Abstract
Background
Phytochemicals like carotenoids, tocopherols, ascorbates and phenols present in the plants are strong antioxidants and have an important role in the health care system. There is growing interest in correlating the phytochemical constituents of a plant with its pharmacological activity. Therefore, the present study investigates the content of total phenolics, flavonoids and the antioxidant activity of four different varieties of Lantana camara L. (Verbenaceae) leaves by using in vitro antioxidant models.
Methods
The leaves of Chandigarh purple variety (CPV), Palampur red variety (PRV), Chandigarh yellow turning pink variety (YTPV) and Chandigarh yellow variety (CYV) Lantana camara were collected and the total phenolic, flavonoid content, antioxidant and free radical scavenging activities were determined in their methanolic extracts.
Results
The phenolic content was found to be highest in the CYV extract (232.99 ± 15.97 mg GAE/ g extract). The content of the flavonoids are in the order of YTPV, PRV, CPV and CYV. The IC50 values for the 2, 2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging test were in the order of CYV (33.30 ± 2.39) < PRV (40.32 ± 2.94) < YTPV (475.33 ± 5.20) < CPV (927.16 ± 2.88 μg/mL). The highest total antioxidant capacity was observed in CYV (222.20 ± 5.05 mg AAE/ g). The Ferric ion reducing antioxidant potential (FRAP) value of the extracts were in the order of CYV > PRV > YTPV > CPV. The IC50 values of 2, 2'-azino-bis-(3-ethylbenzothiazoline-6-sulfonate) (ABTS) scavenging assay for CYV, PRV, YTPV, CPV were 18.25 ± 0.19, 18.24 ± 1.82, 50.43 ± 9.49, 52.84 ± 1.82 μg/mL respectively. PRV extract showed the maximum in vitro lipid peroxidation inhibition effect with an IC50 value of 68.50 μg/mL which is even stronger as compared to the standard Rutin (79.69 μg/mL). The extracts showed a strong correlation between the phenolic content and their antioxidant activities. The highest correlation (r = 0.998, R2 = 0.997) was found between total phenolic content and ABTS scavenging assay.
Conclusion
Among the four varieties investigated, CYV and PRV extracts showed strong antioxidant activities and may be used as a potential source of natural antioxidant against free radical associated diseases.
Keywords: Lantana camara, Verbenaceae, Antioxidant activity, Total phenolic content, DPPH, ABTS
Background
Free radicals are fundamental to many biochemical processes and represent an essential part of aerobic life and metabolism. Reactive oxygen species (ROS) mediated oxidative damage to macromolecules namely lipids, proteins and DNA have been implicated in the pathogenicity of major diseases such as cancer, rheumatoid arthritis, post ischemic reperfusion injury, degeneration process of aging, myocardial infarction, cardiovascular disease etc. [1]. Antioxidants function through protection mechanisms at several levels within cells in human body by inhibiting the formation of free radical species, intercepting radical-chained reactions, converting existing free radicals into less harmful molecules and repairing oxidative damage. Several synthetic antioxidants, such as butylated hydroxyanisol (BHA), butylated hydroxytoluene (BHT) and tert-butylhydroquinone (TBHQ) are commercially available but some adverse effects have been observed with their continuous use [2].
Fundamental biochemical processes of plants are considered to be the primary metabolic processes that generally follow similar mechanism in the cells of all plants and are necessary for each plant to survive and to reproduce. However, the plants produce a large number of compounds called secondary metabolites with diverse physiological and biochemical activities. Secondary metabolites and their derivatives show significant pharmacological activities such as being hepatoprotective, diuretic, anti allergic, anticancer effects and antioxidant [3–5]. Phytochemicals like carotenoids, tocopherols and tocotrienols, ascorbates and phenols are strong antioxidants and have carved out an important role in the health care system. There is renewed interest in finding out new natural antioxidants from living system for application in food, pharmaceutical and cosmetics.
Lantana camara L. is regarded both as a notorious weed and a popular ornamental garden plant and has found various uses in folk medicine in many parts of the world [6]. Different parts of Lantana plant are considered antiseptic, antispasmodic, carminative and diaphoretic [7]. Anti-inflammatory, antipyretic and analgesic properties of extracts of L. camara leaves have also been reported [8]. Verbascoside isolated from the leaves of L. camara exhibited inhibition of protein kinase C and antitumor activity [9].
The different varieties of L. camara have been reported to contain different types and levels of lantadenes and other phytochemicals [10], and may contain different bioactive constituents among these varieties of this Lantana species.
The present work aimed (i) to conduct a quantitative analyses of the antioxidant activity, total phenolic content and total flavonoids in methanolic extracts of different varieties of Lantana camara leaves (ii) to correlate the total phenolic content with the antioxidant activities as such comparative antioxidant study of these different varieties of L. camara has not been reported.
Methods
Chemicals and reagents
Gallic acid, 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2, 4, 6-tripyridyl-s-triazine (TPTZ) and 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonate) (ABTS) were purchased from Sigma-Aldrich. Ascorbic acid, Folin-Ciocalteu’s reagent, sodium carbonate and silica gel F254 TLC plates were from Merck (Mumbai, India). All the chemicals used including solvents were of analytical grade.
Collection of plant material
The leaves of different varieties were collected from different areas in and around Panjab University campus, Chandigarh and red variety was obtained from I.V.R.I. Palampur surroundings. The samples were authenticated and voucher specimens (Voucher specimen No. 20319 I-IV) deposited at the Herbarium, Department of Botany, Panjab University, Chandigarh. The leaves were washed, dried and grinded to make powders and then they were labelled and stored for further use. The different varieties of Lantana camara collected were Chandigarh purple variety (CPV), Palampur red variety (PRV), Chandigarh yellow turning pink variety (YTPV) and Chandigarh yellow variety (CYV).
Methanolic extract preparation
Lantana leaf powders were extracted with methanol (10% w/v) for 24 hours and filtered through four layers of muslin cloth and then through Whatman No. 1 filter paper. The extracts were then subjected to rotary vaporizer (Equitron, Roteva – 8763 RV) to evaporate methanol. The semi-solid residue obtained was blackish brown in color, henceforth called Lantana camara methanolic extract (LCME). The percentage yield (w/w) obtained for different varieties were CPV (14.22%), PRV (16.82%), YTPV (14.46%) and CYV (15.72%).
Estimation of total phenolic content
The total phenols were determined by method of McDonald et al., [11] with slight modifications. 10 mg of gallic acid monohydrate was dissolved in 100 mL of methanol to give a concentration of 100 μg/mL and used as the standard. Different aliquots of 0.1 to 1.0 mL from the stock solution were taken in 10 graduated tubes. To each tube 2.5 mL of 1:1 mixture of Folin- Ciocalteu reagent and distilled water and 2 mL of 7.5% sodium carbonate were added. The mixture was allowed to stand for 30 minutes and the volume was made with water to get a concentration ranging from 1–10 μg/mL. The absorbance of the resulting solutions was measured at 765 nm against reagent blank. A standard calibration curve was prepared by plotting absorbance against concentration and it was found to be linear over this concentration range. 10 mg of extract was dissolved in 10 mL of methanol to get 1 mg/mL solution The concentration of total phenol in the test sample was determined from the calibration graph. The total phenol content in the extract was expressed in terms of gallic acid equivalent (mg GAE /g extract).
Estimation of total flavonoid content
Aluminium chloride colorimetric technique was used for flavonoids estimation [12]. 10 mg of rutin was dissolved in 10 mL of methanol to get 1000 μg/mL solution and was used as standard. Aliquots ranging from 0.01 to 0.08 mL from the above stock solution were taken in different tubes. To each tube 1.5 mL of methanol, 0.1 mL of 10% aluminium chloride, 0.1 mL of 1 M potassium acetate and 2.8 mL of distilled water was added. The reaction mixture was kept at room temperature for 30 min. The absorbance of the resulting solutions was measured at 415 nm against reagent blank. The calibration curve was prepared by plotting absorbance against concentration and it was found to be linear over this concentration range. 10 mg of extracts were dissolved in 10 mL of methanol to get 1 mg/mL solutions respectively. The concentration of total flavonoid in the test sample was determined from the calibration curve. The total flavonoid content in the extract was expressed as rutin equivalent (mg RE/g extract).
Thin layer chromatography analysis of antioxidant activity
DPPH (2,2-Diphenyl-1-picrylhydrazyl) assay [13] was used as a rapid thin layer chromatography screening method to evaluate the antioxidant activity of the methanolic extracts due to free radical scavenging. DPPH is a purple coloured stable free radical, which on reduction gives yellow coloured diphenyl picryl hydrazine compound. 2.5 μL of different methanolic extracts (1 mg/mL) were loaded on silica gel F254 TLC plates (Merck, Germany) which were sprayed with 0.05% DPPH solution in methanol and examined at 30 minutes after spraying. Any antioxidant compound is seen as a yellow spot on a purple background. Vitamin C and gallic acid were used as positive controls.
DPPH radical scavenging activity
The DPPH free radical scavenging activity of the extract, based on the scavenging of the stable 2, 2-diphenyl-1-picrylhydrazyl (DPPH) free radical was determined [13]. It is a discoloration assay, which is evaluated by the addition of the antioxidant to a DPPH solution in methanol and the ability to scavenge the stable free radical of DPPH was measured in the absorbance at 517 nm.
The solution of DPPH in methanol (200 μM) was freshly prepared. Different concentrations of extracts were added to methanolic solution of DPPH. After 30 min at room temperature in dark, the absorbance was recorded at 517 nm. IC50 value denotes the concentration of sample, which is required to scavenge 50% of DPPH free radicals. Radical scavenging activity was calculated by the following formula
IC50 value was determined from the plotted graph of scavenging activity against the different concentrations of extracts, which is defined as the total antioxidant necessary to decrease the initial DPPH radical concentration by 50%. The measurements were triplicated and their scavenging effects were calculated based on the percentage of DPPH scavenged.
Evaluation of total antioxidant capacity
The assay is based on the reduction of Mo (VI) to Mo (V) by the extract and subsequent formation of a green phosphate/Mo (V) complex at acidic pH [14]. 0.3 ml extract was mixed with 3 ml of reagent solution (0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate). The tubes containing the reaction solutions were incubated at 95°C for 90 min. Then the absorbance of the solution was measured at 695 nm using a spectrophotometer against blank after cooling to room temperature. Methanol (0.3 ml) in place of extract was used as blank. The antioxidant activity was expressed as ascorbic acid equivalent (mg AAE/g extract) which served as a positive control.
Ferric ion reducing antioxidant potential (FRAP) assay
A modified method of Benzie & Strain [15] was adopted for the FRAP assay. The stock solutions prepared were 300 mM acetate buffer (3.1 g CH3COONa and 16 mL CH3COOH), pH 3.6, 10 mM TPTZ (2, 4, 6-tripyridyl-s-triazine) solution in 40 mM HCl, and 20 mM FeCl3 solution. The fresh working solution was prepared by mixing 25 mL acetate buffer, 2.5 mL TPTZ and 2.5 mL FeCl3 solution. The temperature of the solution was raised to 37°C before using. Different concentrations of plant extracts (100 μL) were allowed to react with FRAP solution (2900 μL) for 30 min in the dark condition. Readings of the coloured product (ferrous tripyridyltriazine complex) were taken at 593 nm. The standard curve was linear between 200 to 1000 μM FeSO4. Results are expressed in μM Fe (II)/ g extract and compared with BHT as standard.
ABTS radical scavenging assay
The method of Re et al., [16] with slight modifications was adopted for ABTS (2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonate) assay. Briefly, a stock solution of ABTS radical cation was prepared by dissolving ABTS (7 mM) with potassium persulfate (K2S2O8, 2.45 mM). The mixture was left to stand in the dark at room temperature for 16 h (the time required for formation of the radical) before use. For the evaluation of ABTS radical scavenging activity, the working solution was prepared by the previous solution and diluting it in ethanol to obtain the absorbance of 0.700 ± 0.02 at 734 nm. The solvent extracts (0.1 mL) at different concentrations were mixed with the ABTS working solution (1.9 mL) and the reaction mixture was allowed to stand at 30°C for 6 min, then the absorbance was measured by using a UV-visible spectrophotometer at 734 nm, at which point the antioxidants present in the extracts began to inhibit the radical, producing a reduction in absorbance, with a quantitative relationship between the reduction and the concentration of antioxidants present in the tested sample. The radical scavenging activities of the extracts were compared with that of BHT and percentage inhibition is calculated by equation:
In vitrolipid peroxidation inhibition assay
Rat liver was processed to get 10% homogenate in cold phosphate buffered saline, pH 7.4 using polytron homogenizer and filtered to get a clear homogenate. The degree of lipid peroxidation was assayed by estimating the TBARS by using the method of Ohkawa et al., [17] with slight modifications. Different concentrations of the extracts (20–100 μg/ mL) were added to liver homogenate. Lipid peroxidation was initiated by adding 50 μL of 15 mM ferrous sulphate solution to 0.2 mL of homogenate. After 1 h, 1 mL of 15% TCA followed by 1 mL of 0.67% TBA in 50% acetic acid was added to the tubes and heated in boiling water bath for 30 min. The tubes were centrifuged and the intensity of the pink coloured complex was measured at 532 nm. The control was prepared without fractions and rutin was used as the standard. The experimental procedure was approved by the Institutional animal ethics committee, Panjab University, Chandigarh. Experiments involving animals were conducted in accordance with the guidelines for the use of laboratory animals.
Statistical analysis
Assays were performed in triplicate and results are shown as mean ± standard deviation. Linear regression analysis was used to calculate the IC50 values. Pearson’s correlation coefficient was calculated using Microsoft excel 2007. Statistical significance was determined among various treatments with one way ANOVA test using SPSS 16.0 for Windows. A statistical significance of p < 0.05 was considered to be significant.
Results and discussion
Phenolic compounds are considered to be the most important antioxidants and are widely distributed among various plant species. These phenols play important roles in plants such as protection against herbivores and pathogens, cementing material joining phenolic polymers to cell wall polysaccharides [18], regulation of cell growth and cell division [19]. The phenolic content expressed as gallic acid equivalent per gram (GAE /g) in the CYV extract was found to be highest (232.99 ± 15.97 mg GAE/g) followed by PRV (225.15 ± 12.52 mg GAE/g), YTPV (70.91 ± 4.57 mg GAE/g) and CPV (55.57 ± 2.82 mg GAE/g). Phenolic compounds exhibit their antioxidant activity by several mechanisms such as donating hydrogen atoms to free radicals, scavenging other reactive species such as OH•, NO2•, N2O3, ONOOH and HOCl. Some phenolics, mostly the di and polyphenols, can react with O2•‾ or by binding transition metal ions (especially iron and copper), often resulting in forms poorly active in promoting free radical reactions and hence can also interfere with the uptake of metals from the diet [20, 21]. Phenolic acids such as caffeic acid and rosmarinic acid were reported to be among the abundant compounds in the methanolic extracts of aerial tissues of L. camara [22]. Vamanu and Nita showed that rosmarinic acid, a major phenolic compound, in the methanolic extracts of wild edible Boletus edulis mushroom is an effective antioxidant [23].
Flavonoids are water soluble polyphenolic compounds which are extremely common and wide spread in the plant kingdom as their glycosides. The contents of total flavonoid were measured by aluminium chloride method and are expressed in terms of rutin equivalent (RE) as 16.14 ± 0.21 mg RE/ g for CYV, 18.17 ± 0.92 mg RE/g for CPV, 24.60 ± 2.25 mg RE/ g for PRV, 25.22 ± 2.59 mg RE/g for YTPV (Table 1). The flavonoids act through scavenging or chelating process [24, 25]. Ghisalberti mentioned six flavonoids, such as 3-methoxy-, 3, 7 dimethoxy- and 3, 7, 4′-trimethoxyquercetin, pectolinarigénin 7-O-β-D-glucoside, hispidulin and a camaraside glycoside in the leaves and stems of L. camara [7].
Table 1.
Total phenolic content (mg GAE /g extract) and flavonoid content (mg RE /g extract)
| Sample | Total phenolic content (mg GAE / g) ± SD | Flavonoid content (mg RE/ g) ± SD |
|---|---|---|
| CPV | 55.57 ± 2.82b | 18.17 ± 0.92b |
| PRV | 225.15 ± 12.52a | 24.60 ± 2.25a |
| YTPV | 70.91 ± 4.57b | 25.22 ± 2.59a |
| CYV | 232.99 ± 15.97a | 16.14 ± 0.21c |
Results are expressed as mean ± SD (n = 3). Gallic acid equivalent (GAE) and rutin equivalent (R.E.).
Values in the column followed by a different letter superscript (a-c) are significantly different (p < 0.05) and values having same superscript are not statistically significant.
The result of the qualitative DPPH test on TLC plate (F254) is shown in Figure 1. The DPPH scavenging effects are expressed as yellow zones of the various extracts on the purple background as compared to positive controls ascorbic acid (Vitamin C) and gallic acid and blank TLC plate as negative control.
Figure 1.
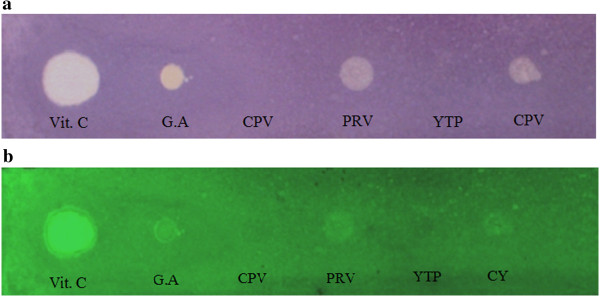
Antioxidant activity by qualitative DPPH test on F 254 TLC plate (Merck). a) in visible light b) in U.V. light at 254 nm. Vit. C (Vitamin C), G.A. (Gallic acid), CPV (Chandigarh Purple), PRV (Palampur red), YTPV (Yellow turning pink), CYV (Chandigarh yellow).
Table 2 shows the IC50 values of leaf extracts for DPPH free radical scavenging activity for CYV (33.30 ± 2.39 μg/ mL), PRV (40.32 ± 2.94 μg/ mL), YTPV (475.33 ± 5.20 μg/ mL), CPV (927.16 ± 2.88 μg/ mL). The lower IC50 value indicates higher antioxidant capacity and hence CYV extract showed significantly higher radical scavenging activity as compared to PRV, YTPV and CPV. The radical scavenging activity is in accordance with the levels of phenolic content in the extracts respectively. Free radical scavenging activity of extracts of black chokeberry and blueberry has been reported to be correlated with its total phenolics content [26].
Table 2.
Effect of methanolic extracts of Lantana camara on different antioxidant models
| Sample | DPPH radical scavenging activity IC 50Value (μg/mL) | Total antioxidant capacity (mg AAE /g) ± SD | FRAP value (mM Fe (II)/g) ± SD |
|---|---|---|---|
| CPV | 927.16 ± 2.88e | 105.70 ± 0.80d | 5.60 ± 0.25d |
| PRV | 40.32 ± 2.94c | 210.30 ± 0.41b | 236.30 ± 6.11c |
| YTPV | 475.33 ± 5.20d | 155.40 ± 2.72c | 17.70 ± 2.95d |
| CYV | 33.30 ± 2.39b | 222.20 ± 5.05a | 339.00 ± 11.13b |
| Vitamin C | 10.73 ± 0.09a | - | - |
| BHT | - | - | 550.00 ± 7.54a |
Results are expressed as mean ± SD (n = 3). Ascorbic acid equivalent (AAE). Values in the column followed by a different letter superscript (a-e) are significantly different (p < 0.05) and values having same superscript are not statistically significant. -, not determined.
Total antioxidant capacity is a better way of depiction of combined effect of phenolics, flavonoids and other reducing compounds in the plant extracts and is expressed in terms of ascorbic acid equivalents (AAE). The phosphomolybdenum method is based on the reduction of Mo (VI) to Mo (V) by the action of antioxidant compounds and the formation of a green phosphate - Mo (V) complex with a maximal absorption at 695 nm. A significantly higher total antioxidant capacity was observed in CYV (222.20 ± 5.05 mg AAE/ g) followed by PRV (210.30 ± 0.41 mg AAE/ g), YTPV (155.40 ± 2.72 mg AAE/ g) and CPV (105.70 ± 0.80 mg AAE/ g) (Table 2). The total antioxidant capacity values follow the same order as that of phenolic content in the extracts respectively.
The ferric ion reducing antioxidant potential (FRAP) of different extracts were estimated from their ability to reduce TPTZ-Fe (III) to TPTZ-Fe (II). The FRAP value of the extracts is in the order of BHT (550.00 ± 7.54 mM Fe (II)/g) > CYV (339.00 ± 11.13 mM Fe (II)/g) > PRV (236.30 ± 6.11 mM Fe (II)/g), YTPV (17.70 ± 2.95 mM Fe (II)/g) followed by CPV (5.60 ± 0.25 mM Fe (II)/g) (Table 2). The differences in the values are found to be significant and are in order with their phenolic content.
Protonated radical scavenging is an important attribute of antioxidants. ABTS, a protonated radical, has characteristic absorbance maxima at 734 nm which decreases with the scavenging of the proton radicals [27]. The extracts were fast and effective scavengers of the ABTS cation radical (Figure 2). The IC50 values for PRV (18.24 ± 1.82 μg/mL), CYV (18.25 ± 0.19 μg/mL), YTPV (50.43 ± 9.49 μg/mL) and CPV (52.84 ± 1.82 μg/mL) are given in Table 3.
Figure 2.
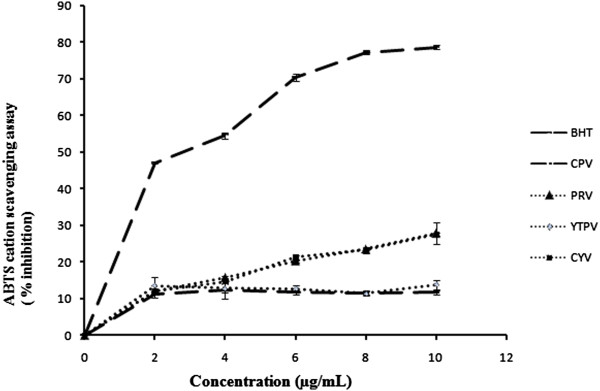
ABTS radical cation scavenging activity.
Table 3.
Effect of methanolic extract of CPV, PRV, YTPV and CYV compared with BHT as standard on ABTS radical scavenging assay
| Conc. (μg/mL) | [IC 50values are mean ± SD of 3 replicates] Inhibition (%) | ||||
|---|---|---|---|---|---|
| BHT | CPV | PRV | YTPV | CYV | |
| 2 | 47.15 ± 0.32 | 11.13 ± 0.45 | 12.05 ± 0.31 | 13.60 ± 2.32 | 12.10 ± 0.73 |
| 4 | 54.61 ± 0.85 | 12.29 ± 0.59 | 15.74 ± 0.58 | 12.82 ± 2.84 | 14.67 ± 0.59 |
| 6 | 70.55 ± 0.95 | 11.66 ± 0.39 | 20.26 ± 0.38 | 12.68 ± 0.91 | 21.28 ± 0.52 |
| 8 | 77.30 ± 0.51 | 11.56 ± 0.17 | 23.47 ± 0.25 | 11.51 ± 0.52 | 23.17 ± 0.77 |
| 10 | 78.71 ± 0.53 | 11.66 ± 0.25 | 27.78 ± 2.92 | 13.79 ± 1.38 | 27.50 ± 0.44 |
| IC 50 (μg/mL) | 4.34 ± 0.03 a | 52.84 ± 1.82 c | 18.24 ± 1.17 b | 50.43 ± 9.49 c | 18.25 ± 0.19 b |
Values in the row followed by a different letter superscript (a-c) are significantly different (p < 0.05) and values having same superscript are not statistically significant.
Lipid peroxidation is the oxidative degradation of polyunsaturated fatty acids and involves the formation of lipid radicals leading to membrane damage. Free radicals induce lipid peroxidation in polyunsaturated lipid rich areas like brain and liver [28] and can also damage DNA, proteins and other biological molecules. Initiation of lipid peroxidation by ferrous sulphate takes place either through ferryl-perferryl complex [29] or through hydroxyl radical by Fenton reaction [30] depending upon the reaction conditions. The extracts showed the in vitro lipid peroxidation inhibition effects in a concentration dependent manner and the results are expressed in terms of percentage inhibition (Figure 3) and IC50 values for the same are calculated from the regression equation respectively (Table 4). PRV extract showed the significantly maximum inhibitory effect with lowest IC50 value of 68.51 ± 0.48 μg/ mL, CYV (89.77 ± 0.72 μg/ mL), YTPV (127.59 ± 3.03 μg/ mL), CPV (242.78 ± 6.96 μg/ mL) as compared to the standard rutin which was showing the inhibitory effect with an IC50 value of 79.68 ± 0.66 μg/ mL. Here, PRV showed higher inhibitory effect than rutin (1 mg/ mL) used as a standard. Lipid peroxidation inhibiting activity of Lantana extracts may be due to the compounds such as rosmarinic acid, caffeic acid etc. Caffeic acid (3,4-dihydroxycinnamic acid) has been shown to prevent the lipid peroxidation of food induced by the free radicals and also in pathological conditions, such as cancer and ageing [31]. Aqueous extract of Lantana camara has been reported to inhibit pBR322 plasmid DNA damage induced by free radicals [32] indicating the presence of compounds with strong antioxidant properties.
Figure 3.
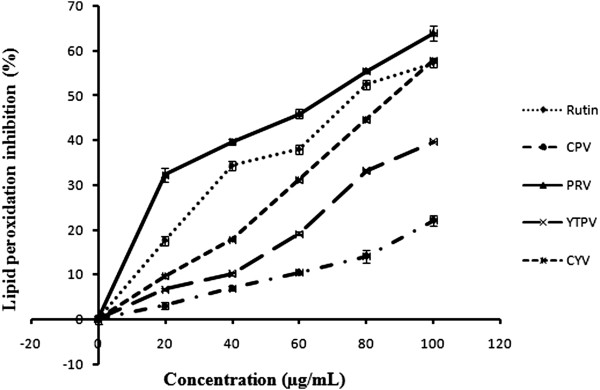
In vitro lipid peroxidation inhibition activity.
Table 4.
Effect of methanolic extract of CPV, PRV, YTPV and CYV compared with Rutin as standard on in vitro lipid peroxidation inhibition assay in rat liver
| Conc. (μg/mL) | [IC 50values are mean ± SD of 3 replicates] Inhibition (%) | ||||
|---|---|---|---|---|---|
| Rutin | CPV | PRV | YTPV | CYV | |
| 20 | 17.62 ± 1.05 | 3.09 ± 0.67 | 32.24 ± 1.56 | 6.76 ± 0.27 | 9.66 ± 0.45 |
| 40 | 34.33 ± 1.00 | 6.99 ± 0.51 | 39.67 ± 0.59 | 10.19 ± 0.34 | 17.8 ± 0.26 |
| 60 | 38.00 ± 1.00 | 10.43 ± 0.38 | 45.91 ± 1.00 | 19.18 ± 0.44 | 31.14 ± 0.36 |
| 80 | 52.47 ± 0.91 | 14.14 ± 1.40 | 55.48 ± 0.50 | 33.23 ± 0.35 | 44.62 ± 0.45 |
| 100 | 57.24 ± 1.03 | 22.14 ± 1.17 | 63.95 ± 1.69 | 39.76 ± 0.09 | 57.86 ± 0.35 |
| IC 50 (μg/mL) | 79.68 ± 0.66 b | 242.78 ± 6.96 e | 68.51 ± 0.48 a | 127.58 ± 3.03 d | 89.77 ± 0.72 c |
Values in the row followed by a different letter superscript (a-e) are significantly different (p < 0.05) and values having same superscript are not statistically significant.
The correlation of total phenolic content with DPPH, FRAP and ABTS scavenging activities are shown in Figure 4(a), (b) and (c) respectively. The Pearson’s correlation coefficient (r) and coefficient of determination (R2) was highest (r = 0.998, R2 = 0.997) between total phenolic content and ABTS activity than that of total phenolic content and DPPH activity (r = 0.994, R2 = 0.988) followed by total phenolic content and FRAP activity (r = 0.974, R2 = 0.949).
Figure 4.
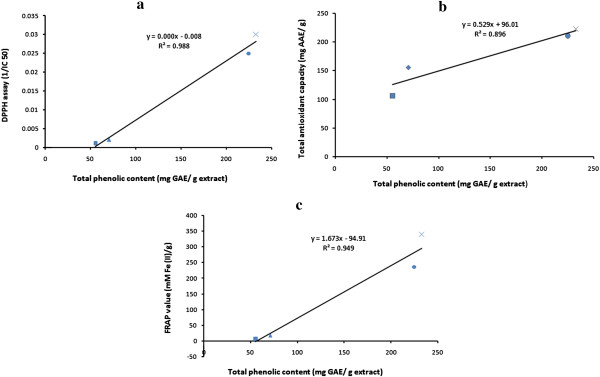
Correlation between total phenolic content and (a) DPPH radical scavenging activity of the extracts. Correlation coefficient, r = 0.994 and coefficient of determination, R2 = 0.988. (b) Total antioxidant capacity of the extracts. Correlation coefficient, r = 0.946 and coefficient of determination, R2 = 0.896. (c) Ferric ion reducing antioxidant potential (FRAP) of the extracts. Correlation coefficient, r = 0.974 and coefficient of determination, R2 = 0.949.
The correlation between total phenolic content with total antioxidant capacity and in vitro lipid peroxidation inhibition activity is shown in Figure 5 (a) and (b) respectively. A correlation (r = 0.946, R2 = 0.833) was observed in Figure 5 (a) and a relatively lower correlation (r = 0.891, R2 = 0.794) in case of Figure 5 (b). Reports have suggested that there is a correlation between the total phenolic content and antioxidant activity of plant extracts [33]. However, no correlation is found between the flavonoid content and the antioxidant activities of the extracts in the present study.
Figure 5.
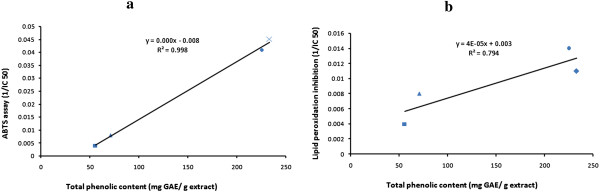
Correlation between total phenolic content and (a) ABTS scavenging activity of the extracts. Correlation coefficient, r = 0.998 and coefficient of determination, R2 = 0.997 (b) Lipid peroxidation inhibition activity of the extracts. Correlation coefficient, r = 0.891and coefficient of determination, R2 = 0.794.
The high correlations confirm the role of phenolic compounds as the main contributor to the antioxidant activities of the Lantana camara leaf extracts. The types and quantities of phenolic compounds might contribute to the varying antioxidant activity of extracts from different varieties. Some phenolic compounds such as salicylic acid, gentisic acid, β-resorcylic acid, coumarin, ferulic acid and 6-Methyl coumarin were identified in L. camara extract [34].
Conclusion
On the basis of the results obtained in the present study, it is concluded that the methanolic leaf extracts of four different varieties of Lantana camara which contains large amount of phenolic compounds, exhibit high antioxidant, free radical scavenging and in vitro lipid peroxidation inhibition activities. A high correlation is found between the total phenolic content and the antioxidant activities using different in vitro antioxidant models in this study. These in vitro assays indicate that these four different varieties has a varying antioxidant activity, and thereby indicate that among them CYV and PRV varieties are good source of antioxidants, which might be useful in preventing the progress of various oxidative conditions. Hence, more queries will be addressed in future studies targeting these varieties to explore the potential and isolation of bioactive compounds responsible for such activities and as chemo preventive and therapeutic agents.
Acknowledgments
Authors would like to thank CSIR, New Delhi for providing financial support and fellowship to Sanjiv Kumar.
Abbreviations
- AAE
Ascorbic acid equivalent
- RE
Rutin equivalent
- GAE
Gallic acid equivalent
- TLC
Thin layer chromatography
- DPPH
2,2-diphenyl-1-picrylhydrazyl
- FRAP
Ferric ion reducing antioxidant potential
- ABTS
2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonate)
- ROS
Reactive oxygen species
- CPV
Chandigarh purple variety
- PRV
Palampur red variety
- YTPV
Yellow turning pink variety
- CYV
Chandigarh yellow variety.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
SO and RS: Designed the study. SK: Carried out the tests and prepared the manuscript. All the authors read and approved the final manuscript.
Contributor Information
Sanjiv Kumar, Email: sanjivbiochem@gmail.com.
Rajat Sandhir, Email: sandhir@pu.ac.in.
Sudarshan Ojha, Email: s_ojha@pu.ac.in.
References
- 1.Mohammad A. Oxidative stress and experimental carcinogenesis. Ind J Exp Biol. 2002;40:656–667. [PubMed] [Google Scholar]
- 2.Wichi HP. Enhanced tumor development of butylated hydroxyanisol (BHA) from the prospective of effect on fore stomach and oesophageal squamous epithelium. Food Chem Toxicol. 1998;26:717–723. [Google Scholar]
- 3.Williams RJ, Spenser JPE, Rice-Evans C. Flavonoids: antioxidants or signalling molecules? Free Radic Biol Med. 2004;36:838–849. doi: 10.1016/j.freeradbiomed.2004.01.001. [DOI] [PubMed] [Google Scholar]
- 4.Mulubagal V, Tsay H. Plant cell cultures – an alternative and efficient source for the production of biologically important secondary metabolites. Int J Appl Sci Eng Tech. 2004;2:29–48. [Google Scholar]
- 5.Borneo R, Leon EA, Aguirre A, Ribotta P, Cantero JJ. Antioxidant capacity of medicinal plants from the Province of Cordoba (Argentina) and their in vitro testing in model food system. Food Chem. 2008;112:664–670. doi: 10.1016/j.foodchem.2008.06.027. [DOI] [Google Scholar]
- 6.Ghisalberti EL. Review Lantana camara L. (Verbenaceae) Fitoterapia. 2000;71:467–486. doi: 10.1016/S0367-326X(00)00202-1. [DOI] [PubMed] [Google Scholar]
- 7.Parrotta JA. Healing Plants of Peninsular India. New York: CAB International; 2001. pp. 710–711. [Google Scholar]
- 8.Forestieri AM, Monforte MT, Ragusa S, Trovato A, Iauk L. Anti-inflammatory, analgesic and antipyretic activity in rodents of plant extracts used in African medicine. Phytother Res. 1996;10:100–106. doi: 10.1002/(SICI)1099-1573(199603)10:2<100::AID-PTR724>3.0.CO;2-I. [DOI] [Google Scholar]
- 9.Herbert JM, Maffrand JP, Taoubi K, Augereau JM, Fouraste I, Gleye J. Verbascoside isolated from Lantana camara, an inhibitor of protein kinase C. J Nat Prod. 1991;54:1595–1600. doi: 10.1021/np50078a016. [DOI] [PubMed] [Google Scholar]
- 10.Sharma OP, Jagdish V, Sharma PD. Comparison of lantadenes content and toxicity of different variety of the lantana plant. J Chem Ecol. 1991;17(11):2283–2291. doi: 10.1007/BF00988008. [DOI] [PubMed] [Google Scholar]
- 11.McDonald S, Prenzler PD, Autolovich M, Robards K. Phenolic content and antioxidant activity of olive extracts. Food Chem. 2001;73:73–84. doi: 10.1016/S0308-8146(00)00288-0. [DOI] [Google Scholar]
- 12.Chang C, Yang M, Wen H, Chern J. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal. 2002;10:178–182. [Google Scholar]
- 13.Blois MS. Antioxidant determinations by the use of a stable free radical. Nature. 1958;181(4617):1199–1200. doi: 10.1038/1811199a0. [DOI] [Google Scholar]
- 14.Prieto P, Pineda M, Aguilar M. Spectrophotometric quantitation of antioxidant capacity through the formation of phosphomolybdenum complex: specific application to determination of vitamin E. Anal Biochem. 1999;269:337–341. doi: 10.1006/abio.1999.4019. [DOI] [PubMed] [Google Scholar]
- 15.Benzie IEF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”. The FRAP assay. Anal Biochem. 1996;239:70–76. doi: 10.1006/abio.1996.0292. [DOI] [PubMed] [Google Scholar]
- 16.Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999;26:1231–1237. doi: 10.1016/S0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- 17.Ohkawa Η, Ohishi Ν, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–358. doi: 10.1016/0003-2697(79)90738-3. [DOI] [PubMed] [Google Scholar]
- 18.Wallace G, Fry SC. Phenolic compounds of the plant cell. International Review of CYVology. 1994;151:229–267. doi: 10.1016/S0074-7696(08)62634-0. [DOI] [PubMed] [Google Scholar]
- 19.Binns AN, Chen RH, Wood HN, Lynn DG. Cell division promotion activity of naturally occurring dehydrodiconiferyl glucosides: Do cell wall components control cell division? Proc Natl Acad Sci U S A. 1987;84:980–984. doi: 10.1073/pnas.84.4.980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zin ZM, Hamid AA, Osman A, Saari N. Antioxidative activities of chromatographic fractions obtained from root, fruit and leaf of Mengkudu (Morinda citrifolia L.) Food Chem. 2006;94:169–178. doi: 10.1016/j.foodchem.2004.08.048. [DOI] [Google Scholar]
- 21.Taubert D, Breitenbach T, Lazar A, Censarek P, Harlfinger S, Berkels R, Klaus W, Roesen R. Reaction rate constant of O2•‾ scavenging by plant antioxidants. Free Rad Biol Med. 2003;35:1599. doi: 10.1016/j.freeradbiomed.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 22.Bangou MJ, Almaraz-Abarca N, Méda NTR, Zeba B, Kiendrebéogo M, Millogo-Rasolodimby J, Nacoulma OG. Polyphenolic composition of Lantana camara and Lippia chevalieri, and their antioxidant and antimicrobial activities. International Journal of Phytomedicine. 2012;4:115–124. [Google Scholar]
- 23.Vamanu E, Nita S. Antioxidant capacity and the correlation with major phenolic compounds, anthocyanin, and tocopherol content in various extracts from the wild edible Boletus edulis mushroom. BioMed Research International. 2013;2013:313905. doi: 10.1155/2013/313905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kessler M, Ubeaud G, Jung L. Anti- and pro-oxidant activity of rutin and quercetin derivatives. J Pharm Pharmacol. 2003;55:131–142. doi: 10.1211/002235702559. [DOI] [PubMed] [Google Scholar]
- 25.Cook NC, Samman S. Flavonoids- chemistry, metabolism, cardioprotective effects, and dietary sources. Nutr Biochem. 1996;7:66–76. doi: 10.1016/0955-2863(95)00168-9. [DOI] [Google Scholar]
- 26.Hwang SJ, Yoon WB, Lee OH, Cha SJ, Kim JD. Radical-scavenging-linked antioxidant activities of extracts from black chokeberry and blueberry cultivated in Korea. Food Chem. 2014;146:71–77. doi: 10.1016/j.foodchem.2013.09.035. [DOI] [PubMed] [Google Scholar]
- 27.Mathew S, Abraham TE. In vitro antioxidant activity and scavenging effects of Cinnamomum verum leaf extract assayed by different methodologies. Food Chemistry and Toxicology. 2006;44:198–206. doi: 10.1016/j.fct.2005.06.013. [DOI] [PubMed] [Google Scholar]
- 28.Coyle JT, Puttfarcken P. Oxidative stress, gluatamate and neurodegenerative diseases. Science. 1993;219:1184. doi: 10.1126/science.6338589. [DOI] [PubMed] [Google Scholar]
- 29.Gutteridge JMC. Age, pigments and free radicals: fluorescent lipid complexes by iron and copper containing proteins. Biochim Biophys Acta. 1985;834:144. doi: 10.1016/0005-2760(85)90149-3. [DOI] [PubMed] [Google Scholar]
- 30.Halliwell B. Superoxide dependent formation of hydroxyl free radicals in the presence of iron chelates. FEBS Lett. 1978;92:321. doi: 10.1016/0014-5793(78)80779-0. [DOI] [PubMed] [Google Scholar]
- 31.Gulcin I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid) Toxicology. 2006;217:213–220. doi: 10.1016/j.tox.2005.09.011. [DOI] [PubMed] [Google Scholar]
- 32.Kalita S, Kumar G, Karthik L, Rao KVB. In vitro antioxidant and DNA damage inhibition activity of aqueous extract of Lantana camara L. (Verbenaceae) leaves. Asian Pac J Trop Biomed. 2012;2(3):S1675–S1679. doi: 10.1016/S2221-1691(12)60476-6. [DOI] [Google Scholar]
- 33.Zhang Y, Wang Z. Phenolic composition and antioxidant activities of two Plomis species: a correlation study. CR Biologies. 2009;332:816–826. doi: 10.1016/j.crvi.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 34.Yi Z, Zhang M, Ling B, Xu D, Ye J. Inhibitory effects of Lantana camara and phenolic compounds from it on growth of Eichhornia crassipes. Yingyong Shengtai Xuebao. 2006;17:1637–1640. [PubMed] [Google Scholar]


