Abstract
AIM: To quantify the intraepithelial lymphocytes (IELs) and to document the membrane expression of CD4, CD8, TCRγδ and adhesion and/or activation-associated molecules (CD103, CD28, CD44, CD69, HLA-DR, CD95/Fas) in the duodenal mucosa of patients with functional dyspepsia (FD) in order to provide arguments for an immunological process in FD.
METHODS: Twenty-six FD patients according to Rome II criteria (20 were H pylori negative) were studied and compared to 12 healthy adults. IELs were isolated from five duodenal biopsy samples, then quantified by microscopy and flow cytometry while the membrane phenotypes were determined by cytofluorometry.
RESULTS: Duodenal histological examination was normal. In H pylori negative patients, the number of IELs was not different from that in healthy controls. Median percentage expression of CD4, CD8, or TCRγδ and CD103, CD44, CD28, CD69 on CD3+ IELs, among the adhesion/activation associated molecules tested, was not different from that in healthy controls. In contrast, the median percentage expression of CD95/Fas [22 (9-65) vs 45 (19-88), P = 0.03] and HLA-DR expressing CD3+ IELs [4 (0-30) vs 13 (4-42), P = 0.04] was significantly lower in the H pylori negative FD group than in healthy controls, respectively. The number of IELs was significantly greater in H pylori positive FD patients than in healthy controls [median ratiofor 100 enterocytes 27.5 (6.7-62.5) vs 10.8 (3-33.3), P = 0.02] due to a higher number of CD8+ CD3+ IELs.
CONCLUSION: In H pylori negative FD patients, the phenotypic characterization of IELs suggests that we cannot exclude a role of IELs in FD.
Keywords: Functional dyspepsia, Intraepithelial T lymphocytes, Gut, CD95/Fas, HLA-DR
INTRODUCTION
Functional dyspepsia (FD) is one of the most common functional gastrointestinal disorders in adults. This clinical syndrome is characterized by chronic or recurrent pain or discomfort centered in the upper abdomen without a cause that is identifiable by conventional diagnostic means[1]. Several mechanisms have been already proposed to explain its symptoms: a delayed gastric emptying found in about 40% of the patients, an impaired proximal gastric accommodation to meals promoting either proximal gastric distension during food intake and/or an early migration of the components of the meal distending the antrum[2,3], a gastric hypersensitivity with a decreased pain threshold during distension that may account for early and/or delayed dyspeptic symptoms in about 40% of the patients[4,5]. However, a poor correlation exists between these findings and symptoms. The pathophysiology of FD is probably multifactorial and not completely understood. In irritable bowel syndrome (IBS), recent findings have pointed out the possible role of an immune dysfunction in the onset of symptoms: a low grade mucosal inflammatory process, undetectable with conventional mucosal histology, exists, particularly in post-infectious IBS[6,7]. An immune activation with the release of several mediators, including cytokines, nitric oxide, histamine and protease which interfere with the function of enteric nerves, has been observed in the intestinal mucosa of IBS patients[8]. Moreover, immune-mediated activation of mastocytes and macrophages in contact with pain-sensitive endings seems to be involved in the development of hypersensitivity in experimental models of visceral pain[9]. As symptoms of IBS and FD often overlap with time, we could speculate that an immune dysfunction may be also involved in the onset of FD in adults.
One of the key clinical features in FD is that symptoms are often meal-related, being either induced or exacerbated by food[10]. The intestinal mucosal barrier consists of intestinal epithelial cells and intestinal intraepithelial lymphocytes (IELs). By their strategic location, IELs are likely to be important in the preservation of mucosal integrity including maintenance of oral tolerance to both particulate and soluble antigens[11-14] and the vast majority of IELs are of T-cell type and more than 70% are CD4-/CD8+ T-cells[15-17].
The present study was carried out to investigate changes in number and phenotype of single cell isolated duodenal IELs, especially phenotypic markers of the IEL activation process, by cytofluorometry, in symptomatic FD patients compared with healthy control adults.
MATERIALS AND METHODS
Subjects
This prospective study concerned 26 consecutive dyspeptic patients, 22 women and 4 men, median age of 44 years (range: 18-69) referred to the Gastroenterology Department of the Rouen University Hospital for upper gastrointestinal endoscopy over a one year period of time. Median duration of symptoms was 4 years (range: 1-35 years). No patient had any history of post-infectious FD. Patients were assessed independently by two gastroenterologists in order to confirm the diagnosis of FD according to Rome II criteria and to look for the following exclusion criteria to participate in the study: (1) predominant symptoms of gastro-esophageal disease (heartburn and/or acid regurgitation), (2) associated abdominal pain and transit disturbances suggestive of irritable bowel syndrome according to Rome II criteria, (3) documented immunodeficiency state and medical history of inflammatory bowel disease, (4) clinical history of lactose intolerance, (5) consumption of non-steroidal anti-inflammatory drugs within two weeks before the study. In order to exclude a celiac disease leading to symptoms mimicking FD, both anti-gliadin and anti-transglutaminase serum antibodies were systematically dosed and normal in all patients.
In eligible dyspeptic patients, research of H pylori was systematically performed both by histological examination of two antral biopsies and by C13 urea breath test. Twenty patients were H pylori negative dyspeptics (both tests negative). In the remaining 6 patients (H pylori positive patients), both histological examination of antral biopsies and C13 urea breath test led to the diagnosis of H pylori infection. The control group consisted of 12 healthy non obese adult volunteers, 9 women and 3 men, median age of 42 years (range 19-76 years). None of them took non-steroidal anti-inflammatory drugs for at least two weeks prior to the study and all were H pylori-negative.
All patients and healthy volunteers received oral and then written information before inclusion. The study was approved by the local Ethical Committee of the Rouen University Hospital (CCPPRB of Haute-Normandie, France). Written consent was obtained from all participants.
Endoscopic duodenal and antral sampling
An upper gastrointestinal endoscopy was performed in all patients and healthy controls. Seven samples were taken in the duodenum, two for conventional histology and five from different sites in the second duodenum for IEL phenotype analysis. Two additional antral biopsy samples were obtained for H pylori detection. Duodenal biopsy specimens for IEL phenotype analysis were placed in calcium and magnesium free Hanks' balanced salt solution (Eurobio, Les Ulis, France) supplemented with antibiotics (penicillin: 100 U/mL, streptomycin: 100 μg/mL) and 4% (vol/vol) fetal calf serum before processed within the first 6 hours following endoscopy.
Flow cytometric membrane characterization of duodenal IELs
Duodenal biopsy samples were treated with 1 mmol/L dithiothreitol (DTT), (Sigma, Saint-Louis, Missouri, USA) for 30 min at 37°C, and 1 mmol/L EDTA for 45 min with continuous agitation at 37°C to remove the epithelial layer. Single cell suspensions were pelleted from the supernatant, and washed once with phosphate buffered saline (PBS) containing 0.5% bovine serum albumin (BSA) and 0.02% sodium azide (1200 r/min, 10 min, at room temperature). For quantitative analysis, duodenal IELs were microscopically counted in a haemocytometer with their number expressed per 100 epithelial cells. IEL viability checked by trypan-blue exclusion was over 95% in all experiments and the stability of IEL over times was constant from one patient to another. As DTT/EDTA treatment has been previously reported to cause loss of β7 integrin expression[18], lymphocytes collected from the supernatant before this treatment were used for CD103 labelling. The following mouse monoclonal antibodies (Mab) used in this study are shown in Table 1.
Table 1.
Monoclonal antibodies used in this study
| Clone | Specificity | Isotype | Label | Source |
| SK7 | CD3 | IgG1 | PE | Becton- Dickinson, Le Pont de Claix, France |
| SK7/SK1/ 2D1/SK3 | CD3/CD8/ CD45/CD4 | IgG1/IgG1/ IgG1/IgG1 | FITC/PE/ Per CP/APC | Becton- Dickinson, Le Pont de Claix, France |
| Ber-ACT8 | CD103 (αE subunit of the αEβ7 integrin) | IgG1 | FITC | Becton- Dickinson, Le Pont de Claix, France |
| CD28.2 | CD28 | IgG1 | FITC | Becton- Dickinson, Le Pont de Claix, France |
| G44-26, | CD44 | IgG2b | FITC | Becton- Dickinson, Le Pont de Claix, France |
| 11F2 | TCR-γδ | IgG1 | FITC | Becton- Dickinson, Le Pont de Claix, France |
| L78 | CD69 | IgG1 | FITC | Becton- Dickinson, Le Pont de Claix, France |
| CR3/43 | HLA-DR | IgG1 | FITC | Dako, Glostrup, Denmark |
| UB2 | CD95/Fas | IgG1 | FITC | Immunotech, Marseille, France |
Ten microlitres of each Mab stock solution was added to 100 μL of cell suspension (approximately 105 cells), then incubated at room temperature for 30 min in the dark before washing once with PBS, 0.5% BSA and 0.02% sodium azide. Flow cytofluorometry data were acquired in a flow cytometer (FACScalibur, Becton-Dickinson) and listmode files were analyzed using the Cell-Quest software (version 3.3, Becton-Dickinson). Cell suspensions were initially visualized in the forward scatter/side scatter (FSC/SSC) profile. Irrelevant isotype-matched control Mab (FITC-labelled IgG1 and PE-labelled IgG2, Becton Dickinson) was used to exclude non-specific binding and to determine the optimal setting of fluorescence quadrants. A gate was drawn around the CD3+ population and used thereafter, excluding most epithelial cells and debris from analysis.
FACScan settings were routinely standardized with beads (Calibrite, Becton Dickinson). Results were obtained as percentages of total CD3+ lymphocytes and absolute cell counts determined using calibrated fluorescent beads (FlowCount fluorospheres, Coulter Immunotech, Marseille, France).
Statistical analysis
Variance comparisons between groups were performed using the “non-parametric” Mann-Whitney test. Significance was expressed as P values, and P < 0.05 was considered statistically significant.
RESULTS
Duodenal histology
The weight of biopsy specimens was not different between those obtained in dyspeptic patients and healthy controls (data not shown, P > 0.05). Duodenal specimens were considered normal after conventional histological examination in all patients, whatever H pylori status. All healthy controls exhibited also normal duodenal histology.
Number and membrane phenotypes of duodenal IELs
The number of IELs did not differ between H pylori negative dyspeptic patients and control subjects, when this number was expressed as the median ratio for 100 enterocytes determined microscopically [12.5% (2.5-50) vs 10.8% (3-33.3), P > 0.05] or as a median absolute number of CD3+ IELs using cytofluorometry [122.4 × 103 (190.2 × 103 - 509 × 103) vs 53.6 × 103 (7.5 × 103 - 756.6 × 103, P > 0.05)]. At variance, duodenal IEL counts were significantly higher (P = 0.02) in H pylori positive patients than in healthy controls, when they were determined microscopically as the median ratio for 100 enterocytes [27.5 (6.7-62.5)] or as a median absolute number using flow cytometry [468.3 × 103 (275 × 103-630 × 103)] (Figure 1).
Figure 1.
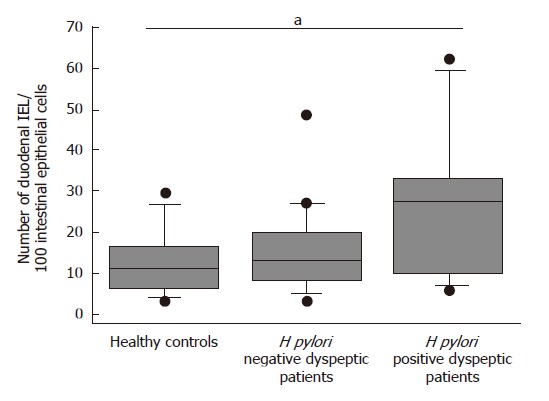
Box- and -wiskers plots of duodenal IEL counts expressed as number of IELs per 100 duodenal epithelial cells (Ec) in healthy controls, H pylori negative and positive dyspeptic patients. aP < 0.05 for H pylori positive vs controls.
Median percentages of CD3+ lymphocytes in H pylori negative dyspeptic patients and healthy controls were 96% (84%-99%) and 93% (80%-96%) respectively (P > 0.05). Neither the percentages of CD3+, CD4+, CD8+ and CD3+ TCRγδ IELs nor the percentages of CD103, CD44, CD28 or CD69 expressing CD3+ IELs differed between H pylori negative dyspeptic patients and healthy controls. However, H pylori positive dyspeptic patients exhibited increased percentages of CD8 expressing CD3+ IELs compared with healthy controls[88% (76%-94%) vs 77% (54%-87%), respectively, P = 0.01]
(Table 2).
Table 2.
Immunophenotype of duodenal intraepithelial lymphocytes in H pylori negative (Hp-) and positive (Hp+) dyspeptic patients and healthy control adults
| IEL | Hp- dyspeptic patients (n = 20) Median % (range %) | Hp+ dyspeptic patients (n = 6) Median % (range %) | Healthy control adults (n = 12) Median % (range %) | P |
| CD3+ | 93 (80-98) | 94.5 (89-99) | 96 (84-99) | aceP > 0.5 |
| CD3+ CD4+ | 8 (3-34) | 8 (3-15) | 10 (2-22) | aceP > 0.5 |
| CD3+ CD8+ | 78 (58-96) | 86 (75-87) | 77 (53-87) | acP > 0.5 |
| bP < 0.01 | ||||
| CD3+ TCRgd+ | 22 (1-36) | 10 (10-30) | 24 (8-60) | aceP > 0.5 |
| CD3+ CD103+ | 91 (58-96) | 81 (75-87) | 90 (80-99) | aceP > 0.5 |
| CD3+ CD44+ | 95 (50-99) | 94 (84-100) | 94 (65-100) | aceP > 0.5 |
| CD3+ CD28+ | 2 (1-11) | 5 (1-10) | 7 (1-9) | aceP > 0.5 |
| CD3+ CD69+ | 97 (68-100) | 99 (96-100) | 96 (91-98) | aceP > 0.5 |
P > 0.5, Hp- vs control;
P > 0.5, Hp- vs Hp+;
P > 0.5, Hp+ vs control;
P < 0.01, Hp+ vs control.
The median percentages of CD95/Fas [22% (9%-65%) vs 45% (19%-88%), P = 0.009] (Figure 2) and HLA-DR expressing CD3+ IELs [4% (0%-30%) vs 13% (4%-42%), P = 0.04] (Figure 3) were significantly lower in the H pylori negative FD group than in healthy controls respectively.
Figure 2.
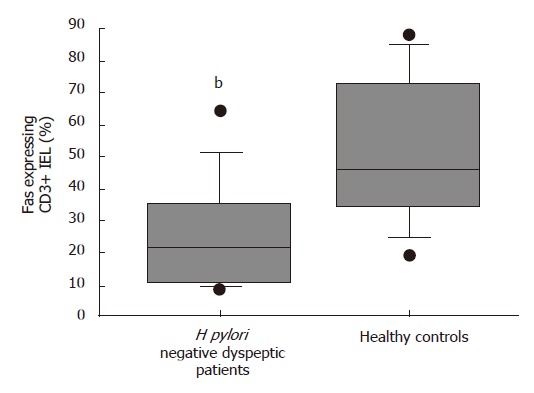
Box- and -wiskers plots of percentages of Fas expressing CD3+ duodenal IELs in healthy controls and H pylori negative dyspeptic patients. bP < 0.01 vs controls.
Figure 3.
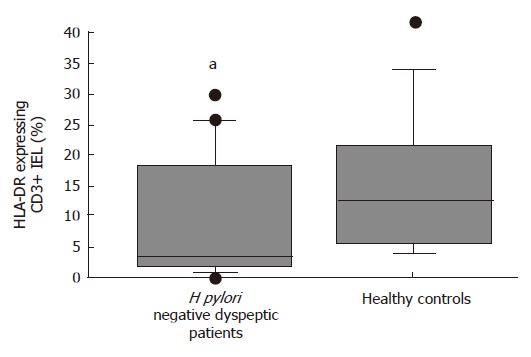
Box- and -wiskers plots of percentages of HLA-DR expressing CD3+ duodenal IELs in healthy controls and H pylori negative dyspeptic patients. aP < 0.05 vs controls.
DISCUSSION
While FD is highly prevalent in the general population, very little is known about the immunopathology of the disease and its underlying mechanisms. In order to check for a possible immune mediated mechanism, by analogy with recent findings in irritable bowel syndrome[7], our study was conducted to quantify and characterize duodenal IELs (particularly membrane expression of adhesion or activation-associated molecules) both in H pylori negative and positive dyspeptic patients by comparison with healthy controls. The results of this study suggest that in FD, besides motor or sensory dysfunctions previously demonstrated, duodenal mucosal immune changes exist. In a series of H pylori negative patients with FD defined according to Rome II criteria, CD95/Fas and HLA-DR, two activation markers, were expressed by a significantly smaller percentage of duodenal CD3+ IELs compared with healthy controls, whereas the number of duodenal IELs was similar in patients with FD and healthy controls. In healthy controls, individual ratios of CD95/Fas expressing CD3+ IEL demonstrated a large range of variations (19%-88%) consistent with other reports[19,20]. Fas/FasL interaction seems closely related to the lymphocyte activation process and the expression of CD95/Fas by intestinal IELs supports the hypothesis that intestinal IELs are activated cells[21-23]. Fas-FasL system has been shown to mediate intestinal lymphocyte apoptosis not only in normal intestine where antigenic challenge continuously activates IELs[20,24-26] but also in some pathological conditions such as celiac disease[27]. Lower ratios of CD95/Fas and HLA-DR-expressing duodenal IELs in dyspeptic patients may reflect the presence of an altered population of primed lymphocytes[28]. With respect to the small sample of patients and due to the inclusion criteria, comparisons of immunopathologic parameters in relation to age, mode of onset, length of history, or predominant symptoms were not performed in the present study.
Duodenal IEL counts, determined either microsco-pically or by flow cytometric counting for CD3+ cells, did not differ between H pylori negative dyspeptic patients and healthy controls. In these two groups, values were very close to those previously reported by others, i.e. 10-20 IELs per 100 villous enterocytes[29-33]. Recent studies have shown that the upper limit of IEL number in the proximal small intestine is 25 IELs/100 epithelial cells[34,35]. In the absence of villus architectural changes, an increased number of IELs can be related to reactions to intraluminal antigens such as H pylori[35] and is considered as the result of an immune response analogous to the response to gluten in patients with celiac disease[36-38]. This is confirmed in our study with the demonstration that H pylori positive patients had higher IEL counts than healthy controls and H pylori negative patients. This quantitative difference in the number of IELs between our two sub-groups of dyspeptic patients is an indirect argument to validate that the patients considered in this study as H pylori negative dyspeptics were really negative. Indeed, this negative status can be discussed as we did not perform H pylori serology (including CagA status) in all patients.
We have confirmed that, like in healthy controls, duodenal IELs in dyspeptic patients are predominant CD3+ CD8+ lymphocytes (> 70% of small bowel IELs)[39-41]. According to a previous study, a significant difference was seen in patients with H pylori gastritis who had an increased of CD8+ CD3+ IEL counts compared with healthy controls[42]. The distribution of CD3+ CD4+ IELs was not different between dyspeptic patients and healthy controls. TCR-γδ CD3+ lymphocytes have been investigated because of their potential role in immune-mediated hypersensitivity via recognition of a variety of both non peptidic and peptidic antigens without requirement for antigen processing/presentation[12]. The median TCR-γδ+ CD3+ percentages are similar to those of normal gut[30,40-44]. Adhesion molecules CD44 and CD103 have also been investigated since they are recognized as co-stimulatory molecules which may regulate interactions between IELs and epithelial cells[45,46]. No difference in their membrane expression was observed between IELs from dyspeptic patients and healthy subjects. In this study, CD44, which belongs to a family of proteins involved in T cell activation and lymphocyte homing to the gut was expressed by the majority of CD3+ IELs as previously reported[13,19,47], and CD103, which is up-regulated in epithelial T lymphocytes and contributes to binding and/or signaling between IELs and epithelial cells was also expressed by most IELs, which is in agreement with previous reports[13,41,48]. Ratios of CD3+ IELs expressing the co-stimulation molecule CD28 were low in both dyspeptic and healthy individuals, which is in agreement with previous reports on freshly isolated small intestinal IELs[13,19,49,50]. As reported in previous studies, most CD3+ IEL expressed CD69, suggesting a "quasi-activated" phenotype in both dyspeptic and healthy adults[13,46,48,51]. Nevertheless, in our study, we could not exclude that differences between duodenal IELs from dyspeptic patients and healthy controls could be partly due to an uncontrolled diet in order to prevent the onset of symptoms following ingestion of some specific nutrients by FD patients. Indeed, the immune responsiveness following oral administration of an antigen is dose-dependent, and depending on dose feeding could lead to clonal anergy or clonal deletion[52]. Shut-down effects of antigen-activated IEL migration to the antigen-sensitized mucosa could alter the population of these antigen-reactive T-cells in the mucosa[53].
Considering the small number of investigated patients and the great scattering of individual values for the two activation markers found to be altered, our findings warrant future studies to determine whether H pylori negative functional dyspepsia is, at least partly, immune-mediated and to ascertain a role of IELs in FD.
Footnotes
S- Editor Liu Y L- Editor Wang XL E- Editor Chen GJ
References
- 1.Madisch A, Miehlke S, Labenz J. Management of functional dyspepsia: Unsolved problems and new perspectives. World J Gastroenterol. 2005;11:6577–6581. doi: 10.3748/wjg.v11.i42.6577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pallotta N, Pezzotti P, Corazziari E. Relationship between antral distension and postprandial symptoms in functional dyspepsia. World J Gastroenterol. 2006;12:6982–6991. doi: 10.3748/wjg.v12.i43.6982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Choung RS, Talley NJ. Novel mechanisms in functional dyspepsia. World J Gastroenterol. 2006;12:673–677. doi: 10.3748/wjg.v12.i5.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tack J, Demedts I, Dehondt G, Caenepeel P, Fischler B, Zandecki M, Janssens J. Clinical and pathophysiological characteristics of acute-onset functional dyspepsia. Gastroenterology. 2002;122:1738–1747. doi: 10.1053/gast.2002.33663. [DOI] [PubMed] [Google Scholar]
- 5.Bouin M, Meunier P, Riberdy-Poitras M, Poitras P. Pain hypersensitivity in patients with functional gastrointestinal disorders: a gastrointestinal-specific defect or a general systemic condition? Dig Dis Sci. 2001;46:2542–2548. doi: 10.1023/a:1012356827026. [DOI] [PubMed] [Google Scholar]
- 6.Parry S, Forgacs I. Intestinal infection and irritable bowel syndrome. Eur J Gastroenterol Hepatol. 2005;17:5–9. doi: 10.1097/00042737-200501000-00002. [DOI] [PubMed] [Google Scholar]
- 7.Chadwick VS, Chen W, Shu D, Paulus B, Bethwaite P, Tie A, Wilson I. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology. 2002;122:1778–1783. doi: 10.1053/gast.2002.33579. [DOI] [PubMed] [Google Scholar]
- 8.Delvaux M. Alterations of sensori-motor functions of the digestive tract in the pathophysiology of irritable bowel syndrome. Best Pract Res Clin Gastroenterol. 2004;18:747–771. doi: 10.1016/j.bpg.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 9.Bueno L, Fioramonti J, Delvaux M, Frexinos J. Mediators and pharmacology of visceral sensitivity: from basic to clinical investigations. Gastroenterology. 1997;112:1714–1743. doi: 10.1016/s0016-5085(97)70056-8. [DOI] [PubMed] [Google Scholar]
- 10.Read NW. Food and hypersensitivity in functional dyspepsia. Gut. 2002;51 Suppl 1:i50–i53. doi: 10.1136/gut.51.suppl_1.i50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Beagley KW, Husband AJ. Intraepithelial lymphocytes: origins, distribution, and function. Crit Rev Immunol. 1998;18:237–254. doi: 10.1615/critrevimmunol.v18.i3.40. [DOI] [PubMed] [Google Scholar]
- 12.Hayday A, Theodoridis E, Ramsburg E, Shires J. Intraepithelial lymphocytes: exploring the Third Way in immunology. Nat Immunol. 2001;2:997–1003. doi: 10.1038/ni1101-997. [DOI] [PubMed] [Google Scholar]
- 13.Mayer L. Mucosal immunity and gastrointestinal antigen processing. J Pediatr Gastroenterol Nutr. 2000;30 Suppl:S4–S12. doi: 10.1097/00005176-200001001-00002. [DOI] [PubMed] [Google Scholar]
- 14.Fujihashi K, Taguchi T, McGhee JR, Eldridge JH, Bruce MG, Green DR, Singh B, Kiyono H. Regulatory function for murine intraepithelial lymphocytes. Two subsets of CD3+, T cell receptor-1+ intraepithelial lymphocyte T cells abrogate oral tolerance. J Immunol. 1990;145:2010–2019. [PubMed] [Google Scholar]
- 15.Ebert EC, Roberts AI. Costimulation of the CD3 pathway by CD28 ligation in human intestinal lymphocytes. Cell Immunol. 1996;171:211–216. doi: 10.1006/cimm.1996.0195. [DOI] [PubMed] [Google Scholar]
- 16.Neutra MR, Mantis NJ, Kraehenbuhl JP. Collaboration of epithelial cells with organized mucosal lymphoid tissues. Nat Immunol. 2001;2:1004–1009. doi: 10.1038/ni1101-1004. [DOI] [PubMed] [Google Scholar]
- 17.Cheroutre H, Madakamutil L. Acquired and natural memory T cells join forces at the mucosal front line. Nat Rev Immunol. 2004;4:290–300. doi: 10.1038/nri1333. [DOI] [PubMed] [Google Scholar]
- 18.Van Damme N, Baeten D, De Vos M, Demetter P, Elewaut D, Mielants H, Verbruggen G, Cuvelier C, Veys EM, De Keyser F. Chemical agents and enzymes used for the extraction of gut lymphocytes influence flow cytometric detection of T cell surface markers. J Immunol Methods. 2000;236:27–35. doi: 10.1016/s0022-1759(99)00243-4. [DOI] [PubMed] [Google Scholar]
- 19.León F, Roldán E, Sanchez L, Camarero C, Bootello A, Roy G. Human small-intestinal epithelium contains functional natural killer lymphocytes. Gastroenterology. 2003;125:345–356. doi: 10.1016/s0016-5085(03)00886-2. [DOI] [PubMed] [Google Scholar]
- 20.Janeway CA, Jones B, Hayday A. Specificity and function of T cells bearing gamma delta receptors. Immunol Today. 1988;9:73–76. doi: 10.1016/0167-5699(88)91267-4. [DOI] [PubMed] [Google Scholar]
- 21.Hongo T, Morimoto Y, Iwagaki H, Kobashi K, Yoshii M, Urushihara N, Hizuta A, Tanaka N. Functional expression of Fas and Fas ligand on human colonic intraepithelial T lymphocytes. J Int Med Res. 2000;28:132–142. doi: 10.1177/147323000002800304. [DOI] [PubMed] [Google Scholar]
- 22.Dhein J, Walczak H, Bäumler C, Debatin KM, Krammer PH. Autocrine T-cell suicide mediated by APO-1/(Fas/CD95) Nature. 1995;373:438–441. doi: 10.1038/373438a0. [DOI] [PubMed] [Google Scholar]
- 23.Yoshikai Y. The interaction of intestinal epithelial cells and intraepithelial lymphocytes in host defense. Immunol Res. 1999;20:219–235. doi: 10.1007/BF02790405. [DOI] [PubMed] [Google Scholar]
- 24.Ebert EC. Do the CD45RO+CD8+ intestinal intraepithelial T lymphocytes have the characteristics of memory cells? Cell Immunol. 1993;147:331–340. doi: 10.1006/cimm.1993.1073. [DOI] [PubMed] [Google Scholar]
- 25.Boirivant M, Pica R, DeMaria R, Testi R, Pallone F, Strober W. Stimulated human lamina propria T cells manifest enhanced Fas-mediated apoptosis. J Clin Invest. 1996;98:2616–2622. doi: 10.1172/JCI119082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.De Maria R, Boirivant M, Cifone MG, Roncaioli P, Hahne M, Tschopp J, Pallone F, Santoni A, Testi R. Functional expression of Fas and Fas ligand on human gut lamina propria T lymphocytes. A potential role for the acidic sphingomyelinase pathway in normal immunoregulation. J Clin Invest. 1996;97:316–322. doi: 10.1172/JCI118418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Di Sabatino A, Ciccocioppo R, Cupelli F, Cinque B, Millimaggi D, Clarkson MM, Paulli M, Cifone MG, Corazza GR. Epithelium derived interleukin 15 regulates intraepithelial lymphocyte Th1 cytokine production, cytotoxicity, and survival in coeliac disease. Gut. 2006;55:469–477. doi: 10.1136/gut.2005.068684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Morimoto Y, Hizuta A, Ding EX, Ishii T, Hongo T, Fujiwara T, Iwagaki H, Tanaka N. Functional expression of Fas and Fas ligand on human intestinal intraepithelial lymphocytes. Clin Exp Immunol. 1999;116:84–89. doi: 10.1046/j.1365-2249.1999.00827.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ferguson A. Intraepithelial lymphocytes of the small intestine. Gut. 1977;18:921–937. doi: 10.1136/gut.18.11.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Snijders F, Meenan J, van den Blink B, van Deventer SJ, ten Kate FJ. Duodenal intraepithelial and lamina propria T lymphocytes in human immunodeficiency virus-infected patients with and without diarrhoea. Scand J Gastroenterol. 1996;31:1176–1181. doi: 10.3109/00365529609036907. [DOI] [PubMed] [Google Scholar]
- 31.Mahadeva S, Wyatt JI, Howdle PD. Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant? J Clin Pathol. 2002;55:424–428. doi: 10.1136/jcp.55.6.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Törnblom H, Lindberg G, Nyberg B, Veress B. Full-thickness biopsy of the jejunum reveals inflammation and enteric neuropathy in irritable bowel syndrome. Gastroenterology. 2002;123:1972–1979. doi: 10.1053/gast.2002.37059. [DOI] [PubMed] [Google Scholar]
- 33.Veress B, Franzén L, Bodin L, Borch K. Duodenal intraepithelial lymphocyte-count revisited. Scand J Gastroenterol. 2004;39:138–144. doi: 10.1080/00365520310007675. [DOI] [PubMed] [Google Scholar]
- 34.Hayat M, Cairns A, Dixon MF, O'Mahony S. Quantitation of intraepithelial lymphocytes in human duodenum: what is normal? J Clin Pathol. 2002;55:393–394. doi: 10.1136/jcp.55.5.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brown I, Mino-Kenudson M, Deshpande V, Lauwers GY. Intraepithelial lymphocytosis in architecturally preserved proximal small intestinal mucosa: an increasing diagnostic problem with a wide differential diagnosis. Arch Pathol Lab Med. 2006;130:1020–1025. doi: 10.5858/2006-130-1020-ILIAPP. [DOI] [PubMed] [Google Scholar]
- 36.Memeo L, Jhang J, Mansukhani M, Hibshoosh H, Rotterdam H, Bhagat G. Duodenal intraepithelial lymphocytosis : manifestation of a host response in patients with H pylori gastritis. Histopathology. 2002;41:174–174. [Google Scholar]
- 37.Hayat M, Arora DS, Dixon MF, Clark B, O'Mahony S. Effects of Helicobacter pylori eradication on the natural history of lymphocytic gastritis. Gut. 1999;45:495–498. doi: 10.1136/gut.45.4.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nahon S, Patey-Mariaud De Serre N, Lejeune O, Huchet FX, Lahmek P, Lesgourgues B, Traissac L, Bodiguel V, Adotti F, Tuszynski T, et al. Duodenal intraepithelial lymphocytosis during Helicobacter pylori infection is reduced by antibiotic treatment. Histopathology. 2006;48:417–423. doi: 10.1111/j.1365-2559.2006.02358.x. [DOI] [PubMed] [Google Scholar]
- 39.Cerf-Bensussan N, Guy-Grand D, Griscelli C. Intraepithelial lymphocytes of human gut: isolation, characterisation and study of natural killer activity. Gut. 1985;26:81–88. doi: 10.1136/gut.26.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Brandtzaeg P, Bosnes V, Halstensen TS, Scott H, Sollid LM, Valnes KN. T lymphocytes in human gut epithelium preferentially express the alpha/beta antigen receptor and are often CD45/UCHL1-positive. Scand J Immunol. 1989;30:123–128. doi: 10.1111/j.1365-3083.1989.tb01196.x. [DOI] [PubMed] [Google Scholar]
- 41.Jarry A, Cerf-Bensussan N, Brousse N, Selz F, Guy-Grand D. Subsets of CD3+ (T cell receptor alpha/beta or gamma/delta) and CD3- lymphocytes isolated from normal human gut epithelium display phenotypical features different from their counterparts in peripheral blood. Eur J Immunol. 1990;20:1097–1103. doi: 10.1002/eji.1830200523. [DOI] [PubMed] [Google Scholar]
- 42.Memeo L, Jhang J, Hibshoosh H, Green PH, Rotterdam H, Bhagat G. Duodenal intraepithelial lymphocytosis with normal villous architecture: common occurrence in H. pylori gastritis. Mod Pathol. 2005;18:1134–1144. doi: 10.1038/modpathol.3800404. [DOI] [PubMed] [Google Scholar]
- 43.Trejdosiewicz LK. What is the role of human intestinal intraepithelial lymphocytes? Clin Exp Immunol. 1993;94:395–397. doi: 10.1111/j.1365-2249.1993.tb08207.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Abuzakouk M, Feighery C, Kelleher D, O'Briain DS, Jones E, Weir D, Casey E, O'Farrelly C. Increased HLA-DR and CD44 antigen expression in the gut: evidence of extraarticular immunological activity in rheumatoid arthritis. J Rheumatol. 1999;26:1869–1876. [PubMed] [Google Scholar]
- 45.Sarnacki S, Bègue B, Buc H, Le Deist F, Cerf-Bensussan N. Enhancement of CD3-induced activation of human intestinal intraepithelial lymphocytes by stimulation of the beta 7-containing integrin defined by HML-1 monoclonal antibody. Eur J Immunol. 1992;22:2887–2892. doi: 10.1002/eji.1830221120. [DOI] [PubMed] [Google Scholar]
- 46.Sillett HK, Southgate J, Howdle PD, Trejdosiewicz LK. Expression of activation and costimulatory elements by human intestinal intraepithelial lymphocytes. Scand J Immunol. 1999;50:52–60. doi: 10.1046/j.1365-3083.1999.00561.x. [DOI] [PubMed] [Google Scholar]
- 47.Salmi M, Jalkanen S. Regulation of lymphocyte traffic to mucosa-associated lymphatic tissues. Gastroenterol Clin North Am. 1991;20:495–510. [PubMed] [Google Scholar]
- 48.Shibahara T, Si-Tahar M, Shaw SK, Madara JL. Adhesion molecules expressed on homing lymphocytes in model intestinal epithelia. Gastroenterology. 2000;118:289–298. doi: 10.1016/s0016-5085(00)70211-3. [DOI] [PubMed] [Google Scholar]
- 49.Kagnoff MF. Immunology of the intestinal tract. Gastroenterology. 1993;105:1275–1280. doi: 10.1016/0016-5085(93)90128-y. [DOI] [PubMed] [Google Scholar]
- 50.Gramzinski RA, Adams E, Gross JA, Goodman TG, Allison JP, Lefrançois L. T cell receptor-triggered activation of intraepithelial lymphocytes in vitro. Int Immunol. 1993;5:145–153. doi: 10.1093/intimm/5.2.145. [DOI] [PubMed] [Google Scholar]
- 51.Ebert EC. Proliferative responses of human intraepithelial lymphocytes to various T-cell stimuli. Gastroenterology. 1989;97:1372–1381. doi: 10.1016/0016-5085(89)90379-x. [DOI] [PubMed] [Google Scholar]
- 52.Chen Y, Inobe J, Marks R, Gonnella P, Kuchroo VK, Weiner HL. Peripheral deletion of antigen-reactive T cells in oral tolerance. Nature. 1995;376:177–180. doi: 10.1038/376177a0. [DOI] [PubMed] [Google Scholar]
- 53.Komoto S, Miura S, Koseki S, Goto M, Hachimura S, Fujimori H, Hokari R, Hara Y, Ogino T, Watanabe C, et al. Effect of specific antigen stimulation on intraepithelial lymphocyte migration to small intestinal mucosa. Clin Exp Immunol. 2005;140:249–257. doi: 10.1111/j.1365-2249.2005.02761.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


