Abstract
Background
Few studies have examined the relationship of lifestyle factors with mortality among colorectal cancer patients.
Methods
Among NIH-AARP Diet and Health study participants we identified 4,213 colon and 1,514 rectal cancer cases through linkage to state cancer registries and determined date and cause of death using the National Death Index. Lifestyle factors were assessed at baseline and included: healthy diet (measured by Healthy Eating Index 2005; HEI-2005), body mass index (BMI), physical activity, alcohol consumption and smoking. We examined the association of factors individually and combined into a lifestyle score with five-year mortality from all-causes, colorectal cancer, and cardiovascular disease (CVD). We estimated relative risks (RRs) and 95% confidence intervals (CIs) using Cox proportional hazards models.
Results
Among colon cancer survivors, smokers had increased risk of total mortality (RR 1.74; 95% CI 1.45–2.08) and colorectal cancer mortality (1.46; 1.17–1.82), compared to never smokers. Obese (BMI ≥30) individuals had increased risk of all death (1.19; 1.02–1.39) and CVD death (1.84; 1.05–3.23), compared to normal weight (BMI 18.5 to <25) individuals. Compared to those with the lowest lifestyle score, those with the highest score had a 34% lower risk of all-cause mortality (0.66; 0.50–0.87).
Among rectal cancer survivors, individuals in the highest quintile of HEI-2005 scores had reduced all-cause mortality (0.60; 0.42–0.86) compared to those in the lowest. Higher combined lifestyle scores were associated with a 46% lower risk of total mortality (0.54; 0.32–0.91).
Conclusion
Healthier lifestyle before cancer diagnosis was associated with improved overall survival after diagnosis with colorectal cancer.
Keywords: Colorectal cancer, survivors, diet, body mass index, smoking, lifestyle
Introduction
The number of colorectal cancer survivors in the United States is estimated at over 1 million [1]. The Institute of Medicine reports numerous gaps in the clinical care of survivors, among them being the need for guidance on “maintaining a healthy lifestyle and preventing recurrent or new cancers” [2]. However, while there is abundant evidence linking diet and lifestyle to colorectal cancer incidence [3], evidence for their effect on patient survival after a cancer diagnosis is only now emerging [4].
Accumulating evidence from observational studies suggests pre- [5] and post-diagnosis body weight [6–11], and pre- [5] and post-diagnosis physical activity [12–15], may have an impact on colorectal cancer survival. Fewer studies have examined the effect of post-diagnosis diet quality [16], or pre-diagnosis alcohol consumption [17], or smoking [17, 18]. Although evidence is limited, the American Cancer Society (ACS) has published Guidelines on Nutrition and Physical Activity for Cancer Survivors [4], which can be summarized as follows: 1) achieve and maintain a healthy weight throughout life; 2) be physically active; and 3) eat a healthy diet, with an emphasis on plant foods. The World Cancer Research Fund and American Institute for Cancer Research Expert Report made similar recommendations that also include limiting alcohol consumption [3].
Since cancer survivors are at increased risk of second cancers and other co-morbid conditions, following an overall healthy lifestyle pattern may be important for improving survival outcomes. However, no studies have evaluated the combined effect of healthy behaviors on cancer survival. Therefore, we examined the relationship of pre-diagnosis lifestyle factors, alone and in combination, on five-year all-cause, colorectal cancer-specific, and cardiovascular (CVD) mortality among colon and rectal cancer cases in the NIH-AARP Diet and Healthy Study cohort.
Materials and Methods
Study Population
Details of the study methods and population have been previously described [19]. Briefly, the NIH-AARP Diet and Health Study participants consisted of AARP members aged 50–71, residing in six states (CA, FL, PA, NJ, NC, and LA) and two metropolitan areas (Atlanta, GA and Detroit, MI). Participants completed mailed questionnaires at baseline in 1995–1996. The study was approved by the Special Studies Institutional Review Board of the US National Cancer Institute (NCI).
Cancer cases were ascertained through December 31, 2006 by linkage to state cancer registries in the study area, and areas where participants tended to move during follow-up (AZ, NV, and TX). More than 90% of cancer cases were identified through this linkage. Registry data include diagnosis date, morphology, stage, grade, and first course of treatment.
We identified 7,107 first primary invasive colorectal cancer cases among the 492,182 men and women who satisfactorily completed the baseline questionnaire and had no history of cancer (except for non-melanoma skin cancer), end-stage renal disease, or self-reported poor health at baseline. We further excluded those: with a colorectal cancer diagnosis <1 year after baseline (n=793); who reported total energy intakes greater than two times the interquartile range (n=59); who had missing height or weight or had body mass index (BMI) less than 18.5 kg/m2 (n=220); had missing physical activity (n=67); or unknown smoking history (n=241). We included 4,213 colon and 1,514 rectal cancer cases in our analysis.
Mortality ascertainment
Mortality and cause of death were ascertained by linkage with the Social Security Administration Death Master File and the National Death Index Plus through December 31, 2008. All-cause mortality was defined as death from any cause while colorectal cancer-specific mortality was defined as having colorectal cancer as the underlying cause of death. CVD death was defined as: disease of the heart (ICD-10 codes I00–I09, I11, I13, I20–I51), hypertension (I10, I12), cerebrovascular diseases (I60–I69), atherosclerosis (I70) or aortic aneurism (I71).
Assessment of Diet and Other Lifestyle Factors
Diet (including alcohol consumption) was assessed with a 124-item food frequency questionnaire, in which participants reported the frequency of food and beverage consumption for the previous 12 months. The instrument used 10 pre-defined categories of intake ranging from “never” to “6+ times per day” for beverages, and “never” to “2+ times per day” for solid foods. Portion size and nutrient intakes were calculated using the 1994–1996 US Department of Agriculture’s Continuing Survey of Food Intakes by Individuals databases.
Dietary quality was determined by applying the Healthy Eating Index 2005 (HEI-2005), which assesses conformance to the 2005 Dietary Guidelines for Americans [20]. The HEI-2005 is energy adjusted using a density method, and scores range from zero to 100 points, with higher scores representing better conformity to dietary guidelines. Participants reported body weight, height, physical activity, and smoking status in the baseline questionnaire. BMI was computed as weight in kilograms (kg) divided by the square of height in meters (m2). Physical activity questions queried the frequency that participants engaged in ≥20 minutes activity that resulted in increased breathing, heart rate, or perspiration. Smoking questions included baseline smoking status and time since quitting for former smokers.
Statistical Analyses
Colon and rectal cancer cases were analyzed separately. Lifestyle factors were examined individually as follows: HEI-2005 scores were categorized into quintiles and alcohol intake was divided into three categories: nondrinking, moderate drinking (≤2 drinks/day for men, or ≤1 drink/day for women), and heavier drinking (>2 drinks/day for men or >1 drink/day for women), with one drink equal to ~13 grams of alcohol. BMI was grouped into three levels: normal weight (18.5 to <25 kg/m2), overweight (25 to <30 kg/m2), and obese (≥30kg/m2). Because underweight individuals may have unique underlying health problems that may affect survival, they were excluded. Physical activity was categorized into five levels (never or rarely, 1–3 times/month, 1–2 times/week, 3–5 times/week, and 5+ times/week) and smoking history was divided into four categories (never smoked, quit 10+ years ago, quit 1–9 years ago, quit <1 year ago or current smoker).
To compute the composite lifestyle score, we dichotomized each lifestyle variable and assigned one point for meeting the recommendation and zero points for not meeting it. Participants received one point for each of the following: BMI from 18.5 to <25 kg/m2, ≥3 episodes/week physical activity, HEI score within the top two quintiles, consuming ≤1 drink/day (women) or 2 drinks/ day (men), and not smoking (including former smokers who had quit at least 1 year before baseline). We then summed the points for all variables to produce a single score with values ranging from zero (worst score) to five (best score).
Relative risks (RRs) and 95% confidence intervals (CIs) were estimated using Cox proportional hazards models, with age as the underlying time metric. Because mortality follow-up began at the age of cancer diagnosis, participants had different lengths of time between baseline measurements and cancer diagnosis (“lag time”). Exploratory analyses revealed interactions between lag time and the composite lifestyle score, but limiting follow-up time to five years post-diagnosis eliminated this interaction. Participants’ follow-up ended at either age at censoring or age at death. Censoring events were reaching five years follow-up post-diagnosis, or at study end (December 31, 2008), whichever was first. Models were adjusted for sex, cancer stage, first course of treatment, and family history of colon cancer. We further adjusted multivariate models for lag time differences in tertiles (1 year to <4.3 years, 4.3 years to <7.3 years, 7.3+ years). In additional analyses we stratified Cox models by cancer stage, sex, smoking, and BMI.
Results
Among 4,213 colon cancer cases, 1,273 cases died during five-year follow-up (856 due to colorectal cancer, 125 to other cancers, 108 to CVD, and 184 deaths from other causes). Among 1,514 rectal cancer cases, 454 were deceased by five years (301 colorectal cancer, 49 other cancer, 43 CVD, and 61 deaths from other causes).
Table 1 presents the distribution of demographic, tumor, treatment, and lifestyle variables across the range of lifestyle scores. Among both colon and rectal cancer cases, those with higher scores tended to be older at diagnosis, women, and more highly educated.
Table 1.
Characteristics of colon and rectal cancer cases in the NIH-AARP Diet and Health Study by composite lifestyle score
| Colon cancer cases (N=4213) | Rectal cancer cases(N=1514) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ≤ 1 | 2 | 3 | 4 | 5 | ≤ 1 | 2 | 3 | 4 | 5 | |
| Participants, n | 461 | 1,168 | 1,359 | 932 | 293 | 184 | 442 | 475 | 327 | 86 |
| Mean age at diagnosis | 68.4 | 69.0 | 69.6 | 70.3 | 70.5 | 68.4 | 68.3 | 68.9 | 69.1 | 69.8 |
| Male, % | 75.7 | 70.7 | 65.1 | 62.5 | 52.9 | 77.2 | 74.4 | 67.4 | 67.9 | 69.8 |
| College graduate, % | 33.4 | 32.9 | 33.0 | 42.4 | 46.8 | 29.9 | 29.0 | 30.7 | 34.9 | 46.5 |
| Family history of colon cancer, % | 10.0 | 10.1 | 11.2 | 9.7 | 13.7 | 5.4 | 6.8 | 7.4 | 7.3 | 9.3 |
| Stage | ||||||||||
| I (localized) | 25.8 | 27.7 | 29.4 | 28.4 | 24.6 | 31.0 | 30.1 | 33.7 | 35.5 | 39.5 |
| II (regional by extension) | 15.6 | 12.9 | 10.9 | 10.9 | 8.5 | 10.9 | 6.8 | 9.5 | 9.8 | 8.1 |
| III (regional w/ lymph node) | 17.1 | 16.8 | 180.0 | 18.6 | 21.8 | 10.9 | 17.7 | 17.1 | 15.0 | 10.5 |
| IV (distant/metastatic) | 11.1 | 11.8 | 10.7 | 11.2 | 9.6 | 11.4 | 11.1 | 8.6 | 8.3 | 2.3 |
| Unknown Stage | 30.4 | 30.8 | 30.9 | 30.9 | 35.5 | 35.9 | 34.4 | 31.2 | 31.5 | 39.5 |
| Treatment | ||||||||||
| Surgery, % | 83.5 | 84.0 | 84.8 | 86.6 | 87.0 | 75.0 | 73.3 | 76.8 | 78.3 | 83.7 |
| Chemotherapy, % | 25.2 | 26.6 | 26.5 | 26.5 | 31.4 | 44.6 | 41.2 | 39.8 | 33.3 | 33.7 |
| Radiation, % | 0.9 | 2.1 | 1.5 | 2.3 | 3.4 | 32.6 | 26.9 | 28.4 | 27.5 | 29.1 |
| Lifestyle Factors | ||||||||||
| Mean Body Mass Index | 28.8 | 29.3 | 28.3 | 26.3 | 22.9 | 28.1 | 29.1 | 27.5 | 25.7 | 23.4 |
| Mean Healthy Eating Index Score | 52.4 | 58.4 | 66.9 | 73.5 | 77.5 | 53.2 | 57.8 | 65.4 | 72.6 | 76.5 |
| Physically Active, %* | 4.6 | 16.0 | 43.7 | 80.4 | 100.0 | 5.4 | 20.8 | 49.3 | 76.2 | 100.0 |
| Nonsmoker, % | 48.6 | 84.7 | 94.4 | 97.2 | 100.0 | 51.6 | 84.4 | 93.7 | 98.2 | 100.0 |
| Moderate Drinker, %** | 27.8 | 78.3 | 92.0 | 97.4 | 100.0 | 22.8 | 74.7 | 88.4 | 97.3 | 100.0 |
At least 20 minutes moderate to vigorous activity, 3 or more times per week
2 drinks or fewer per day for men and 1 drink or fewer per day for women
Table 2 presents the association between lifestyle variables and five-year mortality among colon cancer cases. Pre-diagnosis smoking and obesity were associated with an increased risk of five-year all-cause mortality. Compared to never smokers, those who were current smokers had 74% increased risk of death from any cause within five years, and had 46% increased risk of death from colorectal cancer. Compared to normal weight individuals, those who were obese had about a 19% increased risk of death from any cause, and about 84% increased risk of CVD death. There were also significant trends of decreased CVD mortality with increasing HEI-2005 scores and with alcohol consumption.
Table 2.
Multivariable adjusted risk ratios of five-year mortality by pre-diagnosis lifestyle risk factors among colon cancer cases
| All-cause Mortality | Colorectal Cancer Mortality | CVD Mortality | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Deaths (n) | RR* | 95% CI | Deaths (n) | RR* | 95% CI | Deaths (n) | RR* | 95% CI | |
| Healthy Eating Index | |||||||||
| Quintile 1 (21.8–56.7) | 268 | 1.00 | 170 | 1.00 | 32 | 1.00 | |||
| Quintile 2 (56.8–65.2) | 264 | 1.07 | (0.89–1.28) | 177 | 1.15 | (0.92–1.44) | 18 | 0.51 | (0.28–0.92) |
| Quintile 3 (65.3–71.4) | 239 | 0.99 | (0.82–1.19) | 168 | 1.09 | (0.87–1.38) | 15 | 0.42 | (0.22–0.81) |
| Quintile 4 (71.5–76.9) | 265 | 1.08 | (0.89–1.31) | 179 | 1.16 | (0.91–1.47) | 26 | 0.64 | (0.35–1.15) |
| Quintile 5 (77.0–90.2) | 237 | 0.95 | (0.78–1.16) | 162 | 0.99 | (0.77–1.27) | 17 | 0.45 | (0.23–0.87) |
| p-for-trend | 0.22 | 0.41 | 0.01 | ||||||
| Body Mass Index (m/kg2) | |||||||||
| Normal weight (BMI 18.5–24.9) | 376 | 1.00 | 267 | 1.00 | 22 | 1.00 | |||
| Overweight (BMI 25–29.9) | 543 | 1.02 | (0.88–1.17) | 357 | 0.97 | (0.82–1.15) | 53 | 1.72 | (1.02–2.88) |
| Obese (BMI≥30) | 354 | 1.19 | (1.02–1.39) | 232 | 1.15 | (0.96–1.39) | 33 | 1.84 | (1.05–3.23) |
| p-for-trend | 0.05 | 0.25 | 0.03 | ||||||
| Physical Activity | |||||||||
| Never or rarely | 278 | 1.00 | 179 | 1.00 | 23 | 1.00 | |||
| 1–3 times per month | 202 | 1.02 | (0.85–1.23) | 127 | 0.96 | (0.76–1.22) | 21 | 1.39 | (0.75–2.58) |
| 1–2 times per week | 266 | 0.88 | (0.74–1.04) | 180 | 0.89 | (0.72–1.11) | 19 | 0.77 | (0.42–1.43) |
| 3–4 times per week | 298 | 0.93 | (0.78–1.10) | 202 | 0.98 | (0.79–1.21) | 28 | 0.96 | (0.54–1.71) |
| 5 or more times per week | 229 | 1.04 | (0.87–1.25) | 168 | 1.16 | (0.93–1.45) | 17 | 0.90 | (0.47–1.72) |
| p-for-trend | 0.68 | 0.37 | 0.79 | ||||||
| Alcohol | |||||||||
| Nondrinker | 1022 | 1.00 | 217 | 1.00 | 39 | 1.00 | |||
| Moderate drinker† | 2472 | 0.82 | (0.71–0.93) | 477 | 0.86 | (0.73–1.01) | 50 | 0.47 | (0.31–0.73) |
| Heavier drinker | 719 | 0.92 | (0.77–1.11) | 162 | 1.06 | (0.84–1.33) | 19 | 0.44 | (0.24–0.81) |
| p-for-trend | 0.07 | 0.60 | 0.004 | ||||||
| Smoking | |||||||||
| Never smoked | 366 | 1.00 | 273 | 1.00 | 28 | 1.00 | |||
| Quit 10 or more years ago | 484 | 1.04 | (0.90–1.20) | 329 | 0.95 | (0.80–1.12) | 44 | 1.21 | (0.74–1.99) |
| Quit 9 years ago or less | 207 | 1.29 | (1.09–1.54) | 118 | 1.01 | (0.81–1.27) | 23 | 1.88 | (1.06–3.48) |
| Current smoker | 216 | 1.74 | (1.45–2.08) | 136 | 1.46 | (1.17–1.82) | 13 | 1.41 | (0.70–2.81) |
| p-for-trend | <0.0001 | 0.005 | 0.10 | ||||||
RRs from Cox proportional hazards model with age as time metric, adjusted for lag time, sex, education, family history of colon cancer, cancer stage, first course of treatment (surgery, radiation, chemotherapy) and also mutually adjusted for quintiles of HEI-2005 scores, BMI, physical activity, alcohol, and smoking history.
≤2 drinks/day for men and ≤1 drink/day for women
The associations between individual lifestyle factors and mortality among rectal cancer cases are presented in Table 3. Compared to nonsmokers, those who smoked had 34% increased risk of all-cause mortality, and those with highest HEI-2005 scores had 40% decreased risk of five-year all-cause mortality, compared to those with the lowest scores. There was a significant trend of decreased risk of colorectal cancer specific mortality with increasing HEI-2005 scores. Pre-diagnosis physical activity and alcohol intake were not associated with either all-cause mortality or colorectal cancer-specific mortality among colon or rectal cancer cases.
Table 3.
Multivariable adjusted risk ratios of five-year mortality by pre-diagnosis lifestyle risk factors among rectal cancer cases
| All-cause Mortality | Colorectal Cancer Mortality | CVD Mortality | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Deaths (n) | RR* | 95% CI | Deaths (n) | RR* | 95% CI | Deaths (n) | RR* | 95% CI | |
| Healthy Eating Index | |||||||||
| Quintile 1 (21.8–56.7) | 122 | 1.00 | 83 | 1.00 | 8 | 1.00 | |||
| Quintile 2 (56.8–65.2) | 102 | 0.87 | (0.66–1.15) | 71 | 0.89 | (0.64–1.26) | 11 | 1.37 | (0.53–3.54) |
| Quintile 3 (65.3–71.4) | 98 | 0.96 | (0.71–1.30) | 64 | 0.94 | (0.65–1.37) | 10 | 0.92 | (0.32–2.64) |
| Quintile 4 (71.5–76.9) | 75 | 0.93 | (0.67–1.29) | 42 | 0.81 | (0.53–1.23) | 12 | 1.67 | (0.62–4.45) |
| Quintile 5 (77.0–90.2) | 57 | 0.60 | (0.42–0.86) | 41 | 0.64 | (0.41–0.99) | 2 | 0.28 | (0.06–1.43) |
| p-for-trend | 0.04 | 0.05 | 0.65 | ||||||
| Body Mass Index (m/kg2) | |||||||||
| Normal weight (BMI 18.5–24.9) | 147 | 1.00 | 92 | 1.00 | 16 | 1.00 | |||
| Overweight (BMI 25–29.9) | 198 | 0.85 | (0.68–1.07) | 135 | 0.92 | (0.70–1.22) | 17 | 0.78 | (0.38–1.61) |
| Obese (BMI≥30) | 109 | 1.00 | (0.77–1.30) | 74 | 1.04 | (0.75–1.44) | 10 | 0.97 | (0.42–2.25) |
| p-for-trend | 0.86 | 0.57 | 0.98 | ||||||
| Physical Activity | |||||||||
| Never or rarely | 92 | 1.00 | 59 | 1.00 | 8 | 1.00 | |||
| 1–3 times per month | 72 | 1.08 | (0.78–1.48) | 53 | 1.23 | (0.84–1.80) | 6 | 0.98 | (0.31–3.07) |
| 1–2 times per week | 99 | 0.87 | (0.65–1.16) | 70 | 0.92 | (0.64–1.31) | 10 | 1.08 | (0.41–2.87) |
| 3–4 times per week | 112 | 1.01 | (0.76–1.35) | 69 | 0.95 | (0.66–1.37) | 12 | 1.17 | (0.46–3.00) |
| 5 or more times per week | 79 | 0.84 | (0.61–1.15) | 50 | 0.78 | (0.53–1.15) | 7 | 0.95 | (0.33–2.77) |
| p-for-trend | 0.18 | 0.12 | 0.78 | ||||||
| Alcohol | |||||||||
| Nondrinker | 93 | 1.00 | 67 | 1.00 | 6 | 1.00 | |||
| Moderate drinker† | 246 | 0.96 | (0.75–1.23) | 154 | 0.84 | (0.62–1.14) | 30 | 1.40 | (0.56–3.49) |
| Heavier drinker | 115 | 1.03 | (0.76–1.41) | 80 | 0.97 | (0.66–1.41) | 7 | 0.95 | (0.30–3.01) |
| p-for-trend | 0.69 | 0.90 | 0.80 | ||||||
| Smoking | |||||||||
| Never smoked | 105 | 1.00 | 74 | 1.00 | 8 | 1.00 | |||
| Quit 10 or more years ago | 195 | 1.23 | (0.96–1.58) | 130 | 1.28 | (0.94–1.75) | 15 | 1.01 | (0.41–2.47) |
| Quit 9 years ago or less | 82 | 1.38 | (1.02–1.87) | 53 | 1.29 | (0.89–1.87) | 11 | 2.74 | (1.07–7.03) |
| Current smoker | 72 | 1.34 | (0.97–1.86) | 44 | 1.11 | (0.74–1.68) | 9 | 2.63 | (0.95–7.31) |
| p-for-trend | 0.04 | 0.57 | 0.007 | ||||||
RRs estimated with Cox proportional hazards model with age as time metric and adjusted for lag time, sex, education, family history of colon cancer, cancer stage, first course of treatment (surgery, radiation, chemotherapy), and also mutually adjusted for quintiles of HEI-2005 scores, BMI, physical activity, alcohol, and smoking history.
≤2 drinks/ day for men and ≤1 drink/day for women
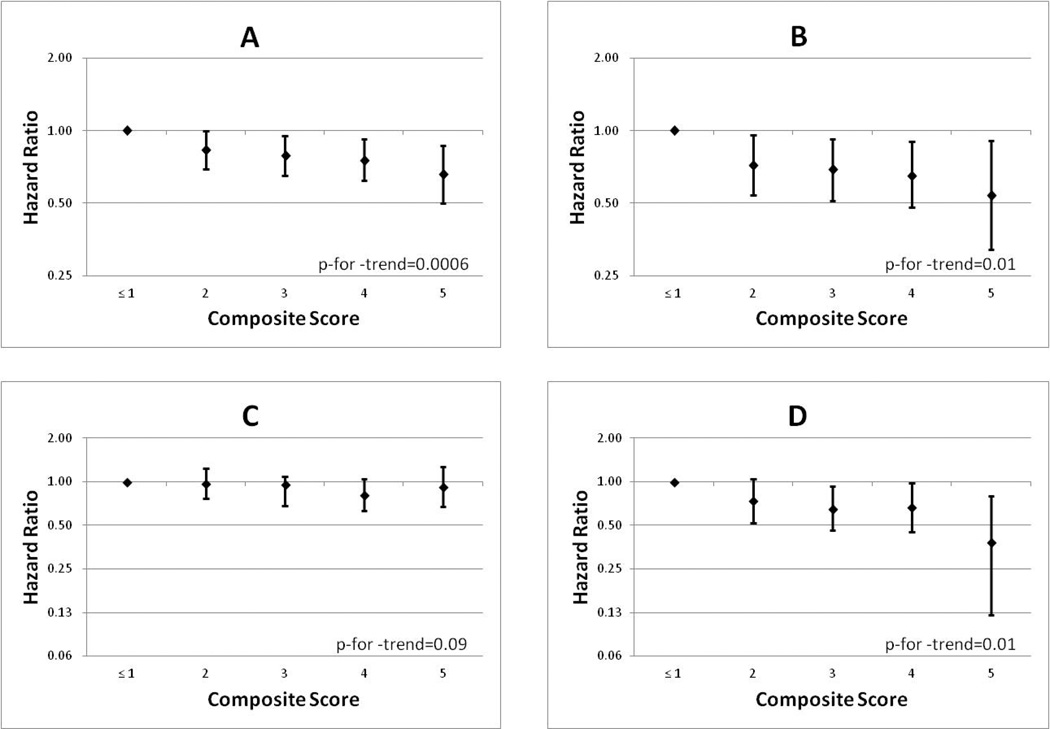
Higher composite lifestyle scores were associated with reduced all-cause mortality (Figure 1) among colon and rectal cancer cases. Compared to those who met 1 or fewer recommendations, colon cancer cases who met all 5 had a 34% reduced risk of death (RR 0.66; 95% CI 0.50–0.87; p-for-trend=0.006, Figure 1A) and rectal cancer cases had a 46% reduced risk of death (RR 0.54; 95% CI 0.32–0.91; p-for-trend=0.01, figure 1B). Higher lifestyle scores were also associated with reduced risk of colorectal cancer mortality among rectal cancer cases (figure 1C), but not among colon cancer cases (Figure 1D). There were no significant interactions between the composite lifestyle score and sex, smoking, BMI, or cancer stage.
Figure 1.
Multivariable* adjusted risk ratios and 95% confidence intervals of composite lifestyle scores and five year all-cause mortality among A) colon cancer cases and B) rectal cancer cases, and five year colorectal-cancer specific mortality among C) colon cancer cases and D) rectal cancer cases in the NIH-AARP Diet and Health Study.
* RRs from Cox proportional hazards model with age as time metric, adjusted for lag time, sex, education, family history of colon cancer, cancer stage, and first course of treatment (surgery, radiation, chemotherapy).
Discussion
In this study we found that a combined lifestyle score was associated with reduced all-cause mortality among both colon and rectal cancer cases. Several modifiable pre-diagnostic lifestyle factors contributed to this association. In colon cancer cases, obesity and smoking were related to an increased risk of all-cause mortality. Smoking was also associated with increased risk of colorectal cancer specific mortality while the HEI-2005, obesity, and alcohol consumption were related to the risk of death due to CVD. In rectal cancer cases, higher HEI-2005 scores were associated with decreased risk of all-cause and colorectal cancer mortality, and smoking was related to an increased risk of all cause and CVD mortality.
Given the well-known negative impact of smoking on health, it is not surprising that pre-diagnosis smoking is associated with increased risk of death among colorectal cancer cases. Similar to our finding, a study of incident colorectal cancer cases identified in the Surveillance Epidemiology and End Results registry in Washington State [17] found that pre-diagnosis smoking was associated with increased risk of all-cause mortality (RR 1.51; 95% CI 1.24–1.83) and colorectal cancer-specific mortality among colorectal cancer cases (RR 1.30; 95% CI 7.09–1.74). In another study of stage III colon cancer cases [18], pre-diagnosis smoking was also associated with increased risk of death from any cause (RR 1.65; 95% CI 1.12–2.42).
In our study pre-diagnosis obesity was associated with poorer overall survival among colon cancer cases. Other studies have also found that pre-diagnosis BMI [6], or body weight, body fat percentage and waist circumference [5] were associated with poorer overall and disease-specific survival among colorectal cancer cases. However, studies of post-diagnosis BMI and mortality in colorectal cancer cases have reported inconsistent results [7–11].
Pre- or post-diagnosis physical activity has been found to be associated with lower risk of mortality among colorectal cancer cases in several studies [5, 12–15]. The lack of association between pre-diagnosis physical activity and mortality in our study could be partly due to differences in measurement instruments, in the time between measurement and diagnosis, or differences in adjustment factors.
In the only other study that examined dietary patterns and survival among colon cancer patients, a post-diagnosis Western dietary pattern (characterized by high intakes of meat, fat, and desserts) was associated with poorer survival [16]. Among rectal cancer cases, we found lower all-cause mortality among those with higher scores on the HEI-2005, while we observed no association among colon cancer cases. This lack of association might be explained by the fact that the HEI-2005 is constructed for overall health rather than cancer prevention or survival. For example, the HEI-2005 awards points for meat consumption without separating red meat (a known colorectal cancer risk factor[19]) from other, leaner sources of animal protein. However, this index incorporates several other features that may partially mitigate these limitations. Specifically, HEI-2005 rewards points for lower consumption of foods thought to contribute to poor health outcomes, such as sugar and saturated fats (i.e. calories from Solid Fats, Alcoholic beverages, and Added Sugars (SoFAAS)). Thus points awarded for consumption of red meat would be somewhat ameliorated by a low score in the SoFAAS category. In addition, the HEI-2005 awards points for consumption of whole fruit and whole grains, which is consistent with ACS and AICR guidelines for cancer prevention and cancer survivors [4,21]. Furthermore, this index has already been shown to be related to colorectal cancer risk in this study population [22], and in contrast to the data-driven approach of Meyerhardt’s study [16], has the ability to be directly compared to other studies that use the same index.
Only one other study has examined alcohol in relationship to survival among colorectal cancer cases [17] and found no association of alcohol intake with all-cause or disease-specific mortality. We found that alcohol intake was related to lower risk of CVD mortality, but not to colorectal cancer mortality among colon cancer cases. More research is needed on dietary patterns and alcohol in relation to cancer survival.
We found significantly reduced risk of five-year all-cause mortality among colon and rectal cancer cases with a higher combined lifestyle score. Energy balance factors such as diet, body weight, and physical activity influence colorectal cancer risk and may also impact cancer recurrence and progression [23]. Proposed mechanisms for the association between energy balance and cancer risk or progression include peptide hormones (i.e. leptin and adiponectin), insulin and insulin-like growth factor 1 signaling, and inflammation [23]. Furthermore, BMI, diet, and physical activity have been shown to be associated with genetic and epigenetic changes in colon and rectal tumors [24] and these changes could have implications for recurrence, progression, and therefore survival.
Our study has several limitations. One important limitation is that lifestyle information was collected only once, before cancer diagnosis, and we do not know how lifestyle changes post-diagnosis might affect survival outcomes among colorectal cancer cases. Clearly, maintaining healthy behaviors in adulthood is consistent with current disease prevention guidelines, and with recommendations for the prevention of comorbidities and improved quality of life among cancer survivors. However currently there is insufficient evidence to recommend lifestyle changes to cancer survivors specifically for the purpose of improving survival outcomes. At most, these results suggest a possible impact of lifestyle behaviors on cancer survival, and support the need for further prospective research on this topic.
This analysis was also limited by the fact that subjects had different lengths of time between the lifestyle assessment and cancer diagnosis (lag time). We therefore adjusted the analyses for lag time and limited follow-up time to 5 years. Also, while we had cancer registry treatment data on first course of therapy within 12 months of cancer diagnosis, we did not have extensive clinical data, including information on recurrence or other non-mortality outcomes. Small sample sizes may have limited our ability to detect associations, and small cell sizes in some analyses could produce spurious associations. Additionally, since true associations of lifestyle factors and cancer survival outcomes are not known, we weighted each recommended lifestyle factor equally when creating a single lifestyle score. Equal weighting may give undo emphasis to weak risk factors and underestimate the effects of others. Therefore, the protective trend associated with higher lifestyle scores should be emphasized rather than the exact magnitude of effect for the combined score. Furthermore, we cannot rule out confounding of health behaviors with unmeasured factors such as socioeconomic status and healthcare access. Strengths of our study include the large sample size that allowed us to examine colon and rectal cancer cases separately, and the collection of multiple health behaviors, which enabled the examination of a lifestyle score computed based on current recommendations for cancer survivors.
In summary, we found that healthier lifestyle before colorectal cancer diagnosis was associated with improved overall survival after diagnosis with colorectal cancer. Our study adds to a growing body of literature demonstrating the importance of maintaining healthy lifestyle before cancer diagnosis. More research is needed to investigate the effect of these lifestyle factors on health in colorectal cancer survivors.
Acknowledgement
Cancer incidence data were collected from following state cancer registries: Georgia Center for Cancer Statistics, Department of Epidemiology, Rollins School of Public Health, Emory University; California Department of Health Services, Cancer Surveillance Section; Michigan Cancer Surveillance Program, Community Health Administration, State of Michigan; Florida Cancer Data System (FCDC) under contract with the Florida Department of Health (FDOH); Louisiana Tumor Registry, Louisiana State University Medical Center in New Orleans; New Jersey State Cancer Registry, Cancer Epidemiology Services, New Jersey State Department of Health and Senior Services; North Carolina Central Cancer Registry; Division of Health Statistics and Research, Pennsylvania Department of Health, Harrisburg, Pennsylvania; Arizona Cancer Registry, Division of Public Health Services, Arizona Department of Health Services; Texas Cancer Registry, Cancer Epidemiology and Surveillance Branch, Texas Department of State Health Services; and Nevada Central Cancer Registry, Center for Health Data and Research, Bureau of Health Planning and Statistics, State Health Division, State of Nevada Department of Health and Human Services. The views expressed herein are solely those of the authors and do not necessarily reflect those of the FCDC or FDOH. The Pennsylvania Department of Health specifically disclaims responsibility for any analyses, interpretations or conclusions.
Funding: Intramural Research Program of the NIH, National Cancer Institute
Footnotes
Financial disclosures: the authors have no financial relationships to disclose.
References
- 1.Surveillance Research Program NCI. Fast Stats: An interactive tool for access to SEER cancer statistics. [Accessed 01/17/2013]; http://seer.cancer.gov/faststats.
- 2.Institute of Medicine (IOM) From Cancer Patient to Cancer Survivor: Lost in Transition. Washington, DC: National Academy Press; 2005. [Google Scholar]
- 3.World Cancer Research Fund (WCRF)/American Institute for Cancer Research (AICR) Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. 2nd ed. Washington, DC: AICR; 2007. [Google Scholar]
- 4.Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin. 2012;62:243–274. doi: 10.3322/caac.21142. [DOI] [PubMed] [Google Scholar]
- 5.Haydon AM, Macinnis RJ, English DR, Giles GG. Effect of physical activity and body size on survival after diagnosis with colorectal cancer. Gut. 2006;55:62–67. doi: 10.1136/gut.2005.068189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Campbell PT, Newton CC, Dehal AN, Jacobs EJ, Patel AV, Gapstur SM. Impact of body mass index on survival after colorectal cancer diagnosis: the Cancer Prevention Study-II Nutrition Cohort. J Clin Oncol. 2010;30:42–52. doi: 10.1200/JCO.2011.38.0287. [DOI] [PubMed] [Google Scholar]
- 7.Dignam JJ, Polite BN, Yothers G, Raich P, Colangelo L, O'Connell MJ, et al. Body mass index and outcomes in patients who receive adjuvant chemotherapy for colon cancer. J Natl Cancer Inst. 2006;98:1647–1654. doi: 10.1093/jnci/djj442. [DOI] [PubMed] [Google Scholar]
- 8.Sinicrope FA, Foster NR, Sargent DJ, O'Connell MJ, Rankin C. Obesity is an independent prognostic variable in colon cancer survivors. Clin Cancer Res. 2010;16:1884–1893. doi: 10.1158/1078-0432.CCR-09-2636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Meyerhardt JA, Catalano PJ, Haller DG, Mayer RJ, Benson AB, 3rd, Macdonald JS, et al. Influence of body mass index on outcomes and treatment-related toxicity in patients with colon carcinoma. Cancer. 2003;98:484–495. doi: 10.1002/cncr.11544. [DOI] [PubMed] [Google Scholar]
- 10.Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Mayer RJ, Nelson H, et al. Impact of body mass index and weight change after treatment on cancer recurrence and survival in patients with stage III colon cancer: findings from Cancer and Leukemia Group B 89803. J Clin Oncol. 2008;26:4109–4115. doi: 10.1200/JCO.2007.15.6687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Meyerhardt JA, Tepper JE, Niedzwiecki D, Hollis DR, McCollum AD, Brady D, et al. Impact of body mass index on outcomes and treatment-related toxicity in patients with stage II and III rectal cancer: findings from Intergroup Trial 0114. J Clin Oncol. 2004;22:648–657. doi: 10.1200/JCO.2004.07.121. [DOI] [PubMed] [Google Scholar]
- 12.Meyerhardt JA, Giovannucci EL, Holmes MD, Chan AT, Chan JA, Colditz GA, et al. Physical activity and survival after colorectal cancer diagnosis. J Clin Oncol. 2006;24:3527–3534. doi: 10.1200/JCO.2006.06.0855. [DOI] [PubMed] [Google Scholar]
- 13.Meyerhardt JA, Heseltine D, Niedzwiecki D, Hollis D, Saltz LB, Mayer RJ, et al. Impact of physical activity on cancer recurrence and survival in patients with stage III colon cancer: findings from CALGB 89803. J Clin Oncol. 2006;24:3535–3541. doi: 10.1200/JCO.2006.06.0863. [DOI] [PubMed] [Google Scholar]
- 14.Meyerhardt JA, Giovannucci EL, Ogino S, Kirkner GJ, Chan AT, Willett W, et al. Physical activity and male colorectal cancer survival. Arch Intern Med. 2009;169:2102–2108. doi: 10.1001/archinternmed.2009.412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Campbell PT, Patel AV, Newton CC, Jacobs EJ, Gapstur SM. Associations of recreational physical activity and leisure time spent sitting with colorectal cancer survival. J Clin Oncol. 2013 Mar 1;31(7):876–885. doi: 10.1200/JCO.2012.45.9735. [DOI] [PubMed] [Google Scholar]
- 16.Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Hu FB, Mayer RJ, et al. Association of dietary patterns with cancer recurrence and survival in patients with stage III colon cancer. JAMA. 2007;298:754–764. doi: 10.1001/jama.298.7.754. [DOI] [PubMed] [Google Scholar]
- 17.Phipps AI, Baron J, Newcomb PA. Prediagnostic smoking history, alcohol consumption, and colorectal cancer survival: the Seattle Colon Cancer Family Registry. Cancer. 2011;117:4948–4957. doi: 10.1002/cncr.26114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McCleary NJ, Niedzwiecki D, Hollis D, Saltz LB, Schaefer P, Whittom R, et al. Impact of smoking on patients with stage III colon cancer: results from Cancer and Leukemia Group B 89803. Cancer. 2011;116:957–966. doi: 10.1002/cncr.24866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McCollough McCullough ML, Gapstur SM, Shah R, Jacobs EJ, Campbell PT. Association between red and processed meat intake and mortality among colorectal cancer survivors. J Clin Oncol. 2013 Aug 1;31(22):2773–2782. doi: 10.1200/JCO.2013.49.1126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schatzkin A, Subar AF, Thompson FE, Harlan LC, Tangrea J, Hollenbeck AR, et al. Design and serendipity in establishing a large cohort with wide dietary intake distributions : the National Institutes of Health-American Association of Retired Persons Diet and Health Study. Am J Epidemiol. 2001;154:1119–1125. doi: 10.1093/aje/154.12.1119. [DOI] [PubMed] [Google Scholar]
- 21.Kushi LH, Doyle C, McCullough M, Rock CL, Demark-Wahenfried W, Bandera EV, et al. American Cancer Society 2010 Nutrition and Physical Activity Guidelines Advisory Committee. CA Cancer J Clin. 2012;62(1):30–37. doi: 10.3322/caac.20140. [DOI] [PubMed] [Google Scholar]
- 22.Reedy J, Mitrou PN, Krebs-Smith SM, Wirfalt E, Flood A, Kipnis V, Leitzmann M, Mouw T, Hollenbeck A, Schatzkin A, Subar AF. Index-based dietary patterns and risk of colorectal cancer: the NIH-AARP Diet and Health Study. Am J Epidemiol. 2008;168:38–48. doi: 10.1093/aje/kwn097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hursting SD, Berger NA. Energy balance, host-related factors, and cancer progression. J Clin Oncol. 2010;28:4058–4065. doi: 10.1200/JCO.2010.27.9935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Slattery ML, Curtin K, Ma K, Edwards S, Schaffer D, Anderson K, et al. Diet activity, and lifestyle associations with p53 mutations in colon tumors. Cancer Epidemiol Biomarkers Prev. 2002;11:541–548. [PubMed] [Google Scholar]