Abstract
Ramoplanin is a glycolipodepsipeptide antibiotic with activity against gram-positive bacteria that is in clinical trials for prevention of vancomycin-resistant Enterococcus (VRE) bloodstream infections and treatment of Clostridium difficile diarrhea. Orally administered ramoplanin suppresses VRE intestinal colonization, but recurrences after discontinuation of treatment have frequently been observed. We used a mouse model to examine the efficacy of ramoplanin for inhibition of VRE colonization and evaluated the etiology of recurrences of colonization. Eight days of treatment with ramoplanin (100 μg/ml) in drinking water suppressed VRE to undetectable levels, but 100% of mice developed recurrent colonization; a higher dose of 500 μg/ml in water was associated with recurrent colonization in 50% of mice. Two of eight (25%) mice treated with the 100-μg/ml dose of ramoplanin had low levels of VRE in their cecal tissues on day 8 despite undetectable levels in stool and cecal contents. Mice that received prior ramoplanin treatment did not develop VRE overgrowth when challenged with 107 CFU of oral VRE 1, 2, or 4 days later. In communal cages, rapid cross-transmission and overgrowth of VRE was observed among clindamycin-treated mice; ramoplanin treatment effectively suppressed VRE overgrowth in such communal cages. Ramoplanin treatment promoted increased density of indigenous Enterobacteriaceae and overgrowth of an exogenously administered Klebsiella pneumoniae isolate. These results demonstrate the efficacy of ramoplanin for inhibition of VRE colonization and suggest that some recurrences occur due to reexpansion of organisms that persist within the lining of the colon. Ramoplanin treatment may be associated with overgrowth of gram-negative bacilli.
The intestinal tract provides a major source for transmission of vancomycin-resistant enterococci (VRE) (5). Oral nonabsorbed antibiotics such as ramoplanin and bacitracin have been shown to effectively suppress VRE intestinal colonization in mice and colonized patients; however, recurrences after discontinuation of treatment have frequently been observed (6, 10, 17-19). Such recurrences may be due to reexpansion of small numbers of VRE that persist in the intestinal tract during treatment or reacquisition of VRE after treatment has eliminated the original colonizing strain or strains. Baden et al. (1) found that recurrences after ramoplanin treatment were often associated with isolation of VRE strains that were genotypically unrelated to pretreatment strains, suggesting that new VRE strains might have been acquired. The concurrent use of antianaerobic antibiotics by many of the study patients was a potential confounding variable because these agents have been shown to facilitate overgrowth of preexisting or newly acquired VRE strains (1, 5). Similarly, the 100% recurrence rate of VRE colonization observed in a previous mouse study could have been due in part to the fact that oral vancomycin or streptomycin treatment was continued during and after discontinuation of oral ramoplanin (18).
After antibiotic treatment, recovery of the indigenous intestinal microflora may be delayed for days or weeks (16). We have previously demonstrated that mice are susceptible to development of high-density VRE stool colonization when VRE are ingested 2 or 5, but not 10, days after completion of treatment with antianaerobic antibiotics (authors' unpublished data). Because ramoplanin and bacitracin both have in vitro activity against gram-positive anaerobes (2, 12), we hypothesized that delayed recovery of competing anaerobes after treatment with such agents might facilitate relapses or reacquisition of VRE colonization. Because VRE may become associated with the lining of the colon in the setting of high-density intestinal colonization (authors' unpublished data), we also hypothesized that persistence of VRE at this site during ramoplanin therapy could provide a source for relapses after treatment.
In this study, we evaluated the efficacy of ramoplanin for decolonization of VRE in mice in the absence of concurrent use of other antibiotics. We examined whether VRE persist within the cecal tissues during ramoplanin treatment. We determined the effect of oral ramoplanin on the indigenous stool microflora and evaluated whether prior ramoplanin treatment facilitates establishment of VRE colonization. We examined the use of ramoplanin as a means to prevent cross-transmission and overgrowth of VRE among mice housed in communal cages. Finally, we examined whether ramoplanin treatment would facilitate colonization with other nosocomial pathogens, including extended-spectrum β-lactamase-producing Klebsiella pneumoniae and Candida glabrata.
MATERIALS AND METHODS
The pathogens studied.
Enterococcus faecium C68, a previously described VanB-type clinical VRE isolate, was used for all VRE experiments (4). The MIC of ramoplanin for VRE C68 was 0.125 μg/ml. Klebsiella pneumoniae P62 is a clinical isolate that produces an SHV-type extended-spectrum β-lactamase (ESBL). C. glabrata A239 is a clinical isolate for which the MIC of fluconazole is 2 μg/ml. We have previously demonstrated that antibiotics that inhibit intestinal anaerobes promote overgrowth of each of the pathogens studied (4, 11, 15; authors' unpublished data).
Quantification of stool pathogens.
Fresh stool specimens were processed as described elsewhere (4). In order to quantify VRE, K. pneumoniae, and C. glabrata, diluted samples were plated onto Enterococcosel agar containing vancomycin (20 μg/ml), MacConkey agar containing ceftazidime (10 μg/ml), or Sabouraud dextrose agar (Becton, Dickinson, and Company, Sparks, Md.) containing piperacillin-tazobactam (16 μg/ml) and linezolid (8 μg/ml), respectively. The plates were incubated in room air at 37°C for 24 or 48 h, and the number of CFU of each pathogen per gram of sample was calculated.
Efficacy of ramoplanin for eradication of VRE colonization.
Female CF1 mice (Harlan Sprague-Dawley, Indianapolis, Ind.) weighing 25 to 30 g were used in all experiments. In order to minimize the risk of cross-contamination, mice were housed in individual cages with plastic filter tops unless otherwise specified. High-density VRE stool colonization was established in mice by administering subcutaneous clindamycin (1.4 mg) once each day for 2 days before and 7 days after orogastric inoculation of 106 CFU of VRE C68 using a stainless steel feeding tube (Perfektum; Popper & Sons, New Hyde Park, N.Y.). After discontinuation of clindamycin, mice received oral ramoplanin (100 or 500 μg/ml of drinking water) or regular drinking water (controls) for 8 days. Six total mice were included in each treatment group. Stool pellets were collected every 3 to 4 days to monitor the density of VRE before, during, and after completion of ramoplanin treatment.
Ramoplanin was administered in drinking water rather than by orogastric gavage in order to avoid exposing the mice to repeated gavage procedures. Based on consumption of approximately 0.5 ml of drinking water by each mouse per day (authors' unpublished data), we estimated that mice receiving the 100- and 500-μg/ml doses would ingest approximately 1.7 and 8.3 mg of ramoplanin/kg of body weight/day, respectively. In comparison, the human doses of 100 or 400 mg twice daily that have been shown to inhibit VRE colonization are equivalent to 3 or 13 mg/kg ramoplanin per day (based on a weight of 60 kg). Whitman et al. (18) previously showed that a 100-μg/ml dose of ramoplanin in drinking water suppressed VRE colonization in mice during a 7-day period of administration despite concurrent oral vancomycin treatment; the concentration of ramoplanin in stool was not assessed.
To evaluate the possibility that ramoplanin-treated mice were being reexposed to VRE from their environment, broth-enrichment cultures for VRE were performed as previously described (13) after contacting cage bottoms and tops, water bottles, and food with premoistened cotton-tipped swabs. To evaluate whether relapses of colonization were due to persistence of VRE within the colon, eight mice that received 8 days of oral ramoplanin treatment (100 μg/ml of water) were euthanized, and portions of cecal contents and cecal lining (sections, 1 by 1 cm) were weighed, homogenized in sterile phosphate-buffered saline (PBS) using a pestle, and cultured for VRE as described above.
Effect of ramoplanin on the indigenous stool microflora.
Five mice were treated with ramoplanin (100 μg/ml) in drinking water for 7 days. Stool samples were collected prior to treatment, on day 7 of treatment, and 3, 6, and 11 days after discontinuation of ramoplanin. Quantitative cultures for facultative and aerobic gram-negative bacilli, enterococci, total anaerobes, Bacteroides species, Lactobacillus species, and Clostridium species were performed by plating serially diluted specimens onto MacConkey agar (Difco Laboratories, Detroit, Mich.), Enterococcosel agar (Becton Dickinson, Cockeysville, Md.), Brucella agar (Becton Dickinson), Bacteroides bile esculin agar, Rogosa agar, and egg yolk agar, respectively. For culture of anaerobes, stool samples were processed inside an anaerobic chamber (Coy Laboratories, Grass Lake, Mich.). Denaturing gradient gel electrophoresis (DGGE) of PCR-amplified bacterial rRNA genes from stool was performed as previously described (7).
Effect of prior ramoplanin treatment on the establishment of VRE colonization.
Four hours, 1 day, 2 days, or 4 days after completing a 7-day course of oral ramoplanin (100 μg/ml of drinking water) or regular drinking water (controls), mice received orogastric inoculation of 107 CFU of VRE in phosphate buffered saline. The density of VRE in stool was monitored before and 1 and 4 days after inoculation. Four mice were included in each treatment group.
Use of ramoplanin to prevent cross-transmission of VRE among mice.
One set of experiments was performed to evaluate the ability of ramoplanin to prevent cross-transmission and overgrowth of VRE among mice in communal cages. High-density VRE stool colonization (∼7 log10 CFU/g) was established in two mice as described above. Each VRE-colonized mouse was placed into a communal cage along with four mice with no previous exposure to antibiotics or VRE; the experimental cage was supplied with oral ramoplanin (100 μg/ml) in drinking water from days 0 to 9 and the control cage was supplied with regular drinking water. All mice were treated with subcutaneous clindamycin (1.4 mg) once daily for 5 days. After 9 days, all mice were separated into individual cages and supplied with regular drinking water. The density of VRE in stool was monitored every 3 to 4 days during and after completion of ramoplanin treatment.
Effect of ramoplanin treatment on the establishment of colonization by C. glabrata or K. pneumoniae.
On day 2 of a 6-day course of oral ramoplanin (100 μg/ml of drinking water) or regular drinking water (controls), mice received orogastric inoculation of 106 CFU of C. glabrata A239 or K. pneumoniae P62. The density of C. glabrata or ceftazidime-resistant K. pneumoniae in stool was monitored every 3 to 4 days. Four mice were included in each treatment group.
Measurement of ramoplanin concentrations in stool.
The concentration of ramoplanin in selected stool samples was measured using an agar well diffusion assay with Clostridium perfringens as the indicator strain (14).
Statistical analysis.
Mean densities (in log10 CFU per gram) were determined across the data collection time points for individual mice, excluding the baseline values. These means were used to create an ordinal composite treatment variable for each treatment group. A one-way analysis of variance of the composite treatment variables was performed. Overall differences and pairwise differences were examined, with P values adjusted for multiple comparisons using the Scheffe correction. For the experiments in which the efficacy of ramoplanin for VRE decolonization was tested, separate analyses were performed during treatment and after completion of treatment. Students t test was used to compare DGGE similarity indices among treatment groups. Computations were performed with the use of Stata software (version 6.0; Stata, College Station, Tex.).
RESULTS
Efficacy of ramoplanin for eradication of VRE colonization.
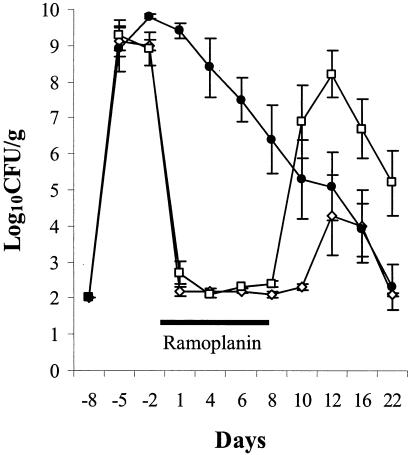
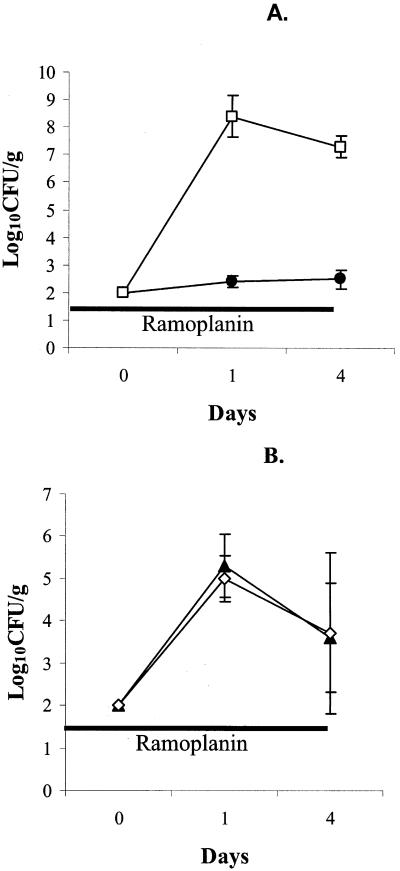
Figure 1 shows the densities of VRE during and after completion of 8 days of ramoplanin treatment. There were no significant differences in the densities of VRE among the treatment groups prior to starting ramoplanin on days −5 and −2 (P = 1). All of the ramoplanin-treated mice developed undetectable levels of VRE in stool during treatment (P < 0.0001 in comparison to saline controls). One hundred percent of mice receiving ramoplanin at 100 μg/ml in drinking water developed a recurrence of colonization after discontinuing treatment; the mean density of VRE rose to a higher level than the saline controls after the recurrence, but the difference was not statistically significant (P = 0.63). Fifty percent of mice receiving ramoplanin at 500 μg/ml developed a detectable recurrence; the mean density of VRE after the recurrence did not differ from the saline controls.
FIG. 1.
Efficacy of oral ramoplanin treatment for decolonization of VRE stool colonization in mice. High-density VRE colonization was established in all mice by administering orogastric VRE on day −8 in conjunction with subcutaneous clindamycin from days −10 to 0. Oral ramoplanin in drinking water (100 [□] or 500 [◊] μg/ml) was given from days 0 to 8 (solid line). Control mice received regular drinking water (•). Error bars represent SE.
During the course of ramoplanin treatment, multiple cultures of cages, food, water, and water bottles were negative for VRE. Of the 8 mice that had cultures of cecal contents and cecal linings taken on day 8 of ramoplanin (100 μg/ml of drinking water) treatment, 8 (100%) had negative stool and cecal content cultures for VRE but 2 (25%) had low levels of VRE (2 to 3 log10 CFU/g) detectable in cecal tissues.
Effect of ramoplanin on the indigenous microflora.
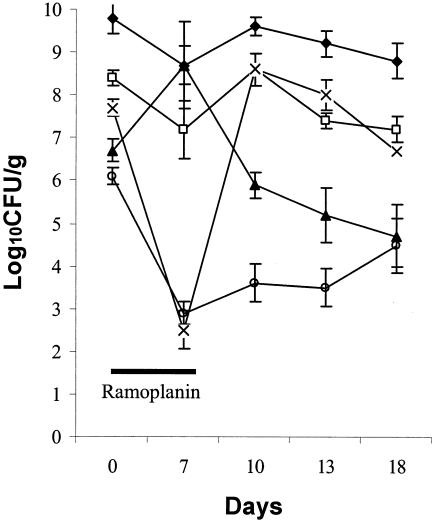
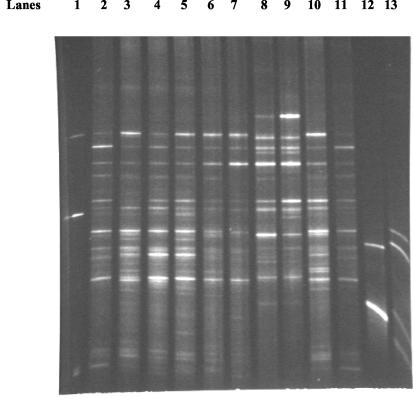
Figure 2 shows the effect of ramoplanin treatment (days 0 to 7) on the indigenous stool microflora. The mean densities of total anaerobes and Bacteroides species were not significantly affected by ramoplanin treatment. The mean density of total facultative and aerobic gram-negative bacilli increased significantly on day 7 of ramoplanin treatment (P < 0.05), but was not significantly different from baseline by 3 days after discontinuation of ramoplanin (day 10). Lactobacillus species were markedly reduced by ramoplanin treatment (P < 0.001), but had returned to pretreatment levels by 3 days after discontinuation of ramoplanin (day 10). Enterococcus species were significantly reduced by ramoplanin treatment (P < 0.001), and remained significantly reduced for at least 11 days after discontinuation of ramoplanin (day 18). Ramoplanin caused relatively little disruption of the stool DGGE patterns (mean similarity indices 72% in comparison to the pretreatment patterns); the effect of subcutaneous clindamycin on the DGGE patterns is shown for comparison (mean similarity indices 17% in comparison to pretreatment patterns) (Fig. 3 shows a representative gel).
FIG. 2.
Mean densities (error bars, SE) of members of the indigenous stool microflora before, during, and after oral ramoplanin treatment from days 0 to 7 (solid line). Symbols: ⧫, total anaerobes; □, Bacteroides; ▴, Enterobacteriaceae; ×, lactobacilli; ○, enterococci.
FIG. 3.
DGGE patterns derived from stool samples before and during antibiotic treatment. Lanes 1, a control pattern containing PCR products obtained from strains of E. coli, Fusobacterium nucleatum, Bacteroides thetaiotaomicron, and Bacteroides uniformis. Lanes 2 and 3, untreated control mice at baseline; lanes 4 and 5, the same control mice 7 days later; lanes 6 and 7, ramoplanin-treated mice at baseline; lanes 8 and 9, the same mice on day 7 of ramoplanin treatment; lanes 10 and 11, clindamycin-treated mice at baseline; lanes 12 and 13, the same mice on day 7 of clindamycin treatment.
Ramoplanin concentrations in stool.
For the 100-μg/ml ramoplanin dose, the mean concentration in stool on day 7 of treatment was 68 μg/g of stool (range, 40 to 75 μg/g; n = 5 mice); no ramoplanin was detectable 3 days after discontinuation of treatment (day 10). For the 500-μg/ml ramoplanin dose, the mean concentration in stool on day 7 was 310 μg/ml (range, 300 to 320 μg/g; n = 5 mice).
Effect of prior ramoplanin treatment on the establishment of VRE colonization.
Mice inoculated with 107 CFU of VRE 4 h or 1, 2, or 4 days after completion of 7 days of ramoplanin treatment did not develop significant overgrowth of VRE in comparison to controls that did not receive ramoplanin (P > 0.5 for each comparison).
Use of ramoplanin to prevent cross-transmission of VRE among mice.
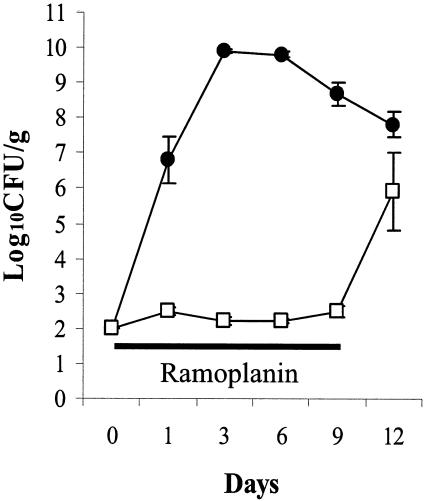
In the absence of ramoplanin treatment, VRE was rapidly transferred from one colonized mouse to 4 clindamycin-treated mice in a communal cage (Fig. 4). With ramoplanin treatment, VRE colonization was rapidly inhibited in the colonized mouse that was added to the communal cage and none of the other mice developed detectable levels of colonization during ramoplanin treatment; after discontinuation of ramoplanin and transfer of mice to individual cages, VRE colonization was detected within 5 days in 4 of 5 mice (80%).
FIG. 4.
Mean densities (error bars, SE) of VRE for groups of mice in communal cages. A mouse with high-density VRE stool colonization (∼7 log10 CFU/g) was placed in a cage along with four mice with no previous exposure to VRE. All mice were treated with subcutaneous clindamycin (1.4 mg) once daily from day 0 to day 5. The control cage received regular drinking water (•), and the experimental cage received ramoplanin in drinking water (□) from days 0 to 9 (solid line). On day 9 all mice were placed in individual cages and ramoplanin was discontinued.
Effect of ramoplanin treatment on the establishment of colonization by C. glabrata or K. pneumoniae.
Ramoplanin facilitated overgrowth of K. pneumoniae P62 (Fig. 5A), but not C. glabrata A239 (Fig. 5B) when these pathogens were inoculated by orogastric gavage on day 2 of a 6-day course of treatment.
FIG. 5.
Effect of oral ramoplanin administration on the establishment of intestinal colonization with ESBL-producing K. pneumoniae (ESBL-KP) (A) or C. glabrata (B) in mice. Mice received 106 CFU of ESBL-KP or C. glabrata by orogastric inoculation on day 0 and either regular drinking water (controls [solid symbols]) or oral ramoplanin in drinking water (open symbols) from days −2 to 4 (solid line). Error bars represent SE.
DISCUSSION
Our data are consistent with previous studies that have demonstrated that oral ramoplanin effectively suppresses VRE intestinal colonization during the course of treatment (18, 19). As previously demonstrated by Whitman et al. (18), 100% of mice treated with 100 μg/ml of ramoplanin in drinking water developed recurrent colonization after treatment was discontinued. Our data suggest that the recurrences of colonization observed by Whitman et al. (18) were not attributable to the fact that oral vancomycin or streptomycin was continued during and after the ramoplanin treatment. In our study, a higher dose of ramoplanin (500 μg/ml of drinking water) was associated with recurrent colonization in only 50% of treated mice. This observation is consistent with the clinical data that suggest that ramoplanin treatment may result in eradication of VRE in some patients (1).
Ramoplanin selectively inhibited the gram-positive colonic microflora of mice. In contrast, administration of oral vancomycin, another agent with minimal in vitro activity against gram-negative bacteria, has been shown to cause marked inhibition of Bacteroides species (8). The overall changes in the microflora associated with ramoplanin were modest in comparison to antianaerobic antibiotics such as clindamycin (Fig. 2), and at least some of the inhibited organisms (e.g., lactobacilli) recovered within 3 days after treatment was discontinued.
Our findings support the hypothesis that some recurrences of VRE colonization after ramoplanin treatment are due to reexpansion of small numbers of organisms that persist in the lining of the colon. We detected VRE in the cecal tissues of two of eight (25%) ramoplanin-treated mice that had undetectable levels of VRE in stool and cecal contents. In addition, prior ramoplanin treatment did not facilitate the establishment of stool colonization after ingestion of VRE. Previous research suggests that organisms that are able to adhere to the mucosal surfaces of the colon (i.e., epithelium or mucous layer) and are adapted to the colonic environment may have a survival advantage over exogenously introduced organisms (9). Minor disruption of the indigenous microflora by ramoplanin could therefore potentially facilitate reexpansion of VRE that are already present, while being insufficient to allow overgrowth of newly introduced strains. As noted previously, antianaerobic antibiotics that are used concurrently with ramoplanin may facilitate acquisition of new VRE strains after decolonization (1, 5).
Although not statistically significant, it is notable that after discontinuation of the 100-μg/ml ramoplanin dose (Fig. 1) the density of VRE among treated mice rose to a level that was higher than among the saline controls (mean ± standard error [SE], 8.2 ± 0.65 versus 5.1 ± 0.95 log10 CFU/g). The failure to achieve statistical significance may be attributable in part to the fact that only six mice were included in the treatment groups. After discontinuation of the 500-μg/ml ramoplanin dose, two of six mice also developed recurrent high-density colonization (>7 log10 CFU/g). Because such overgrowth has implications for the risk of translocation from the intestinal tract and cross-transmission, further studies are needed to evaluate whether similar rebounds of high-density colonization occur among patients.
Because ramoplanin does cause some disruption of the indigenous microflora, use of this agent for VRE could lead to overgrowth of other pathogens that are not inhibited. We found that ramoplanin treatment promoted overgrowth of a strain of ESBL-producing K. pneumoniae (Fig. 5A), but not C. glabrata (Fig. 5B), when these organisms were administered by orogastric inoculation during the course of treatment. Ramoplanin also promoted overgrowth of indigenous Enterobacteriaceae during treatment (Fig. 2). Based on these findings, further studies are warranted to determine whether ramoplanin promotes overgrowth of gram-negative bacilli in patients.
Because ramoplanin effectively suppresses VRE during treatment, this agent could potentially be used to reduce cross-transmission of VRE. For example, ramoplanin treatment of all VRE-colonized patients on high-risk units (including new admissions) could markedly reduce “colonization pressure,” which plays a major role in cross-transmission (3). We have previously shown that patients with high-density stool colonization (≥4 logs) are significantly more likely to contaminate the environment with VRE than those with lower density colonization (5), suggesting that suppression of VRE may effectively lower “colonization pressure” even if small numbers of organisms persist in the intestinal tract. Although the simplified model of cross-transmission that we used (Fig. 4) obviously does not adequately represent clinical settings, our data demonstrate the potential for use of ramoplanin as an infection control measure. Given the risk of recurrence of high-density VRE colonization, ramoplanin treatment might have to be continued for the duration of hospitalization on high-risk units. Because of the risk of recurrence of VRE colonization and possible promotion of overgrowth of gram-negative pathogens, clinical trials are indicated before the use of ramoplanin as an infection control measure can be recommended.
Our study has some limitations. Administration of ramoplanin in drinking water may not reproduce the pharmacokinetics of human dosing. The levels of ramoplanin in the stool of mice were lower than the levels that have been shown to be present in the stool of humans receiving a dose of 400 mg twice daily (∼1,000 to 2,000 μg/g; unpublished data [Genome Therapeutics Corporation, Waltham, Mass.]).
Acknowledgments
This work was supported by an Advanced Career Development Award from the Department of Veterans Affairs to C.J.D.
We thank Genome Therapeutics Corporation for providing ramoplanin.
C.J.D. has served as an advisor for Genome Therapeutics Corporation.
REFERENCES
- 1.Baden, L. R., I. A. Critchley, D. F. Sahm, W. So, M. Gedde, S. Porter, R. C. Moellering, and G. Eliopoulos. 2002. Molecular characterization of vancomycin-resistant enterococci repopulating the gastrointestinal tract following treatment with a novel glycolipodepsipeptide, ramoplanin. J. Clin. Microbiol. 40:1160-1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Berg, R. D. 1980. Inhibition of Escherichia coli translocation from the gastrointestinal tract by normal cecal flora in gnotobiotic or antibiotic-decontaminated mice. Infect. Immun. 29:1073-1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bonten, M. J. M., S. Slaughter, A. W. Ambergen, M. K. Hayden, J. van Voorhis, C. Nathan, and R. A. Weinstein. 1997. The role of “colonization pressure” in the spread of vancomycin-resistant enterococci. An important infection control variable. Arch. Intern. Med. 158:1127-1132. [DOI] [PubMed] [Google Scholar]
- 4.Donskey, C. J., J. A. Hanrahan, R. A. Hutton, and L. B. Rice. 1999. Effect of parenteral antibiotic administration on persistence of vancomycin-resistant Enterococcus faecium in the mouse gastrointestinal tract. J. Infect. Dis. 180:384-390. [DOI] [PubMed] [Google Scholar]
- 5.Donskey, C. J., T. K. Chowdhry, M. T. Hecker, C. K. Hoyen, J. A. Hanrahan, A. M. Hujer, R. A. Hutton-Thomas, C. C. Whalen, R. A. Bonomo, and L. B. Rice. 2000. Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N. Engl. J. Med. 343:1925-1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Donskey, C. J., C. K. Hoyen, S. M. Das, S. Farmer, M. Dery, and R. A. Bonomo. 2001. Effect of oral Bacillus coagulans administration on the density of vancomycin-resistant enterococci in the stool of colonized mice. Lett. Appl. Microbiol. 33:84-88. [DOI] [PubMed] [Google Scholar]
- 7.Donskey, C. J., A. M. Hujer, S. M. Das, N. J. Pultz, R. A. Bonomo, and L. B. Rice. 2003. Use of denaturing gradient gel electrophoresis for analysis of the stool microbiota of hospitalized patients. J. Microbiol. Methods. 1807:1-8. [DOI] [PubMed] [Google Scholar]
- 8.Edlund, C., L. Barkholt, B. Olsson-Liljequist, and C. E. Nord. 1997. Effect of vancomycin on intestinal flora of patients who have previously received antimicrobial therapy. Clin. Infect. Dis. 25:729-732. [DOI] [PubMed] [Google Scholar]
- 9.Freter, R., H. Brickner, J. Fekete, M. M. Vickerman, and K. E. Carey. 1983. Survival and implantation of Escherichia coli in the intestinal tract. Infect. Immun. 39:686-703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hachem, R., and I. Raad. 2002. Failure of oral antimicrobial agents in eradicating gastrointestinal colonization with vancomycin-resistant enterococci. Infect. Control Hosp. Epidemiol. 23:43-44. [DOI] [PubMed] [Google Scholar]
- 11.Hoyen, C. K., N. J. Pultz, D. L. Paterson, D. C. Aron, and C. J. Donskey. 2003. Effect of parenteral antibiotic administration on the establishment of intestinal colonization with extended-spectrum β-lactamase-producing Klebsiella pneumoniae in mice. Antimicrob. Agents Chemother. 47:3610-3612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.O'Hare, M. D., G. Ghosh, D. Felmingham, and R. N. Gruneberg. 1990. In-vitro studies with ramoplanin (MDL 62,198): a novel lipoglycopeptide antimicrobial. J. Antimicrob. Chemother. 25:217-220. [DOI] [PubMed] [Google Scholar]
- 13.Ray, A. J., C. K. Hoyen, S. M. Das, T. F. Taub, E. C. Eckstein, and C. J. Donskey. 2002. Nosocomial transmission of vancomycin-resistant enterococci from surfaces. JAMA. 287:1400-1401. [DOI] [PubMed] [Google Scholar]
- 14.Rolfe, R. D., S. M. Finegold. 1983. Intestinal β-lactamase activity in ampicillin-induced, Clostridium difficile-associate ileocecitis. J. Infect. Dis. 147(2):227-235. [DOI] [PubMed] [Google Scholar]
- 15.Stiefel, U., N. J. Pultz, J. Harmoinen, P. Koski, K. Lindevall, and C. J. Donskey. 2003. Oral administration of recombinant β-lactamase prevents piperacillin-induced overgrowth of nosocomial pathogens in the intestinal tract of mice. J. Infect. Dis. 188:1605-1609. [DOI] [PubMed] [Google Scholar]
- 16.Vollaard, E. J., and H. A. L. Clasener. 1994. Colonization resistance. Antimicrob. Agents Chemother. 38:409-414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weinstein, M. R., H. Dedier, J. Brunton, I. Campbell, and J. M. Conly. 1999. Lack of efficacy of oral bacitracin plus doxycycline for the eradication of stool colonization with vancomycin-resistant Enterococcus faecium. Clin. Infect. Dis. 29:361-366. [DOI] [PubMed] [Google Scholar]
- 18.Whitman, W. S., P. G. Pitsakis, E. DeJesus, A. J. Osborne, M. E. Levison, and C. C. Johnson. 1996. Gastrointestinal tract colonization with vancomycin-resistant Enterococcus faecium in an animal model. Antimicrob. Agents Chemother. 40:1526-1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wong, M. T., C. A. Kauffman, H. C. Standiford, P. Linden, G. Fort, H. J. Fuchs, S. B. Porter, and R. P. Wenzel. 2001. Effective suppression of vancomycin-resistant Enterococcus species in asymptomatic gastrointestinal carriers by a novel glycolipodepsipeptide, ramoplanin. Clin. Infect. Dis. 33:1476-1482. [DOI] [PubMed] [Google Scholar]