Abstract
Mycolic acids represent a major constituent of the mycobacterial cell wall complex, which provides the first line of defense against potentially lethal environmental conditions. Slow-growing pathogenic mycobacteria such as Mycobacterium tuberculosis modify their mycolic acids by cyclopropanation, whereas fast-growing saprophytic species such as Mycobacterium smegmatis do not, suggesting that this modification may be associated with an increase in oxidative stress experienced by the slow-growing species. We have demonstrated the transformation of the distal cis double bond in the major mycolic acid of M. smegmatis to a cis-cyclopropane ring upon introduction of cosmid DNA from M. tuberculosis. This activity was localized to a single gene (cma1) encoding a protein that was 34% identical to the cyclopropane fatty acid synthase from Escherichia coli. Adjacent regions of the DNA sequence encode open reading frames that display homology to other fatty acid biosynthetic enzymes, indicating that some of the genes required for mycolic acid biosynthesis may be clustered in this region. M. smegmatis overexpressing the cma1 gene product significantly resist killing by hydrogen peroxide, suggesting that this modification may be an important adaptation of slow-growing mycobacteria to oxidative stress.
Full text
PDF

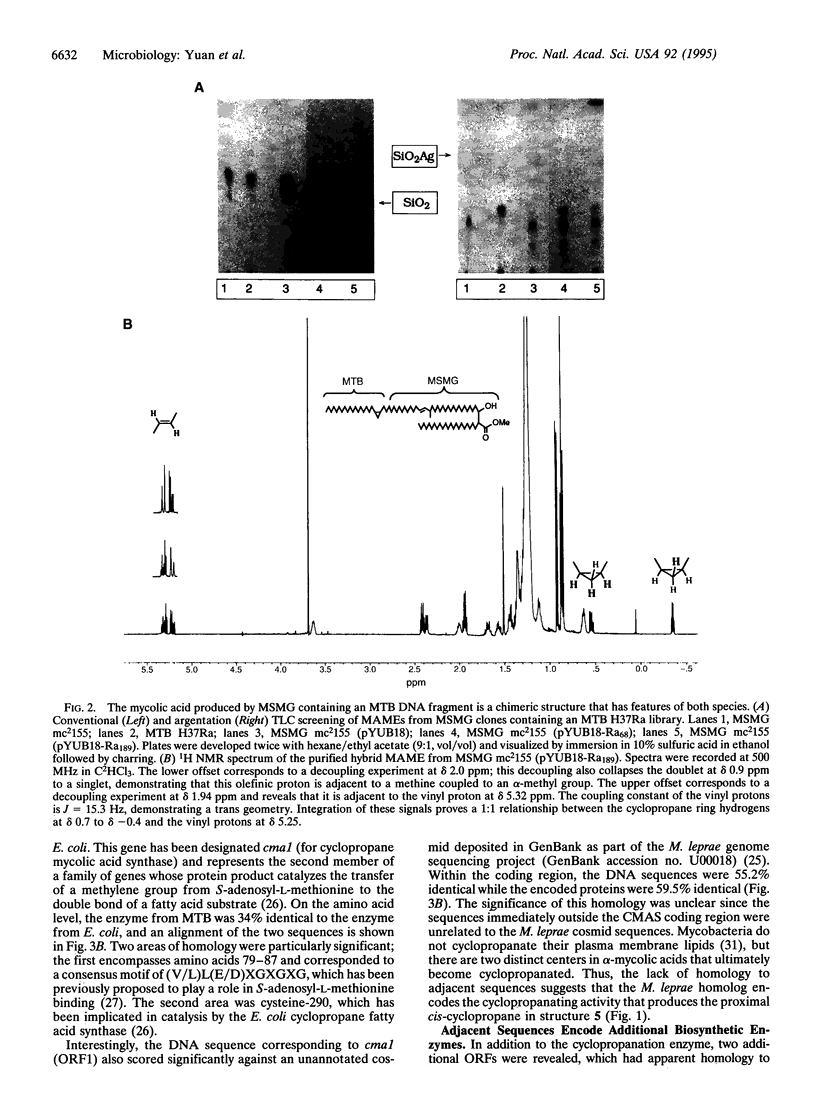


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akamatsu Y., Law J. H. Enzymatic synthesis of 10-methylene stearic acid and tuberculostearic acid. Biochem Biophys Res Commun. 1968 Oct 10;33(1):172–176. doi: 10.1016/0006-291x(68)90274-x. [DOI] [PubMed] [Google Scholar]
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Andersson B. A., Holman R. T. Pyrrolidides for mass spectrometric determination of the position of the double bond in monounsaturated fatty acids. Lipids. 1974 Mar;9(3):185–190. doi: 10.1007/BF02532690. [DOI] [PubMed] [Google Scholar]
- Arrowsmith T. J., Malpartida F., Sherman D. H., Birch A., Hopwood D. A., Robinson J. A. Characterisation of actI-homologous DNA encoding polyketide synthase genes from the monensin producer Streptomyces cinnamonensis. Mol Gen Genet. 1992 Aug;234(2):254–264. doi: 10.1007/BF00283846. [DOI] [PubMed] [Google Scholar]
- Baess I., Mansa B. Determination of genome size and base ratio on deoxyribonucleic acid from mycobacteria. Acta Pathol Microbiol Scand B. 1978 Oct;86B(5):309–312. doi: 10.1111/j.1699-0463.1978.tb00049.x. [DOI] [PubMed] [Google Scholar]
- Banerjee A., Dubnau E., Quemard A., Balasubramanian V., Um K. S., Wilson T., Collins D., de Lisle G., Jacobs W. R., Jr inhA, a gene encoding a target for isoniazid and ethionamide in Mycobacterium tuberculosis. Science. 1994 Jan 14;263(5144):227–230. doi: 10.1126/science.8284673. [DOI] [PubMed] [Google Scholar]
- Belisle J. T., Pascopella L., Inamine J. M., Brennan P. J., Jacobs W. R., Jr Isolation and expression of a gene cluster responsible for biosynthesis of the glycopeptidolipid antigens of Mycobacterium avium. J Bacteriol. 1991 Nov;173(21):6991–6997. doi: 10.1128/jb.173.21.6991-6997.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besra G. S., McNeil M. R., Khoo K. H., Dell A., Morris H. R., Brennan P. J. Trehalose-containing lipooligosaccharides of Mycobacterium gordonae: presence of a mono-O-methyltetra-O-acyltrehalose "core" and branching in the oligosaccharide backbone. Biochemistry. 1993 Nov 30;32(47):12705–12714. doi: 10.1021/bi00210a020. [DOI] [PubMed] [Google Scholar]
- Cronan J. E., Jr, Reed R., Taylor F. R., Jackson M. B. Properties and biosynthesis of cyclopropane fatty acids in Escherichia coli. J Bacteriol. 1979 Apr;138(1):118–121. doi: 10.1128/jb.138.1.118-121.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eiglmeier K., Honoré N., Woods S. A., Caudron B., Cole S. T. Use of an ordered cosmid library to deduce the genomic organization of Mycobacterium leprae. Mol Microbiol. 1993 Jan;7(2):197–206. doi: 10.1111/j.1365-2958.1993.tb01111.x. [DOI] [PubMed] [Google Scholar]
- Garbe T. R., Barathi J., Barnini S., Zhang Y., Abou-Zeid C., Tang D., Mukherjee R., Young D. B. Transformation of mycobacterial species using hygromycin resistance as selectable marker. Microbiology. 1994 Jan;140(Pt 1):133–138. doi: 10.1099/13500872-140-1-133. [DOI] [PubMed] [Google Scholar]
- Hamid M. E., Minnikin D. E., Goodfellow M. A simple chemical test to distinguish mycobacteria from other mycolic-acid-containing actinomycetes. J Gen Microbiol. 1993 Sep;139(9):2203–2213. doi: 10.1099/00221287-139-9-2203. [DOI] [PubMed] [Google Scholar]
- Hiltunen J. K., Wenzel B., Beyer A., Erdmann R., Fosså A., Kunau W. H. Peroxisomal multifunctional beta-oxidation protein of Saccharomyces cerevisiae. Molecular analysis of the fox2 gene and gene product. J Biol Chem. 1992 Apr 5;267(10):6646–6653. [PubMed] [Google Scholar]
- Ingrosso D., Fowler A. V., Bleibaum J., Clarke S. Sequence of the D-aspartyl/L-isoaspartyl protein methyltransferase from human erythrocytes. Common sequence motifs for protein, DNA, RNA, and small molecule S-adenosylmethionine-dependent methyltransferases. J Biol Chem. 1989 Nov 25;264(33):20131–20139. [PubMed] [Google Scholar]
- Jackett P. S., Aber V. R., Lowrie D. B. Virulence and resistance to superoxide, low pH and hydrogen peroxide among strains of Mycobacterium tuberculosis. J Gen Microbiol. 1978 Jan;104(1):37–45. doi: 10.1099/00221287-104-1-37. [DOI] [PubMed] [Google Scholar]
- MITCHISON D. A., SELKON J. B., LLOYD J. VIRULENCE IN THE GUINEA-PIG, SUSCEPTIBILITY TO HYDROGEN PEROXIDE, AND CATALASE ACTIVITY OF ISONIAZID-SENSITIVE TUBERCLE BACILLI FROM SOUTH INDIAN AND BRITISH PATIENTS. J Pathol Bacteriol. 1963 Oct;86:377–386. doi: 10.1002/path.1700860213. [DOI] [PubMed] [Google Scholar]
- Mathur M., Kolattukudy P. E. Molecular cloning and sequencing of the gene for mycocerosic acid synthase, a novel fatty acid elongating multifunctional enzyme, from Mycobacterium tuberculosis var. bovis Bacillus Calmette-Guerin. J Biol Chem. 1992 Sep 25;267(27):19388–19395. [PubMed] [Google Scholar]
- Persson B., Krook M., Jörnvall H. Characteristics of short-chain alcohol dehydrogenases and related enzymes. Eur J Biochem. 1991 Sep 1;200(2):537–543. doi: 10.1111/j.1432-1033.1991.tb16215.x. [DOI] [PubMed] [Google Scholar]
- Qureshi N., Takayama K., Jordi H. C., Schnoes H. K. Characterization of the purified components of a new homologous series of alpha-mycolic acids from Mycobacterium tuberculosis H37Ra. J Biol Chem. 1978 Aug 10;253(15):5411–5417. [PubMed] [Google Scholar]
- Snapper S. B., Lugosi L., Jekkel A., Melton R. E., Kieser T., Bloom B. R., Jacobs W. R., Jr Lysogeny and transformation in mycobacteria: stable expression of foreign genes. Proc Natl Acad Sci U S A. 1988 Sep;85(18):6987–6991. doi: 10.1073/pnas.85.18.6987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snapper S. B., Melton R. E., Mustafa S., Kieser T., Jacobs W. R., Jr Isolation and characterization of efficient plasmid transformation mutants of Mycobacterium smegmatis. Mol Microbiol. 1990 Nov;4(11):1911–1919. doi: 10.1111/j.1365-2958.1990.tb02040.x. [DOI] [PubMed] [Google Scholar]
- Snider D. E., Jr, Roper W. L. The new tuberculosis. N Engl J Med. 1992 Mar 5;326(10):703–705. doi: 10.1056/NEJM199203053261011. [DOI] [PubMed] [Google Scholar]
- Takayama K., Schnoes H. K., Armstrong E. L., Boyle R. W. Site of inhibitory action of isoniazid in the synthesis of mycolic acids in Mycobacterium tuberculosis. J Lipid Res. 1975 Jul;16(4):308–317. [PubMed] [Google Scholar]
- Wang A. Y., Grogan D. W., Cronan J. E., Jr Cyclopropane fatty acid synthase of Escherichia coli: deduced amino acid sequence, purification, and studies of the enzyme active site. Biochemistry. 1992 Nov 17;31(45):11020–11028. doi: 10.1021/bi00160a011. [DOI] [PubMed] [Google Scholar]
- Wheeler P. R., Besra G. S., Minnikin D. E., Ratledge C. Stimulation of mycolic acid biosynthesis by incorporation of cis-tetracos-5-enoic acid in a cell-wall preparation from Mycobacterium smegmatis. Biochim Biophys Acta. 1993 Apr 7;1167(2):182–188. doi: 10.1016/0005-2760(93)90160-b. [DOI] [PubMed] [Google Scholar]
- Wong M. Y., Steck P. A., Gray G. R. The major mycolic acids of Mycobacterium smegmatis. Characterization of their homologous series. J Biol Chem. 1979 Jul 10;254(13):5734–5740. [PubMed] [Google Scholar]