Abstract
Background:
Glycyrrhiza (licorice) an herb, which has medicinal value and was used in ancient times as a remedy for a great diversity of ailments and sweetener. Roots of Glycyrrhiza contain a high concentration of saponin and glycyrrhizin, which are supposed to be sweetening agents. The aim of the study was to compare the efficacy of aqueous and alcoholic licorice root extract against Streptococcus mutans and Lactobacillus acidophilus in comparison to chlorhexidine (CHX).
Materials and Methods:
In the first part of the study, various concentrations of aqueous and ethanolic licorice extract were prepared in the laboratory of Pharmacy College. It was then subjected to microbiological assay to determine its zone of inhibition using agar disk diffusion test and minimum inhibitory concentration (MIC) using serial broth dilution method against S. mutans and L. acidophilus. CHX was used as a positive control.
Results:
MIC of aqueous and ethnolic licorice root extract against S. mutans and L. acidophilus were 25% and 12.5%, respectively. Mean zone of inhibition of the aqueous and alcoholic licorice extracts against S. mutans at 48 h were 22.8 mm and 26.7 mm, respectively. Mean zone of inhibition of the aqueous and alcoholic licorice extracts against L. acidophilus at 48 h were 14.4 mm and 15.1 mm, respectively. Mean zone of inhibition of the CHX against S. mutans and L. acidophilus at 48 h was 20.5 and 13.2, respectively.
Conclusion:
The inhibitory effect shown by alcoholic licorice root extract against S. mutans and L. acidophilus was superior when compared with that of aqueous form and CHX.
Keywords: Chlorhexidine, Glycyrrhiza glabra, licorice
Introduction
Dental caries is a chronic, infectious, transmissible, and biobehavioral disease that extends throughout the life span. The essential process of this disease involves bacterial adherence to tooth surfaces, dental plaque formation, and localized demineralization of tooth enamel by acids of bacterial origin produced from the fermentation of dietary carbohydrates.1
More than a century back, Miller had postulated the chemico-parasitic theory for the etiology of dental caries, until today the modern concepts of cariogram demonstrates microorganisms as one of the major etiological factors for dental caries. Streptococcus mutans are shown to be highly associated with caries in humans. Considerable evidence exist implicating S. mutans as an important etiological agent in initiation of enamel caries, both in laboratory animals and humans. The presence of ≥106 mutans streptococci/ml saliva may indicate a high caries risk or activity.2
Prevention of this disease in the individual can focus on increasing the ability of the host to respond to the insult, decreasing the cariogenicity of the bacterial agents, and altering the diet to be less caries promoting.
Renewed interest in developing an anti-microbial approach for the management of dental caries has evolved as a result of: (1) Identifying certain members of the oral microflora as major cariogens and (2) increased understanding of the specific ecology of these cariogens. In conjunction with this concept, control, and prevention of caries has been sought by reducing the number of bacteria colonizing. Reducing their level in the oral cavity will provide an additional rationale for the prevention of dental caries.2
Research in the field of caries prevention has been focusing on ways for reducing or totally eradicating cariogenic flora from oral cavity. Studies have shown that caries can be prevented by regular tooth brushing and flossing. However, most of the studies have shown it is difficult to eliminate S. mutans from pits, fissures and approximal surfaces by mechanical means alone. For effective caries control, these methods should be combined with the chemoprophylactic agents. These agents, e.g. chlorhexidine (CHX) and antibiotics, act by lowering the number of microorganisms or inhibiting dental plaque formation. However, they have several undesirable side-effects, including tooth staining and emergence of bacterial resistance. These side-effects stimulate the search for alternative agents.1
Ayurveda is the traditional medicinal form, prevalent in India since 2000 BC. The Ayurvedic treatment is entirely based on herbs, which have certain medicinal value or property. In the ancient times, the Indian sages believed that Ayurvedic herbs are one-stop solutions to cure a number of health-related problems and diseases. They conducted thorough study about the same, experimented with herbs to arrive at accurate conclusions about the efficacy of different plants and herbs that have medical value. Most of the Ayurvedic herbs, thus formulated, are free of side-effects or reactions. This is the reason why Ayurveda is growing in popularity across the globe. The Ayurvedic herbs that have medicinal quality provide rational means for the treatment of many internal diseases, which are otherwise considered incurable in other systems of medicine.3 In 2003, according to the World Health Organization, the use of traditional herbal medicines has spread not only in developing countries, but also in the industrialized ones, as a complementary way to treat and prevent illnesses.1
Plant extracts constitute rich sources of novel compounds with a variety of pharmacological activities. In many countries, plant extracts have been traditionally used for the treatment of oral mucosal lesions and periodontal diseases without any scientific validation.1
One such herb, which has medicinal value, is the product “Glycyrrhiza (licorice).” The Egyptians, Greeks, and Romans used the drug as a remedy for a great diversity of ailments and sweetener. In addition to some flavonoids, the roots of Glycyrrhiza contain a high concentration of the triterpene saponin glycyrrhizin, the concentration of which varies between 2.5 and 9%, according to the source and work up of the drug. Glycyrrhizin, which is a diglucuronide of glycyrrhetinic acid, is the active principle to which most, if not all, the positive properties of the drug should be attributed. It is responsible for the sweet flavor of licorice, and because it is a saponin, endows it with emollient and expectorating faculties. The presence of the α, β-unsaturated carbonyl function in the aglycon imparts glycyrrhyzin (and glycyrrhetinic acid) with adrenocorticoid and anti-inflammatory activities. However, in spite of being a saponin, glycyrrhizin does not possess the most deleterious property of saponins, namely that of inducing hemolysis in red blood cells.4
According to the researchers at the University of California at least two compounds extracted from the licorice plant root are potent inhibitors of S. mutans, a major cause of dental caries.5 Hence, this in vitro study was undertaken with an aim to assess the anti-microbial effect of licorice root extract against S. mutans and L. acidophilus in comparison to CHX.
Materials and Methods
A standard procedure for performing the minimum inhibitory concentration (MIC) test was followed. Dried Licorice root pieces were procured from M/s Amruth Kesari Depot, Bangalore, Karnataka.
The materials used in this study are:
-
Test materials used:
- Licorice root
- Aqueous water
- Ethanol
- CHX (0.2%).
-
Microorganism:
- S. mutans American type culture collection (ATCC) 25175
- L. acidophilus ATCC 4356.
The microbial strain selected for the present study was collected from the ATCC, USA.
Brain heart infusion agar.
Vernier Caliper.
Preparation of licorice root aqueous extract
Licorice root specimen was identified by a Botanist and a Pharmacgnosist for their authenticity at the Department of Pharmacognosy, Bapuji College of Pharmacy, Davangere. After washing all, the root specimens were dried for 3 days and powdered. Thus, obtained powder is weighted up to 50 g and then it is mixed with 100 ml of sterile distilled water in a round bottom flask with occasional shaking. The extract was then filtered through a muslin cloth for coarse residue and finally through Whatman No. 1 filters paper and kept in an airtight amber colored container.
Preparation of licorice root ethanolic extract
Licorice extract was prepared by macerating 50 g of dry powder with 100 ml of 70% (w/v) ethyl alcohol for a week in a round bottom flask with occasional shaking. The extract was then filtered through a muslin cloth for coarse residue and finally through Whatman No. 1 filters paper and was stored at 4°C for further use. Stock solutions of crude extracts were prepared by mixing well the appropriate amount of dried extracts with an inert solvent dimethylsulfoxide to obtain the final concentrations.
Microbial analysis
Revival of the organisms
The bacterial strains from the stock were revived by plating on blood agar medium. After overnight incubation at 37°C, isolated colonies were selected and the identities of the organisms were confirmed. Isolated colonies were transferred to sterile brain-heart infusion (BHI) broth for the bacteria and once again incubated overnight. The growth concentration was adjusted to 105 organisms/ml by using 0.5 McFarland’s turbidity standard.
Agar ditch plate method for testing the antibacterial properties
Agar well-diffusion assay was used to evaluate the anti-microbial potential of the extracts. Petridishes containing 18 m1 of BHI agar for S. mutans and L. acidophillus were inoculated with approximately 100 µl of microbial strain using swab technique.
Wells of 8 mm diameter were cut into solidified agar media using a sterilized standard device. One hundred µl of each extract was poured in the respective well and the plates were incubated at 37°C for 48 h. To ensure the consistency of all findings, the experiment was performed and repeated under strict aseptic conditions. The antibacterial activity of each extract was expressed in terms of the mean of diameter of zone of inhibition (in mm) produced by each extract at the end of incubation period.
Minimum inhibitory concentration determination for the aqueous licorice root extract
A volume of 200 µl of the BHI broth was added in each of ten MIC tubes/bacterial strain. In the first MIC tube containing 200 µl broth, 200 µl of stock was added. After mixing well, 200 µl was transferred to the second MIC tube. This was continued till the last (10th) tube. From the last tube 200 µl final solution was discarded. By following this serial dilution, the concentration of the licorice root powder was achieved as the following 50%, 25%, 12.5%, 6.25%, 3.1%, 1.56%, 0.78%, 0.39%, 0.19% and 0.09%, respectively. To each of the ten such prepared MIC tubes with varying concentrations, 200 µl of the earlier prepared strain of S. mutans was added such that the final volume/tube was 400 µl.
After the incubation, the MIC values were determined by visual inspection of the tubes. With each batch of tests, positive and negative controls were put up. Positive control containing broth plus bacterial strain showed turbidity and negative control containing broth only appeared clear. In each series of tubes, the last tube with clear supernatant was considered to be without any growth and taken as MIC value. Turbidity in the MIC tube indicated growth of the bacteria strain implying that the organisms were resistant to the aqueous licorice root extract.
Minimum inhibitory concentration determination for the alcoholic licorice root extract
A solution of 50% concentration was prepared as the stock solution. The working concentration for the extract was achieved as the following 50%, 25%, 12.5%, 6.25%, 3.1%, 1.56%, 0.78%, 0.39%, 0.19% and 0.09%, respectively. A similar procedure of serial dilution as mentioned above was carried out to test the anti-microbial effect of the alcoholic licorice root extract.
All the measurements of zone of inhibition were carried out by a single examiner. Calibration of examiner was done prior to and during the study by re-examining 5% of the samples, to minimize intra-examiner variability. Intra-examiner agreement was determined using kappa statistics (κ) and the score thus obtained (κ = 0.84) was almost perfect, according to Landis and Koch, thus meeting the scientific requirement for validity and reliability.
Statistical analysis
The collected data were classified and tabulated in Microsoft Office Excel. SPSS for windows version 17 software (IBM Corp, Chicago, USA) was employed for statistical analysis. Frequency distributions of responses to the questions were produced. Since the data were continuous type, parametric tests were used for analysis. Mean (χ) and standard deviation were calculated. One-way analysis of variance test was used for multiple group comparisons, followed by Tukey post-hoc for group-wise comparisons, and P < 0.05 was considered as statistically significant.
Results
At the end of 48 h, statistically significant anti-microbial activity was demonstrated by all the test specimens used in this study (P = 0.002).
Graph 1 shows the anti-microbial activity of the licorice root extract (aqueous and alcohol) against S. mutans at 48 h. The inhibitory effect shown by licorice root extracts against S. mutans was superior, when compared with CHX. Among licorice root extract, Alcoholic extract showed maximum inhibitory effect against S. mutans.
Graph 1.
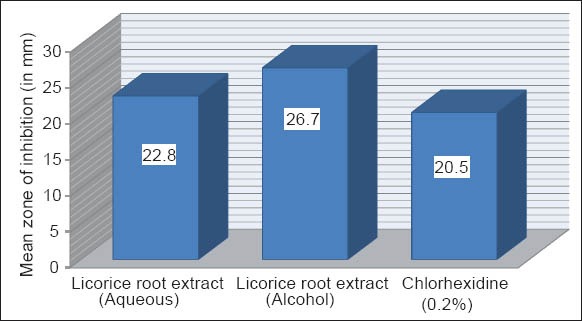
Mean zone of inhibition of the extracts against Streptococcus mutans at 48 h.
Graph 2 shows the anti-microbial activity of the licorice root extract (aqueous and alcohol) against L. acidophilus at 48 h. The inhibitory effect shown by licorice root extracts against S. mutans was superior, when compared with CHX. Among licorice root extract, Alcoholic extract showed maximum inhibitory effect against S. mutans.
Graph 2.
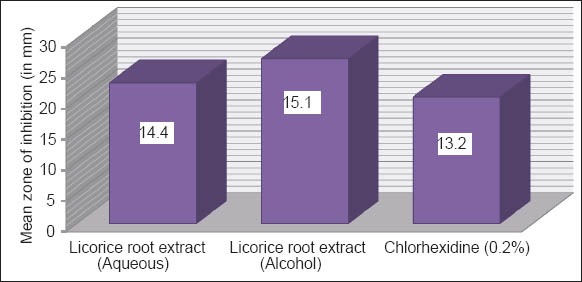
Mean zone of inhibition of the extracts against Lactobacillus acidophilus at 48 h.
Table 1 shows anti-microbial activity of the extracts against S. mutans at 48 h. Alcoholic licorice root extract showed a highest inhibition rate against S. mutans compared with the aqueous form and CHX and this finding was found to be statistically significant.
Table 1.
Anti-microbial activity of the extracts against S. mutans at 48 h.
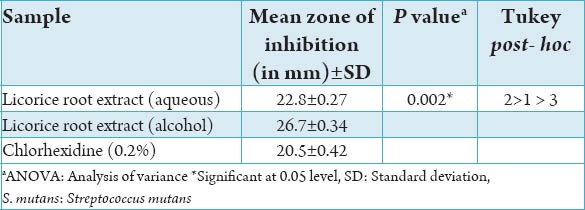
Table 2 shows anti-microbial activity of the extracts against L. acidophilus at 48 h. Alcoholic licorice root extract showed a highest inhibition rate against L. acidophilus compared with the aqueous form and CHX and this finding was found to be statistically significant.
Table 2.
Antimicrobial activity of the extracts against L. acidophilus at 48 h.
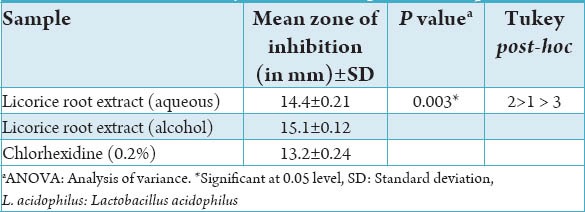
Table 3 shows MIC of aqueous licorice root extract against S. mutans and L. acidophilus. S. mutans showed sensitivity to aqueous licorice root extract in concentrations of 50%, 25% and demonstrated resistance to concentrations of 12.5%, 6.25%, 3.1%, 1.56%, 0.78%, 0.39%, 0.19%, and 0.09% respectively. L. acidophilus showed sensitivity to aqueous licorice root extract only in concentration of 50% and demonstrated resistance to concentrations of 25%, 12.5%, 6.25%, 3.1%, 1.56%, 0.78%, 0.39%, 0.19%, and 0.09%. Hence, the MIC of aqueous licorice root extract for S. mutans was established at 25% and L. acidophilus at 50%.
Table 3.
Minimum inhibitory concentration of aqueous licorice root extract against S. mutans.
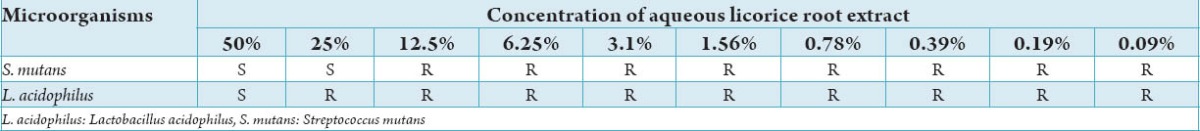
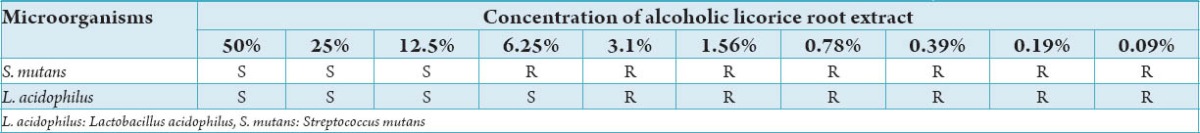
Table 4 shows MIC of alcoholic licorice root extract against S. mutans and L. acidophilus. S. mutans showed sensitivity to aqueous licorice root extract in concentrations of 50%, 25%, 12.5% and demonstrated resistance to concentrations of 6.25%, 3.1%, 1.56%, 0.78%, 0.39%, 0.19%, and 0.09% respectively. L. acidophilus showed sensitivity to aqueous licorice root extract in concentrations of 50%, 25%, 12.5%, 6.25% and demonstrated resistance to concentrations of 3.1%, 1.56%, 0.78%, 0.39%, 0.19%, and 0.09%. Hence, the MIC of alcoholic licorice root extract for S. mutans was established at 12.5% and L. acidophilus at 6.25%.
Table 4.
Minimum inhibitory concentration of alcoholic licorice root extract against L. acidophilus.
Discussion
The current anti-microbial strategies used to treat dental caries have consisted primarily of mechanical removal of dental plaque or generalized killing of oral bacteria with anti-bacterial compounds. These remove all, kill-all approaches have shown limited efficacy, since a cleaned tooth surface provides an equal opportunity for commensal and pathogenic bacteria to re-colonize in the non-sterile environment of the oral cavity. Cariogenic bacteria usually re-dominate the Dental plaque after the treatment and start another cycle of cariogenesis. This study proposes to develop a targeted anti-microbial therapy against S. mutans. By selectively killing or inhibiting the cariogenic bacteria within a pathogenic Dental plaque, a non-pathologic, commensal microbial community could be established. This healthy plaque would then serve as an effective barrier to prevent the subsequent colonization of cariogenic bacteria on the tooth surface, leading to a sustained anti-caries therapeutic effect.
Liquorice or Muleti is a perennial herb or undershrub that is about 1 m high. Its dried peeled or unpeeled underground stems and roots constitute the drug, which is an important constituent of all cough and catarrh syrups, throat lozenges and pastilles. This has been used in medicine for more than 4000 years. Hippocrates (400 BC) mentioned its use as a remedy for ulcers and quenching of thirst. It is grown in some parts of India. However, its commercial cultivation has not yet been possible and the domestic requirement is largely met through imports.6
The compound glycyrrhizic acid, found in liquorice, is now routinely used throughout Japan for the treatment and control of chronic viral hepatitis, and there is a possible transaminase-lowering effect.7
Roots of this shrub contain a number of compounds, the most important being a glucoside, glycyrrhizin, that gives glycyrrhetinic acid on enzyme hydrolysis. Glycyrrhizin has anti-diuretic, anti-inflammatory, expectorant, anti-ulcerous, and antihistaminic properties. Liquorice may be useful in conventional and naturopathic medicine for both mouth ulcers and peptic ulcers.
In India, it is used as a tonic, diuretic, demulcent, expectorant, emenagogue laxative and laxative. It is also used: (1) For allaying coughs and catarrhal infections, (2) in irritable conditions of mucous membrane of urinary organs, (3) sorting out other diseases like sore throat, cough, anorexia and persistent low fever.7
In spite of the use in medicinal field, less light has been shed for the use of licorice in Dentistry. Therefore, this study was aimed to assess the anti-microbial effect of licorice root extracts on caries causative microorganisms i.e., S. mutans and L. acidophilus.
Since there was lack of literature regarding which extract (aqueous or alcoholic) would yield better results in inhibiting selected microorganisms, both extracts were assessed for efficacy.
CHX was considered as positive control in this study. The bisbiguanide CHX, which has been studied extensively for over 25 years, is currently the most potent anti-microbial agent against mutans streptococci and dental caries. Its method of action has been comprehensively reviewed by Hugo, whose classical studies demonstrated that CHX w concentrations is a potent membrane active against both gram-positive and -negative bacteria, including the release of K+, 260 nm-absorbing material and pentoses. It is also an inhibitor of adenosine triphosphatase activity. At higher bactericidal concentrations, CHX induces precipitation of cytoplasmic protein and nucleic acids. It abolishes the activity of the phosphoenolpyruvate-phosphotransferase sugar transport system, and thereby markedly inhibits acid production, in oral streptococci cariogenic bacteria in subjects with a high risk of developing caries.2
It was unanticipated that licorice root extract would yield greater inhibition rates than CHX. Particularly, it was alcoholic licorice root extract, which was superior in inhibiting S. mutans and Lacto bacillus, compared with the other two specimens (aqueous form and CHX).
The reason why mean inhibition rates was more with alcoholic licorice root extract than aqueous form is unknown. However, the reasons may be better solubility of the liquorice compound in alcohol or the very presence of alcohol.
There is less mention in e-literature of the licorice extract against the tested microorganisms. It is also difficult to compare our results with the in vivo studies reported in literature because the present study was an in vitro study. One study by R. Segal et al.4 showed that neither licorice or glycyrrhizin promoted growth or induced plaque formation. In the presence of sucrose, glycyrrhizin inhibited bacterial growth, but the adherence (plaque formation) was markedly inhibited and at 0.5-1% concentration inhibition of plaque formation was almost complete.4
Another study conducted by Toors and Herczog8 aimed to measure acid production from an experimental, non-sugar licorice, its separate constituents and some other foodstuffs in bacterial suspensions. It was found that the experimental licorice proved to be relatively well fermentable by both S. mutans and the plaque-saliva mixture.
With respect to MIC of licorice extract, inhibition of S.mutans and L. acidophilus by alcoholic licorice root extract at lower concentration was superior when compared with aqueous form. This may be due to better dissolving capacity in alcohol, better bioavailability (thus enhancing bioactivity), polarity of antibacterial compounds, which makes the compound more readily extracted by organic solvents.
Conclusion
The inhibitory effect shown by alcoholic licorice root extract against S. mutans and L. acidophilus was superior when compared with that of aqueous form and CHX.
Recommendations
The roots of Glycyrrhiza glabra contain glycyrrhizin, a saponin that is 60 times sweeter than cane sugar. Licorice is a popular alternate sweetener, used by those who cannot use cane sugar. Furthermore, because of its anti-microbial property it appears to offer a measure of protection against dental caries. For these reasons, cultivation and marketing of licorice should be increased, where in inclusion of liquorice can be considered in various food stuffs. Health concerns due to sucrose usage, obesity, and diabetes may also increase demand of such sweeteners. A few developed countries are widely marketing confectionery incorporating this compound and India should follow suit.
If further studies show promise, the licorice compounds could eventually be used as cavity-fighting components in mouthwash and toothpaste. Drug industries can also incorporate such extracts, which can be delivered as syrups or in other products. G. glabra can serve as efficient vehicle for topical oral medications because of its agreeable sweet taste, its excellent dispersing qualities, and its capability to form stable aqueous gels.
Animal studies, in vivo studies and large clinical trials have to be carried out to ascertain the effect of licorice root extract on microorganisms. Research assessing the action of liquorice root extract on periodontal pathogens, other caries causing microorganisms and fungal species would be recommended.
Footnotes
Source of Support: Nil
Conflict of Interest: None
References
- 1.Mittal S, Hiregoudar M, Subramaniam R, Muralikrishna KS, Sakeenabi B, Prashant GM, et al. Dental effect of three herbal extracts against Streptococcus mutans and Lactobacillus acidophilus in comparison to chlorhexidine. J Indian Assoc Public Health Dent. 2011;(17):336–40. [Google Scholar]
- 2.Pallavi SK. Effect of chlorhexidine on mutans Streptococci and dental caries. J Indian Assoc Public Health Dent. 2011;(17):678–683. [Google Scholar]
- 3. [Last accessed on 2011 Oct 09]. Available from: http://www.ayurveda.iloveindia.com/herbology/medicinal-value-of-herbs.html .
- 4.Segal R, Pisanty S, Wormser R, Azaz E, Sela MN. Anticariogenic activity of licorice and glycyrrhizine I: Inhibition of in vitro plaque formation by Streptococcus mutans. J Pharm Sci. 1985;74(1):79–81. doi: 10.1002/jps.2600740121. [DOI] [PubMed] [Google Scholar]
- 5. [Last accessed on 2011 Sep 30]. Available from: http://www.telegraphindia.com/1060206/asp/knowhow/story_5801348.asp .
- 6. [Last accessed on 2011 Sep 30]. Available from: http://www.ppjoy.tripod.com/PDFs/Bk%20Medicinal%20Plants.PDF .
- 7. [Last accessed on 2011 Sep 30]. Available from: http://www.en.wikipedia.org/wiki/Liquorice .
- 8.Toors FA, Herczog JI. Acid production from a nonsugar licorice and different sugar substitutes in Streptococcus mutans monoculture and pooled plaque-saliva mixtures. Caries Res. 1978;12(1):60–8. doi: 10.1159/000260316. [DOI] [PubMed] [Google Scholar]


