Abstract
Inhalants are distinguished as a class primarily based upon a shared route of administration. Grouping inhalants according to their abuse-related in vivo pharmacological effects using the drug discrimination procedure has the potential to provide a more relevant classification scheme to the research and treatment community. Mice were trained to differentiate the introceptive effects of the trichloroethylene vapor from air using an operant procedure. Trichloroethylene is a chlorinated hydrocarbon solvent once used as an anesthetic as well as in glues and other consumer products. It is now primarily employed as a metal degreaser. We found that the stimulus effects of trichloroethylene were similar to those of other chlorinated hydrocarbon vapors, the aromatic hydrocarbon toluene and the vapor anesthetics methoxyflurane and isoflurane. The stimulus effects of trichloroethylene overlapped with those of the barbiturate methohexital, to a lesser extent the benzodiazepine midazolam and to ethanol. NMDA antagonists, the kappa opioid agonist U50,488 and the mixed 5-HT agonist mCPP largely failed to substitute for trichloroethylene. These data suggest that stimulus effects of chlorinated hydrocarbon vapors are mediated at least partially by GABAA receptor positive modulatory effects.
1. Introduction
The term “inhalant” is widely utilized to arbitrarily combine hundreds of chemicals with known or potential abuse liability into a single family based only on their method of administration [1; 2]. The classification of abused inhalants solely based on route of administration, is otherwise probably minimally helpful to the substance abuse research and treatment community. In fact it may even be detrimental to advancing our scientific understanding of the abuse-related effects of these compounds, as grouping around a common means of administration suggests a homogeneity of mechanism and behavioral effects that may be scientifically unwarranted. As an illustration of the inadequacy of this simplistic taxonomic approach, consider the fact that if a similar system were applied to other common drugs of abuse tobacco, crack cocaine and marijuana would be treated as a single category. Instead other classes of abused drugs such as psychomotor stimulants, opioids and benzodiazepines are classified in a more rationale fashion according to their abuse-related pharmacological and molecular mechanism actions. Unfortunately the experimental data necessary to group inhalants according to pharmacological mechanism are not yet adequate to this purpose, forcing reliance on less meaningful sub classification by chemical type, form or product usage [1].
Without question it would be advantageous to develop a pharmacological taxonomy for inhalants more akin to that applied to other classes of abused drugs. Such a framework would permit direction of research efforts toward thoroughly examining prototypic members of pharmacologically-related inhalant families with some confidence that the findings generated would generalize to other compounds within that family. It should also permit development of more targeted therapeutic approaches for treating addictions to related inhalants. Unfortunately the difficulties inherent in developing a meaningful abuse-related pharmacological classification system for inhalants are not inconsequential. Firstly, there are dozens if not hundreds of inhalant chemicals with potential abuse liability in common household products. While this would seem a daunting challenge, inhalant abusers gravitate toward use of products containing a more limited number of major constituents, considerably narrowing the number of potential candidates warranting detailed examination [3; 4]. A second problem is the technical difficulty associated with conducting abuse-related studies with volatile and gaseous compounds that may deter some laboratories. Although some adjustments to techniques, unique equipment and environmental safeguards are required, the detailed methods necessary to address these impediments in in vitro, ex vivo and in vivo experimental paradigms are freely available in published manuscripts [5-9]. The final problem is perhaps the most difficult. Specifically the most consistent finding from in vitro experiments is that inhalants including volatile hydrocarbons, volatile anesthetics and gases such as nitrous oxide all act either directly or indirectly upon multiple targets including ligand-gated ion channel receptors, G-protein coupled receptors and voltage-gated ion channels [10; 11]. Disentangling which of these diverse actions are responsible for the abuse-related behavioral effects of inhalants and which are of lesser importance is critical to establishing a rational pharmacologically based classification system with relevance to the drug abuse research community.
Development and validation of an inhalant categorization system based on pharmacological action require adequate experimental data. Historically the vast majority of abuse-related inhalant research has focused on volatile solvents due to their high abuse liability and toxicity [2]. There are many chemical classes within the umbrella of volatile solvents but only the aromatic and chlorinated hydrocarbons have received meaningful scientific attention aimed at probing their CNS mechanisms. The aromatic hydrocarbon toluene is arguably the most frequently abused of all volatile inhalants and is also the best characterized at the molecular and behavioral level. Toluene enhances recombinant γ-aminobutyric acid type A (GABAA), glycine and serotonin type 3 (5-HT3) receptor activity while attenuating N-methyl-D-aspartate (NMDA), AMPA and nicotinic acetylcholine receptor function [6; 12-19]. Lagging considerably behind toluene in published reports are the chlorinated hydrocarbons exemplified by 1,1,1-trichloroethane, tetrachloroethylene (perchloroethylene) and trichloroethylene. Like toluene, 1,1,1-trichloroethane and trichloroethylene positively modulate GABAA and glycine receptors expressed in Xenopus oocytes [14; 15; 20], enhance GABA-mediated synaptic activity in neurons [21] and alter voltage-sensitive calcium channel function [22]. 1,1,1-trichloroethane also attenuates NMDA-induced currents [17] and facilitates 5-HT3 receptor function in Xenopus oocytes [16]. Perchloroethylene inhibits nicotinic acetylcholine receptors expressed in oocytes [23]. Finally, acute exposure to 1,1,1-trichloroethane decreases DAMGO binding to mu-opioid receptors in discrete brain regions [24].
Some, all or few of these molecular actions may be critical to the abuse-related behavioral effects of aromatic and chlorinated hydrocarbons. Our laboratory has been engaged over the past decade in addressing the mechanism or mechanisms relevant to abuse-related behavioral endpoints. Our primary tool for this purpose is the drug discrimination procedure in mice. Drug discrimination involves training subjects to distinguish the interoceptive state resulting from an experimenter-administered psychoactive drug versus administration of vehicle in order to choose an appropriately reinforced behavioral response [25]. The most common variant in non-human subjects involves pairing reinforcer presentation with responding on one manipulandum, generally a lever, following drug administration and pairing reinforcer presentation with responding on a second lever following vehicle administration. Over successive daily training sessions alternating between drug and vehicle administration, subjects will learn to respond only on the lever that corresponds with the prior drug or vehicle administration. A critical feature of the drug discrimination paradigm is that the interoceptive stimulus effects of drugs are both receptor mediated and often highly specific [26; 27]. In most cases only drugs that produce interoceptive stimulus effects similar to the training drug will elicit responding on the training-drug appropriate lever [28; 29]. In contrast, drugs that produce no CNS effects, as well as drugs that produce CNS effects that are dissimilar to the training drug, elicit vehicle-lever responding [30; 31]. Indeed it is possible using drug discrimination to make surprisingly subtle distinctions such as differentiating between compounds that have the same net effect on receptor function but act upon different binding sites on the receptor [32; 33] or between receptors that differ only in the presence or absence of a particular subunit [34]. Importantly, from a standpoint of clinical relevancy the stimulus effects of drugs in animals are a model of and are highly correlated with the subjective intoxication produced by drugs in humans [35]. These aspects of drug discrimination as well as the relatively high throughput and excellent replicability of the procedure make it an extremely valuable method for pharmacologically classifying inhalants using an abuse-relevant and neurochemically-selective behavioral endpoint.
Our published drug discrimination data with inhalants thus far indicates that the list of receptor systems relevant to the discriminative stimulus effects of volatile solvents is much more restricted than their in vitro activity might suggest. We have previously demonstrated that the discriminative stimulus effects of 1,1,1-trichloroethane are mediated at least partially by classical benzodiazepine and barbiturate-like positive GABAA receptor modulation but appear to not be dependent upon NMDA antagonist activity, nicotinic acetylcholine receptor modulation or mu opioid agonist effects [36; 37]. Likewise our data show that GABAA modulators substitute in mice trained to discriminate toluene although interestingly, unlike 1,1,1-trichlorothane, the stimulus effects of toluene are mimicked more completely by classical benzodiazepines than barbiturates [38]. These results suggest that while there are substantial commonalities between the stimulus effects of aromatic and chlorinated hydrocarbons, potentially important pharmacological differences do exist. The question posed in the present study was whether these pharmacological properties are consistent across other inhalants from the same chemical class. To address this experimentally we trained the chlorinated hydrocarbon, trichloroethylene, as a discriminative stimulus in mice. Trichloroethylene was introduced as a vapor general anesthetic in the 1950’s as a replacement for diethyl ether [39]. Trichloroethylene was itself largely replaced by halothane in the mid 1950’s except for analgesia in childbirth for which it continued to be used into the late 1970’s in the United Kingdom [40]. Trichloroethylene has a documented history of abuse and although once present in many consumer products such as typewriter correction fluid it is today used primarily as a commercial metal degreaser [41-44]
Following initial discrimination training we then compared the stimulus effects of trichloroethylene to other inhalants as well as probed the neurotransmitter systems underlying the stimulus effects of trichloroethylene. It was our hypothesis that the neurochemical effects of trichloroethylene would be more closely aligned with those produced by the chemically related chlorinated hydrocarbon 1,1,1-trichloroethane than those produced by the less chemically similar aromatic hydrocarbon toluene.
2. Materials and methods
2.1. Subjects
Fourteen adult male B6SJLF1/J mice (Jackson Laboratory, Bar Harbor, Maine, USA) served as subjects. We have used this strain for a number of prior inhalant discrimination studies [36; 37; 45-47]. The mice were individually housed under a 12 hr light/dark cycle (lights on 6 AM). Feeding was adjusted to maintain a healthy, stable weight of between 27-35g for the duration of the study. Studies were approved by the Institutional Animal Care and Use Committee of Virginia Commonwealth University and conducted in accordance with the Institute of Laboratory Animal Research “Guide for the Care and Use of Laboratory Animals”.
2.2. Compounds
HPLC grade trichloroethylene, tetrachloroethylene (perchloroethylene), toluene and 1,1,1-trichloroethane were purchased from Sigma-Aldrich Chemicals (Milwaukee, WI, USA). Isoflurane was purchased from Webster Veterinary Supply (Charlotte, NC, USA). Methoxyflurane (Metofane) was obtained from Pitman-Moore (Mundelein, IL, USA). Midazolam HCl was obtained in a generic injectable form from the VCU hospital pharmacy (Hospira, Lake Forrest, IL, USA). Methohexital sodium was purchased as a commercial available lyphophilized powder (Brevital, JHP Pharmaceuticals, Parasippany, NJ, USA). Dizocilpine maleate (+MK-801), cis-4-[Phosphomethyl]-piperidine-2-carboxylic acid (CGS-19755) and 1-(3-Chlorophenyl)piperazine (mCPP) were purchased from Tocris Bioscience (St. Louis, MO, USA). U50,488H was provided by the National Institute on Drug Abuse Drug Supply Program (RTI International, Research Triangle Park, NC, USA). Unless otherwise indicated injected test drugs were dissolved in 0.9% physiological saline. Exposures to all vapors prior to the discrimination test session were 10 min in duration. All injected drugs were administered intraperitoneally (i.p.) at a volume of 10 ml/kg. A 5-min pretreatment time was used for methohexital. A 10-min pretreatment time was used for MK-801, midazolam and ethanol. A 30-min pretreatment interval was used for CGS-19755, U50,488 and mCPP.
2.3. Apparatus
Training and test vapor exposures were conducted in sealed 27-liter chromatography jars. An internal mixing fan rapidly volatilized liquid inhalants introduced into the chamber. The volume of each volatile liquid necessary to generate the appropriate chamber vapor concentrations were calculated using the ideal gas law as derived for vapors at standard laboratory pressure and temperature [48]. Accuracy of calculated vapor concentrations and chamber atmosphere concentration stability were empirically verified periodically using a Miran 1A single wavelength infrared spectrometer. The static exposure chambers and procedures for generating test vapors have been described previously in greater detail [49; 50].
Discrimination training and test sessions were conducted in standard 2-lever mouse operant conditioning chambers housed in sound-attenuating cubicles (Med-Associates, St. Albans, VT, USA).. Unlike injected drugs, the stimulus effects as well as the measurable blood concentrations of some volatile compounds may be only a few minutes in duration following brief vapor exposures [37]. Therefore the operant chambers were located directly adjacent to the vapor exposure apparatus to permit rapid transfer of the mice from the exposure to the test environment. Each operant chamber was equipped with two optical levers with a LED stimulus light above each lever, a houselight in the rear chamber wall and a 0.01 ml electrically-operated liquid dipper. Mice were reinforced with a milk solution consisting by volume of 25% sugar, 25% nonfat powdered milk and 50% tap water.
2.4. Discrimination training and substitution testing
Training sessions were conducted Mon-Fri. Both lever lights and the houselight were illuminated for the duration of the session. Completion of the FR requirement on the active lever resulted in 3 sec of dipper access. Responses while the dipper was elevated had no consequences. Over successive days the session length was decreased from 30 min to 5 min after which double alternation trichloroethylene vapor versus air discrimination training sessions began. During training the correct lever was determined by whether the subject received a 10-min exposure to 6000 ppm trichloroethylene vapor or to air. Following the 10-min exposure the mice were rapidly removed from the chromatography jar, placed into the operant chamber and the training session immediately initiated. During training the FR1 response requirement was increased over successive sessions to FR12. Responding on the inactive lever reset the FR requirement on the correct lever. A mouse were determined to have acquired the 6000 ppm trichloroethylene vapor versus air discrimination when it emitted its first FR on the correct lever in 8 of 10 consecutive training sessions at FR12.
Following acquisition, substitution tests were conducted on Tues and Fri with continued training sessions on Mon, Wed and Thurs. Testing was suspended if an animal did not maintain accurate stimulus control during intervening training sessions as exhibited by incorrect first fixed ratio responding on the intervening training sessions. Test sessions were not recommenced until the first FR was emitted on the correct lever for three consecutive training sessions. Substitution tests with vapors were preceded by exposure to a single concentration of the test vapor. In substitution test sessions with an injected drug, the injection was given at the pretreatment time indicated previously and the animal was exposed to air in the exposure chamber for 10 min prior to the start of the test session. Drug discrimination test sessions were 5 min and completion of the FR requirement on either lever resulted in dipper presentation. Doses or concentrations of each compound were generally tested in ascending order until maximal substitution was produced or response rates were suppressed compared to the air control session. Prior to each vapor concentration-effect curve, air and 6000 ppm trichloroethylene control test sessions were conducted. Control sessions prior to injected drug dose-effect curves were conducted in a similar manner with the addition of injection of the test drug’s vehicle before trichloroethylene or air exposure. From 8 to 12 mice were used to generate each dose or concentration-effect curve.
2.5. Drug discrimination data analysis
Percentage toluene-lever responding, response rates (responses/sec) and first fixed ratio (FFR) completed were recorded for each 5-min test session. Group means (± SEM) were calculated for percentage trichloroethylene-lever selection as well as response rate. Any inhalant concentration or injected-drug dose that suppressed response rates to the extent that the animal did not complete at least one FR during the test session resulted in the exclusion of that mouse’s datum from the group lever-selection analysis, although that datum was included in the response rate analysis. Test concentrations/doses in which fewer than three mice emitted a complete FR during the test session were not plotted on the substitution curve. A criterion of 75% or greater mean toluene vapor-appropriate responding was defined as full substitution, between 25% and 75% as partial substitution and less than 25% as no substitution. When possible EC50 or ED50 values (and 95% confidence limits) for trichloroethylene vapor-lever selection and response rate suppression were calculated based on the linear portion of each mean dose-effect curve [51].
3. Results
All 14 mice acquired the 6000 ppm trichloroethylene vapor versus air discrimination. The degree of substitution of trichloroethylene for the 10-min, 6000 ppm trichloroethylene vapor training condition was a function of exposure duration (Figure 1, upper panel). It required a minimum of 7 min of 6000 ppm trichloroethylene vapor exposure to elicit full substitution for the training exposure duration of 10 min. The stimulus effect of 10 min of 6000 ppm trichloroethylene diminished in a time-dependent fashion following the cessation of vapor exposure (Figure 1, lower panel). It required 60 min following the cessation of vapor exposure before trichloroethylene lever selection returned to vehicle-appropriate levels.
Fig. 1.
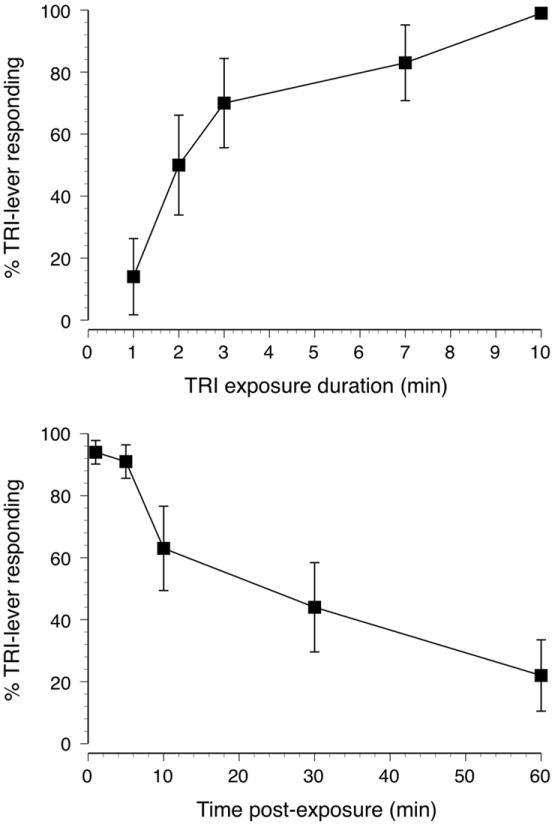
Upper panel shows percentage trichloroethylene-lever selection (±SEM) for tests sessions conducted after exposure to 1, 2, 3, 7 or 10 min of 6000 ppm trichloroethylene vapor. Lower panel shows trichloroethylene-lever selection (±SEM) resulting from delaying the start of the test session for 0, 3, 10, 30 or 60 min after cessation of exposure to 10 min of 6000 ppm trichloroethylene vapor.
The substitution profiles of three chlorinated hydrocarbon vapors are shown in figure 2. Trichloroethylene vapor concentration-dependently substituted for itself. Full substitution was engendered by exposure to 10 min of both 6000 and 8000 ppm trichloroethylene vapor. The EC50 for substitution by trichloroethylene was 2286 ppm [CL: 1847-2829 ppm]. Tetrachloroethylene also concentration-dependently and fully substituted for trichloroethylene with an EC50 of 1855 ppm [CL:1525-2256 ppm]. Lastly, 1,1,1-trichloroethane fully substituted for trichloroethylene with an EC50 of 6561 ppm [CL:4938-8720 ppm]. The substitution concentration-effect curves for three additional volatile compounds in trichloroethylene-trained mice is shown in figure 3. The aromatic hydrocarbon solvent toluene fully substituted for trichloroethylene with an EC50 of 2759 ppm [CL:2188-3478 ppm]. Likewise the volatile anesthetic agents isoflurane and methoxyflurane both fully substituted for trichloroethylene with EC50’s of 4947 ppm [CL:3162-7739 ppm] and 1734 ppm [CL:1455-2066 ppm], respectively.
Fig. 2.
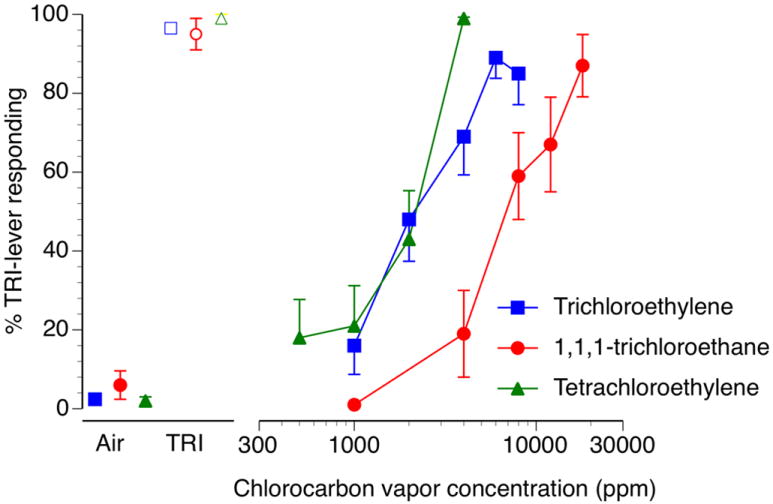
Mean percentage trichloroethylene-lever selection (±SEM) following 10 min of exposure to increasing concentrations of trichloroethylene (blue squares), 1,1,1-trichloroethane (red circles) or tetrachloroethylene (green triangles) in mice trained to discriminate 6000 ppm inhaled trichloroethylene vapor from air. Points above Air (closed symbols) and TRI (open symbols) represent the results of air and 6000 ppm inhaled trichloroethylene exposure control sessions.
Fig. 3.
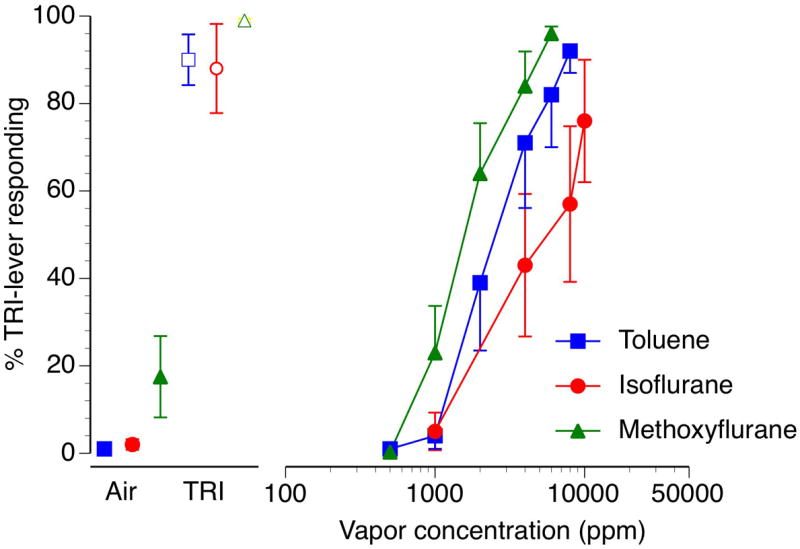
Mean percentage trichloroethylene-lever selection (±SEM) following 10 min of exposure to increasing concentration of toluene (blue squares), isoflurane (red circles) or methoxyflurane (green triangles) in mice trained to discriminate 6000 ppm inhaled trichloroethylene vapor from air. Points above Air (filled symbols) and TRI (open symbols) represent the results of air and 6000 ppm inhaled trichloroethylene exposure control sessions.
The degree of substitution (upper panel) and effects on operant response rates (lower panel) produced by a series of drugs that positively modulate GABAA receptor neurotransmission or attenuate NMDA-receptor activity are shown in figure 4. The classical benzodiazepine midazolam (blue squares) produced a maximum of 48% trichloroethylene-lever selection. Midazolam also suppressed operant response rates in a dose-dependent manner with a 50% reduction in responding resulting from the highest midazolam test dose of 45 mg/kg. The short-acting barbiturate methohexital (red circles) produced dose-dependent substitution for trichloroethylene with an ED50 of 18 mg/kg [CL: 12-27 mg/kg]. A maximum of 70% trichloroethylene -lever selection was engendered at a methohexital dose of 40 mg/kg, but the mean group data may underestimate the degree of substitution as 11 of the 12 mice tested exhibited greater than 75% trichloroethylene-lever selection at one or more methohexital dose (data not shown). Methohexital also dose-dependently suppressed operant responding with an ED50 of 32 mg/kg [CL: 28-38 mg/kg]. The uncompetitive NMDA receptor antagonist channel blocker MK-801 (green triangles) produced a maximum of 45% trichloroethylene-lever responding at a dose of 0.56 mg/kg, which also suppressed operant response rates to the extent that only 4 of 10 mice were capable of emitting one complete fixed ratio value. The competitive NMDA receptor antagonist CGS-19755 (purple diamonds) engendered a maximum of 35% trichloroethylene -lever selection at a dose of 17 mg/kg, which also suppressed operant responding to 17% of vehicle control rates. Lastly, ethanol (black inverted triangles) produced dose-dependent partial substitution for trichloroethylene with an ED50 of 1971 mg/kg [CL: 1559-2493 mg/kg]. A maximum of 67% trichloroethylene -lever responding was produced by a dose of 2500 mg/kg, which also suppressed operant responding to 34% of vehicle control rates.
Fig. 4.
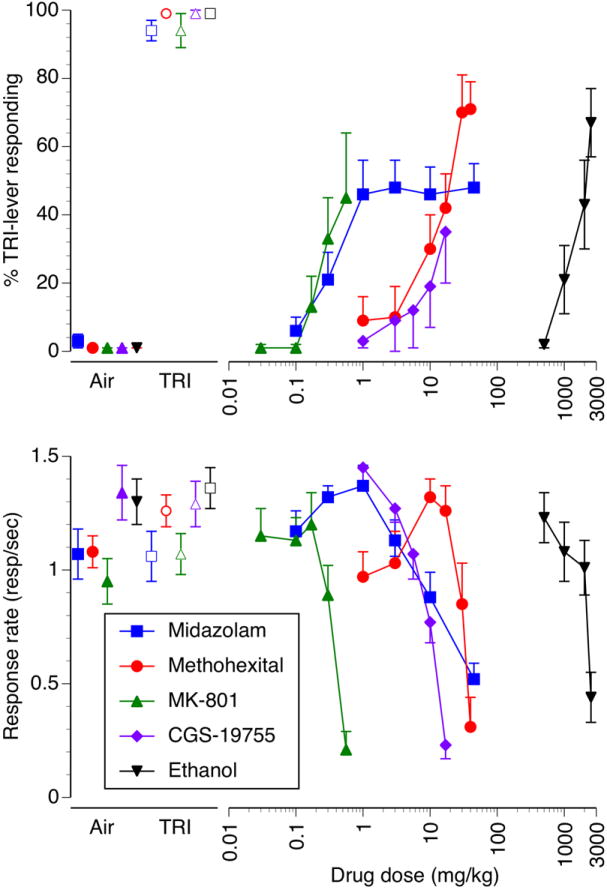
Dose-effect curves for midazolam (blue squares), methohexital (red circles), [+]MK-801 (green triangles), CGS-19755 (purple diamonds) and ethanol (black inverted triangles) in mice trained to discriminate 6000 ppm inhaled trichloroethylene vapor from air. Points above Air (filled symbols) and TRI (open symbols) represent the results of air and 6000 ppm inhaled trichloroethylene exposure control sessions. Mean (± SEM) percentages trichloroethylene-lever responding are shown in the upper panel, mean (± SEM) response rates in responses/sec are shown in the bottom panel.
Substitution and response-rate data generated following pretreatment with mCPP, U50,488 and L701,324 are presented in Table 1. mCPP has affinity for a number different serotonergic and non-serotonergic receptors [52]. mCPP also has ethanol-like discriminative stimulus effects [53] that are likely mediated by 5-HT2C receptor agonism [54-56]. mCPP failed to produce trichloroethylene-like stimulus effects up to a dose of 5.6 mg/kg, which also suppressed mean operant responding to 36% of vehicle control levels and completely suppressed responding in 4 of 8 subjects. The kappa opioid agonist U50,488 also failed to produce trichloroethylene -like discriminative stimulus effects up to a dose of 10 mg/kg, which suppressed responding to 8% of vehicle control rates and abolished all responding in 8 of 10 mice. The NMDA receptor glycine-site antagonist L701,324 did not to substitute for trichloroethylene up to a dose of 10 mg/kg but also failed to suppress operant response rates compared to the vehicle control.
Table 1.
Trichloroethylene lever selection and operant response rates following pretreatment with mCPP (n=8), U50,488 (n=10) or L701,324 (n=8). Numerals in brackets [ ] indicate number of animals out of the group emitting sufficient responses to be included in % trichloroethylene-lever responding determination.
| Drug | Test drug dose | % toluene-lever responding (±SEM) | Response rate in responses/sec (±SEM) |
|---|---|---|---|
| mCPP | Air + vehicle | 1 (1) | 1.1 (0.1) |
| 6000 ppm TRI | 99 (1) | 1.4 (0.1) | |
| 0.1 mg/kg | 0 (0) | 1.3 (0.1) | |
| 1 mg/kg | 0 (0) | 0.9 (0.1) | |
| 3 mg/kg | 0 (0) [4/8] | 0.4 (0.2) | |
| 5.6 mg/kg | 3 (2) [4/8] | 0.4 (0.2) | |
| U50,488 | Air + vehicle | 2 (1) | 1.2 (0.1) |
| 6000 ppm TRI | 99 (1) | 1.2 (0.1) | |
| 1 mg/kg | 2 (1) | 1.3 (0.1) | |
| 3 mg/kg | 7 (6) | 1 (0.1) | |
| 5.6 mg/kg | 22 (22) [4/10] | 0.3 (0.1) | |
| 10 mg/kg | 5 [2/10] | 0.1 (0.1) | |
| L701,324 | Air + vehicle | 1 (1) | 1.3 (0.1) |
| 6000 ppm TRI | 99 (1) | 1.4 (0.1) | |
| 1 mg/kg | 1 (1) | 1.3 (0.1) | |
| 3 mg/kg | 1 (1) | 1.1 (0.1) | |
| 10 mg/kg | 0 (0) | 1.3 (0.1) |
4. Discussion
As we have previously demonstrated with the vapor inhalants toluene[47; 49], 1,1,1-trichloroethane [36; 37] and isoflurane [46], in the present study a 6000 ppm trichloroethylene vapor versus air discrimination could be readily established in mice.The kinetics of the onset and offset of stimulus effects resulting from trichloroethylene administration differed from those of the previous inhalants we have examined. Specifically, the rate of decay of trichloroethylene’s discriminative stimulus was quite slow. Even following the short training exposure duration of 10 min, it required between 30 and 60 min before trichloroethylene-lever responding returned to near that of the air control (Figure 1, bottom panel). This is compared to 10-20 min to reach a similar level of recovery in mice trained to discriminate toluene [38] and less than 10 min for 1,1,1-trichloroethane (unpublished observation). Likewise the onset of stimulus effects resulting from trichloroethylene exposure was also slower than that of 1,1,1-trichloroethane [37]. These findings of a slow onset of effects and extended recovery period following the cessation of exposure are in agreement with the clinical observations of trichloroethylene used as an anesthetic in humans [57] and are not surprising given trichloroethylene has centrally active metabolites including chloral hydrate [58].
Each of the three chlorinated hydrocarbons we examined produced full substitution for trichloroethylene (Figure 2). The potency and efficacy of trichloroethylene and tetrachloroethylene for producing trichloroethylene-like stimulus effects were similar. 1,1,1-trichloroethane also demonstrated similar efficacy for producing trichloroethylene-like stimulus effects but was less potent. The aromatic hydrocarbon toluene, as well as the halogenated ether vapor anesthetic methoxyflurane also produced nearly 100% trichloroethylene-lever selection (Figure 3). A second halogenated ether vapor anesthetic isoflurane had lower potency as well as producing somewhat less substitution than did methoxyflurane. Interestingly methoxyflurane although no longer used as an anesthetic is approved for patient-controlled temporary analgesia and emergency analgesia in Australia. In contrast, isoflurane is a poor analgesic [59] suggesting some differences in mechanism which we may also be detecting in our discrimination assay. The present findings are generally in agreement with previous experiments from our laboratory in which the vapor anesthetic halothane produced full substitution in mice trained to discriminate 1,1,1-trichloroethane vapor from air [37] and both 1,1,1-trichloroethane and tetrachloroethylene produced complete substitution in mice trained to discriminate toluene vapor from air [38]. In aggregate, these data demonstrate there is substantial overlap in the discriminative stimulus effects of a number of different classes of volatile inhalants including aromatic and chlorinated hydrocarbons as well as vapor anesthetics.
The chlorinated hydrocarbon 1,1,1-trichloroethane has been shown to attenuate NMDA-induced seizures [60] as well as inhibit NMDA receptors expressed in Xenopus oocytes [17]. In the present study the uncompetitive NMDA receptor antagonist MK-801 and the competitive NMDA antagonist CGS-19755 elicited a maximum of 45% and 35% trichloroethylene-lever responding, respectively (Figure 4). When the individual animal data was examined only 3 of 10 mice showed full substitution at any MK-801 dose and 2 of 8 at any CGS-19755 dose. In addition, appreciable substitution was only produced at doses that substantially reduced operant responding. In the case of the highest dose of MK-801 (0.56 mg/kg) responding was completely suppressed in 6 of 10 mice. The NMDA glycine-site antagonist L701,324 produced no trichloroethylene-lever selection, but also failed to attenuate responding which may indicate an insufficient test dose range although similar doses have demonstrated behavioral activity in other assays. These data might suggest that NMDA receptor antagonism plays some role, albeit limited, in producing the stimulus effects of trichloroethylene. However, low levels of partial cross substitution between NMDA antagonists and other drugs, particularly GABAA agonists have been reported in other studies [61; 62] suggesting that the data may be attributable to non-specific mechanisms. This latter interpretation is strengthened by data from a prior study in our laboratory in which MK-801, CGS-19755 and L701,324 all produced exclusively vehicle-appropriate responding in mice trained to discriminate 1,1,1-trichloroethane vapor from air [36]. In general, the present results suggest that attenuation of NMDA receptor function plays little if any role in the discriminative stimulus effects of chlorinated hydrocarbons.This overarching finding extends to toluene as well where we have demonstrated that competitive, uncompetitive and glycine-site NMDA antagonists do not elicit toluene-like discriminative stimulus effects [38], again despite very strong evidence that toluene inhibits NMDA receptor function in other assays [6; 13; 17; 18; 60; 63; 64]. Interestingly, although NMDA receptor antagonism does not appear to underlie the discriminative stimulus effects of chlorinated or aromatic hydrocarbons it, in addition to GABAA receptor positive modulation, does appear to be involved in transducing the stimulus effects of the volatile anesthetic isoflurane [46].
Of the mechanistically specific drugs tested, the most robust group mean substitution was produced by the GABAA positive modulator barbiturate methohexital. Methohexital produced 70% mean trichloroethylene -lever selection which is below that normally considered to constitute complete substitution in most studies. However, as we have previously demonstrated when examining GABAergic compounds for cross-substitution in toluene-trained mice, in the present study there were also substantial differences in the most effective methohexital cross-substitution dose across individual subjects [38]. These findings suggest that the most sensitive measure of training-drug like discriminative stimulus effects may be the number of individual subjects demonstrating full substitution regardless of specific test dose, rather than the group mean substitution data which obscures individual differences. When examined on an individual animal basis, at least one dose of methohexital fully substituted for trichloroethylene in 11 of the 12 mice tested. In contrast, an almost equal mean trichloroethylene -lever selection was elicited by ethanol, but individually only 6 of 13 mice demonstrated full substitution at one or more ethanol dose. Mean trichloroethylene-lever selection produced by the benzodiazepine midazolam was even less robust and midazolam only fully substituted for trichloroethylene in 4 of 12 mice at any of the tested doses. The general finding that trichloroethylene has positive GABAA receptor modulator-like effects is consistent with prior drug discrimination studies with 1,1,1-trichloroethane [36; 37; 65; 66]. However, the profile of which GABAA positive modulators produce the most robust substitution does not appear to be identical between trichloroethylene and 1,1,1-trichloroethane. In the present study the rank order of similarity to trichloroethylene was barbiturate > ethanol > benzodiazepine. In contrast in 1,1,1-trichloroethane-trained mice, the rank order of similarity was barbiturate = benzodiazepine ≫ ethanol [36; 37]. Intriguingly, the pattern of substitution of these same GABAA positive modulators in toluene vapor trained mice is yet again somewhat different: benzodiazepine ≫ barbiturate = ethanol [[38]and unpublished observations].
In summary it appears that both chlorinated and aromatic hydrocarbons can, based on in vivo discriminative stimulus effects, be classified primarily as positive GABAA modulators. We must note that this classification is tentative as we have yet to fully exhaust the list of candidate targets suggested by the in vitro literature. Our results do not fully confirm our initial hypothesis that chlorinated hydrocarbons will be more mechanistically similar to each other than to a more distantly related aromatic hydrocarbon. Instead it appears that specific mechanistic interactions at the GABAA receptor complex may differ to some extent even within a given volatile chemical group This suggests that pharmacologically categorizing an inhalant based only on the data of a representative member of the same chemical family may be limited to suggesting the receptors affected rather than pinpointing the exact nature of the interactions at those receptors. It is important to point out that our present results regarding receptor activity are specific for discriminative stimulus effects. They may or may not entirely reflect the receptor systems that are responsible for other behavioral effects of chlorinated hydrocarbons such as rewarding effects, sedation, analgesia and anxiolysis. However, data showing that GABAA positive modulation is an important aspect of the subjective stimulus of chlorinated hydrocarbons could prove useful clinically as it suggests that treatment strategies appropriate for barbiturate and benzodiazepine abuse may also be effective in those abusing volatile hydrocarbons.
References
- 1.Balster RL, Cruz SL, Howard MO, Dell CA, Cottler LB. Classification of abused inhalants. Addiction. 2009;104:878–882. doi: 10.1111/j.1360-0443.2008.02494.x. [DOI] [PubMed] [Google Scholar]
- 2.Balster RL. Abuse potential evaluation of inhalants. Drug Alc Depend. 1987;19:7–15. doi: 10.1016/0376-8716(87)90082-2. [DOI] [PubMed] [Google Scholar]
- 3.Bowen SE, Daniel J, Balster RL. Deaths associated with inhalant abuse in Virginia from 1987 to 1996. Drug Alcohol Depend. 1999;53:239–245. doi: 10.1016/s0376-8716(98)00139-2. [DOI] [PubMed] [Google Scholar]
- 4.Howard MO, Balster RL, Cottler LB, Wu LT, Vaughn MG. Inhalant use among incarcerated adolescents in the United States: prevalence, characteristics, and correlates of use. Drug and alcohol dependence. 2008;93:197–209. doi: 10.1016/j.drugalcdep.2007.08.023. [DOI] [PubMed] [Google Scholar]
- 5.Shelton KL, Balster RL. Inhalant Drug Discrimination: Methodology, Literature Review and Future Directions. In: Glennon RA, Young R, editors. Drug Discrimination: Applications to Medicinal Chemistry and Drug Studies. 1. Wiley; 2011. [Google Scholar]
- 6.Cruz SL, Mirshahi T, Thomas B, Balster RL, Woodward JJ. Effects of the abused solvent toluene on recombinant N-methyl-D-aspartate and non-N-methyl-D-aspartate receptors expressed in Xenopus oocytes. J Pharmacol Exp Ther. 1998;286:334–340. [PubMed] [Google Scholar]
- 7.Wood RW, Rees DC, Laties VG. Behavioral effects of toluene are modulated by stimulus control. Toxicology and applied pharmacology. 1983;68:462–472. doi: 10.1016/0041-008x(83)90291-0. [DOI] [PubMed] [Google Scholar]
- 8.Moser VC, Balster RL. The effects of acute and repeated toluene exposure on operant behavior in mice. Neurobehav Toxicol Teratol. 1981;3:471–475. [PubMed] [Google Scholar]
- 9.Tegeris JS, Balster RL. A comparison of the acute behavioral effects of alkylbenzenes using a functional observational battery in mice. Fundam Appl Toxicol. 1994;22:240–250. doi: 10.1006/faat.1994.1028. [DOI] [PubMed] [Google Scholar]
- 10.Balster RL. Neural basis of inhalant abuse. Drug Alcohol Depend. 1998;51:207–214. doi: 10.1016/s0376-8716(98)00078-7. [DOI] [PubMed] [Google Scholar]
- 11.Bowen SE, Batis JC, Paez-Martinez N, Cruz SL. The last decade of solvent research in animal models of abuse: mechanistic and behavioral studies. Neurotoxicol Teratol. 2006;28:636–647. doi: 10.1016/j.ntt.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 12.Bale AS, Smothers CT, Woodward JJ. Inhibition of neuronal nicotinic acetylcholine receptors by the abused solvent, toluene. Br J Pharmacol. 2002;137:375–383. doi: 10.1038/sj.bjp.0704874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bale AS, Tu Y, Carpenter-Hyland EP, Chandler LJ, Woodward JJ. Alterations in glutamatergic and gabaergic ion channel activity in hippocampal neurons following exposure to the abused inhalant toluene. Neuroscience. 2005;130:197–206. doi: 10.1016/j.neuroscience.2004.08.040. [DOI] [PubMed] [Google Scholar]
- 14.Beckstead MJ, Weiner JL, Eger EI, 2, Gong DH, Mihic SJ. Glycine and gamma-aminobutyric acid(A) receptor function is enhanced by inhaled drugs of abuse. Mol Pharmacol. 2000;57:1199–1205. [PubMed] [Google Scholar]
- 15.Beckstead MJ, Phelan R, Mihic SJ. Antagonism of inhalant and volatile anesthetic enhancement of glycine receptor function. J Biol Chem. 2001;276:24959–24964. doi: 10.1074/jbc.M011627200. [DOI] [PubMed] [Google Scholar]
- 16.Lopreato GF, Phelan R, Borghese CM, Beckstead MJ, Mihic SJ. Inhaled drugs of abuse enhance serotonin-3 receptor function. Drug Alcohol Depend. 2003;70:11–15. doi: 10.1016/s0376-8716(02)00330-7. [DOI] [PubMed] [Google Scholar]
- 17.Cruz SL, Balster RL, Woodward JJ. Effects of volatile solvents on recombinant N-methyl-D-aspartate receptors expressed in Xenopus oocytes. Br J Pharmacol. 2000;131:1303–1308. doi: 10.1038/sj.bjp.0703666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Beckley JT, Woodward JJ. The abused inhalant toluene differentially modulates excitatory and inhibitory synaptic transmission in deep-layer neurons of the medial prefrontal cortex. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2011;36:1531–1542. doi: 10.1038/npp.2011.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Beckley JT, Evins CE, Fedarovich H, Gilstrap MJ, Woodward JJ. Medial prefrontal cortex inversely regulates toluene-induced changes in markers of synaptic plasticity of mesolimbic dopamine neurons. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2013;33:804–813. doi: 10.1523/JNEUROSCI.3729-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Beckstead MJ, Phelan R, Trudell JR, Bianchini MJ, Mihic SJ. Anesthetic and ethanol effects on spontaneously opening glycine receptor channels. J Neurochem. 2002;82:1343–1351. doi: 10.1046/j.1471-4159.2002.01086.x. [DOI] [PubMed] [Google Scholar]
- 21.MacIver MB. Abused inhalants enhance GABA-mediated synaptic inhibition. Neuropsychopharmacology. 2009;34:2296–2304. doi: 10.1038/npp.2009.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shafer TJ, Bushnell PJ, Benignus VA, Woodward JJ. Perturbation of voltage-sensitive Ca2+ channel function by volatile organic solvents. J Pharmacol Exp Ther. 2005;315:1109–1118. doi: 10.1124/jpet.105.090027. [DOI] [PubMed] [Google Scholar]
- 23.Bale AS, Meacham CA, Benignus VA, Bushnell PJ, Shafer TJ. Volatile organic compounds inhibit human and rat neuronal nicotinic acetylcholine receptors expressed in Xenopus oocytes. Toxicol Appl Pharmacol. 2005;205:77–88. doi: 10.1016/j.taap.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 24.Paez-Martinez N, Ambrosio E, Garcia-Lecumberri C, Rocha L, Montoya GL, Cruz SL. Toluene and TCE decrease binding to mu-opioid receptors, but not to benzodiazepine and NMDA receptors in mouse brain. Ann N Y Acad Sci. 2008;1139:390–401. doi: 10.1196/annals.1432.031. [DOI] [PubMed] [Google Scholar]
- 25.Colpaert FC, Niemegeers CJ, Janssen PA. Theoretical and methodological considerations on drug discrimination learning. Psychopharmacologia. 1976;46:169–177. doi: 10.1007/BF00421388. [DOI] [PubMed] [Google Scholar]
- 26.Colpaert FC. Pharmacological specificity of opiate drug discrimination: the effects of training dose and the role of reinforcement in fentanyl-saline discrimination. Psychopharmacology. 1982;76 [Google Scholar]
- 27.Colpaert FC, Slangen JL. Drug Discrimination: Applications in CNS Pharmacology. 1982;448:448. [Google Scholar]
- 28.Glennon RA, Titeler M, Young R. Structure-activity relationships and mechanisms of action of hallucinogenic agents based on drug discrimination and radioligand binding studies. Psychopharmacol Bull. 1986;22:953–958. [PubMed] [Google Scholar]
- 29.Appel JB, Cunningham KA. The use of drug discrimination procedures to characterize hallucinogenic drug actions. Psychopharmacology bulletin. 1986;22:959–967. [PubMed] [Google Scholar]
- 30.Rosecrans JA, Meltzer LT. Central sites and mechanisms of action of nicotine. Neurosci Biobehav Rev. 1981;5:497–501. doi: 10.1016/0149-7634(81)90020-8. [DOI] [PubMed] [Google Scholar]
- 31.Meltzer LT, Rosecrans JA, Aceto MD, Harris LS. Discriminative stimulus properties of the optical isomers of nicotine. Psychopharmacology. 1980;68:283–286. doi: 10.1007/BF00428116. [DOI] [PubMed] [Google Scholar]
- 32.Nicholson KL, Balster RL. Phencyclidine-like discriminative stimulus effects of polyamine modulators of N-methyl-D-aspartate receptor activity in rats. Neurosci Lett. 1998;253:53–56. doi: 10.1016/s0304-3940(98)00606-5. [DOI] [PubMed] [Google Scholar]
- 33.Nicholson KL, Balster RL. The discriminative stimulus effects of N-methyl-D-aspartate glycine-site ligands in NMDA antagonist-trained rats. Psychopharmacology (Berl) 2009;203:441–451. doi: 10.1007/s00213-009-1469-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vinkers CH, Olivier B, Hanania T, Min W, Schreiber R, Hopkins SC, Paterson N, et al. Discriminative stimulus properties of GABAA receptor positive allosteric modulators TPA023, ocinaplon and NG2-73 in rats trained to discriminate chlordiazepoxide or zolpidem. European journal of pharmacology. 2011;668:190–193. doi: 10.1016/j.ejphar.2011.06.054. [DOI] [PubMed] [Google Scholar]
- 35.Schuster CR, Johanson CE. Relationship between the discriminative stimulus properties and subjective effects of drugs. Psychopharmacol Ser. 1988;4:161–175. doi: 10.1007/978-3-642-73223-2_13. [DOI] [PubMed] [Google Scholar]
- 36.Shelton KL, Nicholson KL. GABAA-positive modulator selective discriminative stimulus effects of 1,1,1-trichloroethane vapor. Drug and alcohol dependence. 2012;121:103–109. doi: 10.1016/j.drugalcdep.2011.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shelton KL. Pharmacological characterization of the discriminative stimulus of inhaled 1,1,1-trichloroethane. J Pharmacol Exp Ther. 2010;333:612–620. doi: 10.1124/jpet.109.158949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shelton KL, Nicholson KL. Benzodiazepine-like discriminative stimulus effects of toluene vapor. European journal of pharmacology. 2013;720:131–137. doi: 10.1016/j.ejphar.2013.10.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Galley AH. Trichlorethylene as a general anaesthetic in dental surgery; a report based on 2000 cases. Lancet. 1945;2:597–599. doi: 10.1016/s0140-6736(45)91613-8. [DOI] [PubMed] [Google Scholar]
- 40.Requiem for tichloroethylene? Anaesthesia. 1979;34:903–904. [PubMed] [Google Scholar]
- 41.King GS, Smialek JE, Troutman WG. Sudden death in adolescents resulting from the inhalation of typewriter correction fluid. JAMA : the journal of the American Medical Association. 1985;253:1604–1606. [PubMed] [Google Scholar]
- 42.O’Connor WA. A case of trichlorethylene addiction. British medical journal. 1954;2:451–452. doi: 10.1136/bmj.2.4885.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Derobert L. Death in addictive inhalation of trichloroethylene. Ann Med Leg Criminol Police Sci Toxicol. 1956;36:293–294. [PubMed] [Google Scholar]
- 44.Alapin B. Trichlorethylene addiction and its effects. Br J Addict Alcohol Other Drugs. 1973;68:331–335. doi: 10.1111/j.1360-0443.1973.tb01266.x. [DOI] [PubMed] [Google Scholar]
- 45.Shelton KL. Discriminative stimulus effects of inhaled 1,1,1-trichloroethane in mice: comparison to other hydrocarbon vapors and volatile anesthetics. Psychopharmacology (Berl) 2009;203:431–440. doi: 10.1007/s00213-008-1380-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shelton KL, Nicholson KL. GABA(A) positive modulator and NMDA antagonist-like discriminative stimulus effects of isoflurane vapor in mice. Psychopharmacology (Berl) 2010;212:559–569. doi: 10.1007/s00213-010-1979-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shelton KL, Slavova-Hernandez G. Characterization of an inhaled toluene drug discrimination in mice: effect of exposure conditions and route of administration. Pharmacol Biochem Behav. 2009;92:614–620. doi: 10.1016/j.pbb.2009.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nelson GO. Controlled Test Atmospheres. Principles and Techniques. Ann Arbor: Ann Arbor Press; 1971. [Google Scholar]
- 49.Shelton KL. Inhaled toluene vapor as a discriminative stimulus. Behav Pharmacol. 2007;18:219–229. doi: 10.1097/FBP.0b013e328157f460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bowen SE, Balster RL. Effects of inhaled 1,1,1-trichloroethane on locomotor activity in mice. Neurotoxicol Teratol. 1996;18:77–81. doi: 10.1016/0892-0362(95)02024-1. [DOI] [PubMed] [Google Scholar]
- 51.Tallarida RJ, Murray RB. Manual of Pharmacological Calculations with Computer Programs. 2. New York: Springer-Verlag; 1986. [Google Scholar]
- 52.Hamik A, Peroutka SJ. 1-(m-chlorophenyl)piperazine (mCPP) interactions with neurotransmitter receptors in the human brain. Biological psychiatry. 1989;25:569–575. doi: 10.1016/0006-3223(89)90217-5. [DOI] [PubMed] [Google Scholar]
- 53.Grant KA, Colombo G, Gatto GJ. Characterization of the ethanol-like discriminative stimulus effects of 5-HT receptor agonists as a function of ethanol training dose. Psychopharmacology (Berl) 1997;133:133–141. doi: 10.1007/s002130050383. [DOI] [PubMed] [Google Scholar]
- 54.Fiorella D, Helsley S, Rabin RA, Winter JC. 5-HT2C receptor-mediated phosphoinositide turnover and the stimulus effects of m-chlorophenylpiperazne. Psychopharmacology. 1995;122:237–243. doi: 10.1007/BF02246545. [DOI] [PubMed] [Google Scholar]
- 55.Fiorella D, Rabin RA, Winter JC. The role of the 5-HT2A and 5-HT2C receptors in the stimulus effects of m-chlorophenylpiperazine. Psychopharmacology. 1995;119:222–230. doi: 10.1007/BF02246164. [DOI] [PubMed] [Google Scholar]
- 56.Callahan PM, Cunningham KA. Involvement of 5-HT2C receptors in mediating the discriminative stimulus properties of m-chlorophenylpiperazine (mCPP) Eur J Pharmacol. 1994;257:27–38. doi: 10.1016/0014-2999(94)90690-4. [DOI] [PubMed] [Google Scholar]
- 57.Totonidis S. A role for trichloroethylene in developing nation anaesthesia. Kathmandu Univ Med J (KUMJ) 2005;3:181–190. [PubMed] [Google Scholar]
- 58.Lash LH, Fisher JW, Lipscomb JC, Parker JC. Metabolism of trichloroethylene. Environmental health perspectives. 2000;108(Suppl 2):177–200. doi: 10.1289/ehp.00108s2177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Petersen-Felix S, Arendt-Nielsen L, Bak P, Roth D, Fischer M, Bjerring P, Zbinden AM. Analgesic effect in humans of subanaesthetic isoflurane concentrations evaluated by experimentally induced pain. British journal of anaesthesia. 1995;75:55–60. doi: 10.1093/bja/75.1.55. [DOI] [PubMed] [Google Scholar]
- 60.Cruz SL, Gauthereau MY, Camacho-Munoz C, Lopez-Rubalcava C, Balster RL. Effects of inhaled toluene and 1,1,1-trichloroethane on seizures and death produced by N-methyl-D-aspartic acid in mice. Behav Brain Res. 2003;140:195–202. doi: 10.1016/s0166-4328(02)00323-6. [DOI] [PubMed] [Google Scholar]
- 61.Shelton KL, Balster RL. Effects of abused inhalants and GABA-positive modulators in dizocilpine discriminating inbred mice. Pharmacol Biochem Behav. 2004;79:219–228. doi: 10.1016/j.pbb.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 62.Geter-Douglass B, Witkin JM. Dizocilpine-like discriminative stimulus effects of competitive NMDA receptor antagonists in mice. Psychopharmacology. 1997;133:43–50. doi: 10.1007/s002130050369. [DOI] [PubMed] [Google Scholar]
- 63.Bale AS, Jackson MD, Krantz QT, Benignus VA, Bushnell PJ, Shafer TJ, Boyes WK. Evaluating the NMDA-glutamate receptor as a site of action for toluene, in vivo. Toxicol Sci. 2007;98:159–166. doi: 10.1093/toxsci/kfm080. [DOI] [PubMed] [Google Scholar]
- 64.Lo PS, Wu CY, Sue HZ, Chen HH. Acute neurobehavioral effects of toluene: involvement of dopamine and NMDA receptors. Toxicology. 2009;265:34–40. doi: 10.1016/j.tox.2009.09.005. [DOI] [PubMed] [Google Scholar]
- 65.Bowen SE, Wiley JL, Jones HE, Balster RL. Phencyclidine- and diazepam-like discriminative stimulus effects of inhalants in mice. Exp Clin Psychopharmacol. 1999;7:28–37. doi: 10.1037//1064-1297.7.1.28. [DOI] [PubMed] [Google Scholar]
- 66.Rees DC, Knisely JS, Balster RL, Jordan S, Breen TJ. Pentobarbital-like discriminative stimulus properties of halothane, 1,1,1-trichloroethane, isoamyl nitrite, flurothyl and oxazepam in mice. J Pharmacol Exp Ther. 1987;241:507–515. [PubMed] [Google Scholar]


