artery of the remnant liver often mean a locally far advanced tumor. Although there have been recent reports of major hepatectomy with reconstruction of the hepatic artery being performed safely with acceptable mortality rates (0-8%)[90,91], since it is more dangerous than major hepatectomy without reconstruction[41], at present reconstruction of the hepatic artery together with major hepatectomy is advocated if the tumor can be resected without residual tumor at any surgical margin.
SHORT-AND LONG-TERM OUTCOME
As mentioned above, major hepatectomy is the most suitable surgical procedure for removing hilar bile duct cancer with curative intent, and it is the standard procedure. Table 1 shows the results of major hepatectomy for hilar bile duct cancer over the last 10 years based on the review of the literature[16-19,38,66,73,82,92-101]. Hepatectomy improved the resectability and curability of the surgical resection for hilar bile duct cancer. Launois et al[96] reviewed selected studies and pointed out the positive correlation between the resectability rate and hepatectomy. The rate of major hepatectomy and curability are plotted in Figure 8 and show a significant positive correlation (P = 0.0027, R = 0.7). Recent reports have shown that the 5-year survival rate for hilar bile duct cancer treated by major hepatectomy has improved to 40%.
Table 1.
Review of the literature on major hepatectomy for hilar bile duct carcinoma
| Author | Year | Period | Total resection | Rate of R0 (%) | Major Hx | MH/ resection (%) | BD | PVE | Liver failure (%) | Mortality | 5-yr survival rate (%) |
| Miyagawa[16] | 1995 | 1989-1994 | 37 | 33 | 89 | Done | Done | 0.0 | 0.0 | NA | |
| Pichlmayr[92] | 1996 | 1975-1993 | 125 | 73 | 92 | 74 | No | No | 10.5 | 12.7 | 26.31 |
| Klempnauer[93] | 1997 | 1971-1995 | 151 | 78 | 111 | 74 | No | No | 3.4 | 11.7 | 32.01 |
| Nagino[82] | 1998 | 1977-1996 | 138 | 78 | 109 | 79 | Done | Done4 | 8.0 | 9.7 | 25.82 |
| Burke[94] | 1998 | 1991-1997 | 30 | 83 | 22 | 73 | No | No | NA | 6.6 | 45.0 |
| Ogura[95] | 1998 | 1976-1995 | 66 | 55 | 31 | 47 | Done | No | 15.9 | 2.2 | NA |
| Kosuge[38] | 1999 | 1980-1997 | 65 | 52 | 52 | 80 | Done | Done4 | 10.8 | 9.2 | 34.8 |
| Neuhaus[66] | 1999 | 1988-1998 | 95 | 61 | 66 | 69 | Done | (TAE) | NA | 9.0 | 22.03 |
| Launois[96] | 1999 | 1968-1993 | 40 | 80 | 22 | 55 | No | No | NA (9) | 12.5 | 12.5 |
| Miyazaki[97] | 1999 | 1981-1998 | 93 | 70 | 66 | 71 | Done | Partially | 29.0 | 12.0 | 36.0 |
| Gerhards[98] | 2000 | 1983-1998 | 112 | 14 | 32 | 29 | Done | No | 12.0 | 25.0 | NA |
| Todoroki[99] | 2000 | 1976-1998 | 98 | 14 | 32 | 33 | Done | No | 8.4 | 5.0 | 28.0 |
| Jarnagin[100] | 2001 | 1991-2000 | 80 | 78 | 62 | 78 | Not routine | No | 3.2 | 11.0 | 39.0 |
| Seyama[18] | 2003 | 1989-2001 | 67 | 64 | 58 | 87 | Done | Done | 0.0 | 0.0 | 40.0 |
| Kawasaki[17] | 2003 | 1991-2000 | 79 | 68 | 69 | 87 | Done | Done | 0.0 | 1.3 | 40.0 |
| Kondo[73] | 2004 | 1999-2002 | 40 | 95 | 26 | 65 | Done | Done | 0.0 | 0.0 | 40 (3-yr) |
| Rea[101] | 2004 | 1979-1997 | NA | 80 | 46 | NA | Done | No | 11.0 | 9.0 | 26.0 |
| Hemming[19] | 2005 | 1997-2004 | 53 | 80 | 52 | 98 | Done | Done | 3.7 | 9.0 | 35.0 |
Hx: hepatectomy; NA: not available; R0: microscopic negative margin; BD: biliary drainage; PVE: portal vein embolization. 1, these two reports were from the same center; 2, curative Hx 97 cases; 3, total 95 cases; 4, PVE was started in the middle of the study periods.
Figure 8.
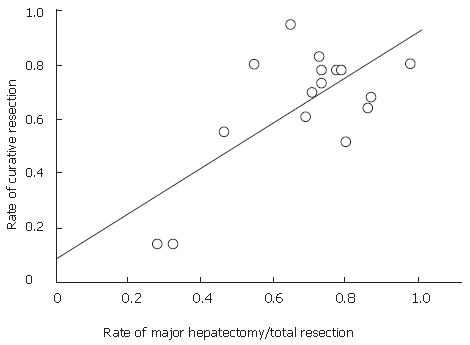
Graph of data from a literature review of the relationship between curative resection and major hepatectomy rate. The studies are listed shown in Table 1. Curative resection is defined as a microscopic ally negative margin. Linear approximation showed a significant correlation between the rate of major hepatectomy and curative resection (P = 0.0027, R = 0.7).
A high rate of postoperative liver failure and high mortality rate have been mentioned as the drawbacks of major hepatectomy for hilar bile duct cancer despite its radicality. Table 1 shows that BD alone does not reduce the rate of postoperative liver failure, and that major hepatectomy can be carried out without liver failure or mortality by using preoperative PVE together with BD. Rates of postoperative liver failure of 10%-20% had have been reported without PVE, and PVE has significantly reduced the rate of postoperative liver failure to nearly zero percent (Figure 9, P = 0.0113). The low mortality rate shows that PVE not only decreased the occurrence of postoperative liver failure but provided a hepatic functional reserve.
Figure 9.
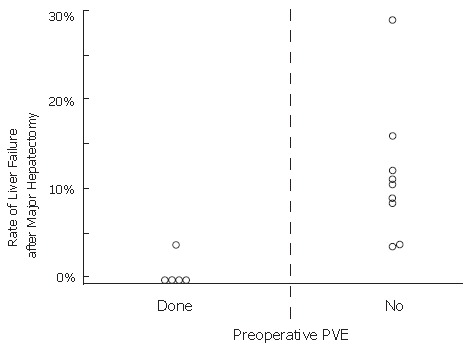
Graph of data from a literature review of the relationship between the preoperative portal vein embolization (PVE) and the rate of postoperative liver failure after major hepatectomy for hilar bile duct cancer. PVE significantly reduced the incidence of postoperative liver failure (P = 0.0113).
Curative resection (R0), lymph node metastasis negative, tumor size smaller than 2 cm, well differentiated histology, and earlier tumor stage have been reported as favorable prognostic factors after resection for hilar bile duct cancer[17-19,38,66,98,100,102]. Combined vascular resection was not a prognostic factor, if curative resection was achieved[39-41,89]. Concomitant PD with hepatectomy did not significantly influence patient survival[17,18]. Therefore, extended radical resection for local advanced bile duct cancer has been justified only under the acceptable low mortality rate. On the other hand, patients with lymph node metastasis have had significantly poorer long-term results, and long-term survival has rarely been expected even when extended lymph node dissection was performed[34].
The reported five-year survival rates after PD for middle and distal bile duct cancer have ranged from 24% to 39%[20-22,24,37,103,104]. Curative resection, lymph node metastasis negative, intraoperative transfusion negative, well differentiated histology, and location (middle or distal) have been pointed out as a favorable prognostic factors for the treatment of middle and distal bile duct cancer. Sakamoto et al[24] emphasized the prognostic significance of the radical margin rather than hepatic margin in the treatment of middle and distal bile duct cancer. They insisted that significance of purchase to the negative hepatic margin is dependent on whether radical margin is negative for the tumor. Survival of patients with middle and distal bile duct cancer and lymph node metastasis is also poor, and Yeo et al[77] showed that radical extended lymph adenectomy provided no survival benefit in a randomized controlled trial. Nor have any studies shown a difference in survival between standard PD and PpPD[83-85].
CONCLUSION
Hemihepatectomy with or without pancreatoduode-nectomy, plus extrahepatic bile duct resection and regional lymphadenectomy can be performed safely and offer a chance for cure of hilar bile duct cancer if preoperative BD followed by PVE is properly indicated. Major hepatectomy with PD and/or vascular reconstruction can be performed without mortality and results in improved resectability, curability, and a 5-year survival rate of 40%. PD is a standard procedure for middle and lower bile duct cancer, and the 5-year survival rate is nearly 30% with acceptable mortality rate. Radical resection with negative margins and no mortality is the goal of surgeons. Survival of patients with lymph node metastasis is still poor despite extended lymph node dissection, and to improve it is a future issue.
Footnotes
S- Editor Liu Y L- Editor Lalor PF E- Editor Lu W
References
- 1.Klatskin G. Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis. An unusual tumor with distinctive clinical and pathological features. Am J Med. 1965;38:241–256. doi: 10.1016/0002-9343(65)90178-6. [DOI] [PubMed] [Google Scholar]
- 2.Bismuth H, Corlette MB. Intrahepatic cholangioenteric anastomosis in carcinoma of the hilus of the liver. Surg Gynecol Obstet. 1975;140:170–178. [PubMed] [Google Scholar]
- 3.Launois B, Campion JP, Brissot P, Gosselin M. Carcinoma of the hepatic hilus. Surgical management and the case for resection. Ann Surg. 1979;190:151–157. doi: 10.1097/00000658-197908000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tompkins RK, Thomas D, Wile A, Longmire WP. Prognostic factors in bile duct carcinoma: analysis of 96 cases. Ann Surg. 1981;194:447–457. doi: 10.1097/00000658-198110000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blumgart LH, Hadjis NS, Benjamin IS, Beazley R. Surgical approaches to cholangiocarcinoma at confluence of hepatic ducts. Lancet. 1984;1:66–70. doi: 10.1016/s0140-6736(84)90002-3. [DOI] [PubMed] [Google Scholar]
- 6.Lai EC, Tompkins RK, Mann LL, Roslyn JJ. Proximal bile duct cancer. Quality of survival. Ann Surg. 1987;205:111–118. doi: 10.1097/00000658-198702000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bengmark S, Ekberg H, Evander A, Klofver-Stahl B, Tranberg KG. Major liver resection for hilar cholangiocarcinoma. Ann Surg. 1988;207:120–125. doi: 10.1097/00000658-198802000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Boerma EJ. Research into the results of resection of hilar bile duct cancer. Surgery. 1990;108:572–580. [PubMed] [Google Scholar]
- 9.Bismuth H, Nakache R, Diamond T. Management strategies in resection for hilar cholangiocarcinoma. Ann Surg. 1992;215:31–38. doi: 10.1097/00000658-199201000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhong L, Xiao SD, Stoker J, Nj Tytgat G. Magnetic resonance cholangiopancreatography. Chin J Dig Dis. 2004;5:139–148. doi: 10.1111/j.1443-9573.2004.00174.x. [DOI] [PubMed] [Google Scholar]
- 11.Uchida M, Ishibashi M, Tomita N, Shinagawa M, Hayabuchi N, Okuda K. Hilar and suprapancreatic cholangiocarcinoma: value of 3D angiography and multiphase fusion images using MDCT. AJR Am J Roentgenol. 2005;184:1572–1577. doi: 10.2214/ajr.184.5.01841572. [DOI] [PubMed] [Google Scholar]
- 12.Tsuzuki T, Ogata Y, Iida S, Nakanishi I, Takenaka Y, Yoshii H. Carcinoma of the bifurcation of the hepatic ducts. Arch Surg. 1983;118:1147–1151. doi: 10.1001/archsurg.1983.01390100021006. [DOI] [PubMed] [Google Scholar]
- 13.Sugiura Y, Nakamura S, Iida S, Hosoda Y, Ikeuchi S, Mori S, Sugioka A, Tsuzuki T. Extensive resection of the bile ducts combined with liver resection for cancer of the main hepatic duct junction: a cooperative study of the Keio Bile Duct Cancer Study Group. Surgery. 1994;115:445–451. [PubMed] [Google Scholar]
- 14.Kawarada Y, Isaji S, Taoka H, Tabata M, Das BC, Yokoi H. S4a + S5 with caudate lobe (S1) resection using the Taj Mahal liver parenchymal resection for carcinoma of the biliary tract. J Gastrointest Surg. 1999;3:369–373. doi: 10.1016/s1091-255x(99)80052-3. [DOI] [PubMed] [Google Scholar]
- 15.Kawasaki S, Makuuchi M, Miyagawa S, Kakazu T. Radical operation after portal embolization for tumor of hilar bile duct. J Am Coll Surg. 1994;178:480–486. [PubMed] [Google Scholar]
- 16.Miyagawa S, Makuuchi M, Kawasaki S. Outcome of extended right hepatectomy after biliary drainage in hilar bile duct cancer. Arch Surg. 1995;130:759–763. doi: 10.1001/archsurg.1995.01430070081016. [DOI] [PubMed] [Google Scholar]
- 17.Kawasaki S, Imamura H, Kobayashi A, Noike T, Miwa S, Miyagawa S. Results of surgical resection for patients with hilar bile duct cancer: application of extended hepatectomy after biliary drainage and hemihepatic portal vein embolization. Ann Surg. 2003;238:84–92. doi: 10.1097/01.SLA.0000074984.83031.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Seyama Y, Kubota K, Sano K, Noie T, Takayama T, Kosuge T, Makuuchi M. Long-term outcome of extended hemihepatectomy for hilar bile duct cancer with no mortality and high survival rate. Ann Surg. 2003;238:73–83. doi: 10.1097/01.SLA.0000074960.55004.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hemming AW, Reed AI, Fujita S, Foley DP, Howard RJ. Surgical management of hilar cholangiocarcinoma. Ann Surg. 2005;241:693–699; discussion 699-702. doi: 10.1097/01.sla.0000160701.38945.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakeeb A, Pitt HA, Sohn TA, Coleman J, Abrams RA, Piantadosi S, Hruban RH, Lillemoe KD, Yeo CJ, Cameron JL. Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors. Ann Surg. 1996;224:463–473; discussion 473-475. doi: 10.1097/00000658-199610000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yeo CJ, Sohn TA, Cameron JL, Hruban RH, Lillemoe KD, Pitt HA. Periampullary adenocarcinoma: analysis of 5-year survivors. Ann Surg. 1998;227:821–831. doi: 10.1097/00000658-199806000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Todoroki T, Kawamoto T, Koike N, Fukao K, Shoda J, Takahashi H. Treatment strategy for patients with middle and lower third bile duct cancer. Br J Surg. 2001;88:364–370. doi: 10.1046/j.1365-2168.2001.01685.x. [DOI] [PubMed] [Google Scholar]
- 23.Nagakawa T, Kayahara M, Ikeda S, Futakawa S, Kakita A, Kawarada H, Matsuno M, Takada T, Takasaki K, Tanimura H, et al. Biliary tract cancer treatment: results from the Biliary Tract Cancer Statistics Registry in Japan. J Hepatobiliary Pancreat Surg. 2002;9:569–575. doi: 10.1007/s005340200076. [DOI] [PubMed] [Google Scholar]
- 24.Sakamoto Y, Kosuge T, Shimada K, Sano T, Ojima H, Yamamoto J, Yamasaki S, Takayama T, Makuuchi M. Prognostic factors of surgical resection in middle and distal bile duct cancer: an analysis of 55 patients concerning the significance of ductal and radial margins. Surgery. 2005;137:396–402. doi: 10.1016/j.surg.2004.10.008. [DOI] [PubMed] [Google Scholar]
- 25.Mimura H, Kim H, Ochiai Y, Takakura N, Hamazaki K, Tsuge H, Sakagami K, Orita K. Radical block resection of hepatoduodenal ligament for carcinoma of the bile duct with double catheter bypass for portal circulation. Surg Gynecol Obstet. 1988;167:527–529. [PubMed] [Google Scholar]
- 26.Nimura Y, Hayakawa N, Kamiya J, Maeda S, Kondo S, Yasui A, Shionoya S. Hepatopancreatoduodenectomy for advanced carcinoma of the biliary tract. Hepatogastroenterology. 1991;38:170–175. [PubMed] [Google Scholar]
- 27.Miyagawa S, Makuuchi M, Kawasaki S, Hayashi K, Harada H, Kitamura H, Seki H. Outcome of major hepatectomy with pancreatoduodenectomy for advanced biliary malignancies. World J Surg. 1996;20:77–80. doi: 10.1007/s002689900014. [DOI] [PubMed] [Google Scholar]
- 28.de Groen PC, Gores GJ, LaRusso NF, Gunderson LL, Nagorney DM. Biliary tract cancers. N Engl J Med. 1999;341:1368–1378. doi: 10.1056/NEJM199910283411807. [DOI] [PubMed] [Google Scholar]
- 29.Lazaridis KN, Gores GJ. Cholangiocarcinoma. Gastroenterology. 2005;128:1655–1667. doi: 10.1053/j.gastro.2005.03.040. [DOI] [PubMed] [Google Scholar]
- 30.Japanese Society of Biliary Surgery. General rules for surgical and pathological studies on cancer of the biliary tract. Tokyo: Kanehara; 2003. [PubMed] [Google Scholar]
- 31.Bhuiya MR, Nimura Y, Kamiya J, Kondo S, Fukata S, Hayakawa N, Shionoya S. Clinicopathologic studies on perineural invasion of bile duct carcinoma. Ann Surg. 1992;215:344–349. doi: 10.1097/00000658-199204000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ogura Y, Takahashi K, Tabata M, Mizumoto R. Clinicopathological study on carcinoma of the extrahepatic bile duct with special focus on cancer invasion on the surgical margins. World J Surg. 1994;18:778–784. doi: 10.1007/BF00298931. [DOI] [PubMed] [Google Scholar]
- 33.Kato M, Nimura Y, Kamiya J, Kondo S, Nagino M, Miyachi M, Kanai M, Igaki H, Maeda S. Carcinoma of the common bile duct with superficial spread to the intrahepatic segmental bile ducts: a case report. Am Surg. 1997;63:943–947. [PubMed] [Google Scholar]
- 34.Yamaguchi K, Chijiiwa K, Saiki S, Shimizu S, Takashima M, Tanaka M. Carcinoma of the extrahepatic bile duct: mode of spread and its prognostic implications. Hepatogastroenterology. 1997;44:1256–1261. [PubMed] [Google Scholar]
- 35.Sakamoto E, Nimura Y, Hayakawa N, Kamiya J, Kondo S, Nagino M, Kanai M, Miyachi M, Uesaka K. The pattern of infiltration at the proximal border of hilar bile duct carcinoma: a histologic analysis of 62 resected cases. Ann Surg. 1998;227:405–411. doi: 10.1097/00000658-199803000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ebata T, Watanabe H, Ajioka Y, Oda K, Nimura Y. Pathological appraisal of lines of resection for bile duct carcinoma. Br J Surg. 2002;89:1260–1267. doi: 10.1046/j.1365-2168.2002.02211.x. [DOI] [PubMed] [Google Scholar]
- 37.Kayahara M, Nagakawa T, Ohta T, Kitagawa H, Tajima H, Miwa K. Role of nodal involvement and the periductal soft-tissue margin in middle and distal bile duct cancer. Ann Surg. 1999;229:76–83. doi: 10.1097/00000658-199901000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kosuge T, Yamamoto J, Shimada K, Yamasaki S, Makuuchi M. Improved surgical results for hilar cholangiocarcinoma with procedures including major hepatic resection. Ann Surg. 1999;230:663–671. doi: 10.1097/00000658-199911000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nimura Y, Hayakawa N, Kamiya J, Maeda S, Kondo S, Yasui A, Shionoya S. Combined portal vein and liver resection for carcinoma of the biliary tract. Br J Surg. 1991;78:727–731. doi: 10.1002/bjs.1800780629. [DOI] [PubMed] [Google Scholar]
- 40.Ebata T, Nagino M, Kamiya J, Uesaka K, Nagasaka T, Nimura Y. Hepatectomy with portal vein resection for hilar cholangiocarcinoma: audit of 52 consecutive cases. Ann Surg. 2003;238:720–727. doi: 10.1097/01.sla.0000094437.68038.a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shimada H, Endo I, Sugita M, Masunari H, Fujii Y, Tanaka K, Misuta K, Sekido H, Togo S. Hepatic resection combined with portal vein or hepatic artery reconstruction for advanced carcinoma of the hilar bile duct and gallbladder. World J Surg. 2003;27:1137–1142. doi: 10.1007/s00268-003-6801-6. [DOI] [PubMed] [Google Scholar]
- 42.Mizumoto R, Suzuki H. Surgical anatomy of the hepatic hilum with special reference to the caudate lobe. World J Surg. 1988;12:2–10. doi: 10.1007/BF01658479. [DOI] [PubMed] [Google Scholar]
- 43.Nimura Y, Hayakawa N, Kamiya J, Kondo S, Shionoya S. Hepatic segmentectomy with caudate lobe resection for bile duct carcinoma of the hepatic hilus. World J Surg. 1990;14:535–543; discussion 544. doi: 10.1007/BF01658686. [DOI] [PubMed] [Google Scholar]
- 44.Ogura Y, Mizumoto R, Tabata M, Matsuda S, Kusuda T. Surgical treatment of carcinoma of the hepatic duct confluence: analysis of 55 resected carcinomas. World J Surg. 1993;17:85–92; discussion 92-3. doi: 10.1007/BF01655714. [DOI] [PubMed] [Google Scholar]
- 45.Nakayama A, Imamura H, Shimada R, Miyagawa S, Makuuchi M, Kawasaki S. Proximal bile duct stricture disguised as malignant neoplasm. Surgery. 1999;125:514–521. [PubMed] [Google Scholar]
- 46.Gerhards MF, Vos P, van Gulik TM, Rauws EA, Bosma A, Gouma DJ. Incidence of benign lesions in patients resected for suspicious hilar obstruction. Br J Surg. 2001;88:48–51. doi: 10.1046/j.1365-2168.2001.01607.x. [DOI] [PubMed] [Google Scholar]
- 47.Mizumoto R, Kawarada Y, Yamawaki T, Noguchi T, Nishida S. Resectability and functional reserve of the liver with obstructive jaundice in dogs. Am J Surg. 1979;137:768–772. doi: 10.1016/0002-9610(79)90090-4. [DOI] [PubMed] [Google Scholar]
- 48.Hatfield AR, Tobias R, Terblanche J, Girdwood AH, Fataar S, Harries-Jones R, Kernoff L, Marks IN. Preoperative external biliary drainage in obstructive jaundice. A prospective controlled clinical trial. Lancet. 1982;2:896–899. doi: 10.1016/s0140-6736(82)90866-2. [DOI] [PubMed] [Google Scholar]
- 49.Blamey SL, Fearon KC, Gilmour WH, Osborne DH, Carter DC. Prediction of risk in biliary surgery. Br J Surg. 1983;70:535–538. doi: 10.1002/bjs.1800700910. [DOI] [PubMed] [Google Scholar]
- 50.Gundry SR, Strodel WE, Knol JA, Eckhauser FE, Thompson NW. Efficacy of preoperative biliary tract decompression in patients with obstructive jaundice. Arch Surg. 1984;119:703–708. doi: 10.1001/archsurg.1984.01390180065011. [DOI] [PubMed] [Google Scholar]
- 51.Pitt HA, Gomes AS, Lois JF, Mann LL, Deutsch LS, Longmire WP. Does preoperative percutaneous biliary drainage reduce operative risk or increase hospital cost? Ann Surg. 1985;201:545–553. doi: 10.1097/00000658-198505000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lai EC, Mok FP, Fan ST, Lo CM, Chu KM, Liu CL, Wong J. Preoperative endoscopic drainage for malignant obstructive jaundice. Br J Surg. 1994;81:1195–1198. doi: 10.1002/bjs.1800810839. [DOI] [PubMed] [Google Scholar]
- 53.Hochwald SN, Burke EC, Jarnagin WR, Fong Y, Blumgart LH. Association of preoperative biliary stenting with increased postoperative infectious complications in proximal cholangiocarcinoma. Arch Surg. 1999;134:261–266. doi: 10.1001/archsurg.134.3.261. [DOI] [PubMed] [Google Scholar]
- 54.Povoski SP, Karpeh MS, Conlon KC, Blumgart LH, Brennan MF. Association of preoperative biliary drainage with postoperative outcome following pancreaticoduodenectomy. Ann Surg. 1999;230:131–142. doi: 10.1097/00000658-199908000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cherqui D, Benoist S, Malassagne B, Humeres R, Rodriguez V, Fagniez PL. Major liver resection for carcinoma in jaundiced patients without preoperative biliary drainage. Arch Surg. 2000;135:302–308. doi: 10.1001/archsurg.135.3.302. [DOI] [PubMed] [Google Scholar]
- 56.Sewnath ME, Karsten TM, Prins MH, Rauws EJ, Obertop H, Gouma DJ. A meta-analysis on the efficacy of preoperative biliary drainage for tumors causing obstructive jaundice. Ann Surg. 2002;236:17–27. doi: 10.1097/00000658-200207000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mansfield SD, Barakat O, Charnley RM, Jaques BC, O'Suilleabhain CB, Atherton PJ, Manas D. Management of hilar cholangiocarcinoma in the North of England: pathology, treatment, and outcome. World J Gastroenterol. 2005;11:7625–7630. doi: 10.3748/wjg.v11.i48.7625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cortes A, Sauvanet A, Bert F, Janny S, Sockeel P, Kianmanesh R, Ponsot P, Ruszniewski P, Belghiti J. Effect of bile contamination on immediate outcomes after pancreaticoduodenectomy for tumor. J Am Coll Surg. 2006;202:93–99. doi: 10.1016/j.jamcollsurg.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 59.Nagino M, Nimura Y, Kamiya J, Kondo S, Kanai M, Miyachi M, Yamamoto H, Hayakawa N. Preoperative management of hilar cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 1995;2:215–223. [Google Scholar]
- 60.Watanapa P. Recovery patterns of liver function after complete and partial surgical biliary decompression. Am J Surg. 1996;171:230–234. doi: 10.1016/S0002-9610(97)89554-2. [DOI] [PubMed] [Google Scholar]
- 61.Singh V, Kapoor VK, Saxena R, Kaushik SP. Recovery of liver functions following surgical biliary decompression in obstructive jaundice. Hepatogastroenterology. 1998;45:1075–1081. [PubMed] [Google Scholar]
- 62.Jagannath P, Dhir V, Shrikhande S, Shah RC, Mullerpatan P, Mohandas KM. Effect of preoperative biliary stenting on immediate outcome after pancreaticoduodenectomy. Br J Surg. 2005;92:356–361. doi: 10.1002/bjs.4864. [DOI] [PubMed] [Google Scholar]
- 63.Kamiya S, Nagino M, Kanazawa H, Komatsu S, Mayumi T, Takagi K, Asahara T, Nomoto K, Tanaka R, Nimura Y. The value of bile replacement during external biliary drainage: an analysis of intestinal permeability, integrity, and microflora. Ann Surg. 2004;239:510–517. doi: 10.1097/01.sla.0000118594.23874.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nakeeb A, Pitt HA. The role of preoperative biliary decompression in obstructive jaundice. Hepatogastroenterology. 1995;42:332–337. [PubMed] [Google Scholar]
- 65.Noie T, Sugawara Y, Imamura H, Takayama T, Makuuchi M. Selective versus total drainage for biliary obstruction in the hepatic hilus: an experimental study. Surgery. 2001;130:74–81. doi: 10.1067/msy.2001.116028. [DOI] [PubMed] [Google Scholar]
- 66.Neuhaus P, Jonas S, Bechstein WO, Lohmann R, Radke C, Kling N, Wex C, Lobeck H, Hintze R. Extended resections for hilar cholangiocarcinoma. Ann Surg. 1999;230:808–818; discussion 819. doi: 10.1097/00000658-199912000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Liu CL, Lo CM, Lai EC, Fan ST. Endoscopic retrograde cholangiopancreatography and endoscopic endoprosthesis insertion in patients with Klatskin tumors. Arch Surg. 1998;133:293–296. doi: 10.1001/archsurg.133.3.293. [DOI] [PubMed] [Google Scholar]
- 68.Rerknimitr R, Kladcharoen N, Mahachai V, Kullavanijaya P. Result of endoscopic biliary drainage in hilar cholangiocarcinoma. J Clin Gastroenterol. 2004;38:518–523. doi: 10.1097/01.mcg.0000123204.36471.be. [DOI] [PubMed] [Google Scholar]
- 69.Sakata J, Shirai Y, Wakai T, Nomura T, Sakata E, Hatakeyama K. Catheter tract implantation metastases associated with percutaneous biliary drainage for extrahepatic cholangiocarcinoma. World J Gastroenterol. 2005;11:7024–7027. doi: 10.3748/wjg.v11.i44.7024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Makuuchi M, Takayasu K, Takuma T, Yamasaki S, Hasegawa H, Nishiura S, Shimamura Y. Preoperative transcatheter embolization of the portal venous branch for patients receiving extended lobectomy due to teh bile duct carcinoma. J Jpn Surg Assoc. 1984;45:1558–1564. [Google Scholar]
- 71.Makuuchi M, Thai BL, Takayasu K, Takayama T, Kosuge T, Gunvén P, Yamazaki S, Hasegawa H, Ozaki H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report. Surgery. 1990;107:521–527. [PubMed] [Google Scholar]
- 72.Kubota K, Makuuchi M, Kusaka K, Kobayashi T, Miki K, Hasegawa K, Harihara Y, Takayama T. Measurement of liver volume and hepatic functional reserve as a guide to decision-making in resectional surgery for hepatic tumors. Hepatology. 1997;26:1176–1181. doi: 10.1053/jhep.1997.v26.pm0009362359. [DOI] [PubMed] [Google Scholar]
- 73.Kondo S, Hirano S, Ambo Y, Tanaka E, Okushiba S, Morikawa T, Katoh H. Forty consecutive resections of hilar cholangiocarcinoma with no postoperative mortality and no positive ductal margins: results of a prospective study. Ann Surg. 2004;240:95–101. doi: 10.1097/01.sla.0000129491.43855.6b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Imamura H, Shimada R, Kubota M, Matsuyama Y, Nakayama A, Miyagawa S, Makuuchi M, Kawasaki S. Preoperative portal vein embolization: an audit of 84 patients. Hepatology. 1999;29:1099–1105. doi: 10.1002/hep.510290415. [DOI] [PubMed] [Google Scholar]
- 75.Takayama T, Makuuchi M. Preoperative portal vein embolization: is it useful? J Hepatobiliary Pancreat Surg. 2004;11:17–20. doi: 10.1007/s00534-002-0800-0. [DOI] [PubMed] [Google Scholar]
- 76.Kitagawa Y, Nagino M, Kamiya J, Uesaka K, Sano T, Yamamoto H, Hayakawa N, Nimura Y. Lymph node metastasis from hilar cholangiocarcinoma: audit of 110 patients who underwent regional and paraaortic node dissection. Ann Surg. 2001;233:385–392. doi: 10.1097/00000658-200103000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yeo CJ, Cameron JL, Lillemoe KD, Sohn TA, Campbell KA, Sauter PK, Coleman J, Abrams RA, Hruban RH. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma, part 2: randomized controlled trial evaluating survival, morbidity, and mortality. Ann Surg. 2002;236:355–366; discussion 366-368. doi: 10.1097/00000658-200209000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Noie T, Bandai Y, Kubota K, Abe H, Makuuchi M. Extended right trisegmentectomy for hilar bile duct carcinoma. Hepatogastroenterology. 1997;44:998–1001. [PubMed] [Google Scholar]
- 79.Shimada K, Sano T, Sakamoto Y, Kosuge T. Safety and effectiveness of left hepatic trisegmentectomy for hilar cholangiocarcinoma. World J Surg. 2005;29:723–727. doi: 10.1007/s00268-005-7704-5. [DOI] [PubMed] [Google Scholar]
- 80.Nagino M, Kamiya J, Arai T, Nishio H, Ebata T, Nimura Y. "Anatomic" right hepatic trisectionectomy (extended right hepatectomy) with caudate lobectomy for hilar cholangiocarcinoma. Ann Surg. 2006;243:28–32. doi: 10.1097/01.sla.0000193604.72436.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Miyazaki M, Ito H, Nakagawa K, Ambiru S, Shimizu H, Shimizu Y, Okuno A, Nozawa S, Nukui Y, Yoshitomi H, et al. Segments I and IV resection as a new approach for hepatic hilar cholangiocarcinoma. Am J Surg. 1998;175:229–231. doi: 10.1016/s0002-9610(97)00295-x. [DOI] [PubMed] [Google Scholar]
- 82.Nagino M, Nimura Y, Kamiya J, Kanai M, Uesaka K, Hayakawa N, Yamamoto H, Kondo S, Nishio H. Segmental liver resections for hilar cholangiocarcinoma. Hepatogastroenterology. 1998;45:7–13. [PubMed] [Google Scholar]
- 83.Di Carlo V, Zerbi A, Balzano G, Corso V. Pylorus-preserving pancreaticoduodenectomy versus conventional whipple operation. World J Surg. 1999;23:920–925. doi: 10.1007/s002689900600. [DOI] [PubMed] [Google Scholar]
- 84.Seiler CA, Wagner M, Sadowski C, Kulli C, Büchler MW. Randomized prospective trial of pylorus-preserving vs. Classic duodenopancreatectomy (Whipple procedure): initial clinical results. J Gastrointest Surg. 2000;4:443–452. doi: 10.1016/s1091-255x(00)80084-0. [DOI] [PubMed] [Google Scholar]
- 85.Tran KT, Smeenk HG, van Eijck CH, Kazemier G, Hop WC, Greve JW, Terpstra OT, Zijlstra JA, Klinkert P, Jeekel H. Pylorus preserving pancreaticoduodenectomy versus standard Whipple procedure: a prospective, randomized, multicenter analysis of 170 patients with pancreatic and periampullary tumors. Ann Surg. 2004;240:738–745. doi: 10.1097/01.sla.0000143248.71964.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Miyagawa S, Makuuchi M, Kawasaki S, Ogiwara M. Second-stage pancreatojejunostomy following pancreatoduodenectomy in high-risk patients. Am J Surg. 1994;168:66–68. doi: 10.1016/s0002-9610(05)80073-x. [DOI] [PubMed] [Google Scholar]
- 87.Seyama Y, Kubota K, Kobayashi T, Hirata Y, Itoh A, Makuuchi M. Two-staged pancreatoduodenectomy with external drainage of pancreatic juice and omental graft technique. J Am Coll Surg. 1998;187:103–105. doi: 10.1016/s1072-7515(98)00127-6. [DOI] [PubMed] [Google Scholar]
- 88.Kubota K, Makuuchi M, Takayama T, Sakamoto Y, Harihara Y, Kimura W. Appraisal of two-staged pancreatoduodenectomy: its technical aspects and outcome. Hepatogastroenterology. 2000;47:269–274. [PubMed] [Google Scholar]
- 89.Kondo S, Katoh H, Hirano S, Ambo Y, Tanaka E, Okushiba S. Portal vein resection and reconstruction prior to hepatic dissection during right hepatectomy and caudate lobectomy for hepatobiliary cancer. Br J Surg. 2003;90:694–697. doi: 10.1002/bjs.4084. [DOI] [PubMed] [Google Scholar]
- 90.Yamanaka N, Yasui C, Yamanaka J, Ando T, Kuroda N, Maeda S, Ito T, Okamoto E. Left hemihepatectomy with microsurgical reconstruction of the right-sided hepatic vasculature. A strategy for preserving hepatic function in patients with proximal bile duct cancer. Langenbecks Arch Surg. 2001;386:364–368. doi: 10.1007/s004230100225. [DOI] [PubMed] [Google Scholar]
- 91.Sakamoto Y, Sano T, Shimada K, Kosuge T, Kimata Y, Sakuraba M, Yamamoto J, Ojima H. Clinical significance of reconstruction of the right hepatic artery for biliary malignancy. Langenbecks Arch Surg. 2006;391:203–208. doi: 10.1007/s00423-006-0026-8. [DOI] [PubMed] [Google Scholar]
- 92.Pichlmayr R, Weimann A, Klempnauer J, Oldhafer KJ, Maschek H, Tusch G, Ringe B. Surgical treatment in proximal bile duct cancer. A single-center experience. Ann Surg. 1996;224:628–638. doi: 10.1097/00000658-199611000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Klempnauer J, Ridder GJ, von Wasielewski R, Werner M, Weimann A, Pichlmayr R. Resectional surgery of hilar cholangiocarcinoma: a multivariate analysis of prognostic factors. J Clin Oncol. 1997;15:947–954. doi: 10.1200/JCO.1997.15.3.947. [DOI] [PubMed] [Google Scholar]
- 94.Burke EC, Jarnagin WR, Hochwald SN, Pisters PW, Fong Y, Blumgart LH. Hilar Cholangiocarcinoma: patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system. Ann Surg. 1998;228:385–394. doi: 10.1097/00000658-199809000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ogura Y, Kawarada Y. Surgical strategies for carcinoma of the hepatic duct confluence. Br J Surg. 1998;85:20–24. doi: 10.1046/j.1365-2168.1998.00532.x. [DOI] [PubMed] [Google Scholar]
- 96.Launois B, Terblanche J, Lakehal M, Catheline JM, Bardaxoglou E, Landen S, Campion JP, Sutherland F, Meunier B. Proximal bile duct cancer: high resectability rate and 5-year survival. Ann Surg. 1999;230:266–275. doi: 10.1097/00000658-199908000-00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Miyazaki M, Ito H, Nakagawa K, Ambiru S, Shimizu H, Okaya T, Shinmura K, Nakajima N. Parenchyma-preserving hepatectomy in the surgical treatment of hilar cholangiocarcinoma. J Am Coll Surg. 1999;189:575–583. doi: 10.1016/s1072-7515(99)00219-7. [DOI] [PubMed] [Google Scholar]
- 98.Gerhards MF, van Gulik TM, de Wit LT, Obertop H, Gouma DJ. Evaluation of morbidity and mortality after resection for hilar cholangiocarcinoma--a single center experience. Surgery. 2000;127:395–404. doi: 10.1067/msy.2000.104250. [DOI] [PubMed] [Google Scholar]
- 99.Todoroki T, Kawamoto T, Koike N, Takahashi H, Yoshida S, Kashiwagi H, Takada Y, Otsuka M, Fukao K. Radical resection of hilar bile duct carcinoma and predictors of survival. Br J Surg. 2000;87:306–313. doi: 10.1046/j.1365-2168.2000.01343.x. [DOI] [PubMed] [Google Scholar]
- 100.Jarnagin WR, Fong Y, DeMatteo RP, Gonen M, Burke EC, Bodniewicz BS J, Youssef BA M, Klimstra D, Blumgart LH. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507–517; discussion 517-519. doi: 10.1097/00000658-200110000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rea DJ, Munoz-Juarez M, Farnell MB, Donohue JH, Que FG, Crownhart B, Larson D, Nagorney DM. Major hepatic resection for hilar cholangiocarcinoma: analysis of 46 patients. Arch Surg. 2004;139:514–523; discussion 523-525. doi: 10.1001/archsurg.139.5.514. [DOI] [PubMed] [Google Scholar]
- 102.Su CH, Tsay SH, Wu CC, Shyr YM, King KL, Lee CH, Lui WY, Liu TJ, P'eng FK. Factors influencing postoperative morbidity, mortality, and survival after resection for hilar cholangiocarcinoma. Ann Surg. 1996;223:384–394. doi: 10.1097/00000658-199604000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Park SJ, Kim SW, Jang JY, Lee KU, Park YH. Intraoperative transfusion: is it a real prognostic factor of periampullary cancer following pancreatoduodenectomy? World J Surg. 2002;26:487–492. doi: 10.1007/s00268-001-0254-6. [DOI] [PubMed] [Google Scholar]
- 104.Yoshida T, Matsumoto T, Sasaki A, Morii Y, Aramaki M, Kitano S. Prognostic factors after pancreatoduodenectomy with extended lymphadenectomy for distal bile duct cancer. Arch Surg. 2002;137:69–73. doi: 10.1001/archsurg.137.1.69. [DOI] [PubMed] [Google Scholar]


