Abstract
Acute lung injury (ALI) is characterized by pulmonary inflammation and edema. Innate immune cells (e.g., neutrophils and macrophages) are major contributors to inflammation in ALI. Less is known regarding the role of T cells. We examined the effects of rapamycin on inflammation in a LPS-induced murine model of ALI. Rapamycin was administered before and after initiation of injury. Inflammatory parameters, including bronchoalveolar lavage cell counts, T cell surface markers (i.e., cytotoxic T lymphocyte antigen 4 [CTLA4] and fork head-winged helix transcription factor [Foxp3]), T cell activation (CD69), IL-6, and IL-10 were analyzed. Rapamycin significantly decreased inflammatory parameters and decreased Foxp3, CTLA4, and CD69 in CD4+ T cells. Rapamycin administration before or after the onset of lung injury, as well as systemically or by pulmonary routes, ameliorates inflammation in ALI.
Keywords: T cells, CTLA4, LPS, acute lung injury, rapamycin
Clinical Relevance
These studies examined the effects of rapamycin on inflammation in a LPS-induced murine model of acute lung injury (ALI). Rapamycin administration before or after onset of ALI, as well as systemically or by pulmonary routes, ameliorates inflammation in ALI. These findings have the potential to affect future therapeutics for ALI.
Acute lung injury (ALI) is a major cause of severe respiratory failure. The pathogenesis of ALI remains ill defined, and the treatment of ALI remains largely supportive (1, 2). In the acute phase, there is evidence of interstitial and alveolar edema with accumulation of neutrophils and macrophages in the alveoli (3–5). Innate immune cells (e.g., neutrophils) contribute to the disruption of the alveolar–capillary barrier and accumulation of protein-rich edema. Less is known regarding the role of adaptive immune cells (e.g., T cells) in ALI. We previously showed that T cell pathways involving cytotoxic T lymphocyte antigen 4 (CTLA4) may modulate inflammation in ALI (6).
Rapamycin is an immunosuppressant that inhibits mammalian target of rapamycin (mTOR) and T cell activation and differentiation (7, 8). Rapamycin administration prevents allograft rejection (9), has entered clinical trials as an anticancer agent (10), and improves lymphangioleiomyomatosis, a progressive cystic lung disease (11). In a model of ALI, rapamycin reduced neutrophil activation (12). To test the impact of rapamycin on T cell activation in ALI, we analyzed a murine model of ALI induced by LPS, a component of the cell wall of gram-negative bacteria. We focused on the impact of rapamycin on T cell–associated molecules, including CTLA4 and fork head-winged helix transcription factor (Foxp3).
Materials and Methods
Mice
Female Balb/c mice (8–10 wk old) were purchased from Harlan Breeders (Indianapolis, IN). The mice were maintained according to the guidelines of the University of California, San Diego Animal Care Program and University of Illinois Animal Care Committee.
Materials
LPS from Escherichia coli 055:B5, purified by gel-filtration chromatography, was purchased from Sigma-Aldrich (St. Louis, MO). Rapamycin was purchased from Eton Bioscience Inc. (San Diego, CA).
Cells Proliferation
Murine spleen cells from Balb/c mice were grown in RPMI 1640 medium with 10% heat-inactivated FBS. Proliferation (2 × 105 cells per well) was determined using a colorimetric immunoassay based on the measurement of BrdU incorporation during DNA synthesis (Roche Diagnostics, Indianapolis, IN). Forty-eight hours after stimulation, cells were pulsed with BrdU solution, denatured with FixDenat solution, and incubated with mouse anti-BrdU mAbs conjugated to peroxidase. After the Ab conjugate was removed, substrate solution was added for 20 minutes. The absorbance was measured at 370 nm using an ELISA plate reader.
Immunoblotting
FACS-sorted cells (described below) were stimulated with LPS (10 μg/ml) and concanavalin A (1 μg/ml) in the presence or absence of rapamycin (50 nM) for 48 hours. Cells were homogenized and lysed in mammalian cellytic lysis buffer (Sigma-Aldrich) containing phosphatase inhibitors (10 mM sodium pyrophosphate, 20 mM β-glycero-glycerophosphate, 100 mM NaF, 5 mM indoleacetic acid, 20 nM okadaic acid) and a protease inhibitor cocktail (Roche Applied Science, Mannheim, Germany). Solubilized proteins were collected by centrifugation and quantified using a protein assay reagent (Bio-Rad, Hercules, CA). Equal amounts of protein of each sample were resolved by SDS gel electrophoresis using a 4 to 15% gradient gel from Bio-Rad and transferred to polyvinylidenedifluoride membranes (Bio-Rad). Immunoblotting was performed using anti-S6K, anti–phospho-p70 S6 kinase (Thr389) (p-S6K), anti–phospho-Akt (Ser473) (p-S473) (Cell Signaling), Akt (pan) (C67E7) Rabbit mAb (panAKT), antitubulin (Sigma-Aldrich), or anti-rabbit or anti-mouse immunoglobulin HRP antibodies.
Murine Model of ALI
Balb/c mice were anesthetized with isoflurane (Minrad, Bethlehem, PA). The tongue was gently extended, and the tip of an otoscope was introduced to reach the trachea. LPS (100 μg) in 50 μl PBS was administered intratracheally through the otoscope as described elsewhere (6). Rapamycin or vehicle (PBS) was administered as described in the figure legends. Mice were harvested at 48 hours after LPS exposure.
Bronchoalveolar Lavage Harvest and Cell Count
Bronchoalveolar lavage (BAL) fluid was obtained by cannulating the trachea and lavaging the lungs three times with 1 ml PBS containing 0.6 mM EDTA. BAL fluid cells were pelleted, and the supernatant was stored at −80°C until use. BAL fluid cells were counted using a hemocytometer and resuspended in RPMI 1640 (5 × 105 cells/ml). Slides for differential cell counts were prepared with Cytospin (Thermo Scientific, Waltham, MA) and fixed and stained with Diff-Quick (Imeb, San Marcos, CA). For each sample, an investigator blinded to the treatment groups performed two counts of 100 cells.
Cytokines and Lung Homogenates
Levels of BAL IL-6 were measured by a multiplexed immunoassay (Invitrogen, Carlsbad, CA). Mean fluorescence intensity was measured by Luminex 100 total system (Luminex, Austin, TX). After the BAL procedure, the lungs were perfused with saline, isolated, and incubated at 37°C for 60 minutes in a digestion mixture consisting of collagenase from Clostridium histolyticum, type IV (1 mg/ml) (Sigma-Aldrich) and DNase I from bovine pancreas (0.5 mg/ml) (Sigma-Aldrich). Cell suspensions were obtained by passing the homogenate through a 70-μm nylon Falcon cell strainer (BD Biosciences, San Jose, CA) to remove pieces of tissue and debris. After lysis of erythrocytes with ammonium chloride potassium bicarbonate lysing buffer, cell pellets were resuspended and washed in RPMI 1640 medium.
Flow Cytometric Analysis
Spleen cells or lung homogenates were resuspended in staining buffer (PBS containing 2% FBS and 0.05% sodium azide). After incubating for 30 minutes at 4°C with anti-mouse CD3 (PECy7) and CD4 (FITC) Abs or their respective immunoglobulin isotypes (eBioscience, San Diego, CA), samples were washed twice with staining buffer and fixed by Fixation/Permeabilization solution (eBioscience). Intracellular staining with anti-mouse CTLA4 (PE) and Foxp3 (APC) Abs was performed. Samples were run on a BD FACSCalibur flow cytometry system (BD Biosciences). Data were analyzed on FlowJo v8 software (Tree Star, Ashland, OR).
Spleen cells were filtered using 70-μM filters, and T cells were labeled with anti-CD4 and anti-CD25 fluorophore antibodies (eBioscience). CD4 was labeled with FITC, and CD25 was labeled with PE (eBioscience). FITC-labeled CD4+CD25− and PE-labeled CD4+CD25+ cells were sorted by flow cytometry and gated to sort for the CD4+CD25+ high population. Cells were stimulated with concanavalin A (1 μg/ml) and LPS (10 μg/ml) in the presence and absence of rapamycin (50 nM) for 48 hours.
Statistical Analysis
Data are expressed as means ± SEM. All parameters were evaluated with the two-tailed unpaired Student t test or compared by one-way ANOVA, followed by the Bonferroni test. A P value < 0.05 was considered significant.
Results
Rapamycin Modifies CTLA4 Expression
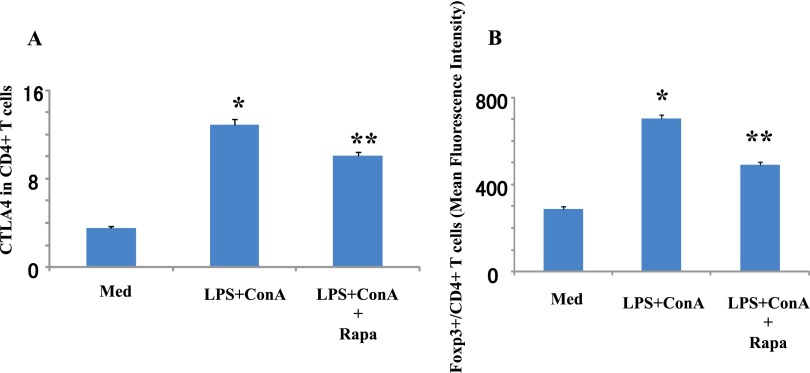
Rapamycin has been shown to decrease adaptive immune responses, but the impact on innate pathways is less well defined. We previously showed that adaptive stimulation increases CTLA4 expression that is modified by rapamycin (13). Here we find that innate stimulation increases CTLA4 expression in CD4+ T cells, which is significantly decreased by rapamycin administration (Figure 1A). Innate stimulation also significantly increases Foxp3 expression in Foxp3+ CD4+ cells, whereas rapamycin decreases Foxp3 expression (Figure 1B).
Figure 1.
Rapamycin decreases T cell markers. Spleen cells from Balb/c mice were harvested and stimulated with LPS (10 μg/ml) and concanavalin A (ConA; 1 μg/ml) for 48 hours. Within 30 minutes of stimulation, rapamycin (Rapa; 10 μg/ml) was added. Expression levels of cytotoxic T lymphocyte antigen 4 (CTLA4) (A) and the ratio of Foxp3+ cells (B) in the CD4+ T cell population were measured by flow cytometry. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus medium (Med). **P < 0.05 versus LPS + ConA.
Rapamycin Decreases Innate Induced Inflammatory Responses
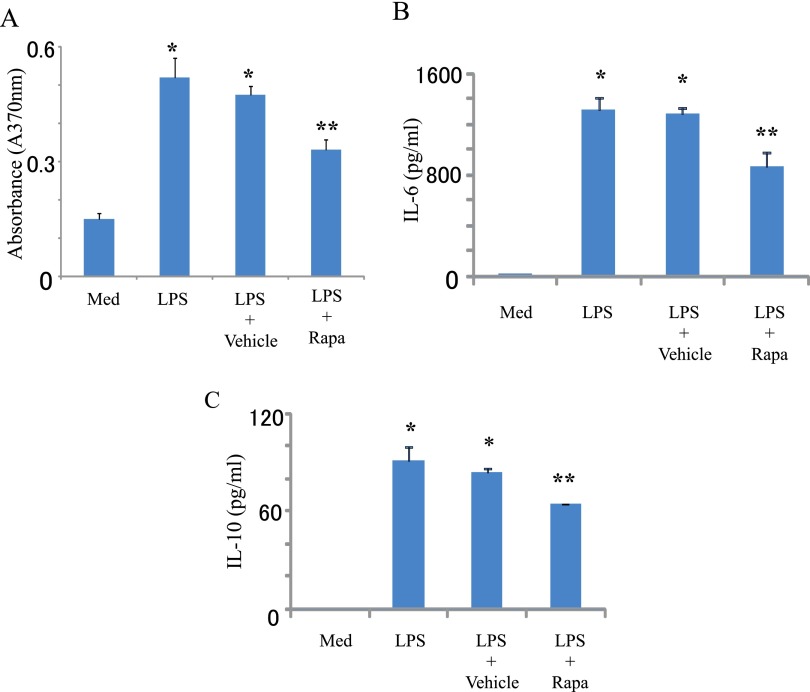
We next examined the impact of rapamycin on innate induced lymphocyte proliferation and cytokine production (Figure 2). Innate stimuli increases cell proliferation in a dose-dependent manner (1–100 μg/ml) (Figure 2 and not shown) as well as IL-6 and IL-10 production, responses that are decreased by rapamycin (Figures 2A–2C).
Figure 2.
Rapamycin decreases LPS-induced proliferation and cytokine production. Spleen cells from Balb/c mice were harvested and stimulated with LPS (2.5 μg/ml). Rapamycin (Rapa; 100 μg/ml) or vehicle (PBS) was added 24 hours after LPS stimulation. After 48 hours, cell proliferation was measured by BrdU assay (A). Supernatant was collected at 48 hours, and IL-6 (B) and IL-10 (C) were measured by a multiplex assay. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus medium (Med). **P < 0.05 versus vehicle.
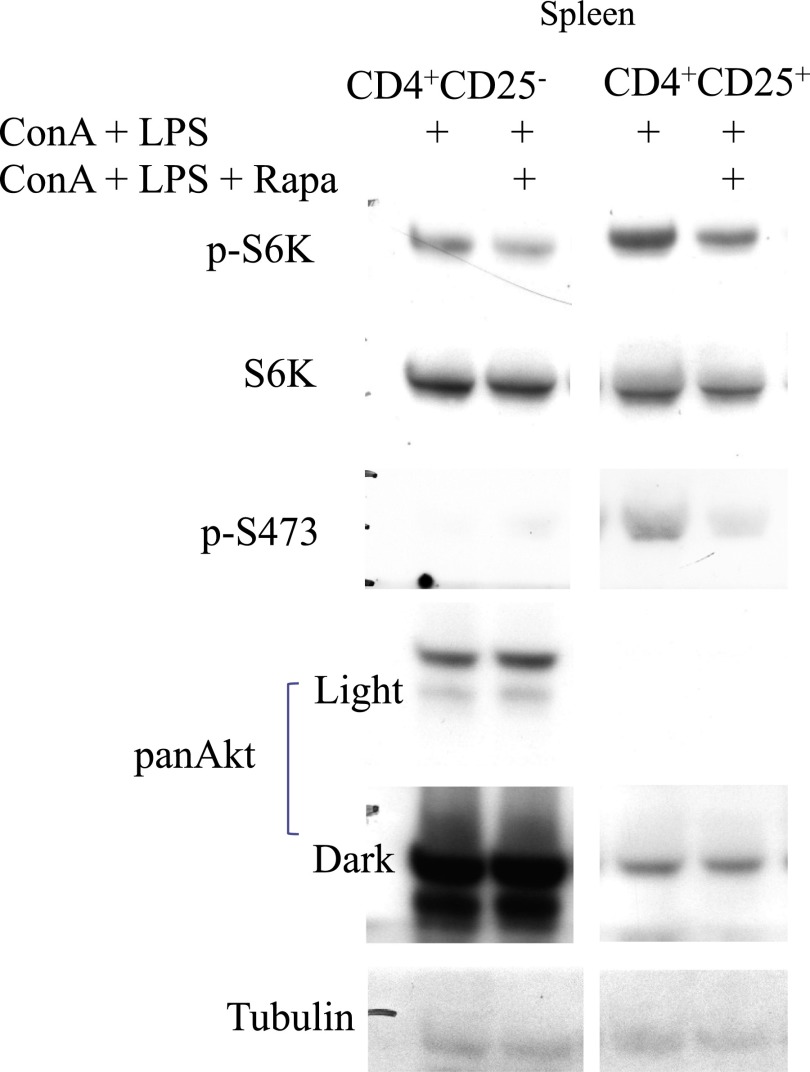
We next focused on the Akt–mTORC1 pathway, a target for rapamycin that is known to facilitate innate immune responses in ALI (14). We analyzed potential differences between CD4+CD25+ cells (FACS-sorted, express CTLA4) and CD4+CD25− cells. FACS-sorted CD4+CD25+ high and CD4+CD25− cells were treated with innate stimuli in the presence or absence of rapamycin and analyzed for protein levels by Western blot (Figure 3). CD4+CD25+ cells exhibit higher phosphorylation of S6K and Akt compared with CD4+CD25− cells. Also, the ratio of p-S6K to total S6K and p-S473 to total Akt is higher in CD4+CD25+ compared with CD4+CD25− cells by densitometry analysis (not shown).
Figure 3.
Analysis of Akt and mTORC1 activity in sorted CD4+CD25+ cells. Spleen cells from Balb/c mice were harvested, stimulated, and FACS sorted for CD4+CD25+ and CD4+CD25− cells, and protein was subjected to Western blotting as described in Materials and Methods.
Rapamycin Decreases Innate Induced Inflammatory Responses In Vivo
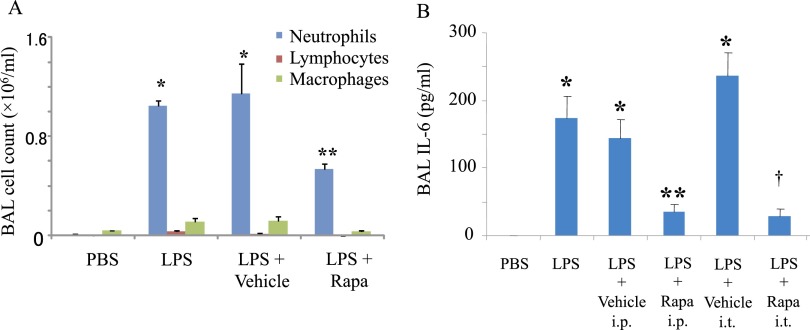
We tested the effect of rapamycin in vivo in a murine model of innate LPS-induced ALI. The LPS model of ALI is characterized by increased neutrophil infiltration and IL-6 secretion in the BAL (Figures 4A and 4B). Administration of rapamycin by a systemic route before LPS exposure (intraperitoneally) significantly decreases pulmonary inflammatory responses (Figures 4A and 4B). We also assessed the impact of timing of rapamycin administration in ALI. Rapamycin administration after LPS exposure also significantly decreases BAL IL-6, a marker of pulmonary inflammation (Figure 4B). Rapamycin significantly decreases pulmonary inflammation whether administered by systemic or local pulmonary (intratracheal) routes.
Figure 4.
Rapamycin decreases LPS-induced pulmonary inflammation. LPS or PBS was administered intratracheally to Balb/c mice. Rapamycin (Rapa; 100 μg) or vehicle (PBS) was administered intraperitoneally 3 hours before LPS exposure (A). Bronchoalveolar lavage (BAL) fluid was collected 48 hours after LPS exposure. BAL cell counts were determined by differential staining. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus PBS group. **P < 0.05 versus vehicle group. Rapamycin (Rapa; 100 μg) or vehicle (PBS) was administered by systemic (intraperitoneal [i.p.]) or local intratracheal (i.t.) routes 3 hours after LPS exposure (B). BAL fluid was collected 48 hours after LPS exposure. IL-6 in BAL was determined by a multiplex assay. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus PBS group. **P < 0.05 versus vehicle i.p. group. †P < 0.05 vehicle i.t. group.
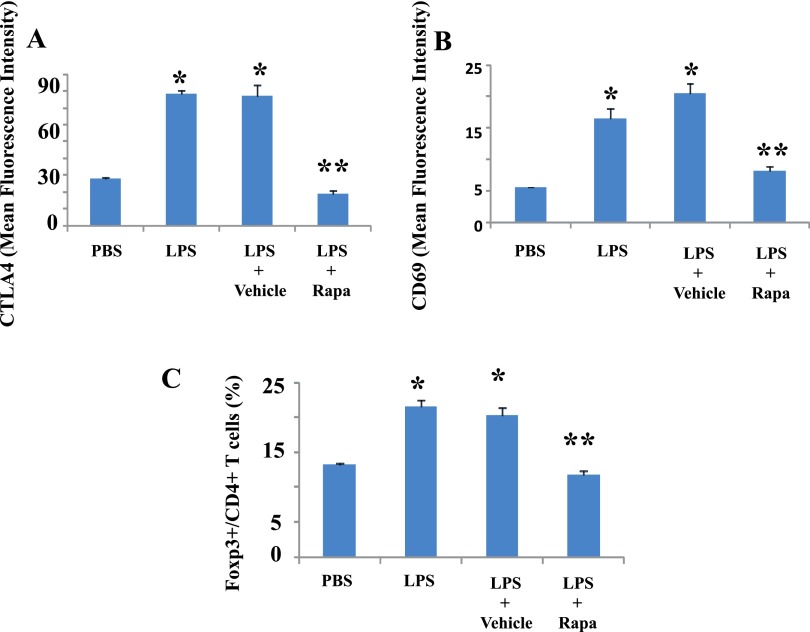
Rapamycin Decreases CTLA4 Expression Induced by Innate Stimuli In Vivo
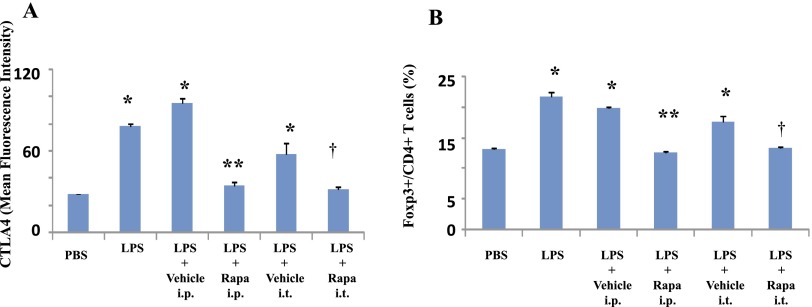
We have previously shown that CTLA4 modulates pulmonary inflammation in ALI (6). LPS induces CTLA4 expression, and we tested whether rapamycin would affect LPS-induced CTLA4 expression in ALI. Flow cytometry analyses indicate that expression of CTLA4 and CD69 (a T cell activation marker) and Foxp3 in pulmonary CD4+ T cells is significantly increased by innate stimuli in vivo (Figures 5A–5C). Rapamycin administration in vivo decreases these T cell markers. We next tested the impact of systemic and local pulmonary administration of rapamycin after LPS exposure on CTLA4 expression. Both routes of rapamycin administration significantly decrease CTLA4 expression and Foxp3+ T cells (Figures 6A and 6B).
Figure 5.
Rapamycin decreases LPS-induced T cell–associated markers. LPS or PBS was administered intratracheally to Balb/c mice. Rapamycin (Rapa; 100 μg) or vehicle (PBS) was administered intraperitoneally 3 hours before LPS exposure. Lungs were isolated 48 hours after LPS exposure. Expression levels (mean fluorescence intensity) of CTLA4 (A) and CD69 (B) and ratio of Foxp3+ cells (C) in the CD4+ T cell population in lung cells were determined by flow cytometry. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus PBS group. **P < 0.05 versus vehicle group.
Figure 6.
Rapamycin diminishes T cell markers by systemic and local routes. LPS or PBS was administered intratracheally to Balb/c mice. Rapamycin (Rapa; 100 μg) or vehicle (PBS) was administered intraperitoneally or intratracheally 3 hours after LPS exposure. Lungs were isolated 48 hours after LPS exposure. Expression levels (mean fluorescence intensity) of CTLA4 (A) and the ratio of Foxp3+ cells (B) in the CD4+ T cell population in lung cells were determined by flow cytometry. Data are shown as mean ± SEM (n = 3 per group). *P < 0.05 versus PBS group. **P < 0.05 versus vehicle i.p. group. †P < 0.05 versus vehicle i.t. group.
Discussion
We analyzed the effect of rapamycin on T cell activation induced in vitro and in vivo. Rapamycin is a specific inhibitor of mTOR, a serine/threonine kinase that regulates protein production essential for cellular proliferation (15). A critical role for mTOR in regulating immune responses is emerging. T cell activation requires at least two signals: signal 1 and costimulatory signals. Signal 1 refers to antigen recognition through the T cell receptor. The activation of T cells is dictated by the net sum of costimulatory signals, such as CD28, PD-1, and CTLA4 (13, 16, 17). mTOR may act to integrate costimulatory signals, which in turn direct the outcome of T cell differentiation and activation.
The PI3K/Akt/mTOR pathway regulates cellular functions, including cell growth, proliferation, differentiation, and survival. The Akt-mTORC1 pathway mediates ALI via increased chemotaxis and proliferation of innate immune cells (14). The Akt–mTORC1 pathway also inhibits T regulatory cell differentiation and proliferation (18). The CD4+CD25+ high cell population (with higher levels of CTLA4 than the CD4+CD25− cells) exhibits higher Akt and mTORC1 activity as assessed by increased phosphorylation of Akt and S6K, the direct target of mTORC1. CD4+CD25+ cells exhibit higher activation than the CD4+CD25− cells. This higher activation may afford better survival and proliferation of CD4+CD25+ cells with or without rapamycin.
We tested whether the T cell–associated molecules CTLA4 and Foxp3 are modulated by rapamycin. CTLA4 is a structural homolog of CD28 and binds the same ligands, CD80 and CD86. CTLA4 is mainly expressed in intracellular recycling vesicles and is transported to the cell surface upon T cell activation (19). CTLA4 signals result in T cell unresponsiveness, whereas CD28 is constitutively expressed on the surface of resting T cells and promotes T cell activation (20). Foxp3 is a transcription factor that has been shown to regulate immunomodulatory functions in immune cells. In this study, rapamycin decreases LPS-induced CTLA4 and Foxp3 expression in CD4+ T cells as well as lymphocyte proliferation and cytokine production. These data suggest that rapamycin may suppress innate-induced cell activation via T cell–modulating pathways that include CTLA4 and FoxP3.
Rapamycin also decreases innate-induced inflammation in vivo. Our results indicate that rapamycin decreases LPS-induced CD69 expression. Because CD69 is a marker for T cell activation, this suggests that rapamycin may decrease inflammation by suppressing T cell activation. Prior studies analyzing different models indicate that rapamycin suppresses T cell activation and modulates the balance of T effector cells and T regulatory cells (15, 18, 21–23).
Prior literature suggests that Foxp3 and CTLA4 exert primarily immunosuppressive effects. Recent work suggests that Foxp3 or CTLA4, under the appropriate timing and conditions, may contribute to a proinflammatory effect. In a prior study, we found that interfering with CTLA4 (by administration of an anti-CTLA4 antibody) did not result in the expected finding of increased inflammation but rather decreased pulmonary inflammation in a model of ALI (6). Another study found that anti-CTLA4 can improve or worsen sepsis depending on the timing of the model and administration (24).
The concept that Foxp3 may exert proinflammatory effects has recently been proposed. Sharma and colleagues (25) identified a new population of Foxp3+ T regulatory cells that are converted to a proinflammatory subtype due to lost expression of the transcription factor Eos. Also, a subset of Foxp3+ cells that are CD161+ has been shown to be capable of producing proinflammatory cytokines (26). In contrast, IL-17, a cytokine presumed to be proinflammatory, may exert protective effects (27). When IL-17 exerts proinflammatory effects, T regulatory cells (Foxp3+ and/or CTLA4+) may exert anti-inflammatory effects to maintain an immune balance. We speculate that, depending on the timing, when IL-17 exerts protective effects, Foxp3+ and/or CTLA4+ cells may regulate immune balance by exerting proinflammatory effects.
Taken together, these studies are consistent with our finding that, depending on the timing and conditions, Foxp3 and CTLA4, classically defined as immunosuppressive, may be decreased in parallel with decreased inflammation. We also underscore that prior analyses of Foxp3 and CTLA4 predominantly examined adaptive, antigen-dependent responses, whereas our studies focus on innate, non–antigen-dependent responses. We evaluate LPS administration in vivo modulating Foxp3 and CTLA4 expression. The use of innate, rather than adaptive, stimuli may contribute to our unexpected finding of decreased Foxp3 and CTLA4 in parallel with decreased inflammation after rapamycin administration. Also, prior analyses of Foxp3 and CTLA4 have not analyzed cells from the lung or BAL. Whether tissue type in addition to innate versus adaptive stimuli and timing or ratio of effector to suppressor cells affects Foxp3 or CTLA4 expression merits further investigation.
Whether before or after LPS administration, rapamycin decreases lung inflammation. Also, both systemic and local administration of rapamycin decreases BAL IL-6, a proinflammatory mediator that coordinates acute-phase responses. Thus, rapamycin influences adaptive pathways in LPS-induced inflammation in vivo by decreasing expression of T cell–related molecules. Prior studies indicate rapamycin’s immunosuppressive effects in transplantation and drug eluting stents. These studies indicate that rapamycin may decrease pulmonary inflammation in ALI by influencing T cell pathways.
Acknowledgments
Acknowledgments
The authors thank Kathryn Reed for assistance with manuscript preparation.
Footnotes
This work was supported by National Institutes of Health grants R01AI053878–10 and 5R01AI075317.
Originally Published in Press as DOI: 10.1165/rcmb.2013-0171OC on March 18, 2014
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000;342:1334–1349. doi: 10.1056/NEJM200005043421806. [DOI] [PubMed] [Google Scholar]
- 2.Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, Stern EJ, Hudson LD. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–1693. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- 3.Harris JF, Aden J, Lyons CR, Tesfaigzi Y. Resolution of LPS-induced airway inflammation and goblet cell hyperplasia is independent of IL-18. Respir Res. 2007;8:24. doi: 10.1186/1465-9921-8-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Morris PE, Glass J, Cross R, Cohen DA. Role of T-lymphocytes in the resolution of endotoxin-induced lung injury. Inflammation. 1997;21:269–278. doi: 10.1023/a:1027393715300. [DOI] [PubMed] [Google Scholar]
- 5.Xie YC, Dong XW, Wu XM, Yan XF, Xie QM. Inhibitory effects of flavonoids extracted from licorice on lipopolysaccharide-induced acute pulmonary inflammation in mice. Int Immunopharmacol. 2009;9:194–200. doi: 10.1016/j.intimp.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 6.Nakajima T, Suarez CJ, Lin KW, Jen KY, Schnitzer JE, Makani SS, Parker N, Perkins DL, Finn PW. T cell pathways involving CTLA4 contribute to a model of acute lung injury. J Immunol. 2010;184:5835–5841. doi: 10.4049/jimmunol.0903238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sakaguchi S, Yamaguchi T, Nomura T, Ono M. Regulatory T cells and immune tolerance. Cell. 2008;133:775–787. doi: 10.1016/j.cell.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 8.Luo H, Duguid W, Chen H, Maheu M, Wu J. The effect of rapamycin on T cell development in mice. Eur J Immunol. 1994;24:692–701. doi: 10.1002/eji.1830240331. [DOI] [PubMed] [Google Scholar]
- 9.Augustine JJ, Bodziak KA, Hricik DE. Use of sirolimus in solid organ transplantation. Drugs. 2007;67:369–391. doi: 10.2165/00003495-200767030-00004. [DOI] [PubMed] [Google Scholar]
- 10.Wendel HG, De Stanchina E, Fridman JS, Malina A, Ray S, Kogan S, Cordon-Cardo C, Pelletier J, Lowe SW. Survival signalling by Akt and eIF4E in oncogenesis and cancer therapy. Nature. 2004;428:332–337. doi: 10.1038/nature02369. [DOI] [PubMed] [Google Scholar]
- 11.McCormack FX, Inoue Y, Moss J, Singer LG, Strange C, Nakata K, Barker AF, Chapman JT, Brantly ML, Stocks JM, et al. National Institutes of Health Rare Lung Diseases Consortium; MILES Trial Group. Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N Engl J Med. 2011;364:1595–1606. doi: 10.1056/NEJMoa1100391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lorne E, Zhao X, Zmijewski JW, Liu G, Park YJ, Tsuruta Y, Abraham E. Participation of mammalian target of rapamycin complex 1 in Toll-like receptor 2- and 4-induced neutrophil activation and acute lung injury. Am J Respir Cell Mol Biol. 2009;41:237–245. doi: 10.1165/rcmb.2008-0290OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Finn PW, He H, Wang Y, Wang Z, Guan G, Listman J, Perkins DL. Synergistic induction of CTLA-4 expression by costimulation with TCR plus CD28 signals mediated by increased transcription and messenger ribonucleic acid stability. J Immunol. 1997;158:4074–4081. [PubMed] [Google Scholar]
- 14.Abraham E. Neutrophils and acute lung injury. Crit Care Med. 2003;31(4 Suppl):S195–S199. doi: 10.1097/01.CCM.0000057843.47705.E8. [DOI] [PubMed] [Google Scholar]
- 15.Janes MR, Fruman DA. Immune regulation by rapamycin: moving beyond T cells. Sci Signal. 2009;2:pe25. doi: 10.1126/scisignal.267pe25. [DOI] [PubMed] [Google Scholar]
- 16.Mark DA, Donovan CE, De Sanctis GT, Krinzman SJ, Kobzik L, Linsley PS, Sayegh MH, Lederer J, Perkins DL, Finn PW. Both CD80 and CD86 co-stimulatory molecules regulate allergic pulmonary inflammation. Int Immunol. 1998;10:1647–1655. doi: 10.1093/intimm/10.11.1647. [DOI] [PubMed] [Google Scholar]
- 17.Mark DA, Donovan CE, De Sanctis GT, He HZ, Cernadas M, Kobzik L, Perkins DL, Sharpe A, Finn PW. B7-1 (CD80) and B7-2 (CD86) have complementary roles in mediating allergic pulmonary inflammation and airway hyperresponsiveness. Am J Respir Cell Mol Biol. 2000;22:265–271. doi: 10.1165/ajrcmb.22.3.3747. [DOI] [PubMed] [Google Scholar]
- 18.Haxhinasto S, Mathis D, Benoist C. The AKT-mTOR axis regulates de novo differentiation of CD4+Foxp3+ cells. J Exp Med. 2008;205:565–574. doi: 10.1084/jem.20071477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Perkins D, Wang Z, Donovan C, He H, Mark D, Guan G, Wang Y, Walunas T, Bluestone J, Listman J, et al. Regulation of CTLA-4 expression during T cell activation. J Immunol. 1996;156:4154–4159. [PubMed] [Google Scholar]
- 20.Rudd CE. The reverse stop-signal model for CTLA4 function. Nat Rev Immunol. 2008;8:153–160. doi: 10.1038/nri2253. [DOI] [PubMed] [Google Scholar]
- 21.Wang Y, Camirand G, Lin Y, Froicu M, Deng S, Shlomchik WD, Lakkis FG, Rothstein DM. Regulatory T cells require mammalian target of rapamycin signaling to maintain both homeostasis and alloantigen-driven proliferation in lymphocyte-replete mice. J Immunol. 2011;186:2809–2818. doi: 10.4049/jimmunol.0903805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Säemann MD, Haidinger M, Hecking M, Hörl WH, Weichhart T. The multifunctional role of mTOR in innate immunity: implications for transplant immunity. Am J Transplant. 2009;9:2655–2661. doi: 10.1111/j.1600-6143.2009.02832.x. [DOI] [PubMed] [Google Scholar]
- 23.Zheng Y, Rudensky AY. Foxp3 in control of the regulatory T cell lineage. Nat Immunol. 2007;8:457–462. doi: 10.1038/ni1455. [DOI] [PubMed] [Google Scholar]
- 24.Inoue S, Bo L, Bian J, Unsinger J, Chang K, Hotchkiss RS. Dose-dependent effect of anti-CTLA-4 on survival in sepsis. Shock. 2011;36:38–44. doi: 10.1097/SHK.0b013e3182168cce. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sharma MD, Huang L, Choi JH, Lee EJ, Wilson JM, Lemos H, Pan F, Blazar BR, Pardoll DM, Mellor AL, et al. An inherently bifunctional subset of Foxp3+ T helper cells is controlled by the transcription factor eos. Immunity. 2013;38:998–1012. doi: 10.1016/j.immuni.2013.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pesenacker AM, Bending D, Ursu S, Wu Q, Nistala K, Wedderburn LR. CD161 defines the subset of FoxP3+ T cells capable of producing proinflammatory cytokines. Blood. 2013;121:2647–2658. doi: 10.1182/blood-2012-08-443473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ke Y, Liu K, Huang GQ, Cui Y, Kaplan HJ, Shao H, Sun D. Anti-inflammatory role of IL-17 in experimental autoimmune uveitis. J Immunol. 2009;182:3183–3190. doi: 10.4049/jimmunol.0802487. [DOI] [PMC free article] [PubMed] [Google Scholar]