Abstract
Eukaryotes package DNA into nucleosomes that contain a core of histone proteins. During DNA replication, nucleosomes are disrupted and re-assembled with newly synthesized histones and DNA. Despite much progress, it is still unclear why higher eukaryotes contain multiple core histone genes, how chromatin assembly is controlled, and how these processes are coordinated with cell cycle progression. We used a histone null mutation of Drosophila melanogaster to show that histone supply levels, provided by a defined number of transgenic histone genes, regulate the length of S phase during the cell cycle. Lack of de novo histone supply not only extends S phase, but also causes a cell cycle arrest during G2 phase, and thus prevents cells from entering mitosis. Our results suggest a novel cell cycle surveillance mechanism that monitors nucleosome assembly without involving the DNA repair pathways and exerts its effect via suppression of CDC25 phosphatase String expression.
DOI: http://dx.doi.org/10.7554/eLife.02443.001
Research organism: D. melanogaster
eLife digest
As a cell prepares to divide, it goes through four distinct stages. First, it grows in size (G1 phase); next it copies its entire DNA content (S phase); then it grows some more (G2 phase); and, last, it splits into two new cells (M phase).
During S phase, groups of histone proteins that normally stick together to tightly package the DNA are pulled apart in order to make the DNA accessible for copying. After the DNA has been duplicated, both copies of the DNA strand need to be repackaged. Therefore, after copying the DNA the cell rapidly reassembles the DNA–histone complexes (called nucleosomes), using a combination of old and newly synthesized histones to do so. A cell can adjust how quickly it copies DNA according to the availability of these histone proteins, which is important because copying DNA without the resources to package it could expose the DNA to damage.
Here, Günesdogan et al. investigate how a cell controls these processes using a mutant of the fruit fly Drosophila melanogaster that completely lacks the genes required to make histones. Cells that lack histones copy their DNA very slowly but adding copies of histone genes back into these flies speeds up the rate at which DNA is copied.
Günesdogan et al. ask whether the slower speed of DNA replication in cells without new histones is connected to preventing DNA damage. However, these cells can still copy all their DNA, despite being unable to package it, so the higher risk of making mistakes is not enough to stop S phase. In fact, indications suggest that DNA damage detection methods continue to work as normal in cells without histones: these cells can get all the way to the end of G2 phase without any problems.
To go one step further and start splitting in two, a cell needs to switch on another gene, called string in the fruit fly and CDC25 in vertebrates, which makes an enzyme required for the cell division process. Normal cells switch on string during G2 phase, but cells that lack histones do not—and therefore do not enter M phase. Günesdogan et al. show that turning on string by a genetic trick is sufficient to overcome this cell cycle arrest and drive the cells into M phase. String could therefore form part of a surveillance mechanism that blocks cell division if DNA–histone complexes are not assembled correctly.
Introduction
Chromatin assembly during DNA replication is crucial for the repackaging of newly synthesized DNA and for maintaining or erasing histone modifications. During this process, pre-existing or so-called parental histones are recycled and assembled into nucleosomes together with de novo synthesized histones (Alabert and Groth, 2012; Annunziato, 2012). To compensate for the high demand of histone proteins during DNA replication, the canonical histones H1, H2A, H2B, H3, and H4, which are encoded by multiple gene copies in higher eukaryotes, are highly and exclusively expressed in S phase of the cell cycle (Marzluff et al., 2008).
The assembly of chromatin is mediated by an interplay of components of the DNA replication machinery and histone chaperones, which mediate the deposition of histones into nucleosomes (Alabert and Groth, 2012; Annunziato, 2012). Apparently, the pace of DNA synthesis is tightly coupled to the assembly of newly synthesized DNA into chromatin. Multiple studies showed that the depletion of the histone chaperones Asf1 and CAF-1 results in a slow down of DNA synthesis during S phase (Hoek and Stillman, 2003; Ye et al., 2003; Nabatiyan and Krude, 2004; Groth et al., 2007; Takami et al., 2007) preceding the accumulation of DNA damage in mammalian cells (Hoek and Stillman, 2003; Ye et al., 2003). Also, diminishing histone supply during S phase through knock down of SLBP, which is required for histone mRNA stability and translation, decreases the rate of DNA synthesis (Zhao et al., 2004). A recent study that targeted SLBP together with FLASH, a factor that is required for histone mRNA transcription and processing (Barcaroli et al., 2006; Yang et al., 2009), revealed that replication fork progression depends on nucleosome assembly potentially through a mechanism based on a feedback from the histone chaperone CAF-1 to the replicative helicase and/or the unloading of PCNA from newly synthesized DNA upon nucleosome assembly (Groth et al., 2007; Mejlvang et al., 2014).
The coupling of replication fork progression and nucleosome assembly might compensate for short-term fluctuations in histone availability (Mejlvang et al., 2014). However, it is still unclear whether chromatin integrity is monitored after or during DNA replication. Genome integrity during S phase is governed by the ATR/Chk1 and ATM/Chk2 checkpoint mechanisms that sense replication stress and DNA damage, respectively (Bartek and Lukas, 2007; Cimprich and Cortez, 2008). Lack of CAF-1 or Asf1 function leads to accumulation of DNA damage and activation of the ATM/Chk2 pathway (Hoek and Stillman, 2003; Ye et al., 2003). These findings led to the hypothesis that chromatin assembly is monitored indirectly through accumulation of DNA lesions in response to stalled replication forks. However, since these chaperones have multiple functions such as unwinding of DNA during replication, in DNA repair (Gaillard et al., 1996; Green and Almouzni, 2003; Schöpf et al., 2012) as well as other nuclear processes (Quivy et al., 2004; Houlard et al., 2006). These multiple functions of these chaperones make it difficult to assess the direct effects of defective chromatin assembly.
Taking advantage of a histone null mutation in a higher eukaryote that recently became available in Drosophila melanogaster (Günesdogan et al., 2010), we directly addressed the requirement of canonical histone supply for DNA replication and cell cycle progression in a developing organism. By reintroducing a defined number of transgenic histone genes into the histone null mutant background, we show that the rate of DNA replication is coupled to the number of histone genes present in the genome and that histone supply is critical to coordinate S phase length with the developmental program. Surprisingly, cells that completely lacked de novo histone synthesis replicate DNA at a reduced rate, but complete S phase and arrest in cell cycle without accumulating DNA damage. This cell cycle arrest is mediated by suppressing the accumulation of transcripts encoding the CDC25 phosphatase String and provides evidence for a chromatin assembly surveillance mechanism that is independent of the known S phase checkpoints.
Results
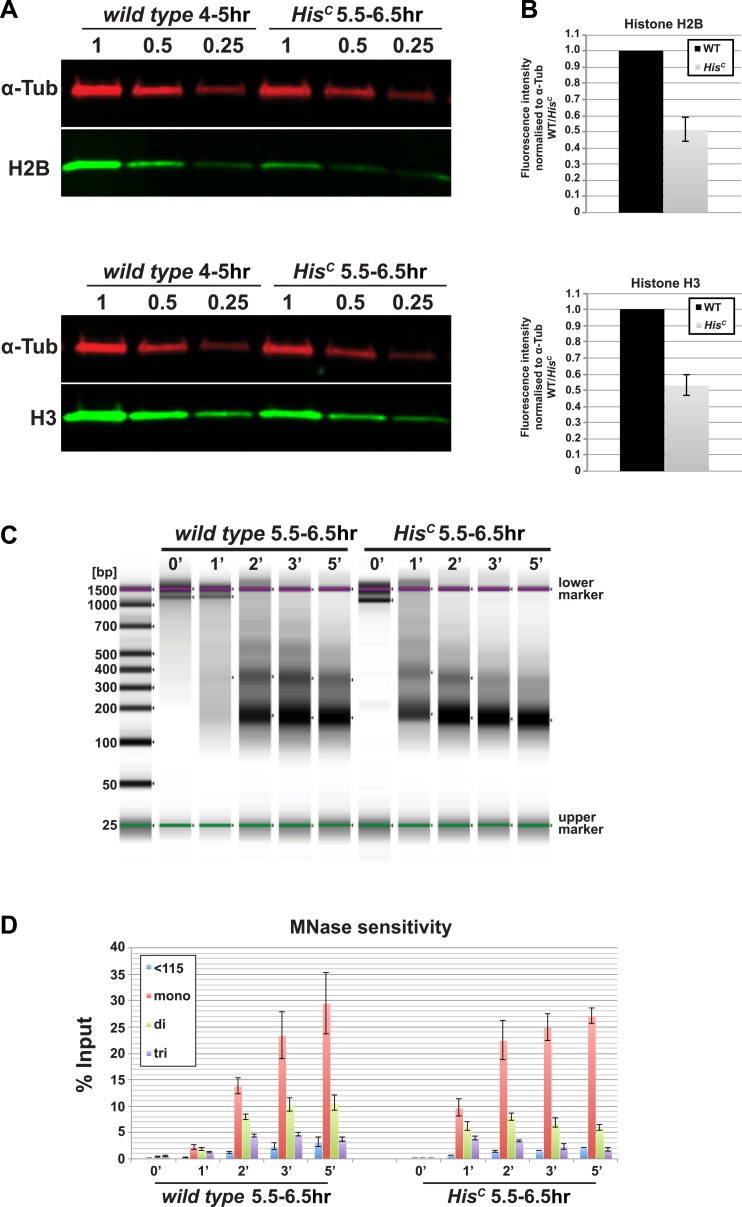
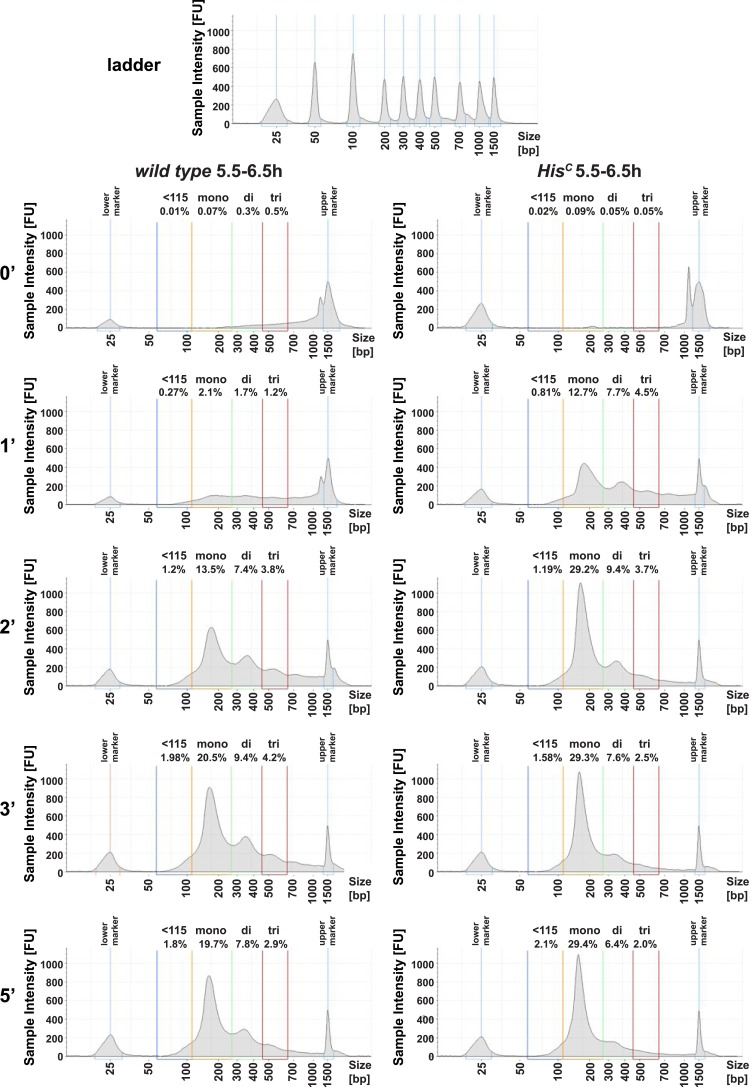
The histone null mutation in D. melanogaster, called Df(2L)HisC, lacks all genes encoding the canonical histones (Günesdogan et al., 2010). Df(2L)HisC homozygous mutant animals (hereafter referred to as HisC mutants) that are derived from heterozygous parents contain only maternal histone mRNA and proteins, which are sufficient to complete the first 14 cell division cycles of the embryo (Günesdogan et al., 2010). HisC mutant embryos arrest before the onset of mitosis in cycle 15 (M15) (Günesdogan et al., 2010). This highly uniform phenotype is likely due to the degradation of maternal histone mRNAs during the first G2 phase of embryogenesis in cell cycle 14 (Marzluff et al., 2008; O'Farrell et al., 1989) combined with the complete lack of zygotic histone gene expression during S phase of cell cycle 15 (S15) (Günesdogan et al., 2010). In order to verify that the lack of histone transcription also results in a diminished pool of histone proteins in S15, we compared the protein levels of histone H2B and H3 of wild type embryos that are in S15 at 4–5 hr after egg laying (AEL) to sorted HisC mutant embryos that are still in S15 at 5.5–6.5 hr AEL (see below and Günesdogan et al., 2010) by quantitative Western blotting (Figure 1A,B). The approximate twofold reduction in the histone levels of HisC mutant embryos is consistent with the fact that these embryos lack synthesis of new histones in S15 but still contain parental histones from chromatin that was assembled during cycle 14. To test whether the reduced supply of histones in HisC mutant embryos leads to a decrease in nucleosome formation, we carried out Micrococcal Nuclease (MNase) digestion assays on chromatin from sorted HisC mutant and wild type sibling embryos (Figure 1C,D, Figure 1—figure supplement 1). The results show that chromatin from HisC mutant embryos is more accessible to MNase than control chromatin, leading to a more rapid generation of mononucleosomal DNA fragments and reflecting a decrease in nucleosome occupancy in chromatin of the HisC mutants.
Figure 1. Nucleosome density is affected in HisC mutant cells.
(A) Fluorescent Western blot analysis for histone H2B and H3 (green) using wild type embryos at 4–5 hr and sorted HisC mutant embryos at 5.5–6.5 hr AEL, respectively. α-Tubulin (α-Tub; red) was used as loading control. A dilution series of each extract was loaded (1, 0.5, 0.25). (B) Quantification of Western blots as shown in (A). Fluorescence measurements of the histone signal was normalised to the α-Tubulin signal and the wild type (WT)/HisC ratio is shown. The histone protein content is ∼twofold reduced in HisC mutant embryos as compared to wild type. Mean values from three independent experiments are shown. Error bars indicate standard error. (C) Gel electrophoresis of time-course (0′–5′) microccocal nuclease (MNase) digestions using sorted HisC mutant and wild type sibling embryos at 5.5–6.5 hr after egg laying, respectively. (D) Quantification of MNase digestion experiments as shown in (C). HisC mutant chromatin is digested more rapidly into mononucleosomal DNA than control chromatin. Mean values from three independent experiments are shown. Error bars indicate standard error.
Figure 1—figure supplement 1. HisC mutant chromatin shows increased MNase sensitivity.
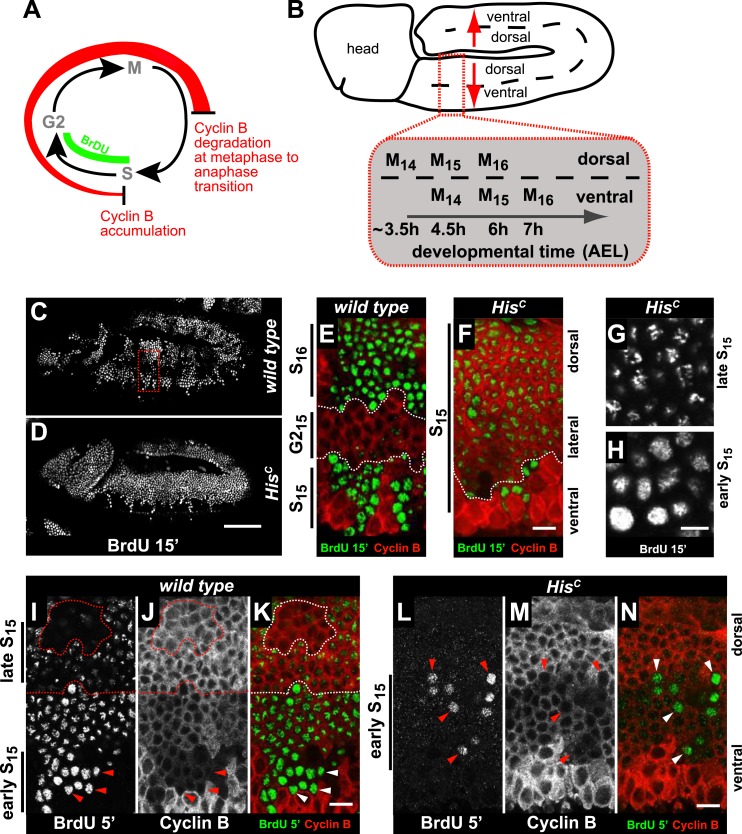
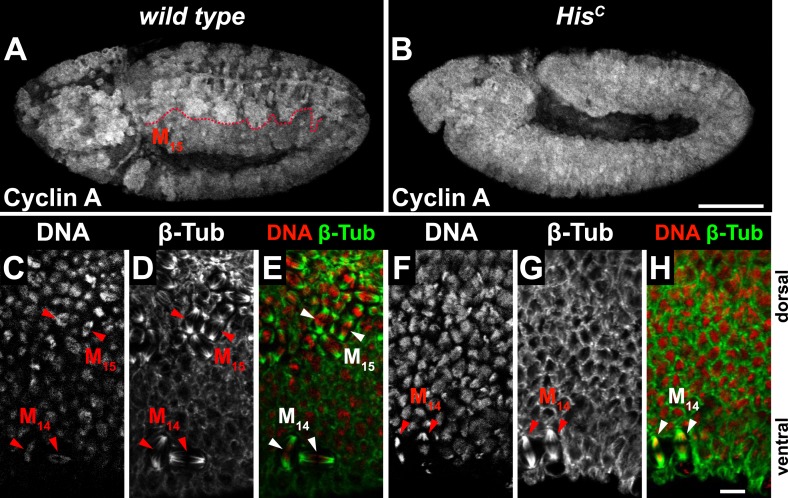
Arrested HisC mutant cells express high levels of mitotic Cyclin B suggesting a cell cycle arrest in G2 phase of cycle 15 (G215) before mitosis (Günesdogan et al., 2010; Lehner and O'Farrell, 1990; Figure 2A). Consistent with this, we did not observe degradation of the mitotic Cyclin A or assembly of mitotic spindles in HisC mutant cells (Figure 2—figure supplement 1). To assess DNA replication in S15, we used BrdU incorporation assays to label newly synthesized DNA (Figure 2). The results show that most cells of HisC mutant embryos entered S15, but the spatial pattern of replicating cells was different from the highly stereotyped wild type pattern (Figure 2B–D). In wild type embryos, about 5 hr AEL (Figure 2C,E), the ventral epidermal cells incorporated BrdU in S15, whereas the lateral cells entered G215 and stopped BrdU incorporation. Dorsal epidermal cells had already passed through M15 and incorporated BrdU in S16. In contrast, the HisC mutant cells failed to reach G215 and the majority of the cells continued to incorporate BrdU at low levels in S15 (Figure 2D,F). To test whether the extended S phase of HisC mutant cells is caused by a reduced rate of DNA synthesis, we shortened the BrdU labelling pulses from 15 to 5 min. In wild type cells, BrdU incorporation was detectable during S15 (Figure 2I–K), whereas in HisC mutant cells BrdU incorporation was detected only in a few cells with low Cyclin B levels indicating that they were still in early S15 (Figure 2L–N). Notably, in HisC mutants ventral cells showed a uniform pattern and dorsal cells a punctuate pattern of BrdU incorporation (Figure 2G,H), which is characteristic for the replication of euchromatin and heterochromatin in early and late S phase, respectively (Shermoen et al., 2010). Although we cannot exclude that the lack of histone synthesis in HisC mutants interfered with firing of individual origins, the results suggest that both early and late replication origins are activated in HisC mutants. In conclusion, the absence of de novo histone supply reduces the rate of DNA synthesis shortly after the initiation of DNA replication, resulting in an extended S phase in mutant cells.
Figure 2. HisC mutant cells extend S phase and slow down DNA synthesis.
(A and B) Schematic models of Cyclin B accumulation and BrdU incorporation during the embryonic cell cycle (A) and its spatial control (B). AEL: after egg laying. (C–N) BrdU pulse labelling for 15 (C–H) or 5 min (I–N) and staining with antibodies against BrdU (green in merge) and Cyclin B (red in merge). (C and D) BrdU was detected in the epidermis of HisC mutant embryos indicating DNA replication but the pattern of replicating cells was distinct from wild type. (E and F) Magnifications of an epidermal region as shown for the wild type embryo (boxed area in C). (E) BrdU labelled wild type cells of the ventral epidermis in S15 (below dashed lines) and of the dorsal epidermis in S16 (above dashed lines). Cells in G215 were BrdU negative with high levels of Cyclin B and located in the lateral epidermis (between dashed lines). (F) In HisC mutant embryos, lateral and dorsal cells re-accumulated Cyclin B and were labelled for BrdU (above dashed line), indicating that mutant cells still replicated DNA during S15. (G and H) Punctate pattern of BrdU incorporation in cells that progressed into late S15 in the dorsal epidermis (G) and uniform incorporation pattern in cells of the ventral epidermis (H). (I–K) In wild type embryos replicating cells in early (ventral, below dashed line) and late S15 (dorsal, above dashed line) were detected after a 5 min BrdU labelling pulse. Some patches of dorsal cells completed S15 and did not incorporate BrdU (encircled). (L–N) In HisC mutant embryos BrdU was detected after a 5 min BrdU labelling pulse only in ventral cells that were in early S phase as indicated by low levels of Cyclin B (arrowheads). Dorsal up (E–N), scale bars: 100 µm (C and D), 10 µm (E–N).
Figure 2—figure supplement 1. The cell cycle arrest of HisC mutant cells is before mitosis.
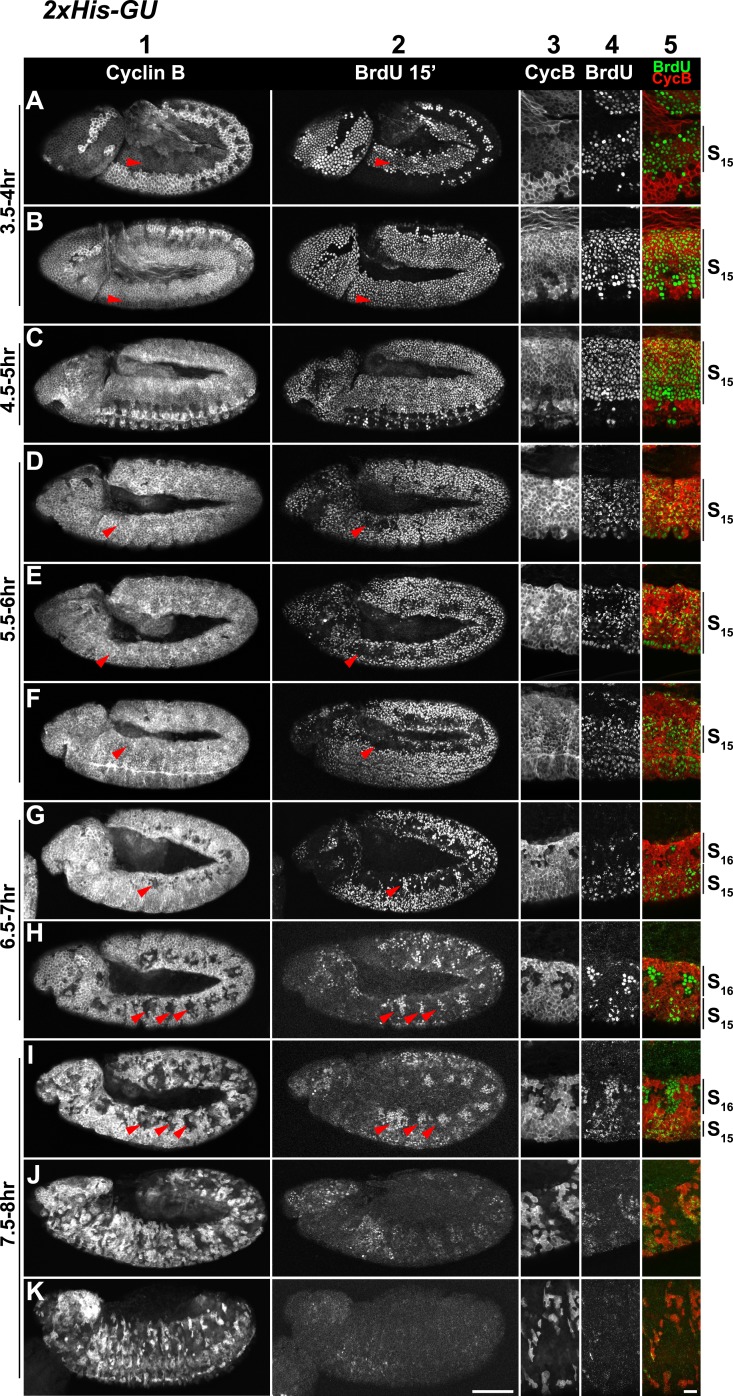
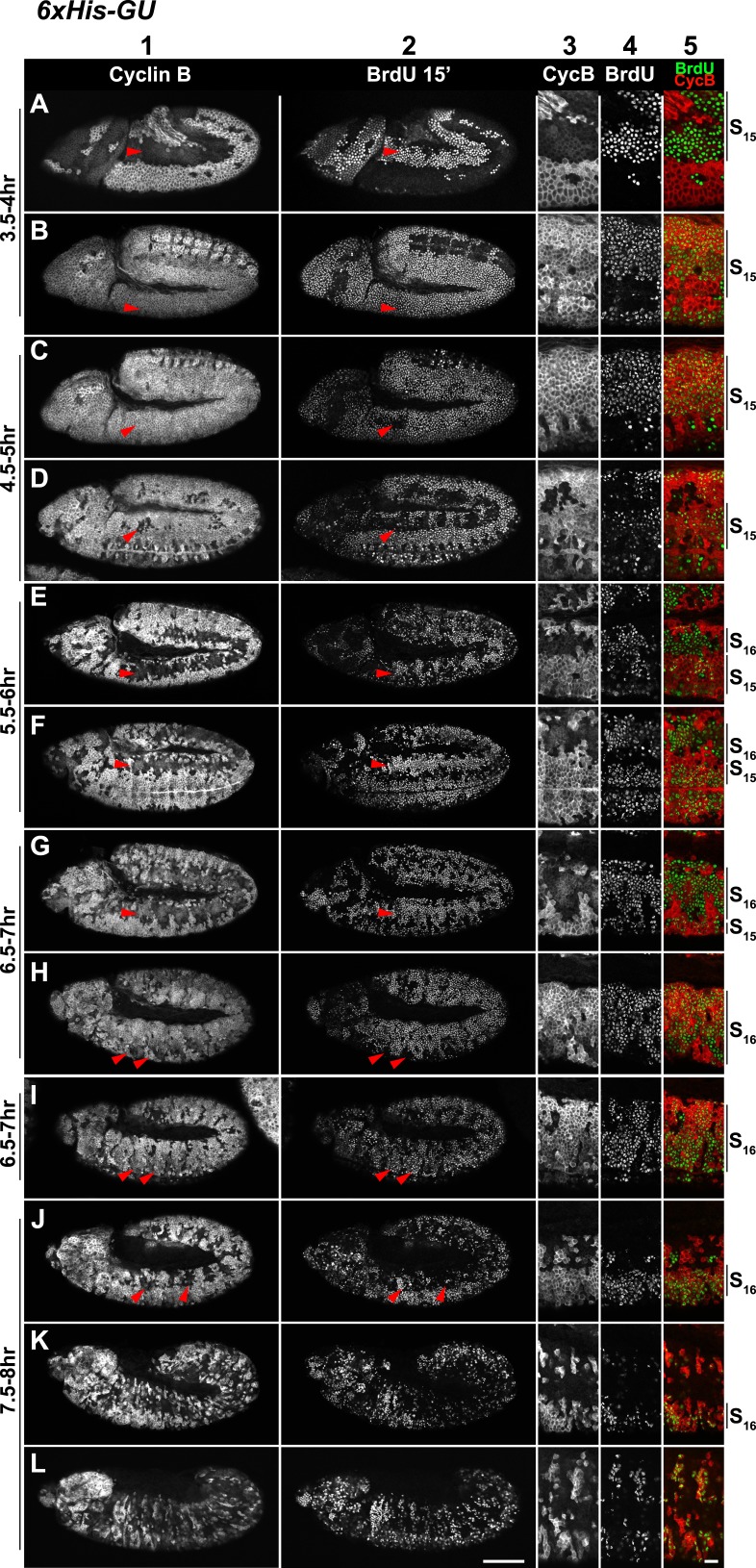
It was previously shown that depletion of histones in cultured cells leads to a slow down of replication fork movement and it was proposed that this mechanism might avoid chromatin assembly defects due to short-term fluctuations in histone availability (Mejlvang et al., 2014). Thus, we asked whether S phase could be faithfully completed under diminished but constant histone supply and whether there is a direct dose dependent relation between histone synthesis and the length of S phase. We reintroduced defined numbers of histone gene units (His-GUs) into the genome of HisC mutant embryos. Embryos carrying either two (2xHis-GU) or six (6xHis-GU) units completed cell cycle 15 as shown by the degradation of Cyclin B in M15 (Figure 3A,B). These embryos were not rescued and they died later towards the end of embryogenesis (Figure 3—figure supplements 1 and 2). Compared to wild type, the onset of M15 was delayed in embryos with 2xHis-GUs and 6xHis-GUs by 2 hr and 1 hr, respectively, which was due to an extended S phase 15 (Figure 3A–E, Figure 3—figure supplements 1 and 2). In contrast, embryos containing 12xHis-GUs were fully rescued and entered M15 at the same time as the wild type cells, that is, 4.5–5 hr AEL as shown previously (Günesdogan et al., 2010). These results establish that the length of S phase directly correlates with the transgene-derived de novo histone supply. In addition, our data indicate that the mechanisms adjusting replication fork movement to the available histone supply allow the completion of S phase under conditions of permanently diminished new histone supply in vivo (Figure 3F).
Figure 3. Histone availability determines the rate of S phase progression but is not required for completion of DNA replication.
(A–D) Immunofluorescent staining with antibodies against Cyclin B. (A) Cyclin B degradation at 6.5–7 hr AEL around the tracheal pits in 2xHis-GU embryos during M15 (arrowheads) shows that these embryos are lagging more than one cell cycle behind as compared to wild type (see also Figure 3—figure supplement 1). (B) Cyclin B degradation in dorsal cells of 6xHis-GU embryos at 5.5–6 hr AEL during M15 (arrowheads), showing that 6xHis-GU reduced the delay in cell cycle progression compared to 2xHis-GUs (see also Figure 3—figure supplement 2). (C) Cyclin B degradation in dorsal cells of wild type embryos at 4.5–5 hr AEL during M15 (arrowheads). (D) Cyclin B degradation at 5.5–6 hr AEL around the tracheal pits in wild type embryos during M16 (arrowheads). (E) BrdU pulse labelling for 15 min of 2xHis-GU and 6xHis-GU embryos at indicated developmental stages and staining with antibodies against BrdU and Cyclin B. Shown is the quantification of embryos that completed S15 (‘S15 exit’, based on lack of BrdU labelling) and progressed into M15 (‘M15 entry’, based on Cyclin B degradation), showing the interdependence of the number of histone genes and cell cycle progression. n: number of embryos. (F) Schematic model showing nucleosome assembly at the replication fork and its dependence on histone supply. (G) DNA quantification of single nuclei stained with DAPI. Wild type nuclei in G215 and early S16 defined 4N and 2N DNA content, respectively. HisC mutant nuclei show mean intensity value of 4N nuclei, suggesting that mutant cells completed genome duplication. n: number of nuclei, p: probability from Student's t test, n.s.: not significant. Scale bars: 100 µm (A–D).
Figure 3—figure supplement 1. Cell cycle progression of HisC mutant embryos with two His-GUs (2xHis-GUs).
Figure 3—figure supplement 2. Cell cycle progression of HisC mutant embryos with six His-GUs (6xHis-GUs).
Several studies showed that mammalian tissue culture cells depleted for CAF-1 accumulate in S phase due to a decreased rate of DNA replication (Hoek and Stillman, 2003; Ye et al., 2003; Nabatiyan and Krude, 2004; Takami et al., 2007) followed by accumulation of DNA damage and activation of the conventional DNA damage checkpoints (Hoek and Stillman, 2003; Ye et al., 2003). However, it remained unclear whether this S phase arrest represents a direct consequence of a failure in chromatin assembly as yeast cells, for example, can complete one round of replication after the depletion of histone H4 (Kim et al., 1988). In order to address whether DNA replication can be completed in the absence of de novo histone synthesis, we quantified the amount of nuclear DNA in DAPI-stained HisC mutant cells and compared it with wild type control cells during early S16 (2N) and G215 (4N), respectively (Figure 3G). The DNA content of HisC mutant cells at 6.5–7.5 hr AEL corresponded to the 4N value of wild type nuclei in G2. This finding indicates that DNA replication in HisC mutant cells has essentially been completed.
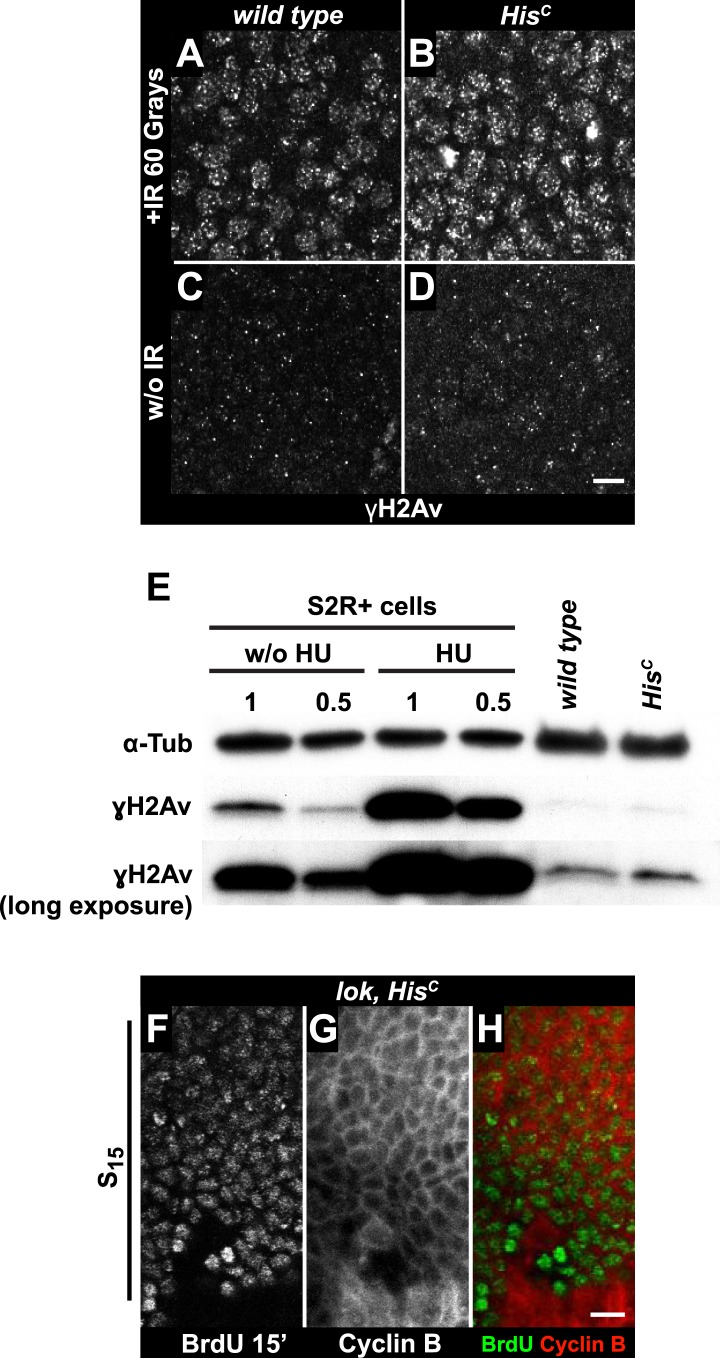
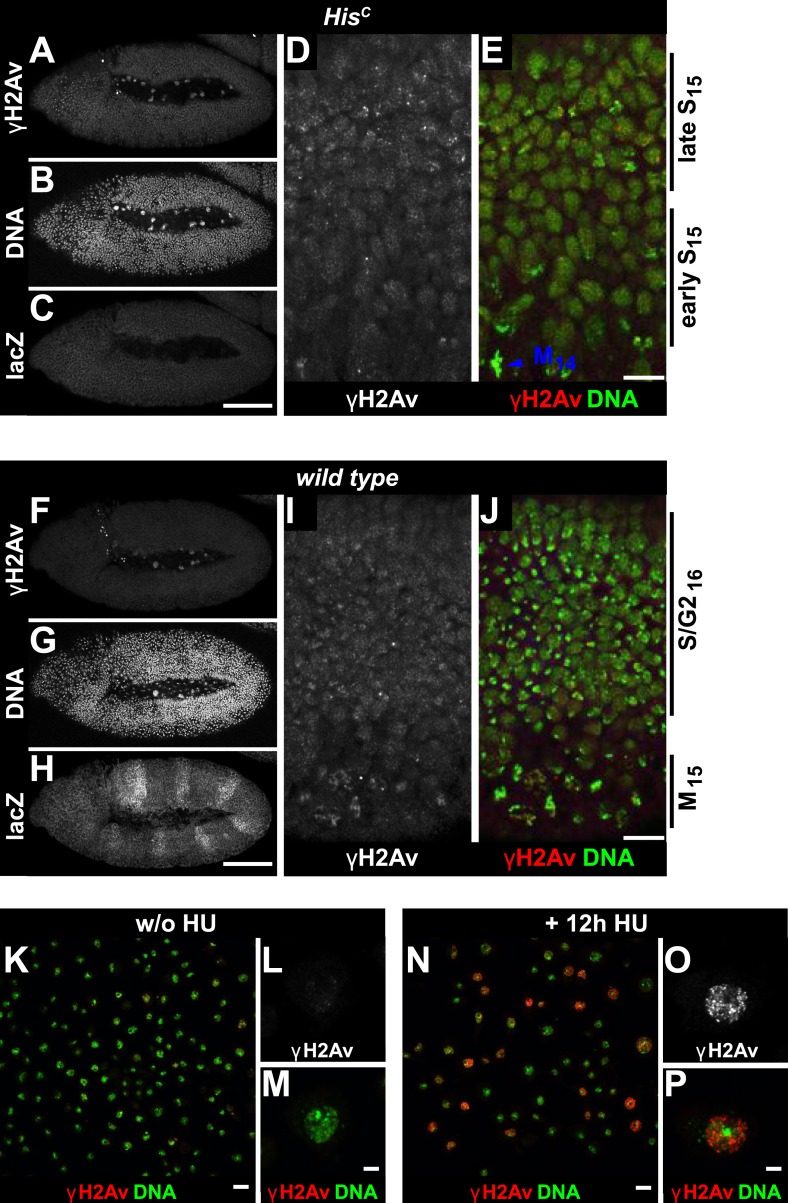
To further explore whether HisC mutant cells accumulate DNA damage and activate the DNA damage checkpoints, we stained for the phosphorylated histone variant H2Av (γH2Av), which is like its vertebrate ortholog γH2AX a marker of DNA damage (Madigan et al., 2002). We found that γH2Av can be induced by ionizing irradiation that causes double strand breaks (DSBs) both in wild type and HisC mutant embryos, showing that the ATM/Chk2 checkpoint mechanism is still functional in HisC mutant cells (Figure 4A,B). We did not observe a difference with respect to γH2Av staining between non-irradiated HisC mutant and wild type cells in early S15 (Figure 4C,D). However, we noted a slight increase in γH2Av staining betwen early and late S15 in HisC mutant embryos (Figure 4—figure supplement 1). To further investigate this increase, we performed Western blot analysis for γH2Av. Drosophila S2R+ cells showed a dramatic increase in γH2Av upon treatment with hyrdoxyurea (HU) (Figure 4E, Figure 4—figure supplement 1 and see below), which was not detectable in HisC mutant embryos undergoing late S15 at 5.5–6.5 hr AEL when compared to wild type sibling embryos (Figure 4E).
Figure 4. The cell cycle arrest of HisC mutant cells does not depend on the ATM/Chk2 DNA damage checkpoint.
(A and B) Wild type and HisC mutant cells responded to ionizing irradiation (IR) by phosphorylation of the variant histone H2Av, detected by a phosphospecific antibody against H2Av (γH2Av). (C and D) Without irradiation, HisC mutant cells did not show elevated γH2Av staining compared to wild type, indicating that mutant cells did not accumulate DNA damage. (E) Western blot for γH2Av using untreated (w/o HU) and HU-treated (HU) S2R+ cells as controls (two dilutions, 1 and 0.5) as well as HisC mutant embryos and wild type embryos at 5.5–6.5 hr AEL. α-Tubulin (α-Tub) was used as a loading control. HU treatment results in a significant increase of γH2Av, which was not observed in HisC mutant embryos. (F–H) BrdU pulse labelling for 15 min and staining with antibodies against BrdU and Cyclin B. lok, HisC double mutant embryos showed a similar phenotype as HisC mutant embryos (see Figure 1F). Dorsal up in (F–H), scale bars: 10 µm.
Figure 4—figure supplement 1. HisC mutant embryos show a moderate increase of γH2Av during S15 progression.
Figure 4—figure supplement 2. HisC mutant embryos do not show accumulation of DNA damage during early embryogenesis.
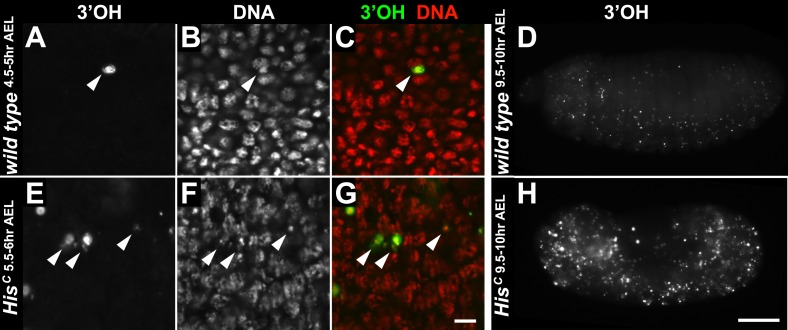
To independently address the extend of DNA damage accumulation in HisC mutant embryos, we used TUNEL assays which support that HisC mutant cells do not accumulate significant levels of DNA damage until much later in development (≥10 hr AEL) when these embryos die (Figure 4—figure supplement 2). Finally, we performed genetic tests using mutants of loki (lok), the Drosophila DNA damage checkpoint kinase chk2 (Xu et al., 2001). Homozygous lok mutants are viable and fertile. In contrast, lok, HisC double mutant embryos exhibited the HisC phenotype (Figure 4F–H). Hence, the cell cycle arrest in HisC mutant embryos is not mediated by the chk2-dependent DNA damage checkpoint pathway.
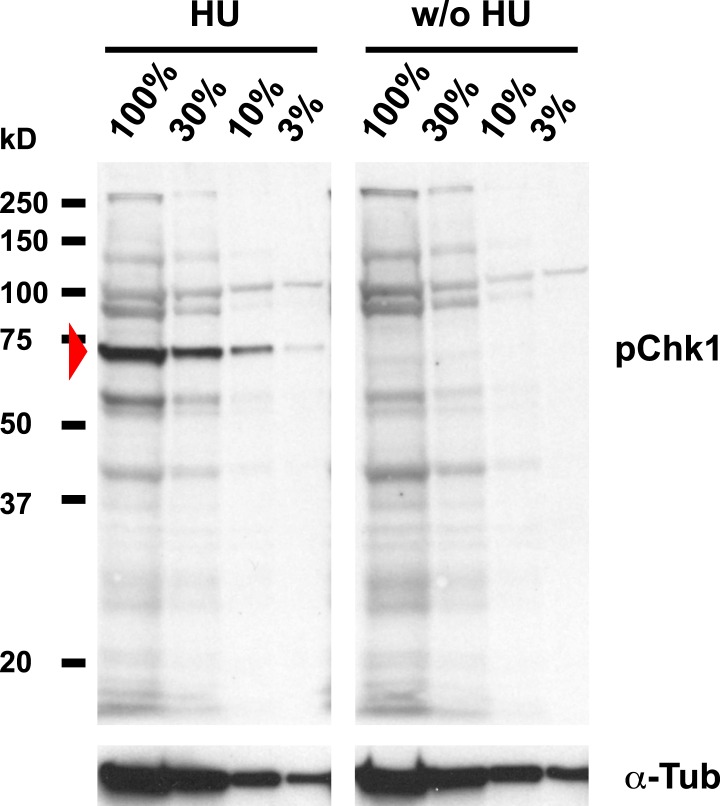
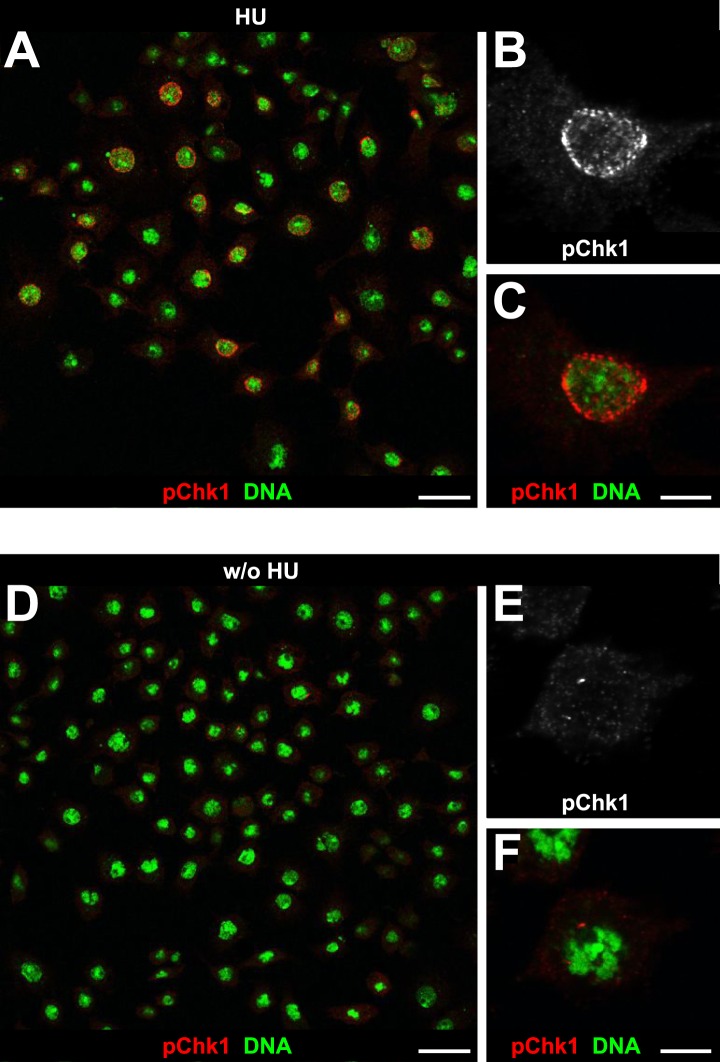
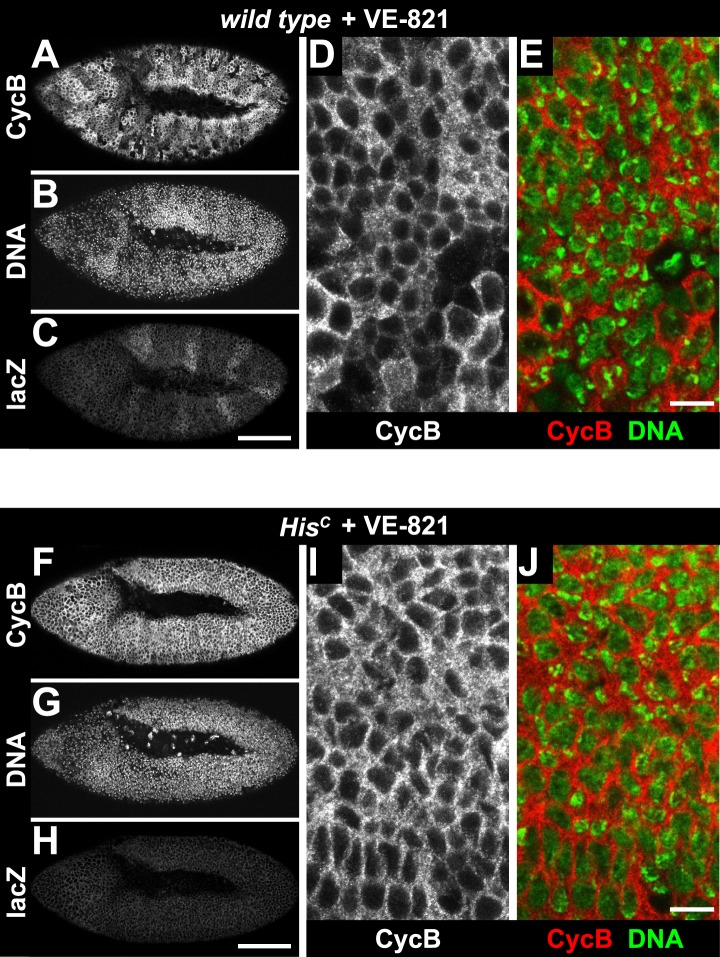
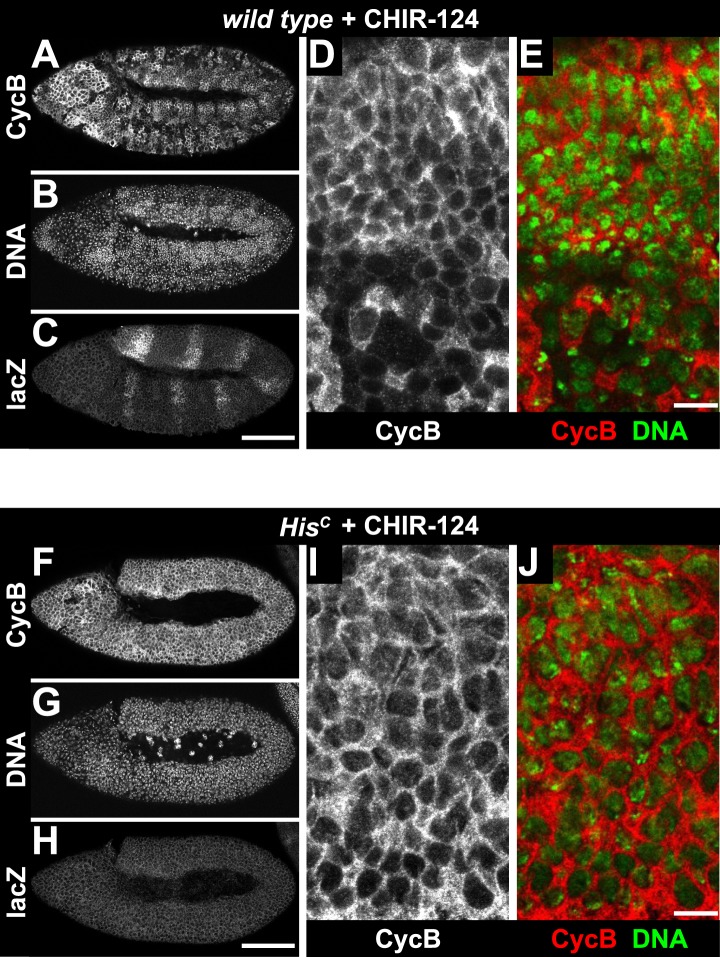
Mutations in the Drosophila ortholog of Chk1 (GRP) show developmental defects prior to cell cycle 15, excluding genetic experiments as we performed for lok (Fogarty et al., 1994; Su et al., 1999). Thus, we tested ATR/Chk1 checkpoint activation by using a phosphospecific antibody that recognizes the ATR-dependent phosphorylation of S345 in human Chk1 in response to replicative stress, for example, UV irradiation or HU treatment (Zhao and Piwnica-Worms, 2001). This antibody is expected to cross-react with Drosophila GRP due to sequence similarity and detected a single band in Western blots (Figure 5—figure supplement 1) as well as a clear signal in immunofluorescence (Figure 5—figure supplement 2) upon HU treatment of Drosophila S2R+ tissue culture cells. To test whether the ATR/Chk1 checkpoint is functional in HisC mutant embryos, we irradiated embryos with UV light (254 nm, UVC), which induces replication stress and replication fork uncoupling (Byun et al., 2005; Cimprich and Cortez, 2008). Wild type embryos and HisC mutant embryos accumulated phosphorylated GRP protein (pGRP) in response to UVC, showing that the checkpoint response is functional in the mutant embryos (Figure 5A–D). Without UVC treatment HisC mutant embryos did not display elevated pGRP levels as compared to wild type (Figure 5E–H), which was also verified by Western blotting of extracts from sorted HisC mutant and wild type sibling embryos (Figure 5I). In addition, treatment of HisC mutant embryos with the ATR inhibitor VE-821 (Prevo et al., 2012) or the Chk1 inhibitor CHIR-124 (Tse et al., 2007) did not result in a release of the cell cycle arrest (Figure 5—figure supplements 3 and 4). In addition to DSBs, replicative stress can induce phosphorylation of H2Av (Figure 4—figure supplement 1K–P), either directly by ATR dependent phosphorylation (Ward and Chen, 2001; Joyce et al., 2011) or through interconversion of single-stranded DNA generated at stalled replication forks into DSBs (Cimprich and Cortez, 2008). Consistent with the notion that the DNA damage checkpoints are functional in HisC mutant embryos we found accumulation of γH2Av in UVC-treated embryos to levels well above the background levels detected in untreated HisC mutant embryos in late S15 (Figure 5—figure supplement 5). Interestingly, UVC-treated embryos were able to enter M15 while displaying levels of γH2Av comparable or above to what we observed in untreated HisC mutant embryos (Figure 5—figure supplement 6). Taken together, these results strongly suggest that HisC mutant cells complete DNA replication in S phase without inducing significant DNA damage or replication stress and that the cell cycle arrest at the G2/M transition in HisC mutant cells is not mediated by the conventional S phase checkpoints.
Figure 5. HisC mutant cells do not activate the ATR/Chk1 DNA damage checkpoint.
(A–D) Wild type and HisC mutant cells responded to UV irradiation (UVC) by phosphorylation of the Drosophila Chk1 ortholog GRP, detected by a phosphospecific antibody for Chk1 (pChk1). Embryos were counterstained for DNA to visualize nuclei (B and D). (E–H) Without irradiation, HisC mutant cells did not show elevated staining for pChk1 compared to wild type, indicating that mutant cells did not activate the ATR/Chk1 checkpoint. (I) Western blot detecting pGRP by an antibody to phosphorylated Chk1 (pChk1) and α−Tubulin (α-Tub). Extracts were prepared from SR2+ tissue culture cells that were either untreated (w/o HU) or treated with HU (HU). Embryos were either sorted wild type controls or HisC mutants. Two dilutions (1 and 0.5) of each sample were loaded. Dorsal up in (A–H), scale bars: 10 µm.
Figure 5—figure supplement 1. Detection of pGRP by a phosphospecific antibody for Chk1 by Western blotting.
Figure 5—figure supplement 2. Detection of pGRP by a phosphospecific antibody for Chk1 by immunofluorescence.
Figure 5—figure supplement 3. Treatment of HisC mutant embryos with the ATR inhibitor VE-821 does not release the cell cycle arrest.
Figure 5—figure supplement 4. Treatment of HisC mutant embryos with the Chk1 inhibitor CHIR-124 does not release the cell cycle arrest.
Figure 5—figure supplement 5. UV irradiation induces phosphorylation of H2Av.
Figure 5—figure supplement 6. Mitotic entry with elevated γH2Av levels.
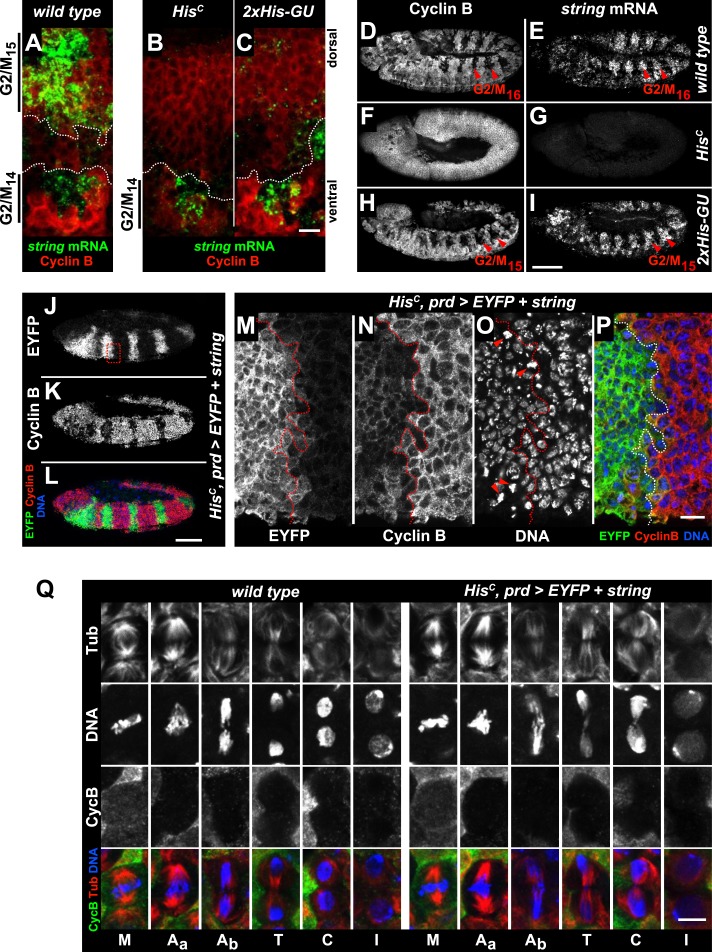
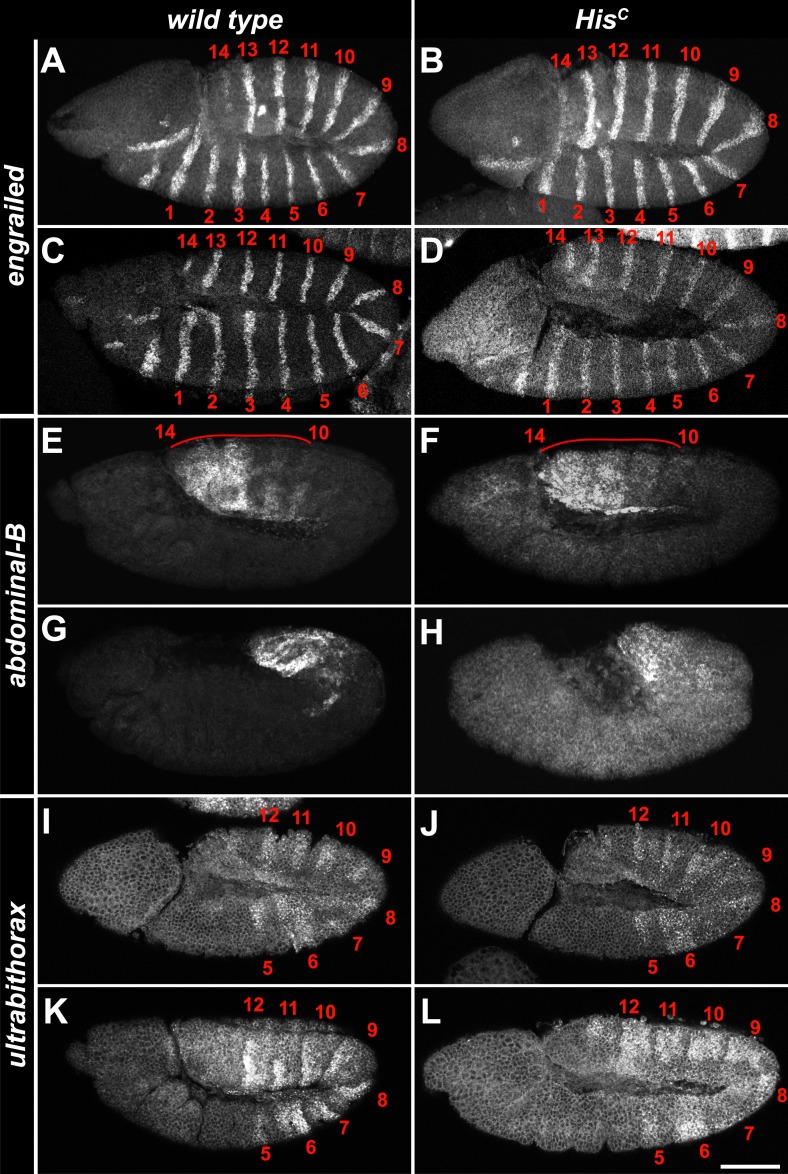
Progression from the G2 phase into mitosis critically depends on the dephosphorylation and activation of Cyclin/Cdk complexes, which is accomplished by a single gene in Drosophila embryos, encoding the CDC25 phosphatase String (Edgar and O'Farrell, 1990). In wild type, string mRNA accumulates during G2 phase and becomes rapidly degraded after cells exit mitosis (Edgar et al., 1994). string transcription is highly dynamic, dictates the pattern of cell divisions during embryogenesis and is controlled by the activity of developmentally regulated transcription factors binding to cis-regulatory sequences spread over >30 kb of the string locus (Edgar et al., 1994). In contrast to wild type embryos which accumulate string mRNA in G215 cells of the dorsal epidermis, HisC mutant embryos failed to accumulate string in the corresponding cells although they showed normal upregulation of string expression in ventral epidermal cells prior to M14 (Figure 6A,B). Given the complex developmental regulation of string, we asked whether string transcription is disturbed in HisC mutant embryos due to misregulation of key patterning genes. However, we observed normal temporal and spatial expression patterns of developmental genes such as the segmentation gene engrailed and the homeotic genes Ultrabithorax and Abdominal-B in HisC mutant embryos (Figure 6—figure supplement 1). Hence, the developmental programme progresses normally in HisC mutant embryos up to the late embryonic stage when they eventually die (Figure 4—figure supplement 2). Similar to HisC mutant embryos, string mRNA was not detectable in dorsal epidermal cells of 2xHis-GU embryos during their extended S15 (Figure 6C). However, when wild type embryos undergo M16 at 6.5–7 hr AEL (Figure 6D,E), HisC mutant embryos still failed to express string (Figure 6F,G) but 2xHis-GU embryos upregulated string expression (Figure 6H,I). This result suggests that the failure of HisC mutant embryos to upregulate string after they finished replication in S15 is not simply a consequence of their extended S phase but rather due to a surveillance mechanism that blocks the G2/M transition because chromatin assembly is not completed. It is interesting to note that the pattern of M15 in 2xHis-GU embryos closely resembled the pattern of M16 in wild type embryos (Günesdogan et al., 2010; Figure 6D,H). This observation provides further support that the developmental programme of these embryos can progress normally, as string mRNA expression readjusts once DNA replication is completed. Thus, sufficient histone supply, as provided by multiple copies of His-GUs, is critical to the coordination of the developmental and cell division programmes during wild type embryogenesis of Drosophila. The results also explain why higher eukaryotes, which undergo rapid mitotic cell divisions during embryonic development, contain multiple His-GUs in their genomes.
Figure 6. The cell cycle arrest of HisC mutant cells depends on string.
(A–I) string RNA in situ hybridisation (green in merge) and staining with an antibody against Cyclin B (red in merge). (A–C) Magnifications of epidermal cells from embryos at 4.5–5 hr AEL. (A) Wild type embryos upregulated string mRNA and accumulated Cyclin B in ventral epidermal cells during G214 (below dashed lines) and in dorsal epidermal cells during G215 (above dashed lines). In lateral cells with low levels of Cyclin B, string is not expressed during S15 (between dashed lines). (B) HisC mutant embryos failed to upregulate string in the dorsal epidermis (above dashed line). (C) 2xHis-GU embryos failed to upregulate string in the dorsal epidermis (above dashed line). (D–I) Whole mount embryos at 6.5–7 hr AEL. (D and E) Dorsal cells of wild type embryos progressed into G216, upregulated string mRNA and degraded Cyclin B during M16 (arrowheads). (F and G) HisC mutant embryos did not degrade Cyclin B and failed to accumulate string mRNA. (H and I) 2xHis-GU embryos accumulated string mRNA in G215 and degraded Cyclin B in M15 (arrowheads). (J–P) HisC mutant embryos expressing UAS-EYFP and UAS-string under the control of prd-GAL4 stained with antibodies against Cyclin B and EYFP. (M–P) Magnifications of epidermal cells from an embryo (boxed in J). (J–P) Epidermal cells within the EYFP, string expression domains degraded Cyclin B and entered M15/S16. Arrowheads in (O) show mitotic cells. (Q) Mitotic progression in wild type M15 and M15 of HisC mutant cells rescued by string expression visualized by staining for α-Tubulin (Tub), DNA, and Cyclin B (CycB). M: metaphase, Aa: anaphase-a, Ab: anaphase-b, T: telophase, C: cytokinesis, I: interphase. Dorsal up (A–C, M–P), scale bars 10 µm (A–C, M–P), 100 µm (D–L), 5 µm (Q).
Figure 6—figure supplement 1. The expression pattern of developmental genes is normal in HisC mutant embryos.
If string activity is the only limiting factor that restricts cell cycle progression in HisC mutant cells, its ectopic expression should drive the G2 to M transition. To test this hypothesis, HisC mutant embryos were forced to express string in a striped pattern under the control of the prd-GAL4 driver using the GAL4/UAS system (Brand and Perrimon, 1993), which was visualised by coexpression of a UAS-EYFP transgene. The string expressing HisC mutant cells entered M15, degraded Cyclin B and reaccumulated Cyclin B after mitosis, whereas cells lacking string expression remained arrested (Figure 6J–P). Mitotic progression in wild type embryos follows a stereotyped pattern characterized by (i) Cyclin B degradation and sister chromatid separation at the metaphase to anaphase transition, (ii) spindle elongation at the transition from anaphase-a into anaphase-b, (iii) the onset of chromatin decondensation in telophase, and eventually by (iv) cytokinesis (Figure 6Q). The string-induced mitosis in HisC mutant embryos was normal up to the metaphase to anaphase transition. During anaphase-b and telophase, however, we observed lagging chromosomes or chromatin bridges in almost all of the cells (95.8%, n = 24). These bridges were eventually resolved during cytokinesis, and the cells entered into interphase of cell cycle 16 (Figure 6Q). Together, these data indicate that string transcription is indeed the limiting downstream factor that restricts cell cycle progression in the absence of de novo histone synthesis. In summary, our study shows that DNA replication and histone availability are tightly coupled. Lack of de novo histone synthesis causes a string-dependent cell cycle arrest in G2 phase, suggesting a novel chromatin assembly checkpoint monitoring chromatin integrity.
Discussion
We used a recently generated null mutation for canonical histones to address the consequences of histone deprivation during metazoan development. In addition to canonical histones, eukaryotes express histone variants that can replace canonical histones in a specific genomic context (Banaszynski et al., 2010). Our results show that these histone variants do not compensate for the lack of canonical histone synthesis with regard to chromatin assembly and cell cycle progression. This could be due to insufficient expression of variant histones from their endogenous promoters as it has been shown for the variant histone H3.3, which can fully replace its canonical counterpart, histone H3, but only if it is expressed from within a histone gene unit like the canonical histone (Hödl and Basler, 2012). Alternatively, it could reflect structural divergence of the histone variants as in the case of His2Av (van Daal et al., 1988) and dBigH1 (Perez-Montero et al., 2013). It will be interesting to test whether individual histone mutations, like a mutation in H2B which does not have a variant histone in Drosophila (Talbert et al., 2012), will cause a similar cell cycle arrest as the histone null mutation HisC.
Our results provide evidence that canonical histone supply directly affects the rate of DNA synthesis (Figure 2L–N). This observation is in line with studies that targeted either histone chaperones (Hoek and Stillman, 2003; Ye et al., 2003; Nabatiyan and Krude, 2004; Groth et al., 2007; Takami et al., 2007) or histone mRNA through SLBP or FLASH (Zhao et al., 2004; Barcaroli et al., 2006; Mejlvang et al., 2014) to interfere with chromatin assembly in tissue culture cells. However, previous work on SLBP in multicellular organisms revealed pleiotropic effects (Sullivan et al., 2001; Lanzotti et al., 2002; Pettitt et al., 2002). Our data illustrate that an extension of the S phase duration caused by diminished histone supply allows a faithful completion of S phase and transition from G2 into M phase of the cell cycle. This S phase extension is likely to be caused by a direct effect of lowered histone availability on replication fork progression (Groth et al., 2007; Mejlvang et al., 2014) and not by a lack of origin firing, although we cannot exclude this possibility completely. It was previously shown that postblastodermal development in Drosophila embryos proceeds largely uncoupled from progression through cell cycles 14–16 (Edgar et al., 1994; Meyer et al., 2002). Therefore, histone availability limits S phase duration and appears to be a critical link between cell division and development.
In the absence of de novo histone synthesis, we find that cells arrest in G2 phase of the cell cycle without activating the known ATM/Chk2 and ATR/Chk1 checkpoints. This observation is in contrast to previous studies on CAF-1, which found that cells arrest in S phase and accumulate DNA damage (Hoek and Stillman, 2003; Ye et al., 2003). This discrepancy might in part be explained by the fact that histone chaperones also have a direct function in DNA repair (Schöpf et al., 2012); and thus, in the presence of an intact DNA repair/chromatin assembly machinery in HisC mutants, DNA is replicated without the accumulation of damage, even when histone supply is restricted to the parental load of histones. Alternatively, the accumulation of DNA damage in histone chaperone-depleted cells might be the consequence of a prolonged replication slow down, since it was shown that neither ATM/Chk2 nor ATR/Chk1 are activated as an immediate consequence of histone deprivation but only after prolonged incubation times (>48 hr) (Mejlvang et al., 2014). Based on our DNA quantification experiments, we found that the bulk of DNA replication in HisC mutants is completed by about 2 hr after entry into S phase, which might differ from the timeframe required to develop significant DNA damage. Interestingly, we find that HisC mutant cells become TUNEL positive by about 6 hr after they enter S phase 15, which might reflect secondary DNA damage and/or cell death. Nevertheless, we found a moderate increase of γH2Av staining during late S phase in HisC mutant embryos. Our data indicate, however, that cells that resolved UVC-induced DNA damage, and therefore entered mitosis can do so with levels of γH2Av comparable to those we observe in HisC mutants. Thus, it is plausible that the slight increase in γH2Av in HisC mutants could result from incomplete turnover of γH2Av rather than directly reflect DNA damage that could activate the S phase checkpoints. Turnover of γH2Av was shown to require the Tip60 chromatin-remodelling complex (Kusch et al., 2004), which may be affected by the altered chromatin structure in HisC mutants. Alternatively, H2Av was recently shown to be phosphorylated independent of ATM/ATR by the chromosomal tandem kinase JIL-1 (Jin et al., 1999; Thomas et al., 2014), which may also be influenced by the changed chromatin topology in HisC mutants.
Both, the ATM/Chk2 and ATR/Chk1 checkpoints are known to act on CDC25 phosphatases by phosphorylation and protein destabilization (Bartek and Lukas, 2007) and it was shown in Drosophila that string transcripts accumulate normally in embryos that suffered from DNA damage (Su et al., 2000). In contrast, we find that HisC mutant cells fail to accumulate string transcripts when arrested in G2. This finding was surprising since it was shown that the temporal and spatial expression pattern of string is essentially unchanged in embryos that are arrested in G2 by mutations in string or in mitotic Cyclins (Edgar et al., 1994). Thus, this difference is likely due to the failure of HisC mutant embryos to assemble chromatin, resulting in a diminished nucleosome density as shown by the presence of excess MNase hypersensitive DNA. Although we cannot rule out that the lower abundance of histone proteins itself directly contributes to the G2 arrest, this possibility seems unlikely since histone levels rapidly decrease in G2 cells where the chromatin assembly surveillance should act (Marzluff et al., 2008). It remains unclear how the presence of unassembled chromatin is linked to the regulation of string, but the effect is specific, since string transcript accumulation is the only limiting factor to overcome the G2 arrest in HisC-mutant embryos. The subsequent mitosis in HisC mutants is completed and cells enter into the next cell cycle. Given that HisC mutant cells enter mitosis with presumably about half of the nucleosomes present in wild type chromatin, the mitotic defects like lagging anaphase chromosomes appear surprisingly mild. These defects could reflect problems in loading of structural components that are required for chromosome condensation and sister chromatid cohesion, like Cohesins and Condensins, which are proposed to require contact to chromatin rather than naked DNA (Bernard et al., 2001; Nonaka et al., 2002; Tada et al., 2011).
Taken together, our results suggest that incomplete chromatin assembly is monitored by a novel surveillance mechanism that can block cell cycle progression at the G2/M transition in Drosophila. Our findings now pave the way to address key questions regarding the orchestration of DNA synthesis and chromatin formation as well as the control of chromatin integrity during cell cycle progression.
Materials and methods
Fly strains
Construction of Df(2L)HisC and histone transgenes was described previously (Günesdogan et al., 2010). The lokP6 Df(2L)HisC double mutant was constructed from lokP6 (Abdu et al., 2002) by meiotic recombination. In order to obtain maternal and zygotic loki mutant embryos, lokP6 Df(2L)HisC/lokP6 animals were crossed inter se. For string expression, we constructed Df(2L)HisC, P{UAS-2xEYFP}AH2 by meiotic recombination and generated embryos of the genotype Df(2L)HisC, P{UAS-2xEYFP}AH2/Df(2L)HisC, P{GAL4-prd.F}RG1/P{UAS-stg.N}4. For sorting of mutant embryos, we constructed Df(2L)HisC, P{GAL4-twi.2xPE}2 and selected EYFP-expressing embryos of the genotype Df(2L)HisC, P{UAS-2xEYFP}AH2/Df(2L)HisC, P{GAL4-twi.2xPE}2. In all other fly strains, Df(2L)HisC was heterozygous over the balancer chromosome CyO, P{ftz/lacB}E3 to identify wild type sibling embryos. 6xHis-GU flies were homozygous for the M{3xHisGU.wt}ZH-86Fb transgene. 2xHis-GU flies were homozygous for the M{1xHisGU.wt}ZH-86Fb transgene (Günesdogan et al., 2010). M{1xHisGU.wt}ZH-86Fb was constructed analogous to M{3xHisGU.wt}ZH-86Fb except that pENTRL4R1-T1 and pENTRR2L3-T2 that both contained ∼500 bp random DNA sequences instead of the His-GU were used for transgene construction.
Embryo collections and staining
Time matched embryonic collections were obtained by restricting egg deposition to 30′ and subsequent aging of embryos at 25°C. Fixation, antibody staining, BrdU incorporation, and RNA in situ hybridisation procedures were described previously (Günesdogan et al., 2010). Different from our standard protocol, we reduced the fixation time to 5 min and used 37% PFA instead of 4% PFA to fix embryos after UVC treatment. For experiments that required visualization of the tubulin cytoskeleton, we fixed as described (Karr and Alberts, 1986). Tissue culture cells were grown in µ-slide eight-well ibidi dishes (IBIDI, Martinsried, Germany) and fixed and treated in these wells by the same procedures as embryos (Günesdogan et al., 2010). Primary antibodies used here were: rabbit anti-Cyclin B (Jacobs et al., 1998) (1: 3000), chicken anti-β-Galactosidase (1:1000; Abcam, Cambridge, UK), H2Av pS137 (γH2Av; 1:500; Rockland, Gilbertsville, PA), rabbit anti-Chk1 phospho-S345 (1:300; Abcam), mouse anti-α Tubulin DM1A (1:500; Sigma-Aldrich, Taufkirchen, Germany), chicken anti-Galactosidase (1:1000; Abcam), sheep anti-Digoxigenin (1:2000; Roche, Mannheim, Germany), and a mouse anti-BrdU antibody (1:80; Becton Dickinson, Heidelberg, Germany). Secondary antibodies were: goat anti-mouse IgG coupled to Alexa Fluor488 (1:400; Life Technologies, Paisley, UK), goat anti-rabbit IgG Alexa Fluor488 (1:400; Life Technologies), goat anti-rabbit IgG Alexa Fluor633 (1:400; Life Technologies), goat anti-rabbit IgG Alexa Fluor647 (1:400; Life Technologies), goat anti-chicken IgY Alexa 568 (1:400; Life Technologies), goat anti-chicken IgY Cy3 (1:400; Jackson ImmunoResearch, West Grove, PA), donkey anti-sheep IgG Biotin-SP (1:500; Jackson ImmunoResearch). DNA was stained with DRAQ-5 (Biostatus Limited, Shepshed, UK) or Vectashield DAPI (Vector Laboratories, Burlingame, CA). Antisense string RNA was obtained by in vitro transcription and detected as described previously (Günesdogan et al., 2010) using biotinylated secondary antibodies. For detection, embryos were incubated for 45′ with ABC reagents (Vector Laboratories), followed by a 5′ incubation with TSA Flourescein reagents (Perkin Elmer, Waltham, MA) diluted 1:50.
Western blots
50–100 wild type or HisC mutant embryos were collected and lysed in 50 µl lysis buffer (50 mM Tris pH8, 150 mM NaCl, 0.5% Triton X-100, 1 mM MgCl2, 0.1 mM EDTA and protease inhibitor [Roche]). Phosphatase inhibitor (Roche) was added for Western blots stained for pChk1 and γH2Av, respectively. 50 µl 4× Laemmli buffer was added and the samples were sonicated for 7 min (30 s interval, power ‘low’) using a Bioruptor (Diagenode, Liège, Belgium) and centrifuged for 10 min at 4°C at maximum speed. The supernatants were denatured at 95°C for 10 min before loading on BioRad 4–20% Mini-PROTEAN TGX gels. PVDF membranes (Merck Millipore, Billerica, MA) were used for blotting. After blotting, membranes were washed with TBS-T (Tris Buffered Saline with 0.1% Tween-20) and blocked for 1 hr in TBS-T and 5% dry milk powder. Primary antibodies diluted in blocking buffer were added and incubated over night at 4°C. Membranes were washed 3× for 5′ in blocking buffer and secondary antibodies were added and incubated for 1 hr at room temperature. Membranes were washed 3× for 5′ in TBS-T. For fluorescent Western blots, images were acquired using an Odyssey infrared imaging system (LI-COR Biosciences, Lincoln, NE, USA). Quantifications were performed using the Image Studio Software (LI-COR Biosciences). For pChk1 and γH2Av Western blots, the ECL Western Blotting Kit (Pierce) was used to detect signals. Primary antibodies were: rabbit anti-Histone H3 (1:20000; Abcam), rabbit anti-Histone H2B (1:1000; Abcam), mouse anti-α Tubulin DM1A (1:2000; Sigma-Aldrich), rabbit anti-Chk1 phospho-S345 (1:1000; Abcam), and H2Av pS137 (γH2Av; 1:500; Rockland). Secondary antibodies were: goat anti-mouse IgG IRDye 680 (1:5000; LI-COR Biosciences), goat anti-rabbit IgG IRDye 800 (1:5000; LI-COR Biosciences), goat anti-rabbit IgG horseradish peroxidase conjugated, and goat anti-mouse IgG horseradish peroxidase conjugated (Sigma-Aldrich).
Micrococcal nuclease assays
70–100 control or HisC mutant embryos were collected and lysed in 50 µl micrococcal nuclease (MNase) digestion buffer (15 mM Tris, pH 7.5; 15 mM NaCl; 60 mM KCl; 0.34 M sucrose; 0.5 mM spermidine; 0.15 mM spermine; 1 mM PMSF; 0.5 mM DTT; 0.1% β-mercaptoethanol, protease inhibitor [Roche]). 200 µl MNase digestion buffer and 1 mM of CaCl2 was added. The suspension was divided in equal aliquots. 500 gel units of MNase (NEB, Hitchin, UK) were added and the samples were incubated at 32°C in a heat block. The reaction was stopped by adding 5 µl of both 0.5 M EDTA and 0.5 M EGTA. 2.5 µl of 10% SDS and 1 µl Proteinase K (10 mg/ml) was added and incubated at 50°C over night. The DNA was purified using Ampure XP beads (Beckman Coulter, Brea, CA). The samples were analysed using an Agilent 2200 Tapestation system with High Sensitivity D1000 screen tapes (Agilent Technologies, Wokingham, UK). Genomic DNA screen tapes (Agilent Technologies) were used to determine the input (time point 0′) concentration. The Tapestation analysis software was used for quantification of following fractions: 60–115 bp (<115 bp), 115–270 bp (mononucleosomes), 270–455 bp (dinucleosomes), 455–650 bp (trinucleosomes).
Irradiation of embryos, hydroxyurea treatment, treatment with inhibitors
Embryos were collected on apple-juice agar plates and aged to 4–5.5 hr AEL (w1118, wild type) or 6–7 hr AEL (HisC mutant embryos). This procedure yielded wild type embryos with cells in G215 and HisC mutant embryos that were arrested in cell cycle progression. After irradiation at 60 Gray in a Torrex 150D (Astrophysics Research Corp., City of Industry, CA) embryos from both collections were aged for 20 min on the apple-juice agar plates and mixed before fixation. UV irradiation (254 nm) was done at 200 mJ/cm2 as described (Zhou and Steller, 2003), and embryos were aged 45 min before fixation. HisC mutant embryos were identified in the stained samples based on their cell division arrest phenotype.
For inhibitor treatment, we used the same procedure as for BrdU incorporation (Günesdogan et al., 2010), replacing the BrdU with 10 µM VE-821 (Selleckchem, Houston, TX) or 10 µM CHIR-124 (Selleckchem) and incubation of 45 min at RT before fixation.
S2R+ tissue culture cell were cultured in Schneiders Medium (Life Technologies), with 10% Fetal Calf Serum. Hydroxyurea (Sigma-Aldrich) was added to a final concentration 10mM and incubated for 12 hr. Extracts were prepared in SDS sample buffer after addition of phosphatase inhibitors (PhosSTOP, Roche) and protease inhibitors (cOmplete EDTA-free, Roche) at approximately 5 × 108 cells per ml.
DNA quantification
Embryos were fixed by heat/methanol treatment and stained for Cyclin B. DNA was stained with DAPI (1:1000; Life Technologies). Cyclin B staining was used to distinguish homozygous mutant Df(2L)HisC embryos and control embryos and to define the cell cycle stage of each cell. Stacks of nuclei were acquired with a 63× objective and a 10× optical zoom with a Leica TCS-SP5 AOBS confocal laser-scanning microscope (z-axis increment: 0.1 µm, 8 bit images, 512 × 512 pixel, 400 Hz scan speed). The gain and offset were adjusted once and then used for one complete experiment, avoiding saturation. Fluorescent measurements were carried out using ImageJ software. To define nuclear circumferences, we used the ‘isodata thresholding’ algorithm followed by manual inspection. This threshold we used in a customized macro (Source code 1) that utilizes the ‘connected threshold grower’ plugin of the ImageJ 3D toolkit to determine nuclear staining intensities in all slices of the z-stack. The absolute nuclear fluorescence intensities were calculated by integration of individual nuclear fluorescence distributed over the image stack. For background detection five regions in between nuclei were analysed and their average was used for background subtraction.
TUNEL assay
TUNEL assays were done as described (Arama and Steller, 2006) with some deviations. DIG-labelled nucleotides were detected with a sheep anti-DIG antibody (Roche) and a donkey biotinylated anti-sheep secondary antibody (Jackson ImmunoResearch). For signal amplification, embryos were incubated for 45′ with ABC reagents (Vector Laboratories), followed by a 5′ incubation with TSA Flourescein reagents (Perkin Elmer) diluted 1:50. Embryos were mounted in ProlongGold (Life Technologies).
Acknowledgements
We thank Christian F Lehner for antibodies, comments, and suggestions; William Theurkauf for flies; Anja Schmidt for help with constructing fly stocks; Nick Brown for providing facilities; Benjamin Klapholz for technical advice; Lena Wartosch for help with Western blots. UG was supported by a fellowship from the Boehringer Ingelheim Fonds and a Marie Skłodowska-Curie fellowship. The work was supported by funds of the Max-Planck-Gesellschaft (HJ).
Funding Statement
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Funding Information
This paper was supported by the following grants:
Max-Planck-Gesellschaft FundRef identification ID: http://dx.doi.org/10.13039/501100004189 to Ufuk Günesdogan, Alf Herzig, Herbert Jäckle.
Boehringer Ingelheim Fonds FundRef identification ID: http://dx.doi.org/10.13039/501100001645 Graduate Student Fellowship to Ufuk Günesdogan.
European Commission FundRef identification ID: http://dx.doi.org/10.13039/501100000780 Marie Sklodowska-Curie fellowship to Ufuk Günesdogan.
Additional information
Competing interests
HJ: Herbert Jäckle has been Vice President of the Max Planck Society–one of eLife's three founding funders–and has attended until recently several meetings of eLife's board of directors.
The other authors declare that no competing interests exist.
Author contributions
UG, Conception and design, Acquisition of data, Analysis and interpretation of data, Drafting or revising the article.
AH, Conception and design, Acquisition of data, Analysis and interpretation of data, Drafting or revising the article.
HJ, Analysis and interpretation of data, Drafting or revising the article.
Additional files
References
- Abdu U, Brodsky M, Schüpbach T. 2002. Activation of a meiotic checkpoint during Drosophila oogenesis regulates the translation of Gurken through Chk2/Mnk. Current Biology 12:1645–1651. doi: 10.1016/S0960-9822(02)01165-X [DOI] [PubMed] [Google Scholar]
- Alabert C, Groth A. 2012. Chromatin replication and epigenome maintenance. Nature Reviews Molecular Cell Biology 13:153–167. doi: 10.1038/nrm3288 [DOI] [PubMed] [Google Scholar]
- Annunziato AT. 2012. Assembling chromatin: the long and winding road. Biochimica et Biophysica Acta 1819:196–210. doi: 10.1016/j.bbagrm.2011.07.005 [DOI] [PubMed] [Google Scholar]
- Arama E, Steller H. 2006. Detection of apoptosis by terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling and acridine orange in Drosophila embryos and adult male gonads. Nature Protocols 1:1725–1731. doi: 10.1038/nprot.2006.235 [DOI] [PubMed] [Google Scholar]
- Banaszynski LA, Allis CD, Lewis PW. 2010. Histone variants in metazoan development. Developmental Cell 19:662–674. doi: 10.1016/j.devcel.2010.10.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barcaroli D, Bongiorno-Borbone L, Terrinoni A, Hofmann TG, Rossi M, Knight RA, Matera AG, Melino G, De Laurenzi V. 2006. FLASH is required for histone transcription and S-phase progression. Proceedings of the National Academy of Sciences of USA 103:14808–14812. doi: 10.1073/pnas.0604227103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartek J, Lukas J. 2007. DNA damage checkpoints: from initiation to recovery or adaptation. Current Opinion in Cell Biology 19:238–245. doi: 10.1016/j.ceb.2007.02.009 [DOI] [PubMed] [Google Scholar]
- Bernard P, Maure JF, Partridge JF, Genier S, Javerzat JP, Allshire RC. 2001. Requirement of heterochromatin for cohesion at centromeres. Science 294:2539–2542. doi: 10.1126/science.1064027 [DOI] [PubMed] [Google Scholar]
- Brand A, Perrimon N. 1993. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118:401–415 [DOI] [PubMed] [Google Scholar]
- Byun TS, Pacek M, Yee MC, Walter JC, Cimprich KA. 2005. Functional uncoupling of MCM helicase and DNA polymerase activities activates the ATR-dependent checkpoint. Genes & Development 19:1040–1052. doi: 10.1101/gad.1301205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cimprich KA, Cortez D. 2008. ATR: an essential regulator of genome integrity. Nature Reviews Molecular Cell Biology 9:616–627. doi: 10.1038/nrm2450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar BA, Lehman DA, O'Farrell PH. 1994. Transcriptional regulation of string (cdc25): a link between developmental programming and the cell cycle. Development 120:3131–3143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar BA, O'Farrell PH. 1990. The three postblastoderm cell cycles of Drosophila embryogenesis are regulated in G2 by string. Cell 62:469–480. doi: 10.1016/0092-8674(90)90012-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty P, Kalpin RF, Sullivan W. 1994. The Drosophila maternal-effect mutation grapes causes a metaphase arrest at nuclear cycle 13. Development 120:2131–2142 [DOI] [PubMed] [Google Scholar]
- Gaillard PH, Martini EM, Kaufman PD, Stillman B, Moustacchi E, Almouzni G. 1996. Chromatin assembly coupled to DNA repair: a new role for chromatin assembly factor I. Cell 86:887–896. doi: 10.1016/S0092-8674(00)80164-6 [DOI] [PubMed] [Google Scholar]
- Green CM, Almouzni G. 2003. Local action of the chromatin assembly factor CAF-1 at sites of nucleotide excision repair in vivo. The EMBO Journal 22:5163–5174. doi: 10.1093/emboj/cdg478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groth A, Corpet A, Cook AJ, Roche D, Bartek J, Lukas J, Almouzni G. 2007. Regulation of replication fork progression through histone supply and demand. Science 318:1928–1931. doi: 10.1126/science.1148992 [DOI] [PubMed] [Google Scholar]
- Günesdogan U, Jäckle H, Herzig A. 2010. A genetic system to assess in vivo the functions of histones and histone modifications in higher eukaryotes. EMBO Reports 11:772–776. doi: 10.1038/embor.2010.124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hödl M, Basler K. 2012. Transcription in the absence of histone H3.2 and H3K4 methylation. Current Biology 22:2253–2257. doi: 10.1016/j.cub.2012.10.008 [DOI] [PubMed] [Google Scholar]
- Hoek M, Stillman B. 2003. Chromatin assembly factor 1 is essential and couples chromatin assembly to DNA replication in vivo. Proceedings of the National Academy of Sciences of USA 100:12183–12188. doi: 10.1073/pnas.1635158100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houlard M, Berlivet S, Probst A, Quivy JP, Héry P, Almouzni G, Gérard M. 2006. CAF-1 is essential for heterochromatin organization in pluripotent embryonic cells. PLOS Genetics 2:e181. doi: 10.1371/journal.pgen.0020181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs HW, Knoblich JA, Lehner CF. 1998. Drosophila Cyclin B3 is required for female fertility and is dispensable for mitosis like Cyclin B. Genes & Development 12:3741–3751. doi: 10.1101/gad.12.23.3741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin Y, Wang Y, Walker DL, Dong H, Conley C, Johansen J, Johansen KM. 1999. JIL-1: a novel chromosomal tandem kinase implicated in transcriptional regulation in Drosophila. Molecular Cell 4:129–135. doi: 10.1016/S1097-2765(00)80195-1 [DOI] [PubMed] [Google Scholar]
- Joyce EF, Pedersen M, Tiong S, White-Brown SK, Paul A, Campbell SD, McKim KS. 2011. Drosophila ATM and ATR have distinct activities in the regulation of meiotic DNA damage and repair. The Journal of Cell Biology 195:359–367. doi: 10.1083/jcb.201104121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karr TL, Alberts BM. 1986. Organization of the cytoskeleton in early Drosophila embryos. The Journal of Cell Biology 102:1494–1509. doi: 10.1083/jcb.102.4.1494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim UJ, Han M, Kayne P, Grunstein M. 1988. Effects of histone H4 depletion on the cell cycle and transcription of Saccharomyces cerevisiae. The EMBO Journal 7:2211–2219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knoblich JA, Sauer K, Jones L, Richardson H, Saint R, Lehner CF. 1994. Cyclin E controls S phase progression and its down-regulation during Drosophila embryogenesis is required for the arrest of cell proliferation. Cell 77:107–120. doi: 10.1016/0092-8674(94)90239-9 [DOI] [PubMed] [Google Scholar]
- Kusch T, Florens L, Macdonald WH, Swanson SK, Glaser RL, Yates JR, III, Abmayr SM, Washburn MP, Workman JL. 2004. Acetylation by Tip60 is required for selective histone variant exchange at DNA lesions. Science 306:2084–2087. doi: 10.1126/science.1103455 [DOI] [PubMed] [Google Scholar]
- Lanzotti DJ, Kaygun H, Yang X, Duronio RJ, Marzluff WF. 2002. Developmental control of histone mRNA and dSLBP synthesis during Drosophila embryogenesis and the role of dSLBP in histone mRNA 3' end processing in vivo. Molecular and Cellular Biology 22:2267–2282. doi: 10.1128/MCB.22.7.2267-2282.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehner CF, O'Farrell PH. 1990. The roles of Drosophila cyclins A and B in mitotic control. Cell 61:535–547. doi: 10.1016/0092-8674(90)90535-M [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madigan JP, Chotkowski HL, Glaser RL. 2002. DNA double-strand break-induced phosphorylation of Drosophila histone variant H2Av helps prevent radiation-induced apoptosis. Nucleic Acids Research 30:3698–3705. doi: 10.1093/nar/gkf496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marzluff WF, Wagner EJ, Duronio RJ. 2008. Metabolism and regulation of canonical histone mRNAs: life without a poly(A) tail. Nature Reviews Genetics 9:843–854. doi: 10.1038/nrg2438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mejlvang J, Feng Y, Alabert C, Neelsen KJ, Jasencakova Z, Zhao X, Lees M, Sandelin A, Pasero P, Lopes M, Groth A. 2014. New histone supply regulates replication fork speed and PCNA unloading. The Journal of Cell Biology 204:29–43. doi: 10.1083/jcb.201305017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer CA, Kramer I, Dittrich R, Marzodko S, Emmerich J, Lehner CF. 2002. Drosophila p27Dacapo expression during embryogenesis is controlled by a complex regulatory region independent of cell cycle progression. Development 129:319–328 [DOI] [PubMed] [Google Scholar]
- Nabatiyan A, Krude T. 2004. Silencing of chromatin assembly factor 1 in human cells leads to cell death and loss of chromatin assembly during DNA synthesis. Molecular and Cellular Biology 24:2853–2862. doi: 10.1128/MCB.24.7.2853-2862.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonaka N, Kitajima T, Yokobayashi S, Xiao G, Yamamoto M, Grewal SI, Watanabe Y. 2002. Recruitment of cohesin to heterochromatic regions by Swi6/HP1 in fission yeast. Nature Cell Biology 4:89–93. doi: 10.1038/ncb739 [DOI] [PubMed] [Google Scholar]
- O'Farrell PH, Edgar BA, Lakich D, Lehner CF. 1989. Directing cell division during development. Science 246:635–640. doi: 10.1126/science.2683080 [DOI] [PubMed] [Google Scholar]
- Perez-Montero S, Carbonell A, Moran T, Vaquero A, Azorin F. 2013. The embryonic linker histone H1 variant of Drosophila, dBigH1, regulates zygotic genome activation. Developmental Cell 26:578–590. doi: 10.1016/j.devcel.2013.08.011 [DOI] [PubMed] [Google Scholar]
- Pettitt J, Crombie C, Schümperli D, Müller B. 2002. The Caenorhabditis elegans histone hairpin-binding protein is required for core histone gene expression and is essential for embryonic and postembryonic cell division. Journal of Cell Science 115:857–866 [DOI] [PubMed] [Google Scholar]
- Prevo R, Fokas E, Reaper PM, Charlton PA, Pollard JR, McKenna WG, Muschel RJ, Brunner TB. 2012. The novel ATR inhibitor VE-821 increases sensitivity of pancreatic cancer cells to radiation and chemotherapy. Cancer Biology & Therapy 13:1072–1081. doi: 10.4161/cbt.21093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quivy JP, Roche D, Kirschner D, Tagami H, Nakatani Y, Almouzni G. 2004. A CAF-1 dependent pool of HP1 during heterochromatin duplication. The EMBO Journal 23:3516–3526. doi: 10.1038/sj.emboj.7600362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schöpf B, Bregenhorn S, Quivy JP, Kadyrov FA, Almouzni G, Jiricny J. 2012. Interplay between mismatch repair and chromatin assembly. Proceedings of the National Academy of Sciences of USA 109:1895–1900. doi: 10.1073/pnas.1106696109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shermoen AW, McCleland ML, O'Farrell PH. 2010. Developmental control of late replication and S phase length. Current Biology 20:2067–2077. doi: 10.1016/j.cub.2010.10.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su TT, Campbell SD, O'Farrell PH. 1999. Drosophila grapes/CHK1 mutants are defective in cyclin proteolysis and coordination of mitotic events. Current Biology 9:919–922. doi: 10.1016/S0960-9822(99)80399-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su TT, Walker J, Stumpff J. 2000. Activating the DNA damage checkpoint in a developmental context. Current Biology 10:119–126. doi: 10.1016/S0960-9822(00)00300-6 [DOI] [PubMed] [Google Scholar]
- Sullivan E, Santiago C, Parker ED, Dominski Z, Yang X, Lanzotti DJ, Ingledue TC, Marzluff WF, Duronio RJ. 2001. Drosophila stem loop binding protein coordinates accumulation of mature histone mRNA with cell cycle progression. Genes & Development 15:173–187. doi: 10.1101/gad.862801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada K, Susumu H, Sakuno T, Watanabe Y. 2011. Condensin association with histone H2A shapes mitotic chromosomes. Nature 474:477–483. doi: 10.1038/nature10179 [DOI] [PubMed] [Google Scholar]
- Takami Y, Ono T, Fukagawa T, Shibahara K, Nakayama T. 2007. Essential role of chromatin assembly factor-1-mediated rapid nucleosome assembly for DNA replication and cell division in vertebrate cells. Molecular Biology of the Cell 18:129–141. doi: 10.1091/mbc.E06-05-0426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talbert PB, Ahmad K, Almouzni G, Ausio J, Berger F, Bhalla PL, Bonner WM, Cande WZ, Chadwick BP, Chan SW, Cross GA, Cui L, Dimitrov SI, Doenecke D, Eirin-López JM, Gorovsky MA, Hake SB, Hamkalo BA, Holec S, Jacobsen SE, Kamieniarz K, Khochbin S, Ladurner AG, Landsman D, Latham JA, Loppin B, Malik HS, Marzluff WF, Pehrson JR, Postberg J, Schneider R, Singh MB, Smith MM, Thompson E, Torres-Padilla ME, Tremethick DJ, Turner BM, Waterborg JH, Wollmann H, Yelagandula R, Zhu B, Henikoff S. 2012. A unified phylogeny-based nomenclature for histone variants. Epigenetics & Chromatin 5:7. doi: 10.1186/1756-8935-5-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas CJ, Kotova E, Andrake M, Adolf-Bryfogle J, Glaser R, Regnard C, Tulin AV. 2014. Kinase-mediated changes in nucleosome conformation trigger chromatin decondensation via poly(ADP-ribosyl)ation. Molecular Cell 53:831–842. doi: 10.1016/j.molcel.2014.01.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tse AN, Rendahl KG, Sheikh T, Cheema H, Aardalen K, Embry M, Ma S, Moler EJ, Ni ZJ, Lopes de Menezes DE, Hibner B, Gesner TG, Schwartz GK. 2007. CHIR-124, a novel potent inhibitor of Chk1, potentiates the cytotoxicity of topoisomerase I poisons in vitro and in vivo. Clinical Cancer Research 13:591–602. doi: 10.1158/1078-0432.CCR-06-1424 [DOI] [PubMed] [Google Scholar]
- van Daal A, White EM, Gorovsky MA, Elgin SC. 1988. Drosophila has a single copy of the gene encoding a highly conserved histone H2A variant of the H2A.F/Z type. Nucleic Acids Research 16:7487–7497. doi: 10.1093/nar/16.15.7487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward IM, Chen J. 2001. Histone H2AX is phosphorylated in an ATR-dependent manner in response to replicational stress. The Journal of Biological Chemistry 276:47759–47762. doi: 10.1074/jbc.M009785200 [DOI] [PubMed] [Google Scholar]
- Xu J, Xin S, Du W. 2001. Drosophila Chk2 is required for DNA damage-mediated cell cycle arrest and apoptosis. FEBS Letters 508:394–398. doi: 10.1016/S0014-5793(01)03103-9 [DOI] [PubMed] [Google Scholar]
- Yang XC, Burch BD, Yan Y, Marzluff WF, Dominski Z. 2009. FLASH, a proapoptotic protein involved in activation of caspase-8, is essential for 3' end processing of histone pre-mRNAs. Molecular Cell 36:267–278. doi: 10.1016/j.molcel.2009.08.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye X, Franco AA, Santos H, Nelson DM, Kaufman PD, Adams PD. 2003. Defective S phase chromatin assembly causes DNA damage, activation of the S phase checkpoint, and S phase arrest. Molecular Cell 11:341–351. doi: 10.1016/S1097-2765(03)00037-6 [DOI] [PubMed] [Google Scholar]
- Zhao H, Piwnica-Worms H. 2001. ATR-mediated checkpoint pathways regulate phosphorylation and activation of human Chk1. Molecular and Cellular Biology 21:4129–4139. doi: 10.1128/MCB.21.13.4129-4139.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, McKillop-Smith S, Müller B. 2004. The human histone gene expression regulator HBP/SLBP is required for histone and DNA synthesis, cell cycle progression and cell proliferation in mitotic cells. Journal of Cell Science 117:6043–6051. doi: 10.1242/jcs.01523 [DOI] [PubMed] [Google Scholar]
- Zhou L, Steller H. 2003. Distinct pathways mediate UV-induced apoptosis in Drosophila embryos. Developmental Cell 4:599–605. doi: 10.1016/S1534-5807(03)00085-6 [DOI] [PubMed] [Google Scholar]