Abstract
Background
Several European countries, including Belgium, still suffer from mild iodine deficiency. Thyroid stimulating hormone (TSH) concentration in whole blood measured at birth has been proposed as an indicator of maternal iodine status during the last trimester of pregnancy. It has been shown that mild iodine deficiency during pregnancy may affect the neurodevelopment of the offspring. In several studies, elevated TSH levels at birth were associated with suboptimal cognitive and psychomotor outcomes among young children. This paper describes the protocol of the PSYCHOTSH study aiming to assess the association between neonatal TSH levels and intellectual, psychomotor and psychosocial development of 4–5 year old children. The results could lead to a reassessment of the recommended cut-off levels of 5 > mU/L used for monitoring iodine status of the population.
Methods
In total, 380 Belgian 4–5 year old preschool children from Brussels and Wallonia with a neonatal blood spot TSH concentration between 0 and 15 mU/L are included in the study. For each sex and TSH-interval (0–1, 1–2, 2–3, 3–4, 4–5, 5–6, 6–7, 7–8, 8–9 and 9–15 mU/L), 19 newborns were randomly selected from all newborns screened by the neonatal screening centre in Brussels in 2008–2009. Infants with congenital hypothyroidism, low birth weight and prematurity were excluded from the study. Neonatal TSH concentration was measured by the Autodelphia method in dried blood spots, collected by heel stick on filter paper 3 to 5 days after birth. Cognitive abilities and psychomotor development are assessed using the Wechsler Preschool and Primary Scale of Intelligence - third edition - and the Charlop-Atwell Scale of Motor coordination. Psychosocial development is measured using the Child Behaviour Check List for age 1½ to 5 years old. In addition, several socioeconomic, parental and child confounding factors are assessed.
Conclusions
This study aims to clarify the effect of mild iodine deficiency during pregnancy on the neurodevelopment of the offspring. Therefore, the results may have important implications for future public health recommendations, policies and practices in food supplementation. In addition, the results may have implications for the use of neonatal TSH screening results for monitoring the population iodine status and may lead to the definition of new TSH cut-offs for determination of the severity of iodine status and for practical use in data reporting by neonatal screening centres.
Keywords: Iodine deficiency, Thyroid stimulating hormone, Child development, Cognitive development, Psychomotor development, Psychosocial development
Background
According to the World Health Organization (WHO), iodine deficiency is the main cause of preventable brain damage [1]. Iodine is important for the production of thyroid hormones (TH), which are essential for the development of the central nervous system [2]. Severe iodine deficiency may lead to perinatal mortality and mental retardation [3]. Maternal iodine deficiency during pregnancy, even at mild to moderate levels, may affect the neurodevelopment of the offspring [4]. Median urinary concentration is used to define classification of the levels of iodine intake as shown in Table 1.
Table 1.
Median UI in school aged children: indicator of iodine nutrition
| Median UI (μg/l) | Iodine nutrition |
|---|---|
| <20 |
Severe iodine deficiency |
| 20-49 |
Moderate iodine deficiency |
| 50-99 |
Mild iodine deficiency |
| 100-199 |
Iodine sufficiency |
| 200-299 |
Iodine intake more than adequate |
| >300 | Iodine excess |
Source: WHO, 2004 [1].
Iodine deficiency is defined through three levels of severity: mild, moderate or severe. Severe iodine deficiency has been observed mainly in Central Africa and Asia and is considered to be disappeared in Europe. However, several European countries, including Belgium, still suffer from mild iodine deficiency (MID) despite implementation of salt iodization programmes as national measures to supress iodine deficiency [5-8]. From 2003 to 2007, the number of European countries which are mildly iodine deficient decreased from 23 to 14 [9] showing that iodine deficiency remains a problem in Europe. Iodine deficiency even re-emerged in countries previously iodine sufficient such as the UK [10]. With respect to Belgium, recent studies found that school aged children were iodine sufficient while women at childbearing age and pregnant women had MID [7,8]. It is a matter of concern as MID during pregnancy could lead to suboptimal cognitive and psychomotor outcomes in the offspring [4].
Thyroid stimulating hormone (TSH) concentration in whole blood measured at birth has been proposed as an indicator of maternal iodine status during the last trimester of pregnancy [11]. TSH controls and stimulates the production of TH. In order to maintain circulating TH levels within the required range, TSH is secreted. If iodine stores are insufficient to produce TH, TSH concentration increases. Figure 1 illustrates the change in neonatal TSH concentration in case of maternal iodine sufficiency or deficiency during pregnancy.
Figure 1.
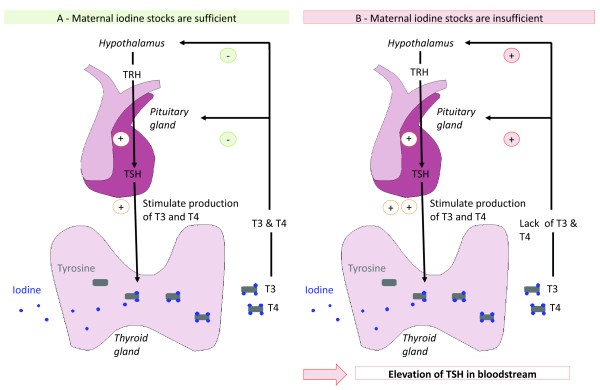
Neonatal thyroid stimulating hormone concentration as indicator of maternal iodine deficiency.Legend: In the thyroid gland, thyroid hormones (TH) T3 and T4 are produced from association of tyrosine and iodine. Liberation of thyroid stimulating hormones (TSH) stimulates TH production and secretion. The lack of TH in the bloodstream leads to the liberation of thyrotropin-releasing hormone (TRH) from the hypothalamus. TRH stimulates the liberation of TSH by the pituitary gland. A - In situation of maternal iodine sufficiency, iodine stocks are sufficient to produce adapted amount of TH and TSH release is inhibited by negative feedback inhibition. B - In situation of maternal iodine deficiency, iodine stocks are insufficient to produce adapted quantity of TH and TSH release is maintained leading to increased TSH concentration in bloodstream.
Several studies showed that elevated TSH levels after birth were associated with suboptimal cognitive and psychomotor outcomes [12-15]. Though, it is hard to claim that the observed impairments in cognitive and/or psychomotor functioning are a direct consequence of MID during gestation. An elevated TSH level at birth can be caused by several factors [16-27] and some of them affect both TSH levels and IQ in childhood [14,23,28-59]. Further studies are needed to evaluate the association between elevated TSH at birth and intellectual, psychomotor and psychosocial development of preschool children taking into account confounding factors.
In order to evaluate the efficiency of national programmes implemented to suppress iodine deficiency, adapted surveillance of the iodine status of the population is important. To monitor the iodine status of a population, median urinary iodine excretion, thyroid size, neonatal TSH concentration in blood and thyroglobulin concentration in blood can be used as indicators [60-63].
In most developed countries, neonatal screening programmes for congenital hypothyroidism are organised allowing early detection and treatment of children with thyroid hormone therapy [64]. When neonatal screening programmes are set up in a country, the WHO recommends the use of TSH results to monitor iodine status in that population [60].
The percentage of neonatal TSH screening results greater than 5 mU/L can be used to define the iodine status of a population (see Table 2) as follows: a frequency below 3% indicates iodine sufficiency, a frequency of 3–19.9% indicates mild iodine deficiency, a frequency of 20–39.9% indicates moderate deficiency and a frequency above 40% indicates iodine deficiency respectively [27,60,61].
Table 2.
Percentage of neonatal TSH screening results >5 mU/L at screening: indicator of iodine nutrition
| Percentage of neonatal TSH > 5 mU/L | Iodine nutrition |
|---|---|
| 0-3% |
Iodine sufficiency |
| 3-19.9% |
Mild iodine deficiency |
| 20 à 39,9% |
Moderate iodine deficiency |
| ≥ 40% | Severe iodine deficiency |
Source: WHO, 1994 [60].
In several studies, the neonatal TSH results have been used to assess the iodine status of the population [65-68]. However the cut-off of 5 mU/L recommended by the WHO has been criticized [69-71]. In addition, a percentage below 3% of the TSH results greater than 5 mU/L was found in populations with MID [27,72] failing to detect MID in those population. Since the proposed cut-off of percentage of TSH screening results >5 mU/L below 3% is not sensitive enough to detect MID, it should be reassessed.
Research objectives
The purpose of the PSYCHOTSH study is to assess the relationship between neonatal TSH levels and intellectual, psychomotor and psychosocial development of children aged 4–5 years old. It is hypothesized that children with a neonatal TSH level (at day 3 to 5) in the highest quintile, used as marker of lower intrauterine iodine supply, have a higher risk to develop psychomotor, cognitive as well as behavioural problems. In addition, the study aims to reassess the cut-off of 5 mU/L of neonatal TSH concentration in whole blood proposed by the WHO [60] to monitor iodine status in a population. The relationship found between TSH level and children’s intellectual, psychomotor and psychosocial development will be used to define a cut-off to indicate iodine deficiency using ROC curve analysis.
Methods
TSH determination
The children were selected from the total sample of neonates screened in 2008 and 2009 by the Brussels newborn screening centre for metabolic disorders (Laboratoire de Pédiatrie, Université Libre de Bruxelles, Brussels). Neonatal TSH level was measured in dried blood spots on filter paper collected by heel stick 3 to 5 days after birth. The measurement of TSH was performed using a time-resolved fluroimmunoassay (Autodelfia method) on filter paper [73].
Subjects
A sample size of 315 children was determined based on a detection probability (or power) of 95%, a significance level alpha of 5% and a correlation factor between TSH and IQ of 0.2. An anticipated drop-out of 20% was taken into account. 380 children aged 4–5 years old with a neonatal TSH concentration in the range 0–15 mU/L were included in the study. Children were stratified by sex and by TSH level using a stratified sampling methodology. The details of the sample are shown in Table 3.
Table 3.
Sample stratification of newborns by neonatal TSH level and by sex
| Neonatal TSH level mU/L | Boys | Girls | Total |
|---|---|---|---|
| 1-0 |
19 |
19 |
38 |
| 1-2 |
19 |
19 |
38 |
| 2-3 |
19 |
19 |
38 |
| 3-4 |
19 |
19 |
38 |
| 4-5 |
19 |
19 |
38 |
| 5-6 |
19 |
19 |
38 |
| 6-7 |
19 |
19 |
38 |
| 7-8 |
19 |
19 |
38 |
| 8-9 |
19 |
19 |
38 |
| 9-15 |
19 |
19 |
38 |
| Total | 190 | 190 | 380 |
For each sex and TSH-interval (0–1, 1–2, 2–3, 3–4, 4–5, 5–6, 6–7, 7–8, 8–9 and 9–15 mU/L) 19 newborns were selected randomly. In addition, the day of collection was taken into account. Exclusion criteria were: 1) neonatal TSH level of 15 mU/L or higher as this is the cut-off of TSH level for suspected congenital hypothyroidism, 2) prematurity (<37 weeks) and low birth weight (<2500 g) as this may induce TSH elevation at birth and alteration of neurodevelopment, 3) neurological disease of the children and 4) non-French speaking children (in order to perform psychological testing in the children’s mother tongue). For each selected infant, 3 replacements (with same TSH level and sex) were randomly selected, in case of refusals or non-contactable individuals.
Approval by Ethical Committee for human subjects and by Privacy Commission
A written informed consent is obtained from the parents before the start of the procedure (i.e. home visit). The study design, the data collection procedures, the consent form and the invitation letter were approved by the Ethical Committee of the Erasme hospital (Université Libre de Bruxelles, Brussels) in accordance with the Code of Ethics of the World Medical Association for experiments involving humans (Declaration of Helsinki). The study was also approved by the Belgian Privacy Commission.
Psychological testing
Psychometric assessment is done by 7 trained clinical psychologists during a home visit. Psychologists are blinded for the neonatal TSH levels of the selected children. Before test administration, the parents are asked to provide a quiet room and leave the psychologist alone with the child. If the child does not feel comfortable being left alone with the psychologist, one adult may stay in the room with the child, outside his or her field of view and without interrupting the psychologist.
Cognitive development is assessed using age-appropriate cognitive tests, the French version of the Wechsler Preschool and Primary Scale of Intelligence - third edition (WPPSI-III) [74,75]. The WPPSI-III is a test administered individually and allows to measure intelligence of children aged 2 years 6 months to 7 years 3 months; it is divided into 2 age groups: 2 years 6 months to 3 years 11 months old and 4 years to 7 years 3 months old. The test contains 14 subtests: 7 verbal tests, 5 performance tests and 2 processing speed tests. These subtests allow calculating a full scale Intelligence Quotient (IQ), a verbal IQ, a performance IQ, a processing speed quotient and an optional score of general language knowledge.
Psychomotor development is assessed using the French adapted version of the Charlop-Atwell Scale of Motor Coordination [76,77]. This individually administered test evaluates gross motor coordination abilities of children of 3½ to 6 years old using 6 subtests. The scale contains a subjective rating (based on quality of performance) in addition to an objective subtest rating (based on accuracy of performance).
Psychosocial development is evaluated using the French version of the Child Behaviour Check List (CBCL) for ages 1½ to 5 years old [78]. This test allows obtaining 3 scores: a total problem score, a score on internalizing problems and a score on externalizing problems. Internalizing problems represent problems of the self, like withdrawal, anxiety or depression. Externalizing problems are problems with others, problems of conflicts or problems with authority, like aggressive behaviour or attention problems. The total problem score includes these 2 scores plus a score on sleeping problems and a score on “other problems”. In order to calculate the scores, the Windows Software Assessment Data Manager is used [78].
Children anthropometric measurements
Next to psychometric assessment, the actual weight, height and head circumference of the child are measured using SECA 815 or SECA 804 weight scales, SECA 214 stadiometers and SECA 212 flexible measuring tapes.
Children urinary iodine concentration
A sample of urine is collected from the child during the home visit. The aim is to determine the current iodine status among the children included in the study. All urine samples are frozen and kept at -80°C until analysis. Analyses of the samples are performed at the Erasme Hospital. Urinary iodine excretion is measured using a modification of the Sandelle-Kolthoff reaction with spectrophotometric detection [79].
Data collection of covariates, effect modifiers and descriptive variables
The variables collected in this study as covariates, effect modifiers and descriptive variables are shown in Table 4.
Table 4.
Covariates, effects modifiers and descriptive variables of the PSYCHOTSH study
| Covariates | Effect modifiers | Descriptive variables | |
|---|---|---|---|
| Association of elevation of TSH and impaired |
Elevation of TSH |
Neurodevelopment and/or psychometric testing |
|
| Neurodevelopment | |||
| Foetus in utero exposure to: |
Foetus in utero exposure to: |
Child related factor: |
Zip code of the house of the child |
| Child bilinguism |
|
||
| Iodine excess |
Iodine containing drugs |
Chronic disease of the child |
Date of birth of the |
| TSH-receptor blocking antibodies from mothers with autoimmune thyroid disease |
|
Attending nursery school |
child, mother and father |
| Height and weight of the child at birth | |||
| Antithyroid drugs |
Exposition of the new-born during neonatal period: |
Previous intellectual assessment |
Actual height, weight of child, mother and father |
| |
|
Child negative life events |
|
| Contrast agents |
Exposition to cold |
Parenting stress |
|
| Organochlorides |
Surgical hypothermia |
Dysfunctional parenting |
Head circumference of the child |
| Lithium |
Delivery by forceps extraction |
|
|
| Cadmium |
|
|
Household composition |
| Maternal smoking |
TSH testing: |
|
Nationality and origin of the child and the parents |
| Maternal alcohol consumption |
Timing of blood sampling |
|
|
| |
TSH assay used |
Parents related factors: |
Maternal/paternal education and employment |
| |
|
Level of education |
Household incomes |
| |
|
Household incomes |
Marital status and custody |
| |
|
Maternal age at birth |
|
| |
|
Parity |
|
| Exposition of the new-born during neonatal period: | |||
| Exposure to iodine-containing antiseptics |
|
Gravidity |
|
| Perinatal anoxia |
|
Pre-pregnancy body mass index |
|
| |
|
Weight gain of the mother during pregnancy |
|
| |
|
Maternal diabetes during pregnancy |
|
| |
|
Maternal diabetes treatment during pregnancy |
|
| |
|
Maternal mental health disorders |
|
| |
|
Maternal poor social support |
|
| Marital discord | |||
Information about covariates and effect modifiers is gathered from the data provided by the ULB newborn screening centre for metabolic disorders. Additional information about pregnancy and about the period between birth and the home visit at 4–5 year old is collected using a self-report questionnaire filled in by the mother and from consultation of the health booklet of the child by the psychologist. In addition, the name of the gynaecologist is requested in the informed consent form in case additional information will be needed regarding the health of the mother during pregnancy.
Concerning pregnancy the following data are collected: exposure to iodine excess (use of iodine- containing antiseptics), intake of iodine-containing drugs, exposure to organochlorides, exposure to cadmium, lithium intake, thyroid disease of the mother, anti-thyroid drug intake, diabetes and treatment of diabetes, alcohol consumption and cigarette smoking, maternal age at birth, reproductive history, parity, gravidity, pre-pregnancy body mass index (BMI), weight gain of the mother during pregnancy.
Concerning the neonatal period the following data are collected: exposure to iodine excess (use of iodine-containing antiseptics), health problems of the newborn, type of delivery, season of birth and perinatal anoxia.
Concerning the period between birth and the home visit the following information is gathered about the parents: maternal/paternal education and employment, household income, marital status, area of residence, maternal age and information about housing.
Concerning the period between birth and the home visit the following information is gathered about the child: breastfeeding, chronic disease, attending nursery school, bilingualism, previous intellectual assessment.
In addition a self-report questionnaire is used to assess several psychological factors supposed to influence mental development: child’s negative life events, maternal mental health, maternal social support, marital discord and parent–child interactions. Some of the questions were adapted from existing questionnaires [80-83].
Discussion and conclusion
Several studies showed that elevated TSH levels after birth were associated with suboptimal cognitive and psychomotor outcomes [12-15]. However, many factors may influence neonatal TSH [16-27] concentration and some of them may affect both TSH levels and neurodevelopment in childhood [14,23,28-59]. The present study aims to evaluate association between elevated TSH at birth and intellectual, psychomotor and psychosocial development of preschool children taking into account confounding factors. This study aims to clarify the effect of MID during pregnancy on the neurodevelopment of the offspring. Therefore, the results may have important implications for future public health recommendations, policies and practices in food supplementation.
In order to evaluate the efficiency of national programmes implemented to suppress iodine deficiency, adapted surveillance of the iodine status of the population is needed. To monitor the iodine status of a population, neonatal TSH whole blood concentration can be used as an indicator [60]. The proposed cut-off percentage below 3% of the TSH results >5 mU/L was shown to be not sensitive enough to detect MID [27,72]. The present study aims to reassess the recommended cut-off of >5 mU/L using ROC curve analysis. Therefore, the results of the study could help to clarify the potential use of neonatal TSH screening results for monitoring the iodine status of populations. Furthermore, the results could lead to the definition of a new neonatal TSH cut-off for monitoring the iodine status of populations. The results could also have an impact on the definition of the cut-off of TSH levels used for reporting by neonatal screening centres.
Abbreviations
MID: Mild iodine deficiency; ROC: Receiver operating characteristic; TH: Thyroid hormone; TRH: Thyrotropin-releasing hormone; TSH: Thyroid-stimulating hormone; WHO: World Health Organization.
Competing interest
All authors declare not having any conflict of interest with regard to this study.
Contributor Information
Caroline Trumpff, Email: Caroline.Trumpff@wiv-isp.be.
Johan Vanderfaeillie, Email: jvdfaeil@vub.ac.be.
Nathalie Vercruysse, Email: nvercr@ulb.ac.be.
Jean De Schepper, Email: Jean.DeSchepper@uzbrussel.be.
Jean Tafforeau, Email: Jean.Tafforeau@wiv-isp.be.
Herman Van Oyen, Email: Herman.VanOyen@wiv-isp.be.
Stefanie Vandevijvere, Email: Stefanie.Vandevijvere@wiv-isp.be.
Acknowledgements
The authors acknowledge the financial support from “Belgian Federal Science Policy Office” (BELSPO) and from the ”Fonds de la Recherche Scientifique Medicale” (F.R.S.M.) for operating costs. All authors contributed to build up the protocol of the study and critically revised the draft versions of the manuscript.
References
- WHO. Iodine status worldwide. Geneva, Switzerland: World Health Organization; 2004. [Google Scholar]
- Corvilain B, Contempre B, Longombe AO, Goyens P, Gervy-Decoster C, Lamy F, Vanderpas JB, Dumont JE. Selenium and the thyroid: how the relationship was established. Am J Clin Nutr. 1993;57:244S–248S. doi: 10.1093/ajcn/57.2.244S. [DOI] [PubMed] [Google Scholar]
- Zimmermann MB, Jooste PL, Pandav CS. Iodine-deficiency disorders. Lancet. 2008;372:1251–1262. doi: 10.1016/S0140-6736(08)61005-3. [DOI] [PubMed] [Google Scholar]
- Trumpff C, De Schepper J, Tafforeau J, Van Oyen H, Vanderfaeillie J, Vandevijvere S. Mild iodine deficiency in pregnancy in Europe and its consequences for cognitive and psychomotor development of children: a review. J Trace Elem Med Biol. 2013;27:174–183. doi: 10.1016/j.jtemb.2013.01.002. [DOI] [PubMed] [Google Scholar]
- WHO. Iodine deficiency in Europe: a continuing public health problem. Geneva, Switzerland: World Health Organization; 2007. [Google Scholar]
- De Benoist B, McLean E, Andersson M, Rogers L. Iodine deficiency in 2007: global progress since 2003. Food Nutr Bull. 2008;29:195–202. doi: 10.1177/156482650802900305. [DOI] [PubMed] [Google Scholar]
- Vandevijvere S, Mourri AB, Amsalkhir S, Avni F, Van Oyen H, Moreno-Reyes R. Fortification of bread with iodised salt corrected iodine deficiency in school-aged children but not in their mothers: A National Cross- sectional survey in Belgium. Thyroid. 2012;22:1046–1053. doi: 10.1089/thy.2012.0016. [DOI] [PubMed] [Google Scholar]
- Vandevijvere S, Amsalkhir S, Mourri BA, Van Oyen H, Moreno Reyes R. Iodine deficiency among Belgian pregnant women not fully corrected by iodine-containing multivitamins: a national cross-sectional study. Br J Nutr. 2013;109(12):2276–2284. doi: 10.1017/S0007114512004473. [DOI] [PubMed] [Google Scholar]
- Zimmermann MB, Andersson M. Prevalence of iodine deficiency in Europe in 2010. Ann Endocrinol (Paris) 2011;72:164–166. doi: 10.1016/j.ando.2011.03.023. [DOI] [PubMed] [Google Scholar]
- Vanderpump MP, Lazarus JH, Smyth PP, Laurberg P, Holder RL, Boelaert K, Franklyn JA. Iodine status of UK schoolgirls: a cross-sectional survey. Lancet. 2011;377:2007–2012. doi: 10.1016/S0140-6736(11)60693-4. [DOI] [PubMed] [Google Scholar]
- Zimmermann MB, Aeberli I, Torresani T, Burgi H. Increasing the iodine concentration in the Swiss iodized salt program markedly improved iodine status in pregnant women and children: a 5-y prospective national study. Am J Clin Nutr. 2005;82:388–392. doi: 10.1093/ajcn.82.2.388. [DOI] [PubMed] [Google Scholar]
- Calaciura F, Mendorla G, Distefano M, Castorina S, Fazio T, Motta RM, Sava L, Delange F, Vigneri R. Childhood IQ measurements in infants with transient congenital hypothyroidism. Clin Endocrinol (Oxf) 1995;43:473–477. doi: 10.1111/j.1365-2265.1995.tb02620.x. [DOI] [PubMed] [Google Scholar]
- Riano Galan I, Sanchez MP, Pilar Mosteiro DM, Rivas Crespo MF. Psycho-intellectual development of 3 year-old children with early gestational iodine deficiency. J Pediatr Endocrinol Metab. 2005;18(Suppl 1):1265–1272. doi: 10.1515/jpem.2005.18.s1.1265. [DOI] [PubMed] [Google Scholar]
- Freire C, Ramos R, Amaya E, Fernandez MF, Santiago-Fernandez P, Lopez-Espinosa MJ, Arrebola JP, Olea N. Newborn TSH concentration and its association with cognitive development in healthy boys. Eur J Endocrinol. 2010;163:901–909. doi: 10.1530/EJE-10-0495. [DOI] [PubMed] [Google Scholar]
- Belcari F, Placidi G, Guzzetta A, Tonacchera M, Ciampi M, Bartoli A, Scaramuzzo RT, Frumento P, Cioni G, Pinchera A, Boldrini A, Ghirri P. Thyroid-stimulating hormone levels in the first days of life and perinatal factors associated with sub-optimal neuromotor outcome in pre-term infants. J Endocrinol Invest. 2011;34:e308–e313. doi: 10.3275/7795. [DOI] [PubMed] [Google Scholar]
- Becks GP, Burrow GN. Thyroid disease and pregnancy. Med Clin North Am. 1991;75:121–150. doi: 10.1016/s0025-7125(16)30475-8. [DOI] [PubMed] [Google Scholar]
- Karlsson FA, Dahlberg PA, Alm J, Larsson A, Felding I. Maternal TSH-receptor antibodies and TSH antibodies in screening for congenital hypothyroidism. Acta Paediatr Scand. 1986;75:756–761. doi: 10.1111/j.1651-2227.1986.tb10286.x. [DOI] [PubMed] [Google Scholar]
- Mengreli C, Maniati-Christidi M, Kanaka-Gantenbein C, Girginoudis P, Vagenakis AG, Dacou-Voutetakis C. Transient congenital hypothyroidism due to maternal autoimmune thyroid disease. Hormones (Athens) 2003;2:113–119. doi: 10.14310/horm.2002.1190. [DOI] [PubMed] [Google Scholar]
- Smallridge RC, Ladenson PW. Hypothyroidism in pregnancy: consequences to neonatal health. J Clin Endocrinol Metab. 2001;86:2349–2353. doi: 10.1210/jcem.86.6.7577. [DOI] [PubMed] [Google Scholar]
- Cheron RG, Kaplan MM, Larsen PR, Selenkow HA, Crigler JF Jr. Neonatal thyroid function after propylthiouracil therapy for maternal Graves’ disease. N Engl J Med. 1981;304:525–528. doi: 10.1056/NEJM198102263040907. [DOI] [PubMed] [Google Scholar]
- Ruppert F, Sulyok E, Varga F, Csaba IF, Petz A. Thyrotropin and prolactin response to ambient temperature in newborn infants. Acta Paediatr Acad Sci Hung. 1982;23:189–194. [PubMed] [Google Scholar]
- Wilber JF, Baum D. Elevation of plasma TSH during surgical hypothermia. J Clin Endocrinol Metab. 1970;31:372–375. doi: 10.1210/jcem-31-4-372. [DOI] [PubMed] [Google Scholar]
- Rashmi, Seth A, Sekhri T, Agarwal A. Effect of perinatal factors on cord blood thyroid stimulating hormone levels. J Pediatr Endocrinol Metab. 2007;20:59–64. doi: 10.1515/jpem.2007.20.1.59. [DOI] [PubMed] [Google Scholar]
- Fisher DA, Klein AH. Thyroid development and disorders of thyroid function in the newborn. N Engl J Med. 1981;304:702–712. doi: 10.1056/NEJM198103193041205. [DOI] [PubMed] [Google Scholar]
- Abuid J, Stinson DA, Larsen PR. Serum triiodothyronine and thyroxine in the neonate and the acute increases in these hormones following delivery. J Clin Invest. 1973;52:1195–1199. doi: 10.1172/JCI107286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose SR, Nisula BC. Circadian variation of thyrotropin in childhood. J Clin Endocrinol Metab. 1989;68:1086–1090. doi: 10.1210/jcem-68-6-1086. [DOI] [PubMed] [Google Scholar]
- Vandevijvere S, Coucke W, Vanderpas J, Trumpff C, Fauvart M, Meulemans A, Marie S, Vincent MF, Schoos R, Boemer F, Vanwynsberghe T, Philips E, Eyskens F, Wuyts B, Selimaj V, Kirkpatrick C, Van Overmeire B, Van Oyen H, Moreno-Reyes R. Neonatal thyroid-stimulating hormone concentrations in Belgium: a useful indicator for detecting mild iodine deficiency? PLoS One. 2012;7:e47770. doi: 10.1371/journal.pone.0047770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao B, Yin G. Effects of high-dose iodine on brain development in mice. Zhonghua Yu Fang Yi Xue Za Zhi. 1997;31:134–136. [PubMed] [Google Scholar]
- Zao J. Iodine deficiency and iodine excess in Jiangsu Province. China: Wageningen University; 2001. [Google Scholar]
- Nishiyama S, Mikeda T, Okada T, Nakamura K, Kotani T, Hishinuma A. Transient hypothyroidism or persistent hyperthyrotropinemia in neonates born to mothers with excessive iodine intake. Thyroid. 2004;14:1077–1083. doi: 10.1089/thy.2004.14.1077. [DOI] [PubMed] [Google Scholar]
- Thomas JV, Collett-Solberg PF. Perinatal goiter with increased iodine uptake and hypothyroidism due to excess maternal iodine ingestion. Horm Res. 2009;72:344–347. doi: 10.1159/000249162. [DOI] [PubMed] [Google Scholar]
- Pavan-Senn CC, Nesi-Franca S, Pelaez J, Pereira RM, Boguszewski MC, Sandrini NR, Lacerda FL. Transient neonatal hypothyroidism due to amiodarone administration during pregnancy–two cases report and review of literature. Arq Bras Endocrinol Metabol. 2008;52:126–130. doi: 10.1590/s0004-27302008000100018. [DOI] [PubMed] [Google Scholar]
- Bartalena L, Bogazzi F, Braverman LE, Martino E. Effects of amiodarone administration during pregnancy on neonatal thyroid function and subsequent neurodevelopment. J Endocrinol Invest. 2001;24:116–130. doi: 10.1007/BF03343825. [DOI] [PubMed] [Google Scholar]
- Magee LA, Downar E, Sermer M, Boulton BC, Allen LC, Koren G. Pregnancy outcome after gestational exposure to amiodarone in Canada. Am J Obstet Gynecol. 1995;172:1307–1311. doi: 10.1016/0002-9378(95)91498-6. [DOI] [PubMed] [Google Scholar]
- De Wolf D, De Schepper J, Verhaaren H, Deneyer M, Smitz J, Sacre-Smits L. Congenital hypothyroid goiter and amiodarone. Acta Paediatr Scand. 1988;77:616–618. doi: 10.1111/j.1651-2227.1988.tb10717.x. [DOI] [PubMed] [Google Scholar]
- Vanhaesebrouck P, Verstraete AG, De PC, Smets K, Zecic A, Craen M. Transplacental passage of a nonionic contrast agent. Eur J Pediatr. 2005;164:408–410. doi: 10.1007/s00431-005-1659-2. [DOI] [PubMed] [Google Scholar]
- Ribas-Fito N, Sala M, Cardo E, Mazon C, De Muga ME, Verdu A, Marco E, Grimalt JO, Sunyer J. Organochlorine compounds and concentrations of thyroid stimulating hormone in newborns. Occup Environ Med. 2003;60:301–303. doi: 10.1136/oem.60.4.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Espinosa MJ, Vizcaino E, Murcia M, Fuentes V, Garcia AM, Rebagliato M, Grimalt JO, Ballester F. Prenatal exposure to organochlorine compounds and neonatal thyroid stimulating hormone levels. J Expo Sci Environ Epidemiol. 2010;20:579–588. doi: 10.1038/jes.2009.47. [DOI] [PubMed] [Google Scholar]
- Grandjean P, Landrigan PJ. Developmental neurotoxicity of industrial chemicals. Lancet. 2006;368:2167–2178. doi: 10.1016/S0140-6736(06)69665-7. [DOI] [PubMed] [Google Scholar]
- Frassetto F, Tourneur MF, Barjhoux CE, Villier C, Bot BL, Vincent F. Goiter in a newborn exposed to lithium in utero. Ann Pharmacother. 2002;36:1745–1748. doi: 10.1345/aph.1C123. [DOI] [PubMed] [Google Scholar]
- Shirkey HC. Human experiences related to adverse drug reactions to the fetus or neonate from some maternally administered drugs. Adv Exp Med Biol. 1972;27:17–30. doi: 10.1007/978-1-4684-3219-0_3. [DOI] [PubMed] [Google Scholar]
- Khashu M, Chessex P, Chanoine JP. Iodine overload and severe hypothyroidism in a premature neonate. J Pediatr Surg. 2005;40:E1–E4. doi: 10.1016/j.jpedsurg.2004.10.028. [DOI] [PubMed] [Google Scholar]
- Chanoine JP, Boulvain M, Bourdoux P, Pardou A, Van Thi HV, Ermans AM, Delange F. Increased recall rate at screening for congenital hypothyroidism in breast fed infants born to iodine overloaded mothers. Arch Dis Child. 1988;63:1207–1210. doi: 10.1136/adc.63.10.1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber G, Vigone MC, Rapa A, Bona G, Chiumello G. Neonatal transient hypothyroidism: aetiological study: Italian Collaborative Study on Transient Hypothyroidism. Arch Dis Child Fetal Neonatal Ed. 1998;79:F70–F72. doi: 10.1136/fn.79.1.f70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gottfried AW. Intellectual consequences of perinatal anoxia. Psychol Bull. 1973;80:231–242. doi: 10.1037/h0034914. [DOI] [PubMed] [Google Scholar]
- Schachter FF, Apgar V. Perinatal asphyxia and psychologic signs of brain damage in childhood. Pediatrics. 1959;24:1016–1025. [PubMed] [Google Scholar]
- Olivieri A, Medda E, De AS, Valensise H, De FM, Fazzini C, Cascino I, Cordeddu V, Sorcini M, Stazi MA. High risk of congenital hypothyroidism in multiple pregnancies. J Clin Endocrinol Metab. 2007;92:3141–3147. doi: 10.1210/jc.2007-0238. [DOI] [PubMed] [Google Scholar]
- Deary IJ, Pattie A, Wilson V, Whalley LJ. The cognitive cost of being a twin: two whole-population surveys. Twin Res Hum Genet. 2005;8:376–383. doi: 10.1375/1832427054936709. [DOI] [PubMed] [Google Scholar]
- Natalucci G, Seitz J, von SK, Bucher HU, Molinari L, Jenni OG, Latal B. The role of birthweight discordance in the intellectual and motor outcome for triplets at early school age. Dev Med Child Neurol. 2011;53:822–828. doi: 10.1111/j.1469-8749.2011.04024.x. [DOI] [PubMed] [Google Scholar]
- Ng SM, Wong SC, Paize F, Chakkarapani E, Newland P, Isherwood D, Didi M. Multivariate analyses of factors that affect neonatal screening thyroid stimulating hormone. J Pediatr Endocrinol Metab. 2011;24:727–732. doi: 10.1515/jpem.2011.234. [DOI] [PubMed] [Google Scholar]
- Petrini JR, Dias T, McCormick MC, Massolo ML, Green NS, Escobar GJ. Increased risk of adverse neurological development for late preterm infants. J Pediatr. 2009;154:169–176. doi: 10.1016/j.jpeds.2008.08.020. [DOI] [PubMed] [Google Scholar]
- Lundgren EM, Tuvemo T, Gustafsson J. Short adult stature and overweight are associated with poor intellectual performance in subjects born preterm. Horm Res Paediatr. 2011;75:138–145. doi: 10.1159/000322015. [DOI] [PubMed] [Google Scholar]
- Caravale B, Tozzi C, Albino G, Vicari S. Cognitive development in low risk preterm infants at 3–4 years of life. Arch Dis Child Fetal Neonatal Ed. 2005;90:F474–F479. doi: 10.1136/adc.2004.070284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietz J, Peter J, Graf R, Rauterberg-Ruland I, Rupp A, Sontheimer D, Linderkamp O. Physical growth and neurodevelopmental outcome of nonhandicapped low-risk children born preterm. Early Hum Dev. 2004;79:131–143. doi: 10.1016/j.earlhumdev.2004.05.001. [DOI] [PubMed] [Google Scholar]
- Goldenberg RL, Dubard MB, Cliver SP, Nelson KG, Blankson K, Ramey SL, Herman A. Pregnancy outcome and intelligence at age five years. Am J Obstet Gynecol. 1996;175:1511–1515. doi: 10.1016/s0002-9378(96)70099-6. [DOI] [PubMed] [Google Scholar]
- Goyen TA, Lui K, Hummell J. Sensorimotor skills associated with motor dysfunction in children born extremely preterm. Early Hum Dev. 2011;87:489–493. doi: 10.1016/j.earlhumdev.2011.04.002. [DOI] [PubMed] [Google Scholar]
- Clapin H, Lewis BD, Greed L, Dawkins H, O'Leary P. Factors influencing neonatal thyroid-stimulating hormone concentrations as a measure of population iodine status. J Pediatr Endocrinol Metab. 2014;27:101–106. doi: 10.1515/jpem-2013-0189. [DOI] [PubMed] [Google Scholar]
- Lawlor DA, Najman JM, Batty GD, O'Callaghan MJ, Williams GM, Bor W. Early life predictors of childhood intelligence: findings from the Mater-University study of pregnancy and its outcomes. Paediatr Perinat Epidemiol. 2006;20:148–162. doi: 10.1111/j.1365-3016.2006.00704.x. [DOI] [PubMed] [Google Scholar]
- Rieger-Fackeldey E, Blank C, Dinger J, Steinmacher J, Bode H, Schulze A. Growth, neurological and cognitive development in infants with a birthweight <501 g at age 5 years. Acta Paediatr. 2010;99:1350–1355. doi: 10.1111/j.1651-2227.2010.01762.x. [DOI] [PubMed] [Google Scholar]
- WHO. Indicators for assessing iodine deficiency disorders and their control through salt iodisation. Geneva: WHO; 1994. WHO/NUT/94.6. [Google Scholar]
- Delange F. Neonatal thyroid screening as a monitoring tool for the control of iodine deficiency. Acta Paediatr Suppl. 1999;88:21–24. doi: 10.1111/j.1651-2227.1999.tb01150.x. [DOI] [PubMed] [Google Scholar]
- Delange F. Neonatal screening for congenital hypothyroidism: results and perspectives. Horm Res. 1997;48:51–61. doi: 10.1159/000185485. [DOI] [PubMed] [Google Scholar]
- Delange F. Screening for congenital hypothyroidism used as an indicator of the degree of iodine deficiency and of its control. Thyroid. 1998;8:1185–1192. doi: 10.1089/thy.1998.8.1185. [DOI] [PubMed] [Google Scholar]
- WHO. Assessment of iodine deficiency disorders and monitoring their elimination: a guide for programme managers. Geneva: World Health Organisation; 2001. [Google Scholar]
- Costante G, Grasso L, Ludovico O, Marasco MF, Nocera M, Schifino E, Rivalta L, Capula C, Chiarella R, Filetti S, Parlato G. The statistical analysis of neonatal TSH results from congenital hypothyroidism screening programs provides a useful tool for the characterization of moderate iodine deficiency regions. J Endocrinol Invest. 1997;20:251–256. doi: 10.1007/BF03350296. [DOI] [PubMed] [Google Scholar]
- Mikelsaar RV, Viikmaa M. Neonatal thyroid-stimulating hormone screening as an indirect method for the assessment of iodine deficiency in Estonia. Horm Res. 1999;52:284–286. doi: 10.1159/000023496. [DOI] [PubMed] [Google Scholar]
- Copeland DL, Sullivan KM, Houston R, May W, Mendoza I, Salamatullah Q, Solomons N, Nordenberg D, Maberly GF. Comparison of neonatal thyroid-stimulating hormone levels and indicators of iodine deficiency in school children. Public Health Nutr. 2002;5:81–87. doi: 10.1079/PHN2001306. [DOI] [PubMed] [Google Scholar]
- Gyurjyan RH, Lugovska R, Vevere P, van der Haar F. Newborn thyrotropin screening confirms iodine deficiency in Latvia. Eur J Clin Nutr. 2006;60:688–690. doi: 10.1038/sj.ejcn.1602364. [DOI] [PubMed] [Google Scholar]
- Burns R, Mayne PD, O'Herlihy C, Smith DF, Higgins M, Staines A, Smyth PP. Can neonatal TSH screening reflect trends in population iodine intake? Thyroid. 2008;18:883–888. doi: 10.1089/thy.2008.0036. [DOI] [PubMed] [Google Scholar]
- Rajatanavin R. Iodine deficiency in pregnant women and neonates in Thailand. Public Health Nutr. 2007;10:1602–1605. doi: 10.1017/S1368980007360990. [DOI] [PubMed] [Google Scholar]
- Gruneiro-Papendieck L, Chiesa A, Mendez V, Bengolea S, Prieto L. Neonatal TSH levels as an index of iodine sufficiency: differences related to time of screening sampling and methodology. Horm Res. 2004;62:272–276. doi: 10.1159/000081786. [DOI] [PubMed] [Google Scholar]
- Travers CA, Guttikonda K, Norton CA, Lewis PR, Mollart LJ, Wiley V, Wilcken B, Eastman CJ, Boyages SC. Iodine status in pregnant women and their newborns: are our babies at risk of iodine deficiency? Med J Aust. 2006;184:617–620. doi: 10.5694/j.1326-5377.2006.tb00417.x. [DOI] [PubMed] [Google Scholar]
- Soini E, Kojola H. Time-resolved fluorometer for lanthanide chelates–a new generation of nonisotopic immunoassays. Clin Chem. 1983;29:65–68. [PubMed] [Google Scholar]
- Wechsler D. WechslerPreschool and Primary Scale of Intelligence - Revised. San Antonio, TX: The Psychological Corporation; 1989. [Google Scholar]
- Wechsler D. Manual for the Wechsler Preschool and Primary Scale of Intelligence. San Antonio, TX: The Psychological Corporation; 1967. [Google Scholar]
- Charlop M, Atwell CW. The Charlop-Atwell scale of motor coordination: a quick and easy assessment of young children. Percept Mot Skills. 1980;50:1291–1308. doi: 10.2466/pms.1980.50.3c.1291. [DOI] [PubMed] [Google Scholar]
- Albaret JM, Noack N. Manuel de l'échelle de coordinations lotrices de Charlop-Atwell. Paris: Editions du Centre de Psychologie appliquée; 1994. [Google Scholar]
- Achenbach TM, Rescorla LA. Manual for the ASEBA Preschool Forms & Profiles. Burlington, VT: University of Vermont, Research Center for Children, Youth & Families; 2000. [Google Scholar]
- Pino S, Fang SL, Braverman LE. Ammonium persulfate: a safe alternative oxidizing reagent for measuring urinary iodine. Clin Chem. 1996;42:239–243. [PubMed] [Google Scholar]
- Coddington RD. The significance of life events as etiologic factors in the diseases of children - II: A study of a normal population. J Psychosom Res. 1972;16:205–213. doi: 10.1016/0022-3999(72)90045-1. [DOI] [PubMed] [Google Scholar]
- Goldberg D, Williams P. A user's guide to the General Health Questionnaire. Berkshire: NFER-NELSON; 1988. [Google Scholar]
- Goldberg DP. In: L'évaluation clinique standardisée en psychiatrie. Pariente P, Guelfi JD, Smith M, Guelfi JD, editor. Paris: Editions Médicales Pierre Fabre; 1978. Manual of the General Health Questionnaire. Traduction française de W. Bettschart & M. Bolognini. [Google Scholar]
- Baillargeron J, Dubois G, Marineau R. Traduction française de l'échelle d'ajustement dyadique. Revue canadienne des sciences du comportement. 1986;18:25–34. [Google Scholar]


