Summary
Background
Neurocysticercosis causes a substantial burden of seizure disorders worldwide. Treatment with either praziquantel or albendazole has suboptimum efficacy. We aimed to establish whether combination of these drugs would increase cysticidal efficacy and whether complete cyst resolution results in fewer seizures. We added an increased dose albendazole group to establish a potential effect of increased albendazole concentrations.
Methods
In this double-blind, placebo-controlled, phase 3 trial, patients with viable intraparenchymal neurocysticercosis were randomly assigned to receive 10 days of combined albendazole (15 mg/kg per day) plus praziquantel (50 mg/kg per day), standard albendazole (15 mg/kg per day), or increased dose albendazole (22·5 mg/kg per day). Randomisation was done with a computer generated schedule balanced within four strata based on number of cysts and concomitant antiepileptic drug. Patients and investigators were masked to group assignment. The primary outcome was complete cyst resolution on 6-month MRI. Enrolment was stopped after interim analysis because of parasiticidal superiority of one treatment group. Analysis excluded patients lost to follow-up before the 6-month MRI. This trial is registered with ClinicalTrials.gov, number NCT00441285.
Findings
Between March 3, 2010 and Nov 14, 2011, 124 patients were randomly assigned to study groups (41 to receive combined albendazole plus praziquantel [39 analysed], 43 standard albendazole [41 analysed], and 40 increased albendazole [38 analysed]). 25 (64%) of 39 patients in the combined treatment group had complete resolution of brain cysts compared with 15 (37%) of 41 patients in the standard albendazole group (rate ratio [RR] 1·75, 95% CI 1·10–2·79, p=0·014). 20 (53%) of 38 patients in the increased albendazole group had complete cyst resolution at 6-month MRI compared with 15 (37%) of 41 patients in the standard albendazole group (RR 1·44, 95% CI 0·87–2·38, p=0·151). No significant differences in adverse events were reported between treatment groups (18 in combined treatment group, 11 in standard albendazole group, and 19 in increased albendazole group).
Interpretation
Combination of albendazole plus praziquantel increases the parasiticidal effect in patients with multiple brain cysticercosis cysts without increased side-effects. A more efficacious parasiticidal regime without increased treatment-associated side-effects should improve the treatment and long term prognosis of patients with neurocysticercosis.
Funding
National Institute of Neurological Disorders and Stroke (NINDS), National Institutes of Health.
Introduction
Neurocysticercosis caused by Taenia solium is regarded as the most frequent cause of acquired epilepsy worldwide.1,2 In the lifecycle of this parasite, human beings harbour the adult tapeworm in their intestines and are the only definitive host. Both human beings and pigs can act as intermediate hosts by harbouring the larvae or cysticerci in their tissues.3 The infection and resulting disease is highly endemic in all developing countries where pigs are raised as a food source.1 Neurocysticercosis is now also increasingly diagnosed in industralised countries because of migration and travel from endemic zones.4
Cyst death after antiparasitic treatment is a result of not only the direct action of the drug, but also of an attack by the host immune system in response to the release of antigens caused by treatment-associated damage, which is most pronounced during the initial days or weeks after the start of antiparasitic treatment.5 Antiparasitic treatment of patients with viable intraparenchymal brain cysts seems to improve the prognosis of their seizure disorders.6–9 However, antiparasitic treatment has suboptimum efficacy, killing roughly 65% of parasites and obtaining complete cyst resolution (no viable parasites remaining) in less than 40% of patients after a course of praziquantel or albendazole.10,11
Praziquantel is a pyrazinoisoquinoline derivative, of which the main pharmacological effects include muscle contractions, paralysis, and tegumentary damage, whereas albendazole is a benzimidazole, of which the main method of action is through selective degeneration of cytoplasmic microtubules resulting in energy depletion, disrupted cell division, and altered glucose intake.12,13 We postulated that combinination of these two antiparasitic drugs would improve the destruction of brain cysts without affecting patient safety, and designed a clinical study to compare treatment with albendazole alone with combined albendazole plus praziquantel. An initial pharmacokinetic substudy showed increased serum albendazole concentrations in patients receiving combination treatment compared with concentrations in those receiving albendazole alone.14 This difference in concentrations was presumed to be due to a pharmacokinetic interaction between praziquantel and albendazole. Hence, the question arose whether any reported superiority of the albendazole–praziquantel combination in elimination of viable cysts would be due to an additional cysticidal effect of praziquantel or due to increased albendazole concentrations arising from an interaction with praziquantel. Therefore, in response to a suggestion by our data and safety monitoring board, we added an increased dose albendazole study group so that we could establish whether any recorded increase in efficacy was a result of increased albendazole concentrations or to the direct action of praziquantel. We also compared seizure rates during periods before and after complete cyst resolution, to assess whether complete cyst resolution resulted in a decrease in seizure frequency.
Methods
Study design and participants
For this double-blind, placebo-controlled, randomised phase 3 clinical trial, we recruited patients from the Instituto Nacional de Ciencias Neurologicas, and the national hospitals Cayetano Heredia, Eduardo Rebagliati, and Guillermo Almenara, Lima, Peru. We did the study at the CNS Parasitic Diseases Research Unit, Universidad Peruana Cayetano Heredia, Lima, Peru.
Inclusion criteria were age between 16 and 65 years; one to 20 viable neurocysticercosis cysts; serological confirmation on western blot; a diagnosis of epilepsy secondary to neurocysticercosis with one or more spontaneous seizures within the previous year but not longer than 10 years, or more than 10 years with seizures but limited to only two to nine total seizure episodes (patients with more than 10 years with seizures were excluded to reduce the hypothetical chances of further seizures initiating from secondary foci, thus not reflecting the effects of antiparasitic treatment, patients with fewer than ten seizures were allowed to be enrolled on the basis that such few events in a long time were unlikely to result in this type of seizures); willingness to remain in hospital for 2 weeks; use of an effective contraception method; normal laboratory values for haematocrit, platelets, white blood cells, and glucose; normal or decreased aspartate transaminase, alanine transaminase, and creatinine (mildly abnormal values such as slightly decreased blood cell counts or increased aspartate transaminase or alanine transaminase up to 2·5 times the normal limit for the reference laboratory were eligible on individual assessment); negative purified protein derivative (PPD) or PPD-positive with negative tuberculosis smears; and negative fecal examination for Taenia spp eggs or Strongyloides spp larvae. Patients should have been on an appropriate antiepileptic drug regimen for at least 1 week before randomisation.
Exclusion criteria were primary generalised seizures; generalised status epilepticus in the past year; a type of neurocysticercosis that could expose the patient to increased risk during the study, specifically basal subarachnoid neurocysticercosis, intraventricular cysts, cysts in brainstem, cysts larger than 30 mm diameter, or untreated ocular cysticercosis (patients with one cyst <20 mm in a lateral ventricle, without hydrocephalus or intracranial hypertension, were eligible); intracranial hypertension that is persistent or progressive or radiologically defined by CT or MRI; previous treatment with albendazole or praziquantel in the past year; active pulmonary tuberculosis; active hepatitis; systemic disease other than neurocysticercosis; unstable condition; hypersensitivity to albendazole or praziquantel; concurrent treatment with cimetidine or theophylline; chronic alcohol or drug misuse; or unwillingness or inability to provide informed consent or to undergo CT scan or MRI examinations.
The study and informed consent forms were approved by the main institutional review board at the Universidad Peruana Cayetano Heredia (IRB Code 51070, FWA 00002541) and at the institutional review boards of every recruiting site. An independent study Data and Safety Monitoring Board reviewed all safety and efficacy data twice per year.
Randomisation and masking
Patients were randomly assigned (1:1:1) to receive combined albendazole (15 mg/kg per day) plus praziquantel (50 mg/kg per day), standard albendazole (15 mg/kg per day), or increased albendazole (22·5 mg/kg per day). Randomisation was done with a computer-generated schedule balanced within four strata based on numbers of cysts (one to two or >three) and concomitant antiepileptic drug (phenytoin or carbamazepine) to prevent confounding due to of these variables. Participants and staff giving the interventions, assessing the outcome, and analysing the data were all masked to group assignments. Patients in the combined treatment group and the standard albendazole group received additional albendazole placebo to allow patient and investigator masking.
Procedures
Treatment was given in hospital. 1 day before antiparasitic treatment, patients were started on dexamethasone (0·1 mg/kg per day) to control intracranial inflammation, with ranitidine (300 mg per day) to prevent gastrointestinal symptoms.
Cell counts, liver function, glucose, creatinine, and electrolytes were monitored at days 4, 7, 11, and 30. Patients were discharged from the hospital on about day 15 and had follow-up visits on days 21, 30, 60, and 90, and then every 3 months until day 540. A follow-up MRI was done on day 180. Patients whose cysts did not completely resolve were offered a further course of antiparasitic treatment with additional 6-month post-treatment MRI. Brain CT was done on day 360 to assess residual calcifications.
Cysticidal efficacy was measured at 6-month MRI as the proportion of patients with complete cyst destruction (no viable parasites after the initial course of therapy) and was compared between treatment groups. No standard definition of what constitutes a resolved cyst exists. To provide a robust criterion, we defined the absence of discernible hyperintense contents on T2 MRI as the marker of final parasite degeneration. Lesions with T2-hyperintense cystic contents were deemed viable cysts, independent of the presence or degree of perilesional inflammation.15 Degenerating cysts, affected by antiparasitic treatment but with persistent T2-hyperintense contents, were not judged to be resolved. This definition is highly conservative and probably underestimates the true efficacy of antiparasitic treatment in all treatment groups.
All radiological assessments were made masked to treatment group. Follow-up MRIs were assessed first by an independent neuroradiologist whose report was limited to establishing the presence or absence of persistent unresolved cysts to guide retreatment decisions. After the last patient had 6-month follow-up MRI, the principal investigator and one neurologist (both experts in neurocysticercosis diagnosis, neither of whom participated in seizure registry or patient management) reviewed the follow-up scans and the neuroradiologist’s assessment of complete cyst resolution. Any discrepancies and all scans showing persisting viable lesions were sent back to the neuroradiologist for confirmation of the number and characteristics of surviving cysts. Treatment group assignment was unmasked only after all analyses had been done and revised by the study data and safety monitoring board.
A diagnosis of seizure is mainly made by interview of the patient or a witness of the event. Patients were instructed to recognise and report compatible events and to record these events in a provided diary, until day 540 of follow-up. A study neurologist reviewed the diary at every visit for neurological events including seizures, and interviewed the patient or witness to establish whether or not the event constituted a seizure. Seizures were classified as per the guidelines of the Classification and Terminology of the International League Against Epilepsy from 1981.16 A seizure was recorded as partial or generalised mainly on the basis of the presence or not of loss of consciousness. Seizure relapse was assessed as the cumulative frequency of seizure events compared during periods before and after complete cyst resolution, and also as a dichotomous outcome. Early seizures (those occurring in the initial 60 days after each course of antiparasitic treatment) were excluded from the main analysis to avoid confusion due to the short-term inflammatory effect associated with treatment-induced cyst destruction.
Outcomes
The primary outcome was the difference in the proportions of patients with complete cyst resolution at 6 months between study groups. Secondary outcomes were the proportions of resolved cysts between study groups and the effect of complete cyst resolution on seizure frequency. Two additional exploratory outcomes were the number of cysts remaining after antiparasitic treatment and the number of severe adverse events.
Statistical analysis
The analysis plan strictly followed the primary and secondary outcomes and statistical methods proposed a priori in the protocol, under close oversight by the Data and Safety Monitoring Board. The primary outcome was assessed with a χ² test. The efficacy rate is presented as the difference between binomial proportions with CIs estimated by conventional exact methods.
The proportions of resolved cysts between treatment groups was measured with a binomial family generalised linear model with a log link. Statistical significance was determined with likelihood ratio tests. Stratified analysis was used to explore the potential effect of number of cysts, choice of antiepileptic drug, presence of seizures, and previous antiparasitic treatment. The size of the test or α level was set to 5%, with 95% CIs, with exact binomial estimates when necessary.
The effect of complete cyst resolution on seizure frequency was established by comparing the frequency of seizure events in follow-up days 61–540 during periods before complete cyst destruction (termed persistent viable infection) and after complete cyst destruction (termed resolved infection). For this cohort analysis, the main exposure factor was whether a patient had resolved all their cysts or not. For this analysis, cysts were assumed to resolve in the initial 2 months after the preceding course of antiparasitic treatment. Follow-up from days 61–180 was allocated to resolved infection or persistent viable infection according to the MRI results. The results of further image examinations were used in the same way as those of the 6-month MRI. The initial 60 days after every retreatment course were censored, and further follow-up to the point of the next image was allocated to infection outcome according to the image results.
The initial 60 days after every course of antiparasitic treatment were censored to account for the acute post-treatment period of cyst degeneration and its effect on seizures. Cyst resolution after antiparasitic treatment takes a variable time, but most of it occurs in the initial weeks. In the pig model, resolved cysts are barely noticeable after 10–12 weeks.5,17 In early trials of anti parasitic treatment, resolved cysts were not visible on brain CT after 3 months.10 In these studies, increased frequency of seizures in the first week of treatment was evident and prompted the concomitant use of steroids. In a randomised trial by our group, we recorded an increase in seizure activity during the first 30 days in patients given albendazole, which turned into reduced seizure frequency after this point.6 This trial also showed that cysts do not resolve by themselves in 6 months. Thus, if a cyst was not visible at 6 months, the most probably scenario is that the cyst resolved in the initial weeks after the onset of antiparasitic treatment, during the censored period (days 1–60).
Seizure rates were computed as incidence density variables (number of seizure-days/time elapsed, for which a seizure-day is a day when one or more seizures occurred) and were analysed with Poisson time-to-event models. Analyses were done perperiod instead of perindividual, in view that some individuals had both periods with and without viable cysts. The protocol planned an intention-to-treat analysis, but because only four patients were lost to follow-up before the main outcome assessment (6-month MRI), so-called as treated results are mainly reported and intention-to-treat results are given for comparison. Patients lost to follow-up contributed with follow-up time until information about the outcome was available. Seizures that happened during days 1–60 after treatment were not included in this analysis because they were probably due to acute antiparasitic-induced inflammation. This analytical approach was designed and executed as proposed in the initial study protocol.
A sample size of 80 individuals per treatment group (n=240) was designed to assess the frequencies of complete cyst resolution in the combination group compared with that in the standard albendazole group, and also in the increased albendazole group compared with in the standard albendazole group. An interim analysis for efficacy was done by an independent biostatistician after the main study outcome had been established in half the required sample size. Although no quantitative rules to stop the trial were predefined on the basis of efficacy assessments, an interim analysis with 50% of the planned total was set to be done with Lan-DeMets spending function with O’Brien-Fleming type boundaries, rejecting the null hypothesis of no difference if the p value was less than 0·003 in a two-sided test with a 0·05 CI. This stopping boundary was conservative, and appropriate for this trial in which the default action is to continue the study. Other stopping rules included frequent serious adverse events of the same sign or symptom, increased frequency of serious adverse events in either treatment group that are possibly related to the study treatment, low accrual, or external information that make the trial unnecessary or unethical. The study personnel were trained to recognise, classify, monitor, and report all adverse events with predesigned case report forms.
The trial is registered with ClinicalTrials.gov, number NCT00441285.
Role of the funding source
Other than suggestions from the Data and Safety Monitoring Board appointed by the National Institute of Neurological Disorders and Stroke, the funders had no role in study design, data collection, data analysis, data interpretation, or decision to submit the paper for publication. All authors had full access to all the data in the study and share the final responsibility for the decision to submit for publication.
Results
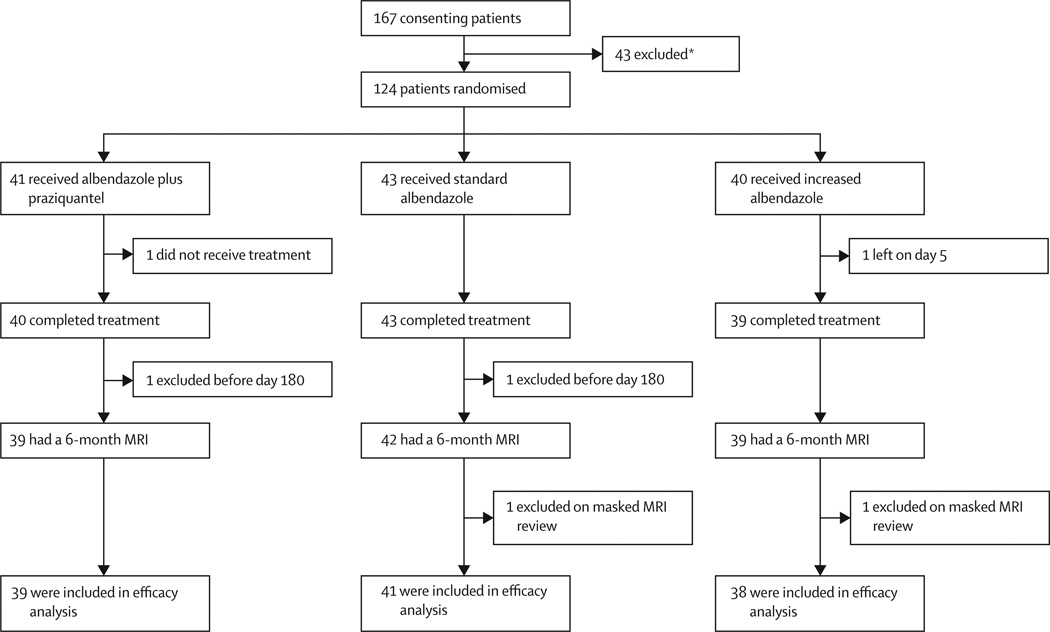
Between March 3, 2010 and Nov 14, 2011, 124 patients were randomly assigned to study groups (41 to receive combined albendazole plus praziquantel, 43 standard albendazole, and 40 increased albendazole). Enrolment was halted by the study data and safety monitoring board after interim analyses showed parasiticidal superiority of one of the treatment groups. Up to this point, 167 patients (100 men, 67 women) had entered the screening phase in whom 43 patients were excluded (figure 1). Of 124 patients who were randomly assigned a study group, 76 were men and 48 were women with a mean age of 34·6 years (SD 13·1, range 16–68). 63 patients (51%) had one to two cysts and 61 (49%) had more than two cysts. 64 (52%) were taking carbamazepine and 60 (48%) were taking phenytoin. Patients had been symptomatic for a mean time of 55·4 months (SD 58·4, range 0·1–392). 103 (83%) patients had had seizures with generalisation, whereas 90 (73%) had partial seizures. 21 (17%) patients had had previous antiparasitic treatment and one (<1%) had had previous neurosurgery. Table 1 shows the baseline characteristics of patients in the three treatment groups. Enrolled and not enrolled patients were statistically similar in terms of sex (p=0·528), age (p=0·292), and length of illness (p=0·809; data not shown).
Figure 1. Trial profile.
*Two individuals had two reasons for exclusion each. Reasons for exclusion: 14 had no viable cysts, eight had more than 20 cysts, three had subarachnoid neurocysticercosis, one had cysts in the eye, two had cysts near the optic nerve, three had cysts in the brain stem, three had haematological abnormalities, one had active tuberculosis, one had more than 10 years with seizures, three had recent therapy with albendazole, one had a history of status epilepticus, and five refused to continue in the study.
Table 1.
Baseline characteristics of enrolled patients
| Albendazole plus praziquantel (n=41) |
Standard albendazole (n=43) |
Increased albendazole (n=40) |
|
|---|---|---|---|
| Men | 26 (63%) | 29 (67%) | 21 (53%) |
| Age (years) | |||
| Mean (SD) | 34 (14) | 35 (13) | 34 (12) |
| Range | 18–68 | 16–63 | 17–65 |
| Number of cysts at baseline | |||
| Mean (SD) | 5 (5) | 4 (4) | 4 (4) |
| Median (range) | 3 (1–19) | 3 (1–18) | 3 (1–18) |
| One or two cysts | 21 (51%) | 22 (51%) | 20 (50%) |
| Three or more cysts | 20 (49%) | 21 (49%) | 20 (50%) |
| Antiepileptic drugs at study onset | |||
| Carbamazepine | 21 (51%) | 22 (51%) | 21 (53%) |
| Phenytoin | 20 (49%) | 21 (49%) | 19 (47%) |
| Had seizures with generalisation | 34 (83%) | 34 (79%) | 35 (88%) |
| Time with seizures (months) | |||
| Mean (SD) | 43 (33) | 55 (61) | 69 (72) |
| Range | 1–121 | 01–294 | 2–392 |
| Previous antiparasitic treatment | 5 (12%) | 6 (14%) | 10 (25%) |
Data are n (%), unless otherwise indicated.
One randomised patient abandoned the study before treatment onset, another left the study during antiparasitic treatment, and two other patients were lost to follow-up before their 6-month MRI control (figure 1). Masked review of baseline and control MRI scans by an independent neuroradiologist excluded two other cases with single small lesions, which were judged to be neuroepithelial cysts of the choroid space, not parasitic in origin (one each in the standard and increased albendazole group). 118 patients were included in the efficacy analysis: 39 in the albendazole plus praziquantel group, 41 in the standard albendazole group, and 38 in the increased albendazole group (figure 1). From day 180 onwards, 18 patients were excluded because of poor compliance with study visits and antiepileptic medication (n=14), for open treatment (n=2), or for refusal to continue in the study (n=2). Seven exclusions were from the standard group—five for poor compliance, one for open treatment, and one for refusal to continue; another seven from the increased albendazole group—five for poor compliance, one for open treatment, and one for refusal to continue; and four from the combination group due to poor compliance.
For cysticidal efficacy, 25 (64%) of 39 patients who received combined albendazole plus praziquantel had complete resolution of all brain cysts at 6 months after treatment compared with 15 (14%) of 41 patients who received standard albendazole (RR 1·75, 95% CI 1·10–2·79, p=0·014). 20 (53%) of 38 patients in the increased albendazole group had complete resolution compared with the standard albendazole group, although this difference was not statistically significant (RR 1·44, 95% CI 0·87–2·38, p=0·151). The proportion of patients with complete cyst resolution in each treatment group was similar for those with only one or two cysts (table 2), but was significantly higher in the combination group for patients with three or more cysts compared with those in the standard albendazole group (RR 14·37, 95% CI 2·07–99·68, p=0·007) and increased albendazole group (RR 2·74, 95% CI 1·21–6·20, p=0·016; overall p<0·0001; table 2, figure 2). We also did an intention-to-treat analysis imputing the potential status of the four patients who did not complete follow-up. The differences in proportions of individuals with complete cyst resolution between the combined treatment group and the increased albendazole compared with standard albendazole were similar to those obtained in the as-treated analyses (RR 1·78, 95% CI 1·11–2·83, p=0·012; 1·51, 0·92–2·48, p=0·101). Retreatment was indicated for the 58 patients whose lesions had not completely resolved (14 in the combination group, 26 in the standard albendazole group, and 18 in the increased albendazole group), and 56 of them received a further course of antiparasitic treatment.
Table 2.
Cysticidal efficacy by treatment group and number of cysts
| Albendazole plus praziquantel (n=39) |
Standard albendazole (n=41) |
Increased albendazole (n=38) |
Overall p value |
|
|---|---|---|---|---|
| One to two cysts | ||||
| Viable cysts at baseline | 27 | 22 | 23 | 0·284 |
| Mean per patient (SD) | 1·4 (0·6) | 1·1 (0·3) | 1·3 (0·6) | 0·281 |
| Cyst range | 1–3* | 1–2 | 1–3* | .. |
| Number of patients | 20 | 20 | 18 | .. |
| Viable cysts at day 180 | 10 | 6 | 3 | 0·237 |
| Mean per patient (SD) | 0·5 (07) | 0·3 (0·5) | 0·2 (04) | 0·162 |
| Cysts resolved | 17/27 (63%) | 16/22 (73%) | 20/23 (87%) | 0·141 |
| Patients cured | 12/20 (60%) | 14/20 (70%) | 15/18 (83%) | 0·287 |
| Three or more cysts | ||||
| Viable cysts at baseline | 171 | 142 | 142 | 0·179 |
| Mean per patient (SD) | 9·0 (4·8) | 6·8 (4·2) | 7·1 (44) | 0·245 |
| Cyst range | 3–19 | 3–18 | 3–18 | .. |
| Number of patients | 19 | 21 | 20 | .. |
| Viable cysts at day 180 | 11 | 112 | 74 | <0·0001 |
| Mean per patient (SD) | 0·6 (1·0) | 5·3 (4·2) | 3·7 (3·1) | 0·0001 |
| Cysts resolved | 160/171 (94%) | 30/142 (21%) | 68/142 (48%) | <0·0001 |
| Patients cured | 13/19 (68%) | 1/21 (5%) | 5/20 (25%) | <0·0001 |
Data are n/N (%), unless otherwise indicated.
In each of these groups, an additional cyst was identified in the MRI of one patient after they had been randomised in the one to two cysts stratum.
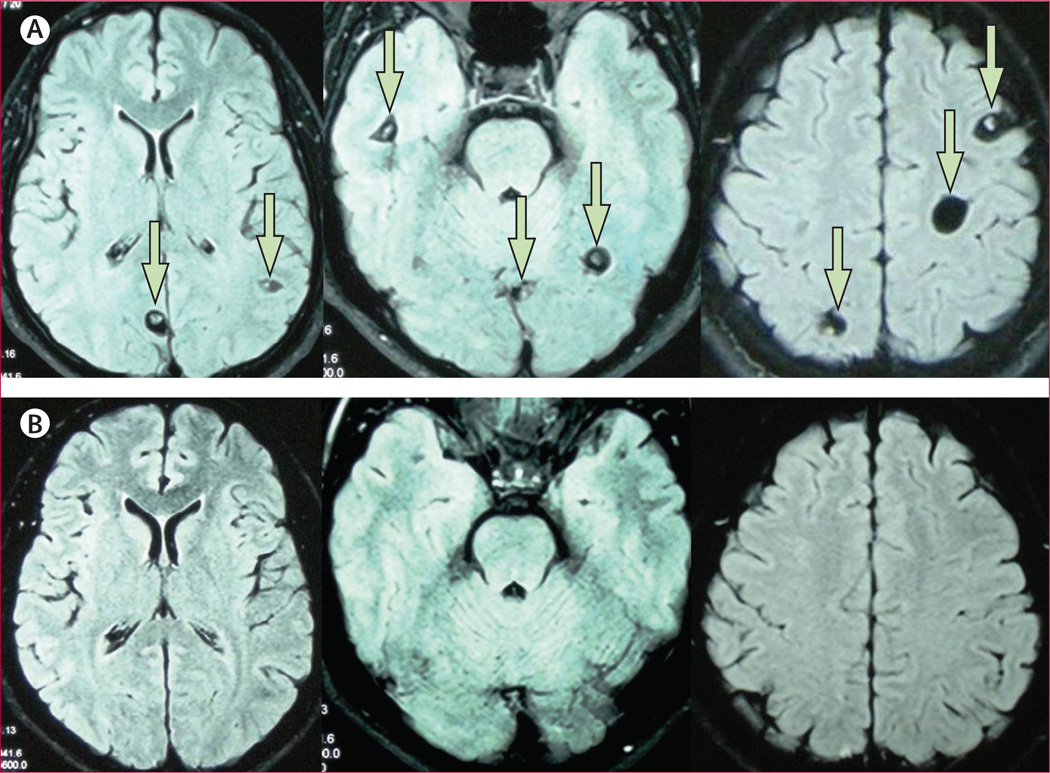
Figure 2. Fluid like attenuated inversion recovery MRI images of a patient with multiple brain cysts before (A) and 6 months after (B) combined antiparasitic therapy with praziquantel and albendazole.
Scans in the upper row are pre-treatment axial images (arrows point to viable cysts) and scans in the bottom row are the corresponding cuts in the 6-month post treatment control MRI scan.
The proportions of cysts resolved followed similar patterns. Combined albendazole plus praziquantel resolved more cysts than did standard albendazole (RR 3·19, 95% CI 2·03–5·01, p<0·0001), and than did increased albendazole (RR 1·68, 95% CI 1·25–2·24, p=0·0005; table 2). All three groups differed significantly from each other (p<0·05). In patients with only one or two cysts, we noted no significant differences between groups, although combined treatment had the lowest efficacy compared with standard albendazole (p=0·468) and compared with increased albendazole (p=0·064; table 2). However, in patients with three or more cysts, combined treatment was significantly more effective than standard albendazole (RR 4·43, 95% CI 2·37–8·26, p<0·0001) and increased albendazole (RR 1·95, 95% CI 1·35–2·82, p<0·0001; table 2)
In intention-to-treat analysis imputing the potential status of the four patients who did not complete the follow-up of the study, all comparisons of the proportion of cysts resolved between the three groups remained highly significant (p<0·0001; data not shown).
297 seizure events (days with seizures) occurred during days 61–540 that were eligible for analyses of the effect of post-treatment cyst resolution (283 partial and 15 generalised, one with both types of seizures). Seizures were less frequent in the periods after complete cyst resolution (resolved infection periods, table 3). The overall seizure rate per year in the resolved infection period was 0·84 and in the persisting viable infection periods was 4·39 (table 3). This difference was significant for partial seizures (table 3). Because the longitudinal nature of this analysis did not account for the three-group randomised trial design, we repeated the analyses after adjustment by trial group, and obtained a slightly larger reduction in partial seizures in patients whose cysts had completely resolved (RR 0·15, 95% CI 0·12–0·20). The rates of generalised seizures per year did not differ significantly between persistent infection and resolved infection periods (table 3).
Table 3.
Seizure frequency and risk by cure status
| Seizure events per day (n) |
Seizure rates per year |
Seizure rate ratios (95% CI) |
|||||
|---|---|---|---|---|---|---|---|
| Persistent infection |
Resolved infection |
Persistent infection |
Resolved infection |
Persistent infection |
Resolved infection | p value | |
| Overall period* | |||||||
| All seizures | 225 | 72 | 4.39 | 0.84 | 1.00 | 0.19 (0.15–0·25) | <0.0001 |
| Partial | 217 | 66 | 4.24 | 0.77 | 1.00 | 0.18 (0.14–0·24) | <0.0001 |
| Generalised | 9 | 6 | 018 | 0.07 | 1.00 | 0.40 (0.14–1.12) | 0.081 |
| Days 61–180† | |||||||
| All seizures | 112 | 32 | 5·88 | 1.62 | 1.00 | 0.28 (0.19–0.41) | <0.0001 |
| Partial | 108 | 30 | 5·67 | 1·52 | 1·00 | 0.27 (0.18–0.40) | <0.0001 |
| Generalised | 5 | 2 | 0·26 | 010 | 1·00 | 0.39 (0.08–1·99) | 0.256 |
| Days 181–540‡ | |||||||
| All seizures | 113 | 40 | 3.51 | 0.61 | 1.00 | 0.17 (0.12–0.25) | <0.0001 |
| Partial | 109 | 36 | 3.38 | 0.55 | 1.00 | 0.16 (0.11–0.24) | <0.0001 |
| Generalised | 4 | 4 | 012 | 0.06 | 1.00 | 0.49 (0.12–1.95) | 0.311 |
18 714 patients per days in non-cured periods and 31 273 patients per days in cured periods.
6960 patients per days in non-cured periods and 7200 patients per days in cured periods.
11 762 patients per days in non-cured periods and 24 073 patients per days in cured periods.
In additional analyses of the proportion of patients with at least one seizure during follow-up, we noted no significant differences between patients whose cysts had completely resolved at 6 months compared with those with persistent viable infection (11 [18%] of 60 vs 15 [26%] of 58, p=0·324). We also noted no differences between treatment groups in the proportions of patients with seizures during the early post-treatment period (days 1–60: 18 [46%] of 39 patients in combination treatment group, 16 [39%] of 41 in standard albendazole group, and 17 [45%] of 38 in increased albendazole group; p=0·792) or in days 61–180 (ten [26%] in combination treatment group, seven [17%] in standard albendazole group, and nine [24%] in increased albendazole group; p=0·624).
We recorded no significant differences in the proportions of severe adverse events by group of therapy (table 4). Most of the reportable severe adverse events corresponded to elective hospital readmissions after a seizure event. In these instances the study team preferred to keep the patients in hospital rather than send them home, which could have increased the response time if further seizures occurred.
Table 4.
Number of severe adverse events by study group
| Albendazole plus praziquantel (n=41) |
Standard albendazole (n=43) |
Increased albendazole (n=40) |
p value* | |
|---|---|---|---|---|
| Seizures | 9 (22%) | 5 (12%) | 6 (15%) | 0.563 |
| Headache | 3 (7%) | 2 (5%) | 6 (15%) | 0.234 |
| Pregnancy | 2 (5%) | 1 (2%) | 3 (8%) | 0.550 |
| Drug-induced hepatitis | 0 | 1 (2%) | 1 (3%) | 0.607 |
| Other† | 4 (10%) | 2 (5%) | 3 (8%) | 0.667 |
Data are n (%).
Kruskal-Wallis rank sum test.
One of the following: dizziness, fever, interruption of therapy and prolongation of admission for personal reasons, intracranial hypertension, motor vehicle accident, spontaneous abortion, urinary tract infection, and vomiting.
Discussion
Findings from this randomised controlled trial have shown the increased antiparasitic efficacy of an albendazole plus praziquantel regimen, and that further seizures are less frequent in individuals with complete cyst resolution after antiparasitic treatment (panel). Neurocysticercosis, particularly intraparenchymal brain cysticercosis, is associated with seizures and epilepsy in most of the world.1,22 The parasitic larvae establish and survive in the brain for a variable period (often years or sometimes decades) because of the protection provided by the blood–brain barrier and by use of a series of active immune evasion mechanisms.23,24 Eventually, the immune equilibrium is broken and the host’s immune system launches a focalised inflammatory reaction that kills the parasitic larvae. The result is either complete disappearance of the cyst or persistence of a calcified scar. Neurocysticercosis-associated seizures can occur at any stage, but they seem more frequent during the period of cyst degeneration.1,23,25
Cysticidal efficacy of present regimens is suboptimum, because they destroy roughly 65% of parasites and clear all cysts in less than 40% of patients after a course of treatment.10,11 This trial showed that combined albendazole plus praziquantel—and increased albendazole (although to a less degree)—kills more cysts than does standard-dose albendazole. This increase in efficacy did not occur in patients with one to two cysts. Cyst damage is likely to lead to the release of parasite antigens and boost the immune response attack, in a process that involves an effect of praziquantel. We selected the combination of albendazole and praziquantel on the basis of their individual efficacy against cysticercosis,6–11 different mechanisms of action,12,13 findings from previous in-vitro and animal studies,26,27 series of treatment of hydatid disease,28 and findings from early studies in neurocysticercosis.18,19 In our initial pharmacokinetics study, albendazole concentrations increased by roughly 50% in the combined treatment group,14 thus we added an increased albendazole group to this trial. The dose and maximum dose were increased by 50%, which in individuals greater than 80 kg approximates to the dose usually applied in Latin America.6,7,10 The increased albendazole regimen did better than did the standard 800 mg per day regimen, but did not reach the efficacy of combined albendazole plus praziquantel.
Antiparasitic treatment of neurocysticercosis accelerates the process of cyst degeneration at the cost of local inflammation,6,25 thus doubts about its effect in seizure prognosis were raised.29–32 The increased antiparasitic efficacy of the combined regimen led to early interruption of enrolment, and seizure analysis in this trial was subsequently underpowered. Despite this limitation, complete cyst resolution was significantly associated with fewer seizures in follow-up, although the reduction in the proportion of patients with at least one additional seizure was not statistically significant. We chose a longitudinal analysis to account for differential follow-up in patients whose cysts resolved completely compared with patients with persistent viable cysts. Other potential outcomes such as the proportion of patients with at least one seizure (or its reciprocal, the proportion of people who remained seizure-free) do not account for this effect. We had previously shown fewer seizures with generalisation in patients receiving antiparasitic treatment than in untreated controls.6 To our knowledge, we provide the first evidence that complete cyst resolution is associated with fewer seizure relapses, which supports the use of antiparasitic treatment as part of the standard of care in viable parenchymal neurocysticercosis. The effect of cyst resolution to reduce the likelihood of future seizures is only partial, and the remainder is probably attributable to other contributing factors such as the baseline frequency and type of seizures, numbers of lesions, extent of local inflammation and scarring, and compliance with antiepileptic treatment. Some cysts could have resolved after day 60, in which case their survival period after day 60 would have been misclassified as resolved infection days, therefore diluting a potential association between persistent infections and further seizures.
Antiparasitic treatment for neurocysticercosis is not free of risks and might be dangerous for some types of neurocysticercosis, such as patients with heavy cyst burdens, or when post-treatment inflammation might trigger or worsen intracranial hypertension or hydrocephalus.33 Combined albendazole plus praziquantel treatment was quite safe in this cohort of patients with one to 20 parenchymal cysts, although larger series are needed to provide further safety data. Our study introduces the combination of albendazole plus praziquantel as an improved antiparasitic regimen for patients with multiple cysts, and shows that complete cyst resolution is associated with fewer seizure relapses after the initial 18 months. Improved antiparasitic treatment should also serve to treat complicated disease, including cisternal or spinal subarachnoid neurocysticercosis, which still cause substantial mortality.1,23,25
Panel: Research in context.
Systematic review
We searched PubMed for articles published in any language between Jan 1, 1980, and Nov 1, 2013, with the search terms “albendazole” or “praziquantel” combined with “Taenia solium”, “cysticercosis”, or “neurocysticercosis”. We identified two trials that used combined albendazole and praziquantel in neurocysticercosis. The first study18 was non-randomised, and reported a roughly two-fold improvement in headache, epilepsy, and MRI findings with combination of albendazole plus praziquantel, and no significant side-effects, although specific doses or side-effects were not listed in the paper. The second study19 was a randomised, placebo-controlled trial that tested the added benefit of 1-day praziquantel treatment in children with a different type of neurocysticercosis (single degenerating cysticercus). The trial showed non-significant benefits in terms of lesion resolution, and no differences in seizures or side-effects. Anecdotal references to non-systematic use of combined albendazole and praziquantel can be found in clinical series.20,21
Interpretation
Albendazole alone or praziquantel alone have suboptimum antiparasitic efficacy, and no controlled studies have assessed their combined use for multicystic parenchymal neurocysticercosis, which are a common presentation in most of the world. Data for the effect of cyst destruction on later seizure relapses is insufficient because of the partial cysticidal effect and the large clinical variability of neurocysticercosis. We previously showed a beneficial effect of antiparasitic treatment on subsequent seizure incidence.6 This study shows that with a combination of albendazole plus praziquantel in patients with multiple brain cysts, antiparasitic efficacy is increased without increased side-effects, and that complete cyst destruction is associated with fewer seizure relapses. Future studies should confirm the safety of this regimen, and assess other factors that contribute to seizure relapses in parasitologically cured patients with neurocysticercosis. A more efficacious parasiticidal regime without increased treatment-associated side-effects should improve the treatment and long term prognosis of patients with neurocysticercosis.
Acknowledgments
This study was funded by the National Institute of Neurological Disorders and Stroke (NINDS), National Institutes of Health, USA, through grant NS054805, with part support from the Fogarty International Center/NIH (training grants D43 TW001140 and TW007393). HHG is supported by a Wellcome Trust International Senior Research Fellowship in Public Health and Tropical Medicine. We thank members of the study data and safety monitoring board for their knowledgeable and constructive suggestions that helped to improve the trial design and performance; Robert H Gilman for his guidance, the enormous effort performed by our clinical coordination team (M Vera, K Fernandez, J Del Carpio, C Castillo, and C Arias), our clinical laboratory team (S Rodriguez, Y Castillo, E Perez, P Berrios, and K Arteaga); and the advice from our consultants A Delgado-Escueta, O H Del Brutto, J Horton, M T Medina, T E Nash, and O Takayanagui. The views expressed in this article are those of the authors only and do not necessarily reflect the official policy or position of the Department of the Navy, Department of Defense, nor the US Government.
Footnotes
Contributors
HHG, AGL, JAB, and EJP conceived and designed the study. IG, MZ, HS, MG, LR, EN, and HU did the trial. AGL and HGG analysed the data. HHG and JAB wrote the report. HHG, IG, AGL, JAB, MZ, DE, HS, MG, LR, EN, HU, and EP reviewed and approved the report.
Declaration of interests
We declare no competing interests. AGL is an employee of the US Government. This work was prepared as part of his duties. Title 17 USC. §105 provides that ‘Copyright protection under this title is not available for any work of the United States Government.’ Title 17 USC. §101 defines a US Government work as a work prepared by a military service member or employee of the US Government as part of that person’s official duties.
References
- 1.Garcia HH, Del Brutto OH. Neurocysticercosis: updated concepts about an old disease. Lancet Neurol. 2005;4:653–661. doi: 10.1016/S1474-4422(05)70194-0. [DOI] [PubMed] [Google Scholar]
- 2.No authors listed. Commission on Tropical Diseases of the International League Against Epilepsy. Relationship between epilepsy, tropical diseases. Commission on Tropical Diseases of the International League Against Epilepsy. Epilepsia. 1994;35:89–93. [PubMed] [Google Scholar]
- 3.Flisser A. Taeniasis and cysticercosis due to T solium . In: Sun T, editor. Progress in clinical parasitology. New York: CRC Press; 1994. pp. 77–116. [PubMed] [Google Scholar]
- 4.Coyle CM, Mahanty S, Zunt JR, et al. Neurocysticercosis: neglected but not forgotten. PLoS Negl Trop Dis. 2012;6:e1500. doi: 10.1371/journal.pntd.0001500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gonzalez AE, Falcon N, Gavidia C, et al. Timeresponse curve of oxfendazole in the treatment of swine cysticercosis. Am J Trop Med Hyg. 1998;59:832–836. doi: 10.4269/ajtmh.1998.59.832. [DOI] [PubMed] [Google Scholar]
- 6.Garcia HH, Pretell EJ, Gilman RH, et al. A trial of antiparasitic treatment to reduce the rate of seizures due to cerebral cysticercosis. N Engl J Med. 2004;350:249–258. doi: 10.1056/NEJMoa031294. [DOI] [PubMed] [Google Scholar]
- 7.Del Brutto OH, Roos KL, Coffey CS, Garcia HH. Metaanalysis: cysticidal drugs for neurocysticercosis: albendazole and praziquantel. Ann Intern Med. 2006;145:43–51. doi: 10.7326/0003-4819-145-1-200607040-00009. [DOI] [PubMed] [Google Scholar]
- 8.Singh G, Rajshekhar V, Murthy JM, et al. A diagnostic and therapeutic scheme for a solitary cysticercus granuloma. Neurology. 2010;75:2236–2245. doi: 10.1212/WNL.0b013e31820202dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Otte WM, Singla M, Sander JW, Singh G. Drug therapy for solitary cysticercus granuloma: a systematic review and metaanalysis. Neurology. 2013;80:152–162. doi: 10.1212/WNL.0b013e31827b90a8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sotelo J, del Brutto OH, Penagos P, et al. Comparison of therapeutic regimen of anticysticercal drugs for parenchymal brain cysticercosis. J Neurol. 1990;237:69–72. doi: 10.1007/BF00314663. [DOI] [PubMed] [Google Scholar]
- 11.Botero D, Uribe CS, Sanchez JL, et al. Short course albendazole treatment for neurocysticercosis in Columbia. Trans R Soc Trop Med Hyg. 1993;87:576–577. doi: 10.1016/0035-9203(93)90095-8. [DOI] [PubMed] [Google Scholar]
- 12.Venkatesan P. Albendazole. J Antimicrob Chemother. 1998;41:145–147. doi: 10.1093/jac/41.2.145. [DOI] [PubMed] [Google Scholar]
- 13.Harnett W. The anthelmintic action of praziquantel. Parasitol Today. 1988;4:144–146. doi: 10.1016/0169-4758(88)90192-5. [DOI] [PubMed] [Google Scholar]
- 14.Garcia HH, Lescano AG, et al. Pharmacokinetics of combined treatment with praziquantel and albendazole in neurocysticercosis. Br J Clin Pharmacol. 2011;72:77–84. doi: 10.1111/j.1365-2125.2011.03945.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jayakumar PN, Chandrashekar HS, Ellika S. Imaging of parasitic infections of the central nervous system. Handb Clin Neurol. 2013;114:37–64. doi: 10.1016/B978-0-444-53490-3.00004-2. [DOI] [PubMed] [Google Scholar]
- 16.No authors listed. Commission on classification and Terminology of the International League Against Epilepsy. Epilepsia. 1981;22:489–501. doi: 10.1111/j.1528-1157.1981.tb06159.x. [DOI] [PubMed] [Google Scholar]
- 17.Gonzalez AE, Falcon N, Gavidia C, et al. Treatment of porcine cysticercosis with oxfendazole: a dose-response trial. Vet Rec. 1997;141:420–422. doi: 10.1136/vr.141.16.420. [DOI] [PubMed] [Google Scholar]
- 18.Guo DM, Xie SP, Jia JP. Therapeutic efficacy of praziquantel, albendazole and a combination of the two drugs in cysticercosis. Zhongguo ji sheng chong xue yu ji sheng chong bing za zhi. 2003;21:187–188. (in Chinese) [PubMed] [Google Scholar]
- 19.Kaur S, Singhi P, Singhi S, Khandelwal N. Combination therapy with albendazole and praziquantel versus albendazole alone in children with seizures and single lesion neurocysticercosis: a randomized, placebocontrolled double blind trial. Pediatr Infect Dis J. 2009;28:403–406. doi: 10.1097/INF.0b013e31819073aa. [DOI] [PubMed] [Google Scholar]
- 20.Wu W, Jia F, Wang W, Huang Y, Huang Y. Antiparasitic treatment of cerebral cysticercosis: lessons and experiences from China. Parasitol Res. 2013;112:2879–2890. doi: 10.1007/s00436-013-3459-3. [DOI] [PubMed] [Google Scholar]
- 21.Cardenas G, Carrillo Mezo R, Jung H, Sciutto E, Hernandez JL, Fleury A. Subarachnoidal neurocysticercosis non responsive to cysticidal drugs: a case series. BMC Neurol. 2010;10:16. doi: 10.1186/1471-2377-10-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Newton CR, Garcia HH. Epilepsy in poor regions of the world. Lancet. 2012;380:1193–1201. doi: 10.1016/S0140-6736(12)61381-6. [DOI] [PubMed] [Google Scholar]
- 23.Mahanty S, Garcia HH. Cysticercosis and neurocysticercosis as pathogens affecting the nervous system. Prog Neurobiol. 2010;91:172–184. doi: 10.1016/j.pneurobio.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 24.White AC, Jr, Robinson P, Kuhn R. Taenia solium cysticercosis: hostparasite interactions and the immune response. Chem Immunol. 1997;66:209–230. [PubMed] [Google Scholar]
- 25.Nash TE, Singh G, White AC, et al. Treatment of neurocysticercosis: current status and future research needs. Neurology. 2006;67:1120–1127. doi: 10.1212/01.wnl.0000238514.51747.3a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Palomares F, Palencia G, Ambrosio JR, Ortiz A, JungCook H. Evaluation of the efficacy of albendazole sulphoxide and praziquantel in combination on Taenia crassiceps cysts: in vitro studies. J Antimicrob Chemother. 2006;57:482–488. doi: 10.1093/jac/dki484. [DOI] [PubMed] [Google Scholar]
- 27.Gonzalez AE, Bustos JA, Jimenez JA, et al. Efficacy of diverse antiparasitic treatments for cysticercosis in the pig model. Am J Trop Med Hyg. 2012;87:292–296. doi: 10.4269/ajtmh.2012.11-0371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Taylor DH, Morris DL. Combination chemotherapy is more effective in postspillage prophylaxis for hydatid disease than either albendazole or praziquantel alone. Br J Surg. 1989;76:954. doi: 10.1002/bjs.1800760927. [DOI] [PubMed] [Google Scholar]
- 29.Kramer LD, Locke GE, Byrd SE, Daryabagi J. Cerebral cysticercosis: documentation of natural history with C T. Radiology. 1989;171:459–462. doi: 10.1148/radiology.171.2.2704811. [DOI] [PubMed] [Google Scholar]
- 30.Del Brutto OH, Santibanez R, Noboa CA, Aguirre R, Diaz E, Alarcon TA. Epilepsy due to neurocysticercosis: analysis of 203 patients. Neurology. 1992;42:389–392. doi: 10.1212/wnl.42.2.389. [DOI] [PubMed] [Google Scholar]
- 31.Vazquez V, Sotelo J. The course of seizures after treatment for cerebral cysticercosis. New Engl J Med. 1992;327:696–701. doi: 10.1056/NEJM199209033271005. [DOI] [PubMed] [Google Scholar]
- 32.Carpio A, Santillan F, Leon P, Flores C, Hauser WA. Is the course of neurocysticercosis modified by treatment with antihelminthic agents? Arch Intern Med. 1995;155:1982–1988. [PubMed] [Google Scholar]
- 33.Garcia HH, Gonzalez AE, Gilman RH. Cysticercosis of the central nervous system: how should it be managed? Curr Opin Infect Dis. 2011;24:423–427. doi: 10.1097/QCO.0b013e32834a1b20. [DOI] [PMC free article] [PubMed] [Google Scholar]